Abstract
Objectives
A thyrotropin (also known as thyroid-stimulating hormone [TSH]) assay using bulk acoustic wave (TSH-BAW) technology is a sensitive and specific test for diagnosing hyperthyroidism; however, the effect of various types of non-thyroidal illness (NTI) have not been evaluated with this assay. The objectives of this study were to compare serum TSH concentrations using the TSH-BAW and a currently available TSH chemiluminescent immunoassay (TSH-CLIA) in hyperthyroid cats, cats with NTI and healthy cats, as well as to compare sensitivity and specificity for diagnosing hyperthyroidism.
Methods
A prospective cross-sectional study was conducted comparing the TSH concentration of 37 hyperthyroid, 32 healthy and 32 NTI cats using the TSH-CLIA and TSH-BAW assays. The effect of disease severity was evaluated with hyperthyroidism and NTI.
Results
The TSH-BAW had a lower sensitivity (78%, 95% confidence interval [CI] 62–90) and negative predictive value (89%, 95% CI 79–95) but higher specificity (97%, 95% CI 89–100) and positive predictive value (94%, 95% CI 79–99) than the TSH-CLIA. The median serum TSH concentration was significantly different between hyperthyroid cats and both healthy and NTI cats with both assays (P <0.01) but was not different between NTI and healthy cats (TSH-CLIA P = 0.168, TSH-BAW P = 0.673). Eight (21.6%) hyperthyroid cats had a detectable TSH-BAW but undetectable TSH-CLIA concentration, with seven (18.9%) having a TSH-BAW within the reference interval. A total of 12 (18.8%) non-hyperthyroid cats (four [12.5%] healthy cats and eight [25%] NTI cats) had an undetectable TSH-CLIA compared with only two (6%) cats (one [3%] healthy cat and one [3%] NTI cat) with the TSH-BAW assay. The proportion of cats with an undetectable serum TSH concentration was significantly higher with the TSH-CLIA than the TSH-BAW in NTI cats (P = 0.008). This was especially evident in NTI cats suffering from moderate to severe illnesses (P = 0.025).
Conclusions and relevance
The TSH-BAW has a high specificity for detecting hyperthyroidism and identifies a normal serum TSH concentration in non-hyperthyroid cats more often than the TSH-CLIA. However, a normal result cannot be used to rule out hyperthyroidism.
Plain language summary
The use of the hormone thyrotropin (also known as thyroid-stimulating hormone [TSH]) can aid in diagnosing feline hyperthyroidism, a common hormonal disease of older cats. A new assay using bulk acoustic wave technology (TSH-BAW) is specific, which means that it helps to rule out the disease. Diseases unrelated to the thyroid gland, called non-thyroidal illnesses (NTIs), can affect the concentration of thyroid hormones, including TSH. The effects of NTIs have not yet been evaluated using the TSH-BAW assay. The objectives of this study were to compare serum TSH concentrations using the TSH-BAW and a currently available TSH chemiluminescent immunoassay (TSH-CLIA) in hyperthyroid cats, cats with NTI and healthy cats, as well as to compare sensitivity (ability to identify cats with the disease) and specificity (ability to identify cats without the disease). We prospectively enrolled 37 hyperthyroid, 32 healthy and 32 NTI cats, and measured their TSH using both assays. We found that the TSH-BAW had a lower sensitivity but a higher specificity than the TSH-CLIA. The median serum TSH concentration was not different between NTI cats and healthy cats. The TSH-CLIA assay falsely diagnosed healthy cats and NTI cats as having hyperthyroidism more often than the TSH-BAW, especially when cats were suffering from moderate to severe NTI. In conclusion, the TSH-BAW assay has a high specificity for detecting hyperthyroidism and identifies a normal serum TSH concentration in non-hyperthyroid cats more often than the TSH-CLIA. However, a normal result cannot be used to rule out hyperthyroidism.
