Abstract
Objectives
This study aimed to evaluate the effects of combined grapiprant and tapentadol on intraoperative physiological parameters, the occurrence of adverse events and postoperative analgesic efficacy in cats undergoing elective ovariohysterectomy, using two multidimensional pain scales.
Methods
A total of 60 mixed-breed female cats were enrolled in a randomized, prospective, blinded study. The animals were evenly distributed into four groups (n = 15 per group). In total, 51 animals completed the study; the control group received placebo (CON; n = 11), while the grapiprant group (GRA; n = 13) received grapiprant (3.8 ± 0.5 mg/kg), the tapentadol group (TAP; n = 14) received tapentadol (5.3 ± 1.2 mg/kg) and the grapiprant-tapentadol group (GT; n = 13) received a combination of grapiprant (4.2 ± 0.5 mg/kg PO) and tapentadol (5 ± 0.6 mg/kg PO) 1 hour before initiation of the standardized anesthesia protocol and surgical procedure. Physiological parameters were monitored during surgery, and postoperative pain was assessed for 6 h after extubation using the Short Form of the UNESP-Botucatu Feline Pain Score (UFESP-SF) and the Feline Grimace Scale (FGS), administered by two blinded evaluators.
Results
No adverse effects or statistically significant differences in physiological parameters were observed between groups. The assessment of pain scores showed good reliability, with intraclass correlation coefficient values of 0.89 for the FGS and 0.91 for the UFESP-SF, supporting inter-rater agreement for both instruments. At 3 h postoperatively, FGS scores differed significantly between the CON and GT groups (P = 0.0363). Rescue analgesia requirements also varied among groups (P = 0.0110): the GT group required rescue at 3 h compared with 1 h in the CON group (P = 0.0007) and 2 h in the GRA group (P = 0.0058).
Conclusions and relevance
The results of this study showed that the analgesic effect of the grapiprant-tapentadol combination lasted up to 3 h in the postoperative period, which was longer than the 2 h of grapiprant and tapentadol alone, without compromising intraoperative physiological stability.
Plain language summary
This study evaluated the efficacy of two non-conventional drugs for postoperative pain management in female cats undergoing ovariohysterectomy. The study compared grapiprant, a non-steroidal anti-inflammatory drug used for inflammatory pain in dogs, with tapentadol, a synthetic opioid effective for both acute and chronic pain in humans. Grapiprant and tapentadol were administered orally, both individually and in combination, to cats undergoing ovariohysterectomy. Antinociceptive effects during surgery were assessed through physiological parameters. Two specific validated pain scales for cats, the Short Form of the UNESP-Botucatu Feline Pain Score and the Feline Grimace Scale, were used to assess analgesia during the first 6 h after surgery. Results demonstrated that the grapiprant–tapentadol combination showed superior efficacy in postoperative acute pain relief compared with either drug alone or placebo. Although this drug combination is novel in feline medicine, this study provides significant contributions to the literature regarding the use of grapiprant and tapentadol in postoperative pain management for female cats.
Keywords
Introduction
Multimodal analgesia, incorporating non-steroidal anti-inflammatory drugs (NSAIDs) and opioids, represents the predominant approach for managing acute inflammatory and visceral pain in cats. 1 However, drug selection for this species requires careful consideration because of the unique metabolic characteristics of cats, which significantly influence both therapeutic efficacy and safety profiles.2,3 Current pain management protocols in cats predominantly utilize meloxicam and tramadol, as demonstrated by survey-based studies conducted across South America.4,5 These agents, however, may induce adverse effects due to cats’ limited metabolic capacity for certain substances, potentially compromising analgesic safety and predictability.2,3
Novel analgesic alternatives are under investigation to address these challenges. Grapiprant, a NSAID belonging to the piprant family, has received approval for osteoarthritis treatment in dogs. 6 Unlike conventional NSAIDs, grapiprant functions as a potent and selective prostaglandin E2 receptor 4 (EP4) antagonist, enabling inflammatory pain inhibition without disrupting the production of homeostatic eicosanoids, thereby reducing the incidence of adverse effects.6,7 Preliminary studies suggest that oral administration of grapiprant in cats demonstrates both safety and potential viability 8 as an alternative to conventional NSAIDs for postoperative pain management in female dogs 9 and cats 10 undergoing ovariohysterectomy (OVH).
