Abstract
Practical relevance:
Cancer is a very common disease in cats, and many feline patients will require chemotherapy as part of their treatment plan, either as a sole agent or as part of multimodality therapy. Although some general practitioners might be reluctant to undertake injectable chemotherapy, oral chemotherapy may feel like a more acceptable treatment option to manage in practice.
Clinical challenges:
The small body size of many cats means that tablet sizes made for the human market are less appropriate, which creates dosing challenges. The administration of oral medication can also be a challenge in some cats.
Audience:
This review is aimed at general practitioners and other clinicians undertaking chemotherapy of feline cancer patients. It summarises the oral medications available for use, their therapeutic indications, dosing strategies, side effects, and health and safety aspects.
Evidence base:
The review includes information from past and recent veterinary literature on the use of oral cytotoxic drugs in cats.
Patient group:
Oral cytotoxic medications may be used in cats for a variety of tumour types, of which haematopoietic tumours, such as lymphoma, would be the most common.
Introduction
Chemotherapy is a treatment that uses drugs to stop the growth of cancer cells, either by killing the cells or preventing them from dividing. Due to its mechanism of action, it is more effective against rapidly dividing tumour cells. Chemotherapy in veterinary medicine is based on experience from human medicine, using lower drug doses to avoid severe side effects and to prioritise good quality of life (QoL). Chemotherapy is well tolerated in cats,1–3 with no significant difference in QoL noted between single agent and multi-agent protocols. 4
Most chemotherapeutic drugs are not specifically licensed for animal use and drug formulations are made for the human market. Chemotherapy can be delivered intravenously, subcutaneously (for specific drugs such as L-asparaginase or cytarabine), orally or, less commonly, by intracavitary or intratumoural routes. While the body size difference between humans and cats is less important for injectable drugs, administration of oral medications to cats can be problematic due to inappropriate tablet sizes. Patient compliance in taking oral medication is also a concern. This article focuses on the different oral cytotoxic drugs available for treating cats with cancer, when they can be used and how to use them safely.
Chemotherapy action and side effects
Most conventional chemotherapy drugs act on rapidly dividing cells, but their effects are not restricted to cancer cells. Rapidly dividing healthy cells are also targeted, leading to chemotherapy-associated side effects. The most common general side effects are gastrointestinal and haematological due to the rapid division of gastrointestinal
epithelium and bone marrow cell precursors. Lower drug doses and differences in hair growth cycles mean alopecia is less obvious in other animals than in humans; however, coat thinning, loss of whiskers or slow hair regrowth after clipping are common occurrences. Certain drugs also have their own specific side effects related to their mechanism of action. Knowledge of anticipated side effects combined with regular monitoring of animals receiving chemotherapy means that side effects can be minimised and QoL maintained (Table 1). Chemotherapy side effects are objectively graded according to the Veterinary Cooperative Oncology Group–Common Terminology Criteria for Adverse Events, so that monitoring can guide clinical decisions regarding continuation, alteration or cessation of treatment. 5 Grade I and II side effects are considered mild to occasionally moderate, whereas grade III and IV side effects are considered moderate to severe, and usually require treatment alterations.
Suggested doses and possible side effects of oral chemotherapy drugs in cats, graded according to the Veterinary Cooperative Oncology Group–Common Terminology Criteria for Adverse Events (VCOG–CTCAE v2) 5

AEs = adverse events; ALT = alanine aminotransferase; EOD = every other day; ETD = every third day; GI = gastrointestinal; MTD = maximum tolerated dose; M, W, F = Monday, Wednesday, Friday; RI = reference interval
Conventional chemotherapy is delivered at maximum tolerated dose (MTD) – the highest dose that does not cause a dose-limiting toxicity. However, an alternative method of delivery is low dose, daily or alternate day administration of cytotoxic drugs to inhibit angiogenesis and alter the immune system (metronomic chemotherapy), which is usually associated with fewer side effects and involves oral medication (Figure 1). Targeted therapy is also available, whereby drugs target specific intracellular pathways and/or cell surface receptors that are altered in cancer cells; normal cells without the altered pathway are spared and side effects are theoretically more limited.
