Abstract
Case summary
An 8-year-old female neutered domestic shorthair cat was referred for further investigations of lethargy, polyuria, polydipsia, hyporexia and weight loss. One month prior, the cat had been diagnosed with normotensive and non-proteinuric chronic kidney disease and a single small well-defined splenic nodule thought to represent a benign process. Specialist abdominal ultrasonography documented changes suggestive of pyelonephritis alongside a solitary splenic mass (2 cm in size). Follow-up imaging 4 weeks later showed progression of the splenic mass (3.5 cm) and a novel marked anaemia. Splenectomy and histopathology were performed, which confirmed a diagnosis of splenic haemangiosarcoma. The cat was started on metronomic thalidomide (5 mg/kg PO q24h; BOVA) 16 days postoperatively. The cat re-presented to the referring veterinary practice 169 days after splenectomy (153 days after starting metronomic thalidomide) owing to lethargy and hyporexia. Abdominal ultrasound identified a solitary hepatic mass and free peritoneal fluid; the cat was subsequently euthanased without further investigations.
Relevance and novel information
This is the first published case report documenting the management of splenic haemangiosarcoma in a cat with splenectomy and metronomic thalidomide. Our observations indicate that survival time falls in line with other chemotherapeutic protocols and that thalidomide side effects were limited to mild polyphagia and moderate sedation, which was tolerated well if the drug was administered in the evening.
Introduction
Haemangiosarcoma (HSA) is an uncommon neoplasm in cats, occurring with similar frequency in visceral and cutaneous forms. Large retrospective studies are rare and no prospective studies have been identified in the literature. Information on treatment options is also limited, with only a few studies describing various chemotherapeutic protocols.1–4 To the authors’ knowledge, this report is the first to describe the treatment of splenic HSA in a cat using splenectomy combined with thalidomide, an immunomodulatory imide with anti-cancerous properties.
Case description