Tapentadol, a synthetic opioid, exhibits a dual mechanism of action through μ-opioid receptor agonism and noradrenaline reuptake inhibition, thereby modulating descending pain pathways. Although sharing similar agonist properties with tramadol, tapentadol demonstrates 120-fold greater potency than tramadol, yet remains 50-fold less potent than morphine.11,12 In cats, tapentadol has demonstrated comparable efficacy to buprenorphine; salivation was reported as the main adverse event after oral administration. 12 In human medicine, tapentadol is used extensively for both acute and chronic pain management, and is available in immediate and extended-release formulations. 13
Despite promising evidence regarding grapiprant and tapentadol, significant knowledge gaps exist concerning their efficacy and safety profiles, both individually and in combination, for postoperative pain management in female cats undergoing elective OVH. Furthermore, limited data exist regarding their influence on intraoperative physiological parameters and potential species-specific adverse effects. We hypothesized that the combination of grapiprant and tapentadol, by acting on different points of the acute pain pathway, produces a synergistic effect that results in adequate analgesia during the postoperative period in female cats undergoing elective OVH. Thus, the present study evaluated the postoperative analgesic efficacy of combined grapiprant and tapentadol in cats undergoing elective OVH, through two multidimensional pain scales, while simultaneously investigating their effects on intraoperative physiological parameters and adverse event occurrence.
Materials and methods
This study received approval from the Animal Use Ethics Committee of the State University of Santa Cruz (CEUA-UESC) protocol number: 027/23.
Animals: inclusion and exclusion criteria
Initially, 60 docile, mixed-breed female cats, aged 6–48 months with a body weight of 2 kg or more, scheduled for elective OVH at the at the UESC Veterinary Hospital were selected. The experimental protocol was explained to the caregivers of the animals, and study inclusion was contingent upon obtaining signed informed consent. Before surgery, all cats underwent clinical examination, complete blood count and abdominal ultrasonography to exclude pregnancy. Only clinically healthy, non-pregnant animals with normal clinical and laboratory parameters, classified as status 1 according to the American Society of Anesthesiologists (ASA), were included in the study. Exclusion criteria were body weight under 2 kg, signs of fear or anxiety during clinical examination, pregnancy or lactation, behavioral changes during the perioperative period, incomplete administration of the experimental medication and ASA status greater than 1. These exclusion criteria aimed to reduce variability, facilitate animal handling and enhance the reliability of pain assessments.
Animals were admitted 24 h preoperatively for environmental acclimation and familiarization with study evaluators. Cats were housed in individual cages within an enriched, species-appropriate environment following cat friendly practice guidelines. 14 Animals were maintained on their standard diet provided in small portions, had ad libitum access to water and underwent a 10-h preoperative fasting period.
Experimental groups
Cats were randomly assigned to four groups before the start of the study using an online software program (www.randomizer.org). The study design proposed the following dosing regimen for each group: CON (control, placebo, n = 15); GRA (grapiprant 4 mg/kg, n = 15); TAP (tapentadol 5 mg/kg, n = 15); and GT (grapiprant 4 mg/kg plus tapentadol 5 mg/kg, n = 15). All medications were administered orally in gelatin capsules 1 h preoperatively, followed by 5 ml of water administered via syringe by a designated investigator. To preserve experimental integrity, the remaining team members were blinded to the treatment allocation.
The doses were defined based on scientific literature7,8,11,12,15 and adjusted to minimize side effects and ensure practicality in administration. The capsules were properly prepared by an external pharmaceutical laboratory.
Anesthetic-surgical protocol
All groups received intramuscular (IM) premedication with 0.05 mg/kg acepromazine (Acepromazin; Syntec), previously diluted in 0.9% sodium chloride solution (0.2 ml of acepromazine in 0.8 ml of 0.9% NaCl). At 20 mins after premedication, the cephalic vein was catheterized for continuous administration of lactated Ringer’s solution (5 ml/kg/h) throughout the surgical procedure. Intravenous (IV) antibiotic prophylaxis was administered as a single dose (cephalothin sodium 30 mg/kg IV, Cefariston; Blau Farmacêutica).