Introduction
Feline hyperthyroidism (FHT) is the most common endocrinopathy in senior cats and is diagnosed in 10% of cats aged over 10 years.1,2 The diagnosis of hyperthyroidism is made by measuring serum total thyroxine (TT4) concentration. However, 10–30% of hyperthyroid cats have a serum TT4 concentration within the reference interval (RI).3 –6 Hyperthyroid cats with a TT4 concentration within the RI may reflect the severity of the disease (mild hyperthyroidism), the normal fluctuations of serum thyroid hormone levels, medications that interfere with the assay measurement of thyroid hormones and/or the presence of non-thyroidal illness (NTI). NTIs can lower the concentration of circulating TT4, with the severity of the decrease corresponding with disease severity.7 –10 NTIs are identified in 20–30% of hyperthyroid cats with a falsely lowered serum TT4 concentration.4,8
When hyperthyroidism is suspected but the serum TT4 concentration is not increased, further tests are required to confirm the diagnosis. Diagnostic options include measurement of other thyroid hormones, stimulation and suppression tests, or thyroid scintigraphy. A serum free thyroxine (fT4) concentration above the RI cannot be used alone to diagnose FHT, as 6.3–12% of cats with NTI will have a high fT4 concentration.4,9 The measurement of serum thyroid-stimulating hormone (TSH; also known as thyrotropin) in cats can also be problematic considering the absence of a feline-specific assay. The chemiluminescent immunoassay (TSH-CLIA) has been validated in cats. The lowest limit of detection of the TSH-CLIA is 0.03 ng/ml, which cannot differentiate between decreased and low-normal concentrations in cats. An undetectable TSH is seen in 98% of hyperthyroid cats, but also in 25–30% of euthyroid cats.6,11 This limitation makes the TSH-CLIA a poor screening test for hyperthyroidism in cats. The serum TSH concentration is significantly less affected by NTIs compared with other thyroid hormones, which is advantageous in a population of cats with a falsely normal serum TT4 concentration. TSH concentrations are similar between ill and healthy cats and usually remain within the RI. A higher proportion of undetectable TSH concentrations is observed in cats with severe illnesses.6,10,11
A TSH assay using bulk acoustic wave (TSH-BAW) technology recently became available, with a limit of detection of 0.008 ng/ml, with 0.01 ng/ml as the low end of the normal RI. 12 Based on this, 12% of healthy cats have an unmeasurable TSH-CLIA (<0.03 ng/ml) but normal TSH-BAW (⩾0.01 ng/ml). 12 In healthy cats, 25.6% had undetectable TSH-CLIA whereas only 1.3% had undetectable TSH-BAW. 6 Therefore, the TSH-BAW assay could better differentiate between healthy and hyperthyroid cats, and potentially help the diagnosis of hyperthyroidism when the serum TT4 concentration is normal. In that study, the TSH-BAW assay demonstrated a better correlation to hyperthyroidism and had a high specificity (98.9%) and positive predictive value (PPV) (86.9%) for the diagnosis of hyperthyroidism. 6 This assay was also studied in a small group of 12 cats with chronic kidney disease. 6 The effect of NTI on the TSH-BAW assay has not been evaluated other than in this small cohort of cats with a single illness.
The objectives of this prospective cross-sectional study were two-fold: first, to compare serum TSH concentrations using the TSH-CLIA and TSH-BAW assays in hyperthyroid cats, cats with NTIs and healthy cats; and second, to evaluate the sensitivity and specificity of the TSH-BAW for the diagnosis of hyperthyroidism. We hypothesized that the TSH-BAW would identify normal TSH concentrations in non-hyperthyroid cats more often than the TSH-CLIA and would have a higher specificity.
Materials and methods
This prospective cross-sectional study was performed on cats presenting to the Virginia-Maryland College of Veterinary Medicine Veterinary Teaching Hospital. It was approved by the institution’s Institutional Animal Care and Use Committee. Written informed consent was obtained from the owners before participation in the study.
Case selection
Client-owned hyperthyroid cats, NTI cats and healthy cats were prospectively enrolled between January 2022 and July 2023. All cats underwent a complete physical examination, complete blood count (CBC), serum biochemistry and thyroid hormone measurements (TT4 and TSH) using both TSH immunoassays. Urinalysis (UA) was performed on hyperthyroid cats and cats with NTI when urine could be collected safely via cystocentesis or free-catch methodology (NOSORB Litter; Catco Veterinary Products).
Hyperthyroidism was confirmed by the presence of a high serum TT4 concentration as well as consistent scintigraphy changes as previously described. 13 Cats were included in the healthy group based on an unremarkable physical examination and blood work (CBC, serum biochemistry, ± UA) and absence of clinical signs consistent with hyperthyroidism. Cats were included in the NTI group if they had an illness other than hyperthyroidism.