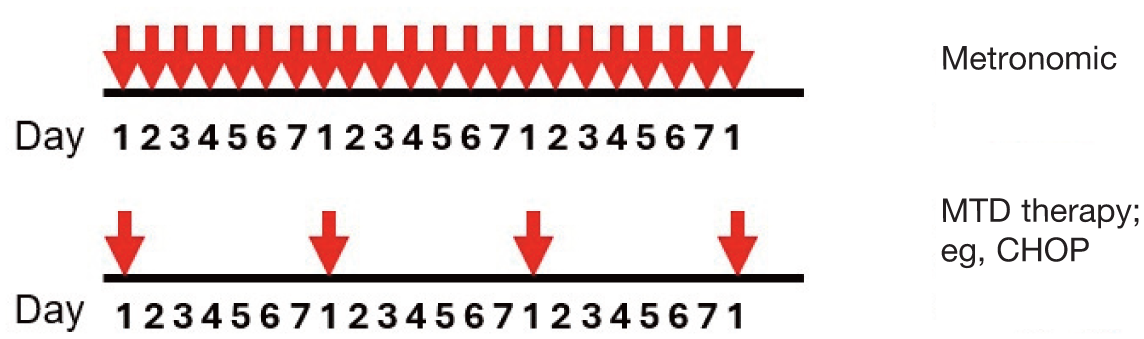
Diagram illustrating the dosing frequencies of metronomic (eg, daily, every other day) and maximum tolerated dose (MTD) (eg, weekly or biweekly) chemotherapy. CHOP = cyclophosphamide, doxorubicin, vincristine, prednisolone
Health and safety aspects of chemotherapy
Chemotherapeutic drugs can be mutagenic, carcinogenic and teratogenic, so health and safety guidelines are required to avoid the exposure of veterinary/nursing staff and clients to the harmful substances in drug prep-arations. 25 Practices must comply with the current legislation relevant to their state or territory: for example, in the UK, this would be the Health and Safety at Work Act 1974, Management of Health and Safety at work regulations 1999, Control of Substances Hazardous to Health (COSHH) Regulations 2002, and in the USA, relevant information on USP <800> Hazardous Drugs – Handling in Healthcare Settings, which can be found in Gabay. 26
Prior to embarking on chemotherapy, a frank and open conversation should be had with the owners to investigate whether there are any vulnerable family members more at risk – for example, young children, breastfeeding or pregnant women, or immunocompromised people. Based on these discussions, some families may decide not to go ahead with treatment.
Dispensing and administering chemotherapeutic drugs
Injectable drugs
While this review focuses on oral medications, the authors wish to also highlight the health and safety aspects of injectable drugs. Since environmental contamination during drug preparation and administration has been reported in human27–29 and veterinary hospi-tals, 30 a class II biological safety cabinet is highly recommended for the preparation of chemotherapy drugs for injectable administration. If this is not possible, venting aids and/or a closed system (eg, Spiros, ICU Medical; PhaSeal, Chemfort) are recommended to prevent aerosol formation when drawing up and administering injectable drugs to patients.25,31 Nursing staff and other employees handling chemotherapeutic drugs in practice should be trained on the proper use of these devices.
Oral drugs
Oral medications are administered at a veterinary practice or given on a more frequent basis by owners at home. A number of safety precautions should be followed when dispensing these drugs and owners should also be informed about how the chemotherapeutic drugs should be stored and handled, and how any tablets or capsules spat out, which may have exposed active drug due to a damaged outer protective layer, should be dealt with (see the ‘Health and safety guidelines for chemotherapeutic agents’ box on the following page). In addition, owners should be provided with advice on how to administer oral medication to their cat, including relevant tips to ensure successful swallowing (see the ‘Tips for administering oral medications’ box).
Handling waste products and bodily fluids of cats being administered chemotherapeutic drugs
Some chemotherapeutic agents are prodrugs that need to be processed in the body to reach their active state; they are most often metabolised prior to excretion in urine or faeces. Drug metabolites may be inactive or retain some cytotoxicity, so intact active drug may be excreted in small quantities. It is therefore assumed that the urine and faeces of pets receiving chemotherapy may contain trace amounts of active drug. Similarly, other bodily fluids such as saliva or vomit may be contaminated. This two-fold risk of exposure, either via the cat’s saliva or via handling urine, faeces, vomitus or sebum/cerumen, all require precautions to be taken, and these can vary depending on whether the cat is contained indoors or has outdoor access. Steps for minimising exposure via a patient’s waste products and other bodily fluids can be found in the ‘Health and safety guidelines for chemotherapeutic agents’ box.
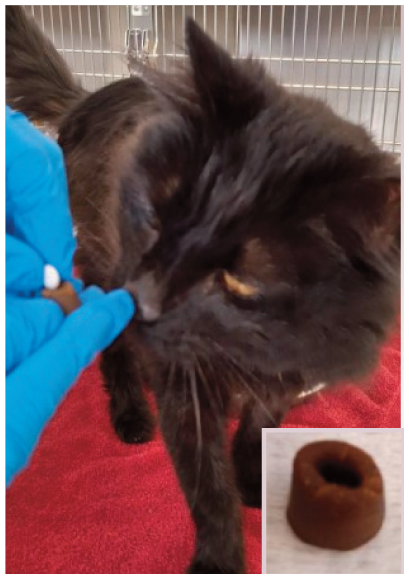
Pill Assist (Royal Canin) can be used to administer oral chemotherapy to cats

Pill popper tools for administering oral medication

Use of syringe and water to assist swallowing after tablet administration


Childproof container for dispensing oral medication, labelled with cytotoxic tape

Litter tray liners help to contain excreted waste and minimise aerosol formation if rinsing out the tray

The period over which drugs may be excreted varies according to the class of drug, although most data are extrapolated from human medicine and most relate to injectable drugs. Excreted drug concentrations in urine drop off rapidly in the first 3 days after admin-istration, whereas those in faeces may remain for 5–7 days. 25 The period of risk in human patients is approximately 4, 3 and 2 days for cyclophosphamide, lomustine and chlorambucil, respectively. 32 There is no such specific information for cats about any drugs. As such, a ‘chemo barrier’ period of 7 days is generally recommended, during which time owners should adhere to health and safety guidelines regarding the handling of waste products.