At 5 mins before the induction of anesthesia, a constant rate infusion (CRI) of remifentanil (20 μg/kg/h IV, Remifas; Cristália) was started. Induction was performed with propofol (1 mg/kg/min IV, Provive; União Química) until tracheal intubation was possible. For endotracheal intubation, cats were placed in sternal recumbency and 0.2 ml of lidocaine hydrochloride monohydrate without vasoconstrictor (Xylestesin; Cristália) was applied to the laryngeal mucosa. After confirming successful intubation via capnography (detection of a normal CO2 waveform), all animals received oxygen flow rate of 200 ml/kg/min or above through an open system (Baraka circuit) and were kept warm using a thermal mattress. Anesthesia was maintained using total IV anesthesia with CRIs of propofol (0.5 mg/kg/min) and remifentanil (20 μg/kg/h). The CRI of propofol was adjusted intraoperatively based on hemodynamic fluctuations, either increased or decreased by 0.1 mg/kg/min in response to changes in heart rate and/or blood pressure. When these parameters exceeded 20% above baseline values, rescue analgesia was administered via an IV bolus of remifentanil (1.5 μg/kg). The doses of propofol administered during induction, throughout the intraoperative period, total cumulative dose and the number of rescue boluses of remifentanil were recorded for subsequent analysis.
Other variables registered included the following: duration of anesthesia, defined as the time from the start to the end of the maintenance CRI; surgical duration, from the time of incision to the completion of dermorrhaphy; extubation time, defined as the time from the end of the maintenance CRI to the removal of the endotracheal tube; and time to initial pain assessment, defined as the time elapsed between experimental treatment administration and initial pain assessment.
All ovariohysterectomies were performed by the same surgeon using a 5 cm ventral midline incision to access the uterine horns. Polyglactin 910 (2-0) suture material was used for ovarian pedicle ligatures, abdominal wall and subcutaneous tissue closure, while skin closure was performed using simple interrupted sutures with monofilament nylon (3-0).
Sedation score
Sedation was evaluated using the feline multiparametric sedation score adapted from Rutherford et al 16 at six time points: baseline (before experimental drug administration), before IV catheterization and at 0.5, 1, 3 and 6 h after extubation (see Appendix A in the supplementary material).
Intraoperative physiological parameter monitoring
Physiological parameters were recorded using a multiparameter monitor (LifeWindow LW9x; Digicare), heart rate (HR; in beats/min) by continuing electrocardiogram, respiratory rate (RR; in breaths/min) and end-tidal carbon dioxide (ETCO2) by capnography, blood oxygen saturation (SpO2) by pulse oximetry, and indirect systolic, mean and diastolic arterial blood pressure (SBP, MAP and DBP, respectively) using an oscillometric method, with a size 2 cuff placed over the radial artery of the left forelimb. Core body temperature was measured with an esophageal thermometer. Parameters were recorded at the following time points: preoperative baseline (M0), after the linea alba incision (M1), after the first pedicle ligation (M2), after the second pedicle ligation (M3), after the uterine stump ligation (M4) and after skin closure (M5).
Pain assessment
Pain was evaluated by two blinded, trained observers using the Feline Grimace Scale (FGS) 17 and Short Form of the UNESP-Botucatu Feline Pain Score (UFESP-SF). 18 Assessments were conducted at baseline and at 1, 2, 3, 4, 5 and 6 h after extubation.
Rescue analgesia
Animals exhibiting pain scores of 4/10 or higher on FGS or 4/12 or higher on UFEPS-SF received buprenorphine (20 μg/kg IM, Beniv; Ourofino). Administration of a second reduced dose (10 μg/kg IM) within the 6-h evaluation interval was considered supplemental rescue analgesia. Regardless of the application of rescue analgesia, cats continued to be evaluated until 6 h after extubation.
Postoperative management
Upon completion of the pain assessment period, all animals received a single dose of sodium dipyrone (15 mg/kg IM, Dipyrone 50%, 500 mg/mL; Ibasa) and meloxicam (0.2 mg/kg SC, 0.2% Maxicam; Ourofino). Cats were discharged 8 h postoperatively (with the option for earlier discharge) and returned to their caregivers with prescribed meloxicam (0.1 mg/kg PO q24h, Maxicam tablets, 0.5 mg; Ourofino) for 4 days and postoperative care instructions.