An 8-year-old female neutered domestic shorthair cat was presented for investigation of lethargy, polyuria, polydipsia, hyporexia and weight loss. One month earlier, the cat had been evaluated elsewhere and diagnosed with normotensive, non-proteinuric chronic kidney disease (CKD), International Renal Interest Society stage I, along with a small, well-defined splenic nodule considered likely benign. Medical management was initiated with maritopant (1 mg/kg PO q24h, Cerenia; Zoetis) and mirtazapine (2 mg/cat PO q48h, Mirtazapine; Summit); however, the cat’s clinical signs progressed regardless.
Physical examination was unremarkable. The cat’s body weight was 4.7 kg (1 kg loss in 5 months) with a body condition score of 5/9 and moderately reduced muscle condition. Complete blood count (CBC), serum biochemistry, venous blood gas, SNAP feline leukaemia virus (FeLV)/feline immunodeficiency virus (FIV) (IDEXX), serum cobalamin, feline-specific pancreatic lipase immunoreactivity and Toxoplasma serology were performed. Urine was collected via cystocentesis for urinalysis and culture (Table 1). Total thyroxine (T4) was assessed by the referring veterinary surgeon and was within normal limits.
Clinical parameters on first presentation
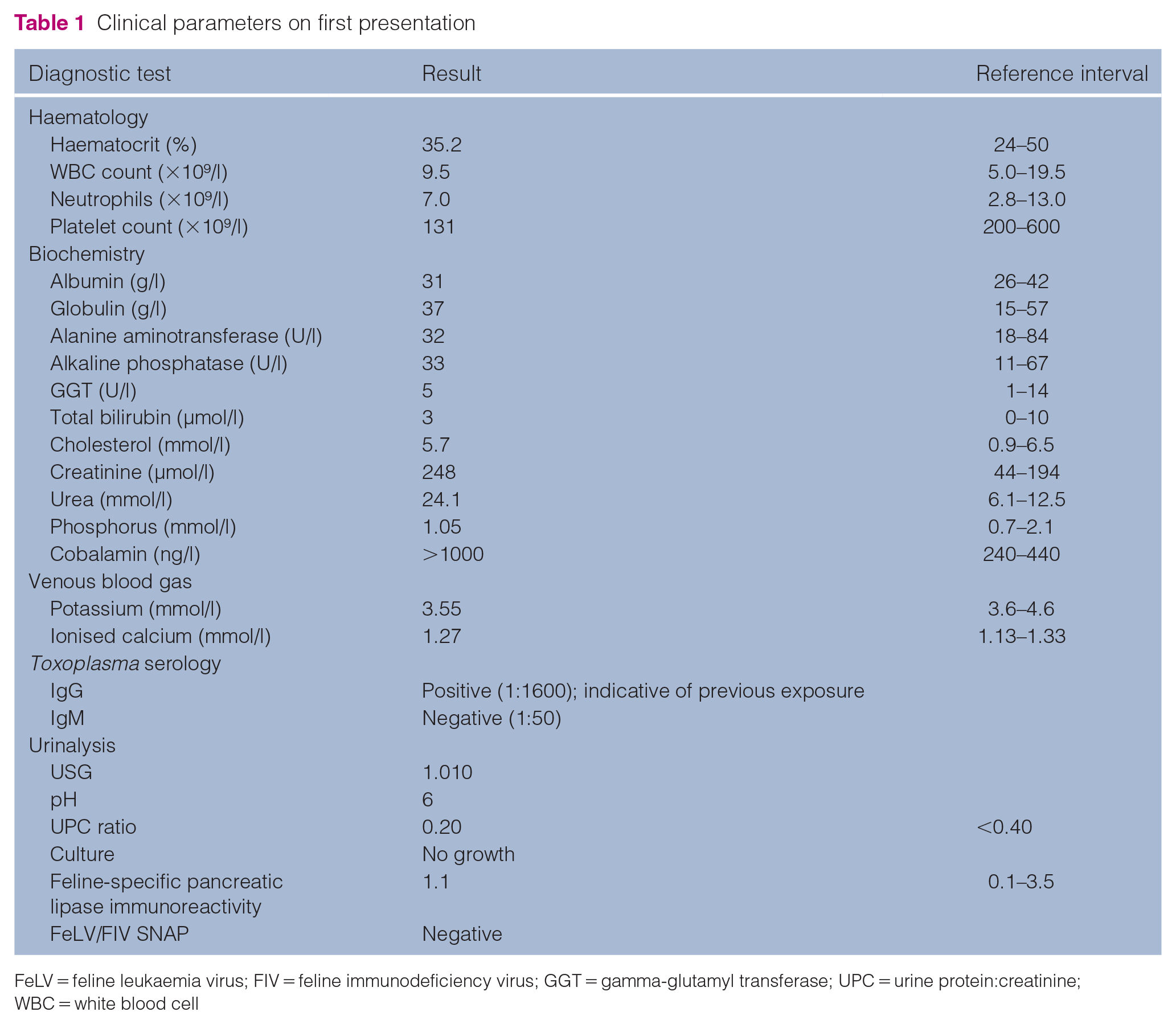
FeLV = feline leukaemia virus; FIV = feline immunodeficiency virus; GGT = gamma-glutamyl transferase; UPC = urine protein:creatinine; WBC = white blood cell
Bicavitary imaging was performed under sedation. Three-view thoracic radiography was unremarkable. Abdominal ultrasonography documented a single mass within the visceral aspect of the ventral splenic extremity, which was rounded, small (2 cm in size), well-defined, heterogeneous and had multiple intralesional tubular structures with strong colour Doppler signal (Figure 1a). Renal ultrasonography documented bilateral changes consistent with either interstitial nephritis and/or pyelonephritis, with associated focal retroperitonitis. Infiltrative renal neoplasia was considered less likely. Ultrasound-guided fine-needle aspiration of the splenic mass and kidneys was performed. The splenic aspirates were hypocellular and harvested normal splenic elements. Renal cytology was also unremarkable. Repeat abdominal ultrasonography was recommended to monitor the splenic mass progression. The cat was prescribed a 6-week course of marbofloxacin (2 mg/kg PO q24h, Marbocare; Animalcare) to treat presumptive pyelonephritis, while maropitant and mirtazapine were continued as previously prescribed.