Practical considerations for oral chemotherapy administration
Dosing to body surface area
The calculated dose of most chemotherapy drugs is based on body surface area (BSA), which more closely correlates with physiological processes than body weight (BW) 33 and is used for drugs with a narrow therapeutic window. The formula to calculate BSA includes a constant, K, which is 10 for cats and 10.1 for dogs, and is:
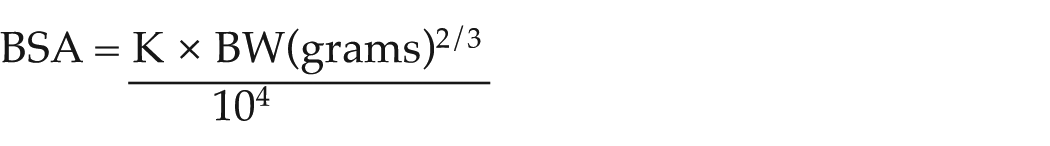
However, the accuracy of the constant is debated, as it derives from work in puppies and kittens of less than 4 months old, with no gut content, 34 which may not be entirely applicable to larger adult dogs and cats. Nonetheless, tables are readily available (British Small Animal Veterinary Association Formulary 35 ) with the alculation already performed to obtain the equivalent BSA for a particular weight (Table 2). Online resources such as the Chemopet BSA Calculator (chemopet. co.uk/bsa-calculator) are also available and provide a rapid way to convert weight into BSA.
BSA (m2) for equivalent weight, derived from the BSA conversion formula for cats

BSA = body surface area
For certain drugs (eg, melphalan, tocer-anib), doses for cats are based on BW (mg/kg) to avoid relative overdoses. Other significant differences in liver metabolic pathways between cats and dogs indicate that drug doses are often lowered in cats to avoid excessive toxicity. 36 Since drug dosing is linked to metabolism and excretion of drugs, severe liver or renal dysfunction may impact activation or excretion, leading to inadequate efficacy or severe toxicity. The latter is more common in cats, as most animals affected by cancer are elderly and some may have early renal disease/insufficiency. Drugs that are renally excreted may need to be dose-reduced or avoided, depending on the severity of the disease. 37 Dehydration, often caused by gastrointestinal side effects such as vomiting or diarrhoea, may worsen renal function in a cat that was previously coping well.
Tablet sizes
No oral liquid chemotherapy options are currently available (apart from the US Food and Drug Administration [FDA]-approved liquid imatinib – Imkeldi; Shorla Oncology), but most drugs are available in several sizes of tablets or capsules. Depending on the recommended dose, the small size of cats can present dosing challenges due to the requirement to avoid splitting tablets for health and safety reasons. Compounding pharmacies (eg, Nova Labs, Chemopet) may be used to reformulate drugs to a size appropriate for a cat; however, variation and inconsistencies in the precision of reformulations have been reported for lomustine in dogs, 38 famciclovir in cats 39 and in compounded formulations of chlorambucil, melphalan and cyclophosphamide. 40 An alternative to drug reformulation may be to extend the dosing interval between drug administrations, so that the same equivalent dose is given over a set period (eg, 1 or 2 weeks) but the number of tablets given in the same period is reduced (see Case B, for example).
Common oral cytotoxic drugs for cats
Common oral cytotoxic drugs available for cats are outlined in Table 1.
Alkylating agents
Most oral chemotherapy options used for cats are alkylating agents. These are antitumor drugs that covalently bind alkyl groups to cellular macromolecules and form inter- or intra-strand cross-links in DNA. Alkylating agents include cyclophosphamide, chlorambucil, lomustine, melphalan, procarbazine and temozolomide (Figure 7). Some drugs, such as chlorambucil and melphalan, should be refrigerated when stored. 40

Common alkylating agents used in cats, illustrating tablet/capsule size: (a) cyclophosphamide 50 mg and reformulated 10 mg capsule; (b) chlorambucil 2 mg; (c) lomustine 40 mg and reformulated 10 mg capsules; and (d) melphalan 2 mg
Cyclophosphamide
Cyclophosphamide is a nitrogen mustardcontaining prodrug that is available in both injectable and tablet (50 mg in the UK and USA; 25 mg in the USA) form (Figure 7a). Metabolic activation occurs via microsomal mixed-function oxidases predominantly in the liver and may be impaired by significant liver disease. Cyclophosphamide activation produces phosphoramide mustard and acrolein, both of which are excreted via the kidneys into urine. Phosphoramide mustard is the most active cyclophosphamide metabolite and is capable of bifunctional alkylation and cross-linking.