Cats that had received medications known to alter thyroid hormone concentrations (reported in cats, dogs or humans) in the previous 14 days or general anesthesia in the previous 48 h were excluded. These medications include corticosteroids, non-steroidal anti-inflammatory drugs, all anti-thyroid medications and/or iodine-restricted diets, tricyclic antidepressants, selective serotonin reuptake inhibitors, sulfonamides and phenobarbital.14 –28 Hyperthyroid cats were excluded if their history, physical examination or blood work revealed abnormalities that could not be explained by hyperthyroidism. Abnormalities consistent with hyperthyroidism included low body condition score, tachycardia, heart murmur, vomiting and diarrhea, low creatinine, mild to moderate elevation of alanine transaminase and alkaline phosphatase, dilute urine (urine specific gravity <1.035) and proteinuria. 29 Hyperthyroid cats were also excluded if they did not undergo thyroid scintigraphy. Healthy and NTI cats had to be aged 5 years or older for enrollment, to mimic a feline hyperthyroid population.3,29 –31 Healthy and NTI cats were excluded if the TT4 concentration was higher than 46 nmol/l at enrollment. Healthy and NTI cats had their TT4 concentration re-evaluated at least 3 months from enrollment, either at our institution or at the referring clinic, to ensure it remained below 46 nmol/l or below the high end of the RI if analysis was performed elsewhere.
The Severity of hyperthyroidism in the cats was categorized based on serum TT4 concentration and defined as mild (TT4 = 51–96 nmol/l), moderate (TT4 = 96.1–166 nmol/l) and severe (TT4 ⩾166 nmol/l), as modified from Peterson et al. 11
TT4 and TSH assays
Blood samples from all cats were collected via saphenous or jugular venipuncture, placed into plain non-additive tubes, allowed to clot and centrifuged (1500 × g for 10–15 mins) within 1 h of collection. The serum was immediately separated and stored at 4 °C until analysis 24 h later.
Serum thyrotropin was measured using the TSH-CLIA assay with the Immulite 2000 (Siemens Healthineers) and the TSH-BAW assay with the TRUFORMA Point-of-Care Diagnostic Platform (Zomedica). The TSH-CLIA has a lower limit of detection of 0.03 ng/ml (RI <0.03–0.3). The TSH-BAW is a bulk acoustic wave technology that has a lower limit of detection of 0.008 ng/ml (RI 0.01–0.301).6,12 The TSH-BAW assay was performed according to the manufacturer’s instructions, using the furnished material (pipettes and cartridges), by two of the investigators (CB and SMD). 32
TT4 was measured in the same serum sample using the feline TT4 assay with the same Immulite 2000. The TT4 assay has a lower limit of detection of 6.4 nmol/l (RI 18.9–50.1).
Severity of NTI
Cats in the NTI group were further divided into two groups based on the severity of their illness: mild vs moderate to severe. Cats were considered to have mild illness if they were asymptomatic or had mild clinical signs that could be treated on an outpatient basis. Moderate to severe illness was defined as cats requiring hospitalization. These cats were evaluated for systemic inflammatory response syndrome (SIRS) criteria. In cats, SIRS is determined by fulfillment of at least two of the following criteria: rectal temperature higher than 103.5 ° F (39.7 °C) or lower than 100 ℉ (37.8 °C), heart rate above 225 beats/min or below 140 beats/min, respiratory rate above 40 breaths/min, white blood cell count greater than 19,500 cells/µl or lower than 5000 cells/µl, and band neutrophil fraction above 5%. 33
Cats were categorized based on types of disease (see supplementary material): endocrine, renal, respiratory, neoplastic, gastrointestinal, hepatic, infectious and miscellaneous. ‘Miscellaneous’ consisted of diseases that did not fit into other disease categories. Cats with comorbidities were categorized into only one category based on the most clinically significant disease at the time of enrollment.
Statistical analysis
Normal probability plots were inspected to assess the distribution properties of age, TSH-BAW, TSH-CLIA and serum TT4 concentration. Subsequently, continuous variables were summarized as median (range) while categorical variables were summarized as counts and percentages. Variables were compared between groups (FHT vs NTI vs healthy) using the Kruskal–Wallis test (continuous variables) or Fisher’s exact test (categorical variables). Where appropriate, P values were adjusted for multiple comparisons using Dunn’s or Bonferroni’s correction. The sensitivity and specificity of TSH-BAW and TSH-CLIA for detecting hyperthyroidism were computed as simple binomial proportions with exact Clopper–Pearson 95% confidence intervals (CIs). All cats were used for this calculation. Additional associations between variables (two at a time) were assessed using Fisher’s exact test or the Wilcoxon rank-sum test. McNemar’s χ2 test was used to compare proportions within groups of cats. Statistical significance was set to P <0.05. All analyses were performed using SAS version 9.4.
With the TSH-BAW assay, a TSH concentration below 0.01 ng/ml was considered suppressed (below the RI) and a TSH concentration below 0.008 ng/ml was considered undetectable.