(a) Transverse ultrasound image illustrating the splenic mass at initial assessment image with power Doppler. Assessment documented a well-defined, heterogeneous mass with multiple intralesional tubular structures displaying a strong colour Doppler signal within the gate. The callipers, located between the visceral and parietal aspects of the spleen, measure the size of the mass (2 cm). (b) Transverse ultrasound image illustrating the splenic mass at follow-up conducted 4 weeks after initial evaluation. Assessment documented new peripheral tubular structures with a strong colour Doppler signal within the gate, consistent with peripheral neovascularisation
The patient presented for re-examination 4 weeks later with fluctuating hyporexia alongside acutely worsening lethargy. A physical examination identified ongoing weight loss (a further 300 g). Repeat CBC and biochemistry was performed (Table 2). CBC documented a marked non-regenerative anaemia (haematocrit 14.4% [reference interval (RI) 27–50]; reticulocyte count 11.9 ×109/l [RI <100]). Haemotropic mycoplasma PCR was negative. Repeat abdominal ultrasonography under sedation identified significant enlargement (3.5 cm) and increased cavitation of the splenic mass, with marked peripheral tubular structures with strong colour Doppler (associated with suspected haemorrhage) (Figure 1b). Considering the mass progression, splenic neoplasia was considered most likely. The marked non-regenerative anaemia was suspected to be secondary to acute intra-lesional haemorrhage. Splenectomy was recommended and performed uneventfully. Histopathological evaluation of the spleen identified medium to large elongated to spindle-shaped cells with ovoid or elongated nuclei. These had finely to moderately stippled chromatin and 1–3 prominent nucleoli with moderate anisokaryosis and anisocytosis; 32 mitotic figures were seen in 10 high-power fields (× 400; 2.37 mm2). These changes were most consistent with a splenic sarcoma; differential diagnoses included other spindle cell sarcomas. Given the extensive areas of haemorrhage, necrosis and areas with vascular clefts, the most likely differential diagnosis was a splenic HSA. The cat’s haematocrit increased to 27.4% (RI 27–50) during the 13 days after surgery.
Clinical parameters at second presentation

GGT = gamma-glutamyl transferase; WBC = white blood cell
Metronomic thalidomide (4.2 mg/kg PO q24h; BOVA; informed signed consent was gained from the owner before administration of this off-license medication) was initiated 16 days postoperatively, alongside the continuing course of marbofloxacin to treat presumptive pyelonephritis (as before). At re-examination 2 weeks later, the cat was clinically well and had gained 200 g of body weight. Signs of moderate sedation and mild polyphagia straight after the first few doses of thalidomide were reported. Repeat CBC and biochemistry tests were performed at this re-examination (Table 3). Marbofloxacin was discontinued and no thalidomide dose adjustments were made.
Repeat clinical parameters 2 weeks after starting thalidomide

WBC = white blood cell
The cat presented 4 weeks later for re-examination with a few days’ history of worsening polyuria and polydipsia. No sedation or polyphagia after thalidomide administration were reported at this checkup. In-house CBC and serum biochemistry tests identified an improved mild anaemia (haematocrit 24.76% [RI 33.7–55.4]; reticulocyte count was not assessed) and static moderate azotaemia (creatinine 274.5 µmol/l [RI 44–194]; urea 28.7 mmol/l [RI 6.1–12.5]). Ongoing polyuria and polydipsia were presumed to be a result of the cat’s underlying CKD; further investigations into differential diagnoses of this were declined by the owner. No treatment adjustments were made at this time.
Three months later the cat presented with a 4-week history of progressive lethargy and hyporexia. A repeat CBC documented worsened mildly regenerative moderate anaemia (haematocrit 20.7% [RI 24–46]; reticulocyte count 74,648/ul [RI 0–150,000]). Abdominal ultrasonography performed at the primary care practice identified a newly detected hepatic mass and peritoneal fluid, suspected to represent haemorrhage. Further investigations, including abdominocentesis, were declined and the cat was euthanased 169 days after splenectomy (153 days after starting metronomic thalidomide).
Discussion
Haemangiosarcomas are an uncommon diagnosis in cats, accounting for less than 2% of non-haematopoietic malignant neoplasms. 3 Splenic neoplasms are uncommon, with a prevalence in the range of 2–40%.4–6 Splenic abnormalities identified using ultrasound, including small nodules, are generally incidental and considered related to non-specific illness or systemic disease. However, the presence of masses larger than 1 cm may suggest malignancy.5–8
Available data reporting prognoses for cats with visceral HSA are limited, with median survival times (STs) in the range of 77–896 days, reported in cases treated with metronomic chemotherapy (with and without surgery).4,6,9,10 Anaemia has been reported as a common finding in up to 82% of cats with visceral HSA. 2
Recommended treatment currently involves splenectomy combined with adjunctive chemotherapy. 1 Published cases report variable use of chemotherapeutic agents, including doxorubicin alone, metronomic oral cyclophosphamide alone, metronomic oral cyclophosphamide and mitoxantrone, a vincristine, doxorubicin and cyclophosphamide protocol (doses and intervals unavailable), carboplatin (dose and interval unavailable), and metronomic cyclophosphamide and thalidomide.1–4
Initial management with splenectomy was recommended for this case, particularly because of the cat’s anaemia, which was suspected to be secondary to intra-lesional splenic haemorrhage and improved postoperatively. Selection of adjunctive chemotherapy was guided by both patient- and owner-related factors. Doxorubicin-induced nephrotoxicity is widely reported, with progressive increases in serum creatinine commonly reported; although reduced dosing has been trialled, cure rates may be reduced by up to 50%.11–13 Given the cat’s pre-existing CKD and the risk of nephrotoxicity, doxorubicin was excluded. In addition, the owners declined intravenous chemotherapeutic protocols, necessitating oral chemotherapeutic options. The use of oral metronomic cyclophosphamide was considered, either solely or combination; however, the reported incidence of myelosuppression and nephrotoxicity in 9–20% of cases precluded its use.14-16 As such, the decision was made to treat the cat with metronomic oral thalidomide, based on reports of beneficial response in dogs with stage I–II splenic HSA 17 and a case report documenting its use alongside cyclophosphamide and meloxicam in a cat with urinary bladder HSA. 4 Further, nephrotoxicity has not been reported as a clinical concern in patients receiving thalidomide. 18 Non-steroidal anti-inflammatory drugs were avoided because of the cat’s underlying CKD.