In dogs, the most recognisable side effect of cyclophosphamide – sterile haemorrhagic cystitis – is caused by acrolein in urine contacting the bladder wall. This side effect is very rare in cats, however, and the dose-limiting toxicity is neutropenia. 9 A urine sample pretreatment is useful for a baseline assessment of haematuria, but after that the authors rarely check urine samples in cats to monitor for cystitis. In multi-agent protocols such as CHOP (cyclophosphamide, doxorubicin, vincristine, prednisolone), the recommended dose of cyclophosphamide is 200–250 mg/m2;7,8 however, dose escalation studies have revealed that 460 mg/m2 given orally or intravenously every 3 weeks could be safe to administer.9,10 Metronomic cyclophosphamide has been evaluated in cats with a variety of neoplastic diseases, and the best response to treatment in most cats was stable disease. A good toxicity profile is obtained with a median cyclophosphamide dose of 14 mg/m2 (range 6–27 mg/m2) given q24h or q48h. 6
Chlorambucil
Chlorambucil is a nitrogen mustard derivative that passively diffuses into cells and has direct bifunctional alkylating ability. It is a prodrug converted in the liver to its active metabolite, phenylacetic acid. Chlorambucil targets B lymphocytes and is considered a slow-acting immunosuppressive agent that may require 2 weeks to reach therapeutic efficacy. It is only available in oral (2 mg tablet) form (Figure 7b). The recommended dosage is typically 2 mg/cat q48h or q72h for smaller cats. 11 Pulse doses of 20 mg/m2 every 2 weeks or 15 mg/m2 q24h for 4 days every 3 weeks can also be used when owners are unable to administer tablets.13,41 Chlorambucil is very well tolerated with few side effects, although occasional neutropenia/thrombocytopenia and rare hepatotoxicity have been reported. 12 After prolonged therapy (months to years), severe myelosuppression, particularly thrombocytopenia, may be observed, with prolonged recovery after the discontinuation of treatment. 41 Gastrointestinal toxicity is uncommon but may be seen with higher pulse doses, 42 and Fanconi syndrome has been reported in four cats treated with chlorambucil and prednisolone. 43 Neurotoxicity has also been reported but appears very rare. 42
Lomustine
Lomustine (cyclohexyl chloroethyl nitrosourea – CCNU) is available only as 40 mg tablets in the UK (Figure 7c) and is usually reformulated for use in cats. In the USA, it is available as 5 mg, 10 mg and 40 mg, and in Australia as 10 mg and 40 mg tablets. It is a nitrosourea-based agent that is highly lipid soluble and enters cells by passive diffusion. Under aqueous conditions and at physiological pH, CCNU will spontaneously decompose to a reactive centre capable of DNA alkylation, and DNA–DNA or DNA–protein cross-links. CCNU and its metabolites have high lipid solubility, allowing them to distribute widely in tissues and cross the blood– brain barrier. 44
In cats, lomustine is used to treat lymphoma,14,45 mast cell tumours (MCTs) 46 and histiocytic diseases at a dose range of 30–60 mg/m2 every 4–6 weeks.14,46,47 As lomustine crosses the blood–brain barrier, it may also be useful for treating brain tumours, although no specific information is available for cats. The dose-limiting toxicity in cats is neutropenia, with variable nadirs (7–28 days) that may be prolonged. 48 A 4–6 week interval between administrations is often required, which is much longer than for dogs (3–4 weeks). Hepatotoxicity, however, which is a problem for dogs, appears to be less common in cats, 49 and the authors do not routinely dispense liver supplements alongside lomustine. Lomustine can be used in cats as a single agent, combined with other drugs in multi-agent protocols 50 or with abdominal radiotherapy to treat gastrointestinal lymphoma. 51
Melphalan
Melphalan (L-phenylalanine mustard) is a nitrogen mustard-containing DNA crosslinking agent with direct alkylating activity (no activation required). It has a similar structure and pharmacology to chlorambucil but is actively transported into tumour cells by amino acid transporters. Melphalan has been used as a single agent in the treatment of multiple myeloma in cats at a dosage of 0.1 mg/kg q24h for 10 days, with the dosage being reduced to 0.05 mg/kg q24h there-after. 16 Given that melphalan is only available in 2 mg tablets (Figure 7d), this protocol often requires reconstitution from a compounding pharmacy, although the dosage may not be ideal as 65% of cats experienced adverse events (mainly haematological), with treatment discontinued in 15% of cats. 16 A pulse protocol of 7 mg/m2 q24h for 5 days every 3 weeks evaluated in dogs 52 was as well tolerated as the daily protocol; however, this has not been evaluated in cats. Melphalan has been evaluated in combination with mechlorethamine, vincristine and prednisolone (MOMP protocol) at a dosage of 2 mg weekly for the treatment of resistant feline lymphoma 15 with a favourable toxicity profile and no hospitalisations secondary to treatment.