For TT4 values, TSH-BAW and TSH-CLIA values below 6.4 nmol/l were given a value of 6.3, values below 0.008 ng/ml were given a value of 0.007 and values below 0.03 ng/ml were given a value of 0.028 for statistical analysis, respectively.
Results
A total of 136 cats presented for enrollment, consisting of 66 hyperthyroid cats, 37 healthy cats and 33 NTI cats. A total of 29 hyperthyroid cats were excluded: 22 for concurrent NTI, five for incomplete data and two for declining confirmation with scintigraphy. Five healthy cats were excluded, three due to having a TT4 concentration above 46 nmol/l and two due to incomplete data. One NTI cat was excluded due to having a TT4 concentration above 46 nmol/l.
A total of 101 cats were included in the study, including 37 hyperthyroid, 32 healthy and 32 NTI cats. The median age of the hyperthyroid, healthy and NTI cats was 12 years (range 6–17), 9 years (range 5–16) and 11 years (range 5–25), respectively. The median age of the hyperthyroid cats was significantly higher than the healthy cats (P = 0.001), but there was no significant difference between the NTI and healthy cats (P = 0.10) or the NTI and hyperthyroid cats (P = 0.12).
Breeds consisted of 78 domestic shorthairs, 18 domestic longhairs, two Siamese and one of each of the following breeds: Devon Rex, Maine Coon and Bengal. Of the cats, 55 were spayed females and 46 were castrated males. There was no difference between groups regarding the breed and sex distribution.
Follow-up TT4 concentrations were available for 28 healthy cats. All were within the RI, except one cat that had a TT4 below the RI (18.5 nmol/l). Two healthy cats were noted to have a small thyroid slip on physical examination. Their serum TT4 concentrations at enrollment were 35.8 nmol/l and 34.7 nmol/l, respectively, and at recheck they were 34 nmol/l and 30.8 nmol/l, respectively. For seven of the healthy cats, it was not noted whether a thyroid nodule was palpable. All those cats’ serum TT4 concentrations were within the RI at recheck at least 3 months later. The four cats that did not have follow-up TT4 concentrations performed did not have a palpable thyroid nodule at enrollment.
A follow-up TT4 concentration was available for 14 NTI cats. All were within the RI. One cat was rechecked sooner at 1 month after enrollment and had a TT4 concentration below 6.4 nmol/l. One cat in the NTI group had a small palpable thyroid slip. This cat’s serum TT4 concentration was 9.9 nmol/l at enrollment and 35.9 nmol/l at follow-up. The rest of the NTI cats did not have a palpable thyroid nodule.
Regarding severity of hyperthyroidism, nine cats had mild disease (median TT4 84.4 nmol/l [range 55.1–94.3]), 12 cats had moderate disease (median TT4 126.5 nmol/l [range 99–157]) and 16 cats had severe disease (median TT4 224 nmol/l [range 179–453]).
Regarding disease severity in NTI cats, 15 and 17 were categorized as mild disease and moderate to severe disease, respectively. All the cats experiencing a moderate to severe disease fulfilled at least two of the SIRS criteria. The types of illnesses in NTI cats with mild disease were chronic kidney disease (n = 9), diabetes mellitus (n = 2), chronic pancreatitis (n = 1), chronic enteropathy (n = 2), feline immunodeficiency virus (n = 1), chronic nasal disease (n = 2), fever (n = 1), urinary tract infection (n = 1) and colonic mass (n = 1), with some cats suffering from more than one illness. The illnesses of NTI cats with moderate to severe disease were acute pancreatitis/cholangiohepatitis (n = 3), diabetic ketoacidosis (n = 3), acute or worsening gastrointestinal illness (n = 2), round cell neoplasia (n = 1), fever of unknown origin (n = 1), pyelonephritis (n = 1), systemic fungal disease (n = 1), blood loss anemia (n = 1), sepsis/bacteremia (n = 2), pancytopenia (n = 1) and secondary hepatic lipidosis (n = 1).
Serum TT4 and TSH with both assays for all cats
The median serum concentrations of TT4 and TSH measured with both the TSH-CLIA and TSH-BAW assays are reported in Table 1.
Total thyroxine (TT4), thyroid-stimulating hormone-chemiluminescent immunoassay (TSH-CLIA) and bulk acoustic wave (TSH-BAW) assay in hyperthyroid cats, healthy cats and cats with non-thyroidal illness (NTI)

Data are median (range). P <0.05 was considered statistically significant; values with the same superscript letter within rows indicate significant differences. RIs: TT4 18.9–50.1 nmol/l, TSH-CLIA <0.03–0.3 ng/ml and TSH-BAW 0.01–0.3 ng/ml
RI = reference interval
Serum TT4
The median serum TT4 concentrations of the hyperthyroid cats were significantly higher than those of the healthy and NTI cats (P <0.001) (Table 1). The median serum TT4 concentration was significantly lower in the NTI cats compared with healthy cats (P = 0.008) (Table 1).