Statistical analysis
Sample size was determined using R version 4.3.0 (2023.06.2 Build 561) using the pwr.anova.test function for four-group (k) with alpha = 0.05, power (beta = 0.8) and effect size (f = 0.5). Analysis indicated that 12 cats per group would be sufficient to detect significant differences. 19 An additional three cats per treatment group were included to account for an anticipated dropout rate of 20%. 19
Statistical analysis was carried out using R version 4.3.0 (2023.06.2 Build 561). Graphs were produced using GraphPad Prism version 8.0.2 (263). Normality was assessed using the Shapiro–Wilk test. One-way ANOVA was applied to the following variables: body weight, age, induction propofol, intraoperative propofol, total propofol, anesthesia time, surgical time, extubation time and time to initial pain assessment. Physiological parameters – HR, SBP, MAP, ETCO2 and temperature – were analyzed using repeated measures ANOVA followed by Tukey’s test, while RR, DBP and SpO2 were analyzed using Friedman’s test with Dunn’s post-hoc analysis. The administered dosages of grapiprant and tapentadol were compared using an unpaired t-test.
To evaluate inter-rater reliability in pain assessment, the intraclass correlation coefficient (ICC) was calculated along with its 95% confidence interval (CI). Sedation scores were analyzed using the Kruskal–Wallis test. For statistical purposes, the mean pain score derived from the assessments of both observers, including all measurements recorded before and after the administration of initial rescue analgesia, was considered. These data were analyzed using the Kruskal–Wallis test with Dunn’s post-hoc analysis. Furthermore, Kaplan–Meier survival analysis with the Log-rank test was used to compare the time to rescue analgesia between groups during both the intraoperative and postoperative periods. Supplemental rescue analgesia requirements between groups were compared using the χ2 test. Statistical significance was set at P <0.05 for all analyses.
Results
Nine cats were excluded from the study because of behavioral changes during the perioperative period, which precluded stress-free handling, despite docile temperament during screening. The final study population comprised 51 animals. There were no significant differences among the groups in body weight or age (Table 1). The mean ± SD of the administered doses in each experimental group were as follows: the GRA group received 3.8 ± 0.5 mg/kg of grapiprant; the TAP group received 5.3 ± 1.2 mg of tapentadol; and the GT group received 4.2 ± 0.5 mg/kg of grapiprant and 5 ± 0.6 mg/kg of tapentadol. No statistically significant differences were observed in the administered doses of grapiprant (P = 0.0625) or tapentadol (P = 0.3410). No adverse effects such as salivation, mydriasis or vomiting were observed after administration of grapiprant or tapentadol, either alone or in combination.
Body weight and age of cats undergoing elective ovariohysterectomy

Data are mean ± SD or median (range)
CON = control group; GRA = grapiprant group; GT = grapiprant and tapentadol group; TAP = tapentadol group
Sedation score
The GRA and GT groups showed significantly higher sedation scores 0.5 h after extubation compared with other treatment groups (P = 0.0120). However, no significant differences were observed among the other treatment groups (Table 2).
Sedation scores in female cats undergoing elective ovariohysterectomy

Data are median (interquartile range). Different letters indicate significant differences between GRA and GT groups (P = 0.0120). Assessment timepoints at 0.5 h, 1 h, 3 h and 6 h were after extubation
CON = control group; GRA = grapiprant group; GT = grapiprant and tapentadol group; IV = intravenous; TAP = tapentadol group
Anesthetic-surgical variables
Table 3 provides a summary of the results for anesthetic and surgical variables. The propofol induction dose was significantly higher in the GRA group compared with both the CON and GT groups (P = 0.0001), and the time to extubation was significantly longer in the GT group compared with both the CON and GRA groups (P = 0.001).
Anesthesia duration, surgical time and time to extubation in female cats undergoing elective ovariohysterectomy

Data are mean ± SD. Different letters indicate significant differences between groups (P <0.05)
CON = control group; GRA = grapiprant group; GT = grapiprant and tapentadol group; TAP = tapentadol group
Intraoperative physiological parameters
Inter-group comparison revealed no statistically significant differences between monitored physiological parameters. However, intra-group variations were observed, with parameters increasing during periods of heightened nociceptive stimulation (M2, M3 and M4) compared with baseline values (M0) (detailed in Appendix B in the supplementary material).
Intraoperative rescue analgesia requirements
No significant differences were observed between groups regarding the need for intraoperative rescue analgesia (P = 0.1516) (Figure 1).