Procarbazine
Procarbazine is a prodrug that requires metabolic activation to generate active metabolites, predominantly in the liver. 53 Procarbazine’s active metabolite leads to O 6 -methylguanine DNA lesions, causing DNA strand breaks and apoptotic cell death in mismatch repair proficient cells. 54 Procarbazine is not often used in cats but has been reported as part of multi-agent first-line (alongside prednisolone, L-asparaginase, doxorubicin, vinblastine, cyclophosphamide – VAPC) and rescue (alongside mechlor-ethamine, vincristine and prednisolone – MOPP) chemotherapy protocols for lymphoma at a dosage of 10 mg q24h for 14 days, with a favourable toxicity profile for both.17,55
Temozolomide
Due to toxicity, temozolomide is currently not recommended for use in cats. Temozolomide is a prodrug that acts in a non-cell cyclespecific manner by methylation of purine bases. 56 It does not require metabolic
activation as it spontaneously decomposes to the active agent. 57 As a small lipophilic drug, it is one of the few chemotherapy drugs that can penetrate the blood–brain barrier and work in the central nervous system, so is used to treat high-grade glioma in dogs. 58 Information about its use in cats is limited, but a pilot study described the treatment of various malignancies. 18 Despite evidence of temozolomide’s efficacy, the study had to be discontinued due to unacceptable adverse events, including grade III and IV haematological and gastrointestinal toxicities, and grade III pleural and pericardial effusions, leading to euthanasia secondary to toxicity in 40% of the cats. Additional studies looking into optional dosing and cumulative side effects are required before this drug can be recommended for treating cats.
Antimetabolites
Hydroxyurea
Hydroxyurea inhibits ribonucleotide reductase, resulting in the depletion of deoxyribonucleotide pools, and also inhibits DNA synthesis. 59 Hydroxyurea use in cats is rarely reported but is most often recommended for treating bone marrow diseases, such as poly-cythaemia vera (PV). A cat with presumed PV was treated with hydroxyurea initially at 20 mg/kg q24h and later reduced to 10 mg/kg q48h for 2 years with no significant side effects. 19 A case series of six cats diagnosed with PV reported weekly maintenance dosages of hydroxyurea of 300–875 mg/cat/ week over 10–79 months. The most common side effects were transient vomiting and anorexia, and neutropenia at the higher induction doses. 20 Another cat with a grade I meningioma, treated with prednisolone and hydroxyurea initially at 25 mg/kg q24h for 2 weeks, and then 25 mg/kg q48h for 1 year, reported no significant side effects. 60
Tyrosine kinase inhibitors
Tyrosine kinase inhibitors (TKIs) are targeted therapies that block the signal transduction pathways of protein kinases in cancer cells and prevent downstream signalling. In veterinary medicine, these were developed as targeted therapy for canine mast cell tumours (MCTs) as mast cells express the cell membrane receptor KIT (for a list of the cellular targets of tyrosine kinase inhibitors and their full names, see Table 3). The intracellular tyrosine kinase enzyme part of the receptor is often mutated in MCT, leading to constitutional downstream activation. Some TKIs cross react to inhibit other cellular and stromal kinases (Table 3), and some can be antiangiogenic by blocking VEGFR2, thus targeting other tumour types.
Cellular targets of tyrosine kinase inhibitors

Toceranib
Toceranib (Palladia; Zoetis) is licensed in dogs for treating non-resectable or recurrent Patnaik grade II/III MCT, and in the UK can only be used off-licence in cats. In the USA, Palladia has full FDA approval and can legally be given off-label to both dogs and cats. Although its primary target is KIT, it also targets PDGFR, Flt3 and VEGFR. 61 It has been used off-license for various tumour types in dogs,62–64 and more recently in cats, for the treatment of oral squamous cell carcinoma (SCC),65,66 MCT, 67 mammary carcino-ma 68 and pancreatic carcinoma. 69 Clinical benefit rates (complete response [CR], partial response [PR] and stable disease [SD] combined) are variable, ranging from 45% to 86%. Toceranib is well tolerated in cats21,70 and, unlike in dogs, development of proteinuria and hypertension are not significant side effects. 71 Toceranib is available as 50 mg, 15 mg or 10 mg tablets, with the last most suitable for cats (Figure 8). The authors usually prescribe 2.5–3 mg/kg q48h or on Monday, Wednesday and Friday (ie, 10 or 15 mg per cat according to size).