In total, 14 (43.7%) cats with NTI had serum TT4 concentrations below the RI (<18.9 nmol/l), with five (15.6%) having an undetectable value (<6.4 nmol/l). All healthy cats had a serum TT4 concentration within the RI at initial enrollment.
The median serum TT4 concentrations were not significantly different between hyperthyroid cats that had a TSH-BAW within the RI (152.5 nmol/l, range 55.1–212) and those that had a TSH-BAW below the RI (142 nmol/l, range 69.4–453) (P = 0.869) (Figure 1).

Serum total thyroxine (TT4) concentrations in hyperthyroid cats with TT4 below the reference interval (RI) or within the RI (18.9–50.1 nmol/l), measured via the thyroid-stimulating hormone-bulk acoustic wave assay. Median TT4 was not significantly different between the groups (P = 0.869). Boxes represent the interquartile range from the 25th to 75th percentile; the horizontal bar in each box represents the median value; the whiskers indicate the range of values with the exception of outliers as indicated by circles
Sensitivity, specificity, positive predictive value and negative predictive value of both TSH assays for the diagnosis of FHT
The TSH-BAW assay had a sensitivity and specificity for the diagnosis of FHT of 78% (95% CI 62–90) and 97% (95% CI 89–100), respectively. The PPV and negative predictive value (NPV) were 94% (95% CI 79–99) and 89% (95% CI 79–95), respectively. The sensitivity and specificity of the TSH-CLIA assay were 95% (95% CI 82–99) and 81% (95% CI 70–90), respectively. The PPV and NPV were 74% (95% CI 60–86) and 96% (95% CI 87–100), respectively.
TSH-CLIA vs TSH-BAW
The median serum TSH concentrations were significantly different between hyperthyroid and healthy cats (P <0.001) and between hyperthyroid and NTI cats (P <0.001) with both assays, but were not different between healthy and NTI cats (TSH-CLIA P = 0.168, TSH-BAW P = 0.673) (Figure 2).

Thyroid-stimulating hormone (TSH) concentrations measured using the bulk acoustic wave (TSH-BAW) assay in hyperthyroid cats, healthy cats and cats with non-thyroidal illness (NTI). The median TSH concentrations were significantly different between hyperthyroid and euthyroid cats (healthy and NTI cats) with both assays (P <0.001), and were not different between healthy and NTI cats (thyroid-stimulating hormone-chemiluminescent immunoassay P = 0.168, TSH-BAW P = 0.673). Boxes represent the interquartile range from the 25th to 75th percentile; the horizontal bar in each box represents the median value; the whiskers indicate the range of values with the exception of outliers as indicated by circles; the upper and lower dotted lines represent the upper and lower values of the reference interval of the TSH-BAW assay (0.01–0.301 ng/ml)
The proportion of cats with an undetectable serum TSH concentration with the TSH-CLIA assay (<0.03 ng/ml) but a normal serum TSH concentration with the TSH-BAW assay (⩾0.01 ng/ml) was 17.8% (18 cats, including 11 non-hyperthyroid cats). The number of cats with undetectable serum TSH concentrations was significantly higher in the hyperthyroid cats compared with the two other groups via both assays (P <0.001).
With the TSH-CLIA assay, the serum TSH concentrations were undetectable (<0.03 ng/ml) in 35 (95%) hyperthyroid cats. With the TSH-BAW assay, the serum TSH concentrations were below the RI (<0.01 ng/ml) in 29 (78.4%) hyperthyroid cats and undetectable (<0.008 ng/ml) in 28 (75.7%) hyperthyroid cats. TSH-BAW detected hyperthyroidism in 29/35 cats. In this population of hyperthyroid cats, eight (21.6%) cats had an undetectable serum TSH concentration with the TSH-CLIA assay (<0.03 ng/ml) but a detectable serum TSH concentration with the TSH-BAW assay (>0.008 ng/ml). Of these cats, seven (18.9%) had a serum TSH-BAW concentration within the RI (⩾0.01 ng/ml). One hyperthyroid cat had a TSH-CLIA within the RI but an undetectable TSH-BAW.