Thalidomide, α-(N-phthalimido) glutarimide, is a racemic derivative of glutamic acid consisting of two enantiomers (R and S). 19 Under physiological conditions, the enantiomers undergo rapid chiral interconversion: the S-isomer inhibits certain cytokine release from mononuclear blood cells and the R-isomer is responsible for sedative effects.20–22 Metabolism into species-specific active metabolites is responsible for thalidomide’s activity and may explain its species-dependent effects. 2 Thalidomide has been investigated and used in several human cancers because of its antiangiogenic, immunomodulatory and cytokine-modulatory properties.23,24 It inhibits several cytokines, including tumour necrosis factor-alpha (TNF-α), interleukins (IL) 1-beta (β), 6 and 12, and granulocyte macrophage-colony stimulating factor (GM-CSF).25,26 The use of metronomic thalidomide for the treatment of canine HSA has been described and shown to have comparable STs to other therapies.19,27 However, further research is needed in cats, as only a single study currently documents its use in cats (administered alongside cyclophosphamide and meloxicam). 4 The dosing regimen in this case was based on current recommendations for tumour-bearing dogs. 28
Metronomic thalidomide was well tolerated overall in this case, with only mild adverse effects reported, namely polyphagia and moderate sedation. To minimise the impact of lethargy on the cat’s daily activities, thalidomide was administered in the evening. The frequency of side effects decreased over time. Lethargy is a recognised side effect of thalidomide in dogs and humans.22,28. Polyphagia, although unreported in companion animals, is an uncommon side effect in humans and thalidomide use has been reported in the management of cancer cachexia in humans.29,30 It is important to note that thalidomide is a teratogenic substance and should be handled with care, particularly by pregnant practitioners and pet carers. 31
Although there was no evidence of metastasis at diagnosis in this case, high metastatic rates in the range of 60–77% have been reported in cats with HSA, most commonly affecting the abdominal lymph nodes (33%) and liver (27%).3,9,32 The cat in this report was euthanased 169 days after splenectomy (153 days after starting metronomic thalidomide) because of a presumptive liver mass and haemoabdomen, which may have represented metastatic disease. It remains uncertain whether concurrent progression of CKD also contributed to the cat’s decline, prompting euthanasia. Previously reported STs for cats with visceral HSA are in the range of 77–896 days.2–4,9 A ST exceeding 896 days has been reported in a cat with urinary bladder HSA treated with metronomic thalidomide (alongside cyclophosphamide and meloxicam); however, the significance of this rare HSA, which may carry more favourable prognosis, is unknown when compared with other visceral HSAs. 4 Excluding that case, the reported ST is in the range of 77–197 days.2,3,9 The ST of 169 days after splenectomy in our case falls within this range, highlighting that adjunctive metronomic thalidomide can be safe (no major side effects were observed) and has the potential to offer similar STs as other, more intensive chemotherapeutic protocols. Further research is needed to support this. Comparative prognosis to cats undergoing splenectomy without chemotherapy is challenging, as 71% of such cases are euthanased within 1 day of diagnosis. 2 A notable limitation in this case is the use of a thalidomide dose previously only reported in dogs; therefore, the efficacy and safety of thalidomide in cats should be assessed in prospective studies involving larger cohorts of patients in the future.
Conclusions
This report discusses the first published case of a cat with splenic HSA treated with splenectomy and metronomic oral thalidomide. Our findings indicate that thalidomide was generally well tolerated if administered in the evening and that side effects were mild to moderate. These findings highlight that further research is needed to assess the use and safety of this metronomic chemotherapy protocol for cats with visceral HSA. Given the promising ST documented in this case, further studies comparing this protocol to alternative chemotherapeutic protocols are recommended in the future.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Off license medications
Informed signed consent was gained from the owner before administration of off license medications.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