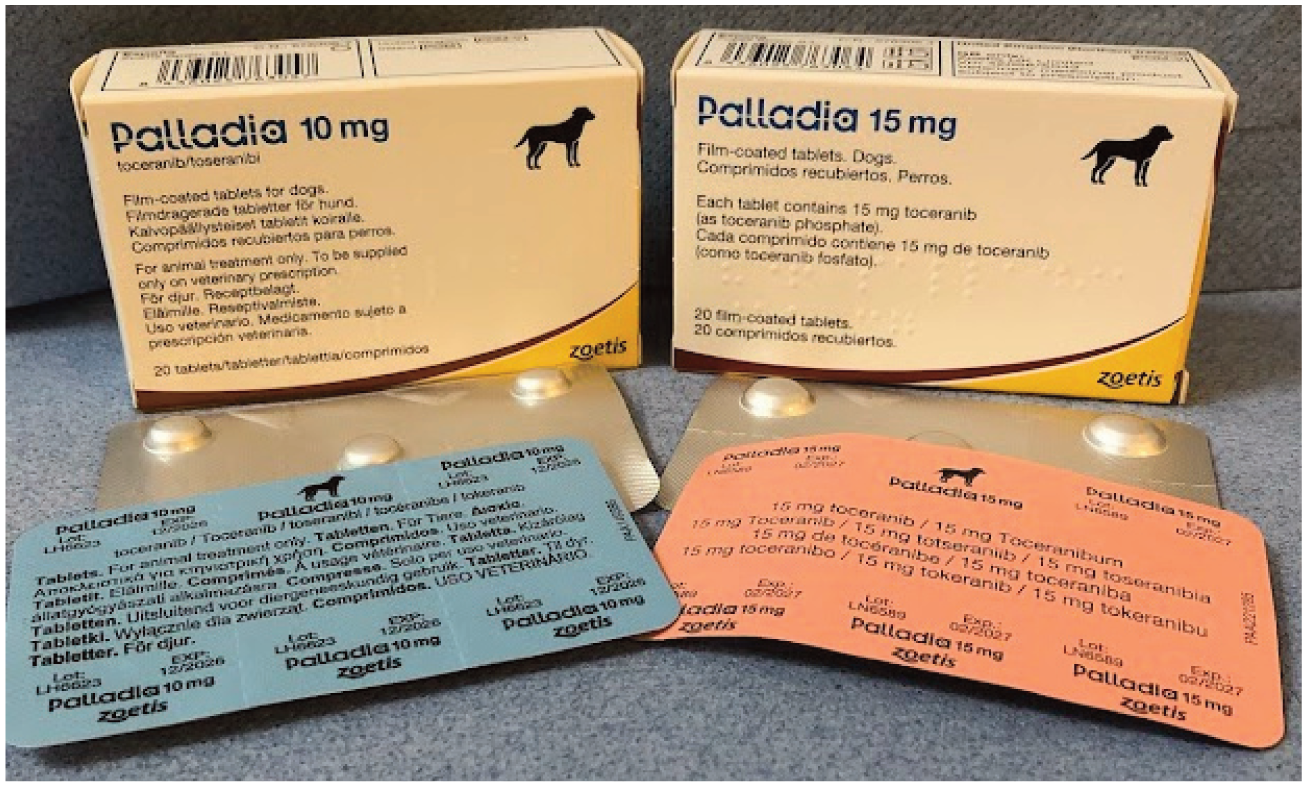
The most commonly used tablet sizes of the tyrosine kinase inhibitors, toceranib (Palladia) suitable for cats: blister packs of 10 mg and 15 mg tablets
Masitinib
Masitinib mesylate (Masivet), similarly to toceranib, targets KIT and other receptors such as PDGFR α/β, FGFR 3 and Lyn. 72 In the UK, masitinib is licensed to treat non-resectable or recurrent Patnaik grade II/III MCT with a KIT mutation in dogs, but it is not licensed in the USA. Masitinib, available as 50 mg and 150 mg tablets, appears to be safe and well tolerated in cats at a dosage of 50 mg q24h or q48h. 22 The most common side effects reported in one study were gastrointestinal (particularly vomiting and less commonly diarrhoea), neutropenia and proteinuria, which was occasionally moderate to severe. 22 Proteinuria only developed in cats treated with daily masitinib and resolved once the drug was discontinued. 22 Increases in serum creatinine were also noted but remained within the reference interval.