With the TSH-CLIA assay, the serum TSH concentrations were undetectable in 12 (18.8%) non-hyperthyroid cats, eight (25%) of which had an NTI. With the TSH-BAW assay, serum TSH concentrations were below the RI in one (3%) healthy cat and one (3%) cat with NTI. The serum TT4 concentration and TSH-CLIA in the healthy cat with an undetectable TSH-BAW was 27.4 nmol/l and 0.121 ng/ml, respectively. A repeat serum TT4 concentration 3 months later was 23.3 nmol/l. The serum TT4 and TSH-CLIA concentrations in the NTI cats with undetectable TSH-BAW were below 6.4 nmol/l and below 0.03 ng/ml, respectively. A repeat serum TT4 concentration was less than 6.4 nmol/l 1 month later. The proportions of undetectable serum TSH concentrations were not different between healthy cats and NTI cats with either assay (TSH-CLIA P = 1, TSH-BAW P = 1). The proportion of cats with an undetectable serum TSH concentration was significantly higher with the TSH-CLIA than the TSH-BAW in NTI cats (P = 0.008), but not in healthy cats (P = 0.179).
The median serum TSH concentrations were not statistically different between cats with mild illness and moderate to severe illness with both assays (TSH-CLIA P = 0.330, TSH-BAW P = 0.467) (Table 2, Figure 3). A total of 28 cats with NTI survived, whereas four cats were euthanized at the discretion of their owners due to a poor prognosis. Median serum TSH concentrations were not significantly different with either assay between survivors and non-survivors (TSH-CLIA P = 1, TSH-BAW P = 0.955). With the TSH-CLIA, the serum TSH concentration was undetectable in two (13.3%) cats with mild disease and six (35.3%) cats with moderate to severe disease. With the TSH-BAW assay, only one cat had an undetectable serum TSH concentration and it was in the moderate to severe group. The proportion of cats with an undetectable serum TSH concentration measured with either assay was not significantly different between groups of ill cats (TSH-CLIA P = 0.229, TSH-BAW P = 1). Cats in the moderate to severe group had a higher proportion of undetectable TSH-CLIA concentrations compared with the TSH-BAW (P = 0.025). There was no significant difference between assays in the mild group (P = 0.248). Regarding type of disease, median serum TT4, TSH-BAW and TSH-CLIA concentrations were also not significantly different (see supplementary material).
Total thyroxine (TT4) and thyroid-stimulating hormone (TSH) measured using the chemiluminescent immunoassay (TSH-CLIA) and bulk acoustic wave (TSH-BAW) assay in non-thyroidal illness cats categorized by severity of illness

Data are median (range). P <0.05 was considered statistically significant. RIs: TT4 18.9–50.1 nmol/l, TSH-CLIA <0.03–0.3 ng/ml and TSH-BAW 0.01–0.301 ng/ml
RI = reference interval
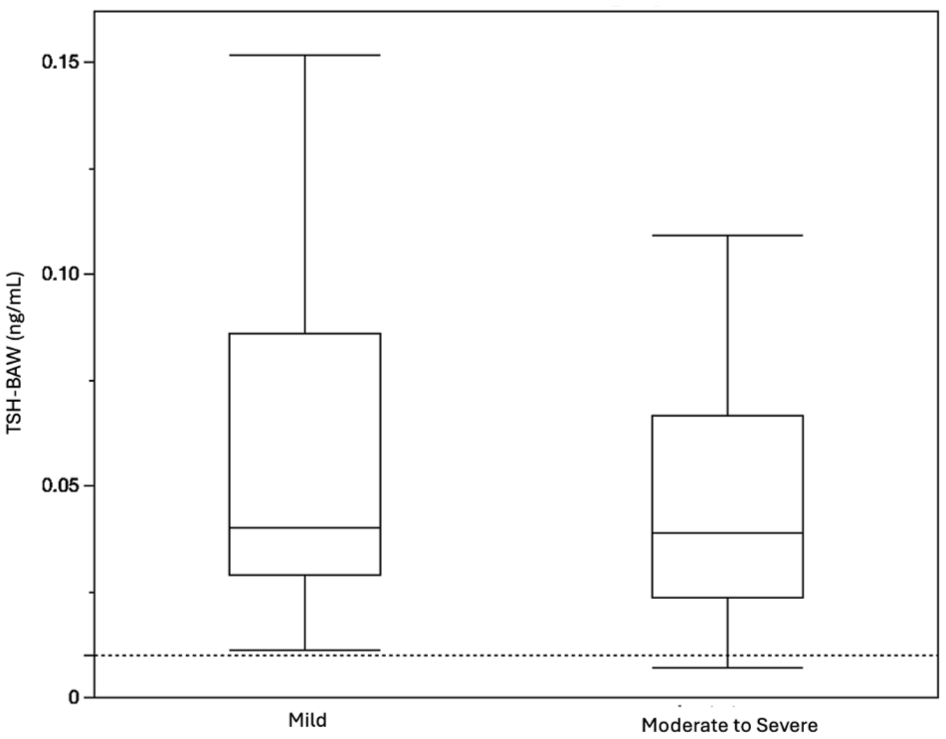
Thyroid-stimulating hormone (TSH) concentrations measured using the bulk acoustic wave (TSH-BAW) assay in cats with non-thyroidal illness categorized according to disease severity. There was no statistical difference between groups (P = 0.467). Boxes represent the interquartile range from the 25th to 75th percentile; the horizontal bar in each box represents the median value; the whiskers indicate the range of values; the dotted line represents the low end of the reference interval of the TSH-BAW assay (0.01–0.301 ng/ml)
Discussion