Kaplan–Meier survival plot based on the number of female cats requiring intraoperative rescue analgesia. Assessment time points: before incision (M0), after linea alba incision (M1), first ovarian pedicle ligation (M2), second ovarian pedicle ligation (M3), uterine stump ligation (M4) and skin closure (M5). CON = control group; GRA = grapiprant group; GT = grapiprant and tapentadol group; TAP = tapentadol group
Postoperative pain assessment
For the FGS, the ICC was 0.890 (95% CI 0.867–0.910); for the UFES-SF, it was 0.902 (95% CI 0.881–0.920), indicating a high level of agreement between evaluators.
The median pain scores measured using the FGS at 3 h after extubation were significantly higher in the treatment group compared with the CON group (P = 0.0363). The UFESP-SF showed similar patterns to the FGS during the first 2 h after extubation; however, no significant differences between groups were detected at any assessment time point (Figure 2).
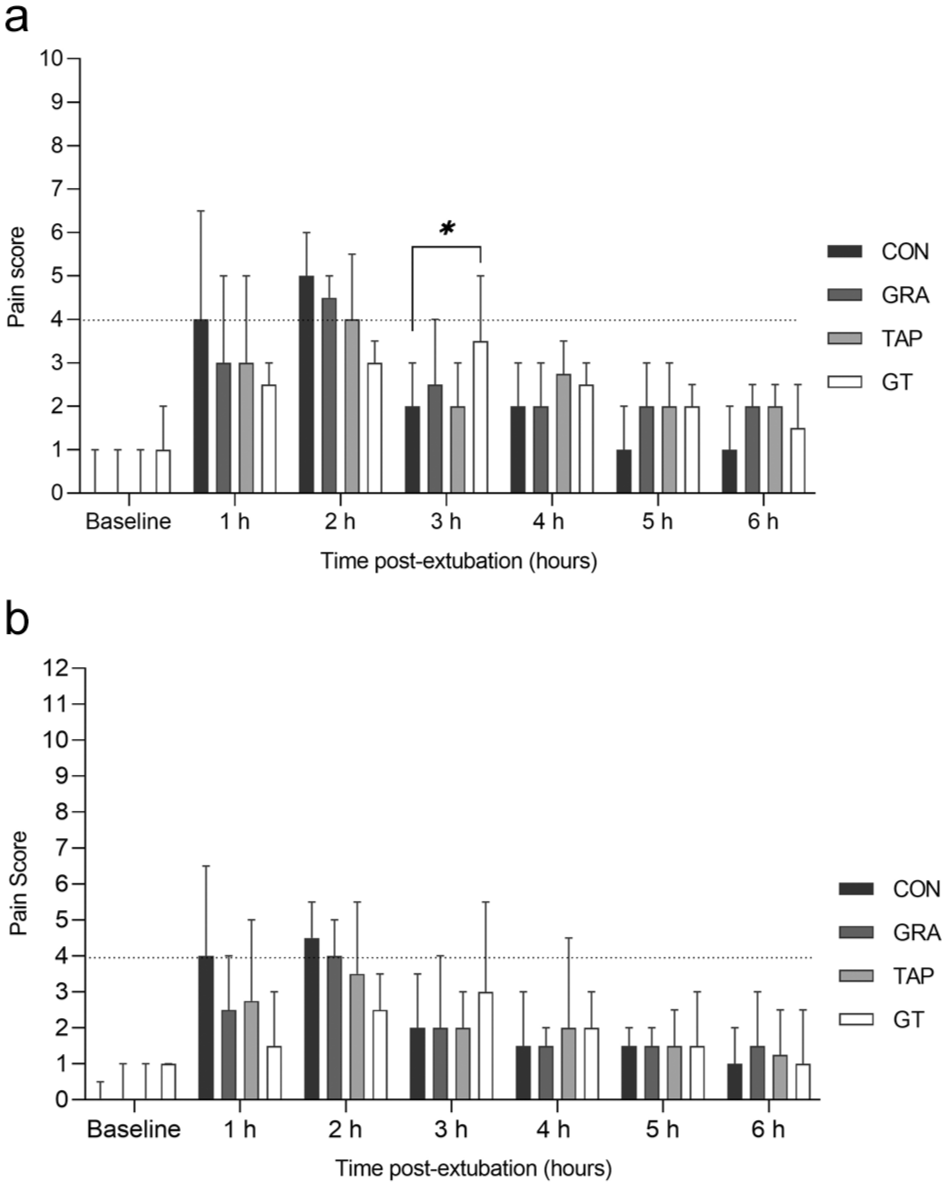
(a) Median and interquartile ranges of Feline Grimace Scale (FGS) scores. *Indicates significant difference (P = 0.0363). (b) Median and interquartile ranges of the Short Form of the UNESP-Botucatu Feline Pain Scale (UFESP-SF) scores. The dashed lines represent the cutoff score for rescue analgesia (⩾4) for FGS and UFESP-SF. CON = control group; GRA = grapiprant group; GT = grapiprant and tapentadol group; TAP = tapentadol group
Rescue analgesia
When comparing the time to first postoperative rescue analgesia administration, significant differences between the groups were found (P = 0.0110). The median time to first rescue analgesia was 1 h for the CON group, 2 h for the GRA and TAP groups, and 3 h for the GT group. The GT group demonstrated significantly longer analgesia compared with both the CON (P = 0.0007) and GRA (P = 0.0058) groups. Only one cat in the TAP group did not require buprenorphine during the 6-h evaluation period. All animals in the remaining groups required rescue analgesia at various time points (Table 4). In addition, three cats from both the CON and TAP groups and two from the GRA group required supplemental rescue analgesia, with no significant differences between groups (P = 0.2610).