Imatinib
Imatinib mesylate (Gleevec) is an inhibitor of several tyrosine kinases and targets BCR-ABL, KIT, PDGFR, CSF1R, ABL1, ABL2, DDR1/2 and Lck,23,73 but it is not licensed for use in either cats or dogs. It is well tolerated in cats, with only mild and occasional constitutional and gastrointestinal side effects reported, 24 and has been used off-licence to treat feline MCT with mutated KIT.74,75 Rare liver enzyme elevations of variable severity (grade II–IV) have been noted.23,24 The most common dose is 10 mg/kg given q24h. It is only available as 100 mg and 400 mg tablets and therefore needs to be reformulated for cats, although a liquid paediatric form has recently been tested. 76
Other chemotherapy-related oral medications
Prednisolone
Prednisolone is the preferred oral corticosteroid drug in cats, available as 1 mg and 5 mg tablets and liquid solutions (Figure 9). Cats achieve significantly higher plasma concentrations when administered oral prednisolone vs prednisone, suggesting that cats either have lower prednisone absorption and/or decreased prednisone conversion to prednisolone. 77 Diabetes mellitus can develop with glucocorticoid treatment in cats, 78 so those receiving long-term corticosteroids as a part of their chemotherapy should be carefully monitored for the development of relevant clinical signs, and owners should be counselled regarding other possible risk factors such as obesity. 79

Other oral drugs commonly used in feline chemotherapy (from left to right): prednisolone 1 mg and 5 mg tablets, prednisolone liquid and meloxicam suspension
Glucocorticoids are most often used to treat lymphoid neoplasia. Use of single agent corticosteroids is considered palliative, with a median survival time (MST) of 50 days reported for naive canine nodal high-grade lymphoma. 48 Exact survival times for cats treated similarly
are not available but are likely to be equally short; however, one study reported a survival time of 60 days for cats with extranodal lymphoma treated with palliative corticosteroids. 80 Glucocorticoids induce multidrug resistance to future treatment with cytotoxic agents, and a recent study in feline lymphoma cell lines reported decreased sensitivity to doxorubicin and vincristine after 72 h of treatment with prednisolone. 81 Similarly, in cats who achieved complete remission from extranodal lymphoma with chemotherapy treatment, reduced survival times were observed in those who had received pretreatment with corticosteroids. 80 Initial use of prednisolone is therefore discouraged in patients in whom chemotherapy is likely to be pursued in the future.
Non-steroidal anti-inflammatory drugs
In the treatment of certain cancers, nonsteroidal anti-inflammatory drugs (NSAIDs) may be more beneficial than corticosteroids, as prostaglandins are involved in many physiological and pathological processes, including pain, inflammation and cancer progression. 82 NSAIDs inhibit prostaglandins by the cyclo-oxygenase (COX) enzymes, and leukotrienes through 5-lipoxygenase (5-LOX) enzymes. 83 Several feline cancers, including mammary carcinoma,84,85 oral SCC 86 and urothelial carcinoma 87 express COX-2, and, as such, there is rationale for their use in the palliative setting or combined with other drugs.
The licensed NSAID meloxicam, most commonly used in cats, is available as a liquid formulation (0.5 mg/ml) (Figure 9). When dosed at 0.02 mg/kg q24h, and combined with toceranib q48h, it was found to be safe and well tolerated for treating various feline malignancies. 88 The other NSAID licensed for cats, robenacoxib, inhibited primary feline injection site sarcoma (FISS) cells in vitro. 89
Main indications and tumour types for using oral chemotherapy in cats
Chemotherapy is used as a single treatment modality for systemic chemosensitive neoplasias such as lymphoma, but for more chemoresistant solid tumours it is usually combined with surgery and/or radiation therapy. For some tumour types a single drug is particularly effective, but for most cancers a combination of drugs in an established protocol is more reliable, combining different modes of action and maximising the chance of a response. Resistance to chemotherapy is one of the main causes of treatment failure, and this can be caused by reduced drug uptake or increased drug efflux, changes in drug metabolism, increased capacity to repair drug-induced DNA damage or increased resistance to apoptosis. 90 Multi-agent protocols tend to be more effective as they are less likely to be affected by the mechanisms of resistance. It is important, however, that the drugs do not interfere with each other or that the resulting side effects combine to be excessive. Most protocols include both injectable and oral medications.
Lymphoma
Lymphoma is one of the most common tumours in cats, with alimentary lymphoma being the most common anatomical subtype. Alimentary lymphoma is further subclassified into low-grade, intermediate–high-grade and large granular lymphocyte lymphoma, each of which has distinct clinical and histopathological features. Lymphoma is highly chemosensitive, and chemotherapy is the main treatment modality.
Low-grade alimentary lymphoma is an indolent subtype with a good prognosis when treated with chlorambucil and prednisolone. Progression-free survival (PFS) times of 2–3 years can be achieved,12,13 either in pulse doses of 20 mg/m2 every 2 weeks 13 or metronomic doses of 2 mg q48h or q72h. 11
Intermediate–high-grade lymphoma and large granular lymphocyte lymphoma carry a guarded to poor prognosis and often respond best to multi-agent protocols including injectable agents (CHOP- or COP-based protocols). However, single-agent lomustine has been reported for the treatment of intermediate to
large cell gastrointestinal lymphoma. 14 The response rate was 50%, with an MST of 330 days for responders and 108 days for all cats, which is comparable with previous reports using multi-agent protocols.91,92 Large granular lymphocyte lymphoma is a particularly aggressive subtype of large cell lymphoma, carrying a worse response to treatment and prognosis, with MSTs of 90 days reported for cats treated with lomustine and 21 days for the entire patient population. 93
Carcinomas
Oral SCC and mammary carcinomas are commonly diagnosed in cats. Most are treated surgically; however, due to their aggressive biological behaviour and metastatic potential, adjuvant MTD chemotherapy is often pursued. For unresectable carcinomas, treatment with oral toceranib has been reported.66,68 Measurable response rates (CR and PR) are approximately 13–35%, with higher clinical benefit rates (CR, PR, SD) of 56–64%. The main benefit of toceranib in this setting, therefore, is delaying disease progression rather than objective reduction of tumour burden, and, as such, it is only appropriate for patients who are clinically well and still enjoying a good QoL.