This study shows that the TSH-BAW assay is a specific test for the diagnosis of hyperthyroidism. Our data did not show a significant effect from NTI on the performance of the assay. It can therefore be used to confirm hyperthyroidism in cats. Only one healthy cat and one NTI cat had a TSH-BAW result below the RI, and both were below the limit of detection. The NTI cat had undetectable serum TT4 and TSH-CLIA concentrations, and the serum TT4 concentration remained undetectable 1 month later. It is possible that this cat’s illness was suppressing TSH-BAW. The one healthy cat with an undetectable TSH-BAW was unlikely to have early hyperthyroidism as the serum TT4 concentration was mid-range and remained normal 3 months later. However, thyroid scintigraphy was not performed in these cats to exclude hyperthyroidism.
In hyperthyroid cats with an increased serum TT4 concentration, the sensitivity and specificity of the TSH-BAW immunoassay were 78% and 97%, respectively. This result is similar to the previously reported specificity of 98.9%. 6 As expected, this assay is more specific than the TSH-CLIA assay (88% in our study, 70–80% in the literature).6,11,34,35 This allows better differentiation between true hyperthyroid cats and euthyroid cats that have a TSH below 0.03 ng/ml. It can also identify euthyroid cats more often (97% of the time vs 81.5% only with the TSH-CLIA).
In our study, the median serum TSH concentration of euthyroid cats did not differ between assays, whether they were healthy or experiencing NTI. This suggests TSH is less affected by NTI, as has been demonstrated previously.6,10 However, it is not completely immune to the effect of NTI, as one study found that undetectable TSH-CLIA concentrations were associated with severe illnesses as well as a higher risk of mortality. 10 Our results showed a trend in the same direction for the TSH-CLIA assay; however, it did not reach statistical significance, likely due to the small number of cats within each NTI group. The proportion of undetectable TSH-CLIA in NTI cats was higher than TSH-BAW, but the same was not seen for healthy cats. It was also significantly higher in NTI cats with moderate to severe disease, but not mild disease. This suggests that the TSH-BAW assay might be less affected by NTI compared with the TSH-CLIA. The lack of difference in illness severity in TSH concentrations in both assays could be due to type II error, however. This is also consistent with another study evaluating the TSH-BAW assay in euthyroid cats and cats with chronic kidney disease. 6 A future study with a larger population of ill cats with different disease severities is needed to further assess the effects of NTI on the TSH-BAW assay. It has been shown that TSH-CLIA concentrations can decrease with increasing severity of illness and predict survival. 10 Whether this is true for TSH-BAW – and whether it may even be superior to TSH-CLIA for this purpose – also remains to be determined.
The sensitivity of the TSH-BAW assay is lower than the TSH-CLIA based on our results; however, this difference is not statistically significant. This sensitivity applies only to our cat population and can be explained by several factors, including the antibody used, the variability between lots, the limitations of methodologies for screening tests when the analyte is at a very low concentration and other undetermined limits of the assay. In addition, the TSH-CLIA assay is incapable of measuring values below 0.03 ng/ml. Therefore, some hyperthyroid cats will have a normal TSH-BAW concentration (⩾0.01 ng/ml) but a serum TSH-CLIA concentration below 0.03 ng/ml. Interestingly, all hyperthyroid cats had a serum TSH concentration below 0.014 ng/ml when measured using the TSH-BAW assay. A serum TSH concentration below this threshold was rarely seen in non-hyperthyroid cats (two healthy, four with NTI). Based on our results, hyperthyroidism is unlikely if the TSH is at the high end of the RI, but a TSH-BAW at the low end of the RI (<0.014 ng/ml) cannot be used to exclude hyperthyroidism.