Cats receiving initial rescue analgesia within the first 6 h after extubation for the management of postoperative pain after undergoing elective ovariohysterectomy

Data are n (%)
CON = control group; GRA = grapiprant group; GT = grapiprant and tapentadol group; TAP = tapentadol group
Discussion
This study represents the first randomized, blinded, controlled clinical trial investigating the efficacy of combined grapiprant and tapentadol for postoperative pain management in cats. The absence of prior studies on this combination underscores the significance of our findings and highlights the need to develop novel multimodal approaches to pain management in this species. Our findings demonstrated that the grapiprant–tapentadol combination provided superior postoperative analgesia for up to 3 h compared with either agent alone. This finding suggests that multimodal analgesia may significantly reduce the need for early rescue analgesia, a crucial aspect in optimizing postoperative pain management in cats. The interaction mechanisms between tapentadol and NSAIDs in cats are not yet fully understood. However, studies in murine models have demonstrated antinociceptive synergy between tapentadol and ketorolac, potentially arising from convergence on a common second messenger pathway. This synergy appears to stem primarily from the distinct mechanisms of action of these drugs, which collectively enhance analgesic efficacy. Further evidence suggests this interaction may involve opioid receptor activation and modulation of adenosine triphosphate-sensitive K+ channels. 20 These findings support the hypothesis that multimodal analgesic strategies combining tapentadol and NSAIDs could improve pain management while minimizing adverse effects.
To date, three studies have evaluated the analgesic efficacy of grapiprant in cats undergoing elective OVH, comparing it with carprofen, 21 dipyrone 22 and robenacoxib. 10 In our study, grapiprant-induced analgesia was limited to 2 h postoperatively, shorter than the approximate duration of 3 h reported in previous studies.21,22 Nonetheless, our results, as well as those from previously published research, differ from the findings of Pisack et al, 10 who demonstrated that grapiprant’s efficacy was non-inferior to robenacoxib analgesia for up to 24 h postoperatively. This discrepancy may be attributed to methodological differences, such as variations in the timing and frequency of pain assessments, as well as the pain scales used and placebo group. Furthermore, the pharmacokinetics of grapiprant’s action may be influenced by the time required for the synthesis, externalization and blockade of EP4 receptors on the membranes of nociceptive terminals, as suggested by previous studies.21,23,24 This combination of factors may partially explain the lack of early analgesic efficacy observed in the GRA group.
To date, no published data exist regarding tapentadol’s analgesic efficacy in cats undergoing elective OVH. In our study, tapentadol provided postoperative analgesia for up to 2 h, comparable to the duration observed in experimental thermal antinociception models in cats. 12 In an experimental model conducted on dogs, tapentadol induced dose-dependent antinociception; 25 however, in another clinical study, it did not improve postoperative analgesia. 26 Furthermore, in rabbits undergoing orchiectomy, tapentadol demonstrated effective analgesia, providing adequate pain control in the postoperative period. 27 This study provides novel data on tapentadol’s efficacy in cats, paving the way for future research into its pharmacokinetics and dose-dependent protocols, particularly considering its dual mechanism of action and potential synergies with other drugs. One cat in the TAP group did not require rescue analgesia, which may be attributed to the analgesic effect provided during the anesthetic protocol, a reduced pain perception or an individual’s favorable response to tapentadol’s analgesic properties. However, it is important to note that the absence of a rescue analgesia requirement does not necessarily indicate a complete lack of pain but rather suggests that analgesic management was adequate to maintain the animal’s wellbeing throughout the evaluated period.