Metronomic cyclophosphamide has been used as an adjuvant therapy of cats with mammary carcinoma and compared with cats treated with surgery alone or surgery with adjuvant doxorubicin. 94 The MST (approximately 14 months) was not significantly different to that of the other groups, and, consequently, the true benefit of adjuvant metronomic chemotherapy in this setting remains controversial.
Sarcomas
As for carcinomas, most sarcomas in cats are treated surgically, but for aggressive sarcomas such as FISS or visceral haemangiosarcoma, adjuvant chemotherapy often forms part of a multimodal approach to address disease progression and potential metastasis.95,96 Cyclophosphamide has been used in combination with doxorubicin to treat feline non-resectable fibrosarcomas, with half of the cats achieving a reduction in tumour burden of >50%, and improved survival times for responders. 97 Oral lomustine has been evaluated in a phase II clinical trial treating FISS in the gross disease setting, with a response rate of 25% and a median duration of response of 82.5 days. 98 Toceranib has also been used in a prospective clinical trial to treat unresectable FISS, with no measurable responses observed, 99 despite previous promising in vivo responses to imatinib and masitinib in FISS cell lines.100,101
Multiple myeloma
Multiple myeloma is a rare neoplasia in cats, but it can be treated effectively with oral cytotoxic drugs, achieving response rates of 87– 100% with melphalan, cyclophosphamide or chlorambucil (each as a single agent), alongside prednisolone. 16 In dogs, the treatment of choice is melphalan and prednisolone; in cats, cyclophosphamide has a similar efficacy to melphalan with a more favourable toxicity profile.16,102 Outcomes are variable, with MSTs of 4–13 months reported. A small study divided cats into ‘aggressive’ and ‘less-aggressive’ multiple myeloma subtypes based on calcium levels, azotaemia, anaemia, Bence Jones proteinuria, pathological fractures and persistent hyperglobulinaemia after 8 weeks of treat-ment. 103 MSTs of 5 days were obtained for the aggressive group compared with 387 days for the less aggressive group.
Histiocytic diseases
Feline histiocytic diseases are rare and not well described in the literature, but are generally divided into histiocytic sarcoma, feline progressive histiocytosis and pulmonary Langerhans cell histiocytosis. 104 Prognosis is guarded to poor and treatment protocols are
not well established; however, limited case examples have been treated with oral therapies including lomustine, toceranib, masiti-nib, chlorambucil, cyclophosphamide, prednisolone and NSAIDs.105–107 Responses and survival times were highly variable, with PFS times ranging from 36 to 1095 days, and MSTs ranging from 10 to 490 days. 105
Mast cell tumours
MCTs in cats may present as cutaneous, 108 splenic/visceral 109 or intestinal 110 forms, each with variable prognoses and treatment options. For non-metastatic cutaneous and splenic MCTs, surgery remains the treatment of choice.109,111 However in non-resectable or metastatic MCTs, chemotherapy – either as a primary treatment or adjuvant – may be required. Alkylating agents, such as lomustine and chlorambucil, and TKIs, such as tocer-anib, have been used to treat splenic and intestinal MCTs108,109 with questionable benefit. The MST for splenic MCTs treated with splenectomy and adjuvant chemotherapy was not longer than that for cats treated with splenectomy alone (853 days vs 856 days), and for gastrointestinal MCTs, the MST was no different than for cats treated with steroids alone (541 days vs 555 days). Toceranib was used to treat different types of MCT 67 with a clinical benefit rate of 80% and median response duration of 32 weeks. Lomustine was used at a dose of 50–60 mg/m2 in the treatment of (mainly cutaneous) MCTs with an overall response rate of 50% and a median response duration of 168 days. 46



Key Points
✜ Oral chemotherapy is most useful for lymphoid malignancies in cats but can also be suitable for various carcinomas, sarcomas and MCTs.
✜ Oral medication may be the only cytotoxic therapy required or it may be part of a multi-agent protocol (eg, CHOP).
✜ Tablet size may present dosing challenges as tablets cannot be split or crushed for health and safety reasons.
✜ Owners should be fully advised on health and safety aspects of administering chemotherapy to their cat.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors of this commissioned Clinical Spotlight review received an honorarium; as for all JFMS articles, this Clinical Spotlight article went through peer review.
Ethical approval
This work did not involve the use of animals and therefore ethical approval was not specifically required for publication in JFMS.
Informed consent
This work did not involve the use of animals (including cadavers) and therefore informed consent was not required. For any animals or people identifiable within this publication, additional informed consent for publication was obtained.