When compared with the study by Peterson et al, 6 our sensitivity results are lower. In that study, the sensitivity of the TSH-BAW assay when combined with TT4 measurement was 84.6%, compared with 90.5% when the TSH was used alone for the diagnosis. The difference in sensitivity between the two studies could relate to study design for inclusion criteria in the hyperthyroid group, the number of cats in each study and possible intrinsic differences in populations. As the serum TT4 concentration was not different between cats with normal vs subnormal serum TSH concentrations in our study, it is unlikely this difference is due to the severity of hyperthyroidism in the two study populations. In the study by Peterson et al, 6 the TSH-BAW concentrations were not significantly different between subclinical hyperthyroid cats compared with cats with mild, moderate or severe hyperthyroidism. However, with the TSH-CLIA and TSH-BAW assays, a higher proportion of cats with early/mild forms of FHT had detectable serum TSH concentrations compared with cats with more severe forms of FHT. However, some cats with severe hyperthyroidism still have a normal TSH concentration. Therefore, an undetectable TSH-BAW cannot be used as a stand-alone test for diagnosing FHT.
Based on the NPV and sensitivity, approximately 10–20% of hyperthyroid cats can have a normal TSH-BAW concentration and be misdiagnosed as not having the disease, whereas a detectable TSH-CLIA would misdiagnose in approximately 5% of hyperthyroid cats. The TSH-BAW has a higher specificity and PPV than the TSH-CLIA. With the TSH-BAW assay, approximately 5% of euthyroid cats could be falsely diagnosed with the disease, whereas this could occur in approximately 20–25% of euthyroid cats with the TSH-CLIA assay. Therefore, TSH-BAW plays a role in helping confirm hyperthyroidism, whereas TSH-CLIA could be better at ruling out the disease. A TSH-BAW concentration is most useful in addition to the TT4 if suspicion of hyperthyroidism is high but the TT4 alone is not enough to confirm the diagnosis. A subnormal TSH-BAW concentration would likely confirm whether hyperthyroidism is present. If TSH-BAW is normal, then either further testing for hyperthyroidism or monitoring TT4 and TSH-BAW concentrations over time to determine if they become more suggestive of hyperthyroidism can be performed. Our study did not evaluate the manufacturers’ RIs: further studies with a larger number of cats are needed to validate the interval. Changes in the RIs could impact the sensitivity of the TSH-BAW.
This study has several limitations. Our NTI group most likely lacked power to fully assess the effect of severe diseases (and mortality) on the TSH-BAW. In addition, categorization of illnesses by severity is strongly subjective. Objectivity was attempted by using the already known SIRS criteria; however, they are known to be imprecise and lack specificity (even if these limitations seem to be less dramatic in cats). 36 Another limitation is that thyroid scintigraphy was not used to confirm euthyroidism. Considering that serum TT4 concentration is highly sensitive for diagnosing FHT,4,6,29,31,37 we elected to confirm euthyroidism based on a normal serum TT4 concentration both at inclusion and at least 3 months later. By repeating a serum TT4 measurement, we could detect most hyperthyroid cats that were subclinical or ill at the time of inclusion. Unfortunately, some cats could not be rechecked (four healthy and 19 NTI cats), due to either loss to follow-up or death of the patient. Therefore, it is possible that subclinical hyperthyroid cats could have been included in the euthyroid groups. Lastly, we did not include subclinical hyperthyroid cats in our study, which are a subgroup of cats that can benefit from TSH measurement for the diagnosis of hyperthyroidism.
Conclusions
The novel TSH-BAW assay (TRUFORMA; Zomedica) has a high specificity for the diagnosis of FHT and identifies a normal serum TSH concentration in non-hyperthyroid cats more often than the TSH-CLIA. However, a normal result cannot be used to rule out hyperthyroidism.
Supplemental Material
Supplementary table
Total thyroxine (TT4) and thyroid-stimulating hormone (TSH) using chemiluminescent assay (TSHCLIA) and bulk acoustic wave (TSH-BAW) in NTI cats.
Footnotes
Acknowledgements
The authors thank Dr Stephen Were for his help with the statistical analysis.
Author note
This paper was presented in part at the 2024 American College of Veterinary Internal Medicine Forum conference.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by Zomedica and Virginia Tech Foundation.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals and procedures that differed from established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient. The study therefore had prior ethical approval from an established (or ad hoc) committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
Supplementary material
The following file is available as supplementary material:
Supplementary table: Total thyroxine (TT4) and thyroid-stimulating hormone (TSH) using chemiluminescent assay (TSH-CLIA) and bulk acoustic wave (TSH-BAW) in NTI cats.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