Regarding the ethical considerations of including the control group, hourly pain assessments were conducted, and when the cats reached the cutoff value on either of the two pain scales used, rescue analgesia was administered immediately. Assessments continued until the end of the study period. Notably, this study observed that cats in the GT group required rescue analgesia later than the other groups. This finding suggests that the drug combination exerted a beneficial effect during the postoperative period, specifically reducing the need for rescue analgesia within the first 3 h after surgery.
In the first 2 h postoperatively, the GT group had the lowest pain scores compared with the TAP and GRA groups, while the CON group had the highest scores. Since all scores were maintained, a significant difference was observed only in the FGS at the third postoperative assessment point (3 h) with a very large effect size (d = 1.25). This result considers that all cats in the CON group had already received rescue analgesia, making the reduction in pain scores expected. Although human studies suggest that large effect sizes may represent only minimal changes in pain scores, 28 in our study, this was reflected by a reduced need for rescue analgesia.
The GT group in the present study had significantly longer extubation times, most likely due to the residual sedative effect of tapentadol. A plausible explanation is that the Tmax of oral tapentadol in cats has been reported to be approximately 2.3 h (range 0.5–8.0), 15 which may have contributed to a sustained sedative effect at the time of extubation. This prolongation is unlikely to be related to the duration of continuous propofol infusion, as no statistically significant differences were observed between groups for this variable. Nevertheless, according to the literature, 29 infusions lasting longer than 30 mins may independently delay extubation, which cannot be entirely ruled out as a contributing factor.
The absence of salivation, the main adverse effect reported, can be attributed to the administration of tapentadol in gelatin capsules, preventing taste aversion in animals.
This study represents a significant advancement in feline analgesia research, being the first controlled clinical trial to demonstrate the effects of combined grapiprant and tapentadol for postoperative pain management. The findings suggest that this combination may offer a safe and effective alternative, minimizing the need for early rescue medication. Furthermore, this study expands our understanding of multimodal approaches to feline pain management, providing clinical evidence that may influence future veterinary analgesia guidelines. Future studies should focus on determining optimal plasma concentrations, evaluating multidose protocols and investigating extended-duration analgesia (>6 h) to better understand the role of this combination in clinical pain management for cats.
A possible limitation of this study is the lack of application of methods or tools that use autonomic changes to recognize nociception or postoperative pain, such as the parasympathetic tone index, since this method is more sensitive to activation of the sympathetic system than physiological parameters in situations where HR does not undergo relevant clinical changes, which may compromise the accurate assessment of anesthetic state and pain in cats. A further limitation of our study includes the use of only one sedation scale. Another limitation was the lack of evaluation of hematological and biochemical variables, as well as the development of methemoglobinemia or Heinz bodies resulting from propofol administration in the cats included in the study. Furthermore, it remains unknown whether the combination of grapiprant and tapentadol could induce such effects.
Conclusions
The preoperative administration of grapiprant and tapentadol in healthy cats undergoing elective OVH provided effective postoperative analgesia for up to 3 h without adverse effects, despite leading to prolonged extubation times. Moreover, the inclusion of a control group in this study enabled an objective assessment of the analgesic duration of grapiprant and tapentadol, confirming their efficacy for 2 h.
Supplemental Material
Appendix A
Feline Multiparametric Sedation Score. Appendix B: Mean ± standard error of intraoperative physiological parameters.
Supplemental Material
Supplemental Material
ARRIVE Essential 10 compliance questionnaire.
Footnotes
Acknowledgements
The authors wish to express their gratitude to CNPq and CAPES for funding the master’s scholarship of the author EAA. We would like to thank Marcia Souza, from the Veterinary Hospital at UESC, for her invaluable assistance and technical support and Lucas José Luduvério Pizauro for the help provided with the statistical part of the study.
Supplementary material
The following files are available as supplementary material:
1. Appendix A: Feline Multiparametric Sedation Score. Appendix B: Mean ± standard error of intraoperative physiological parameters.
2. ARRIVE Essential 10 compliance questionnaire.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The State University of Santa Cruz financed this project (grant number 073.6762.2024.0001739-06).
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
