Abstract
Objectives
It is often suggested in the literature that the anatomy of the extrahepatic portal vein (EHPV) in dogs and cats is similar. Nevertheless, variations and contradictions in the tributaries of the EHPV in cats have been described. Therefore, the aim of this study was to describe the normal anatomical variations of the tributaries of the EHPV in a large cohort of cats.
Methods
A retrospective, descriptive, cross-sectional study of abdominal CT angiography (CTA) scans was performed. Cats that underwent CTA between January 2020 and July 2024 were reviewed in consensus by three observers. Cats with vascular anomalies or CTA scans in which the EHPV or its tributaries could not be accurately evaluated were excluded.
Results
A total of 52 CTA scans were included. The splenic vein (SV) was consistently present in all cats as the most caudally inserting vein in the EHPV. The left gastric vein (LGV) entered the EHPV directly in 42/52 cats and, in 36 of those, the LGV was the most cranial vein entering the EHPV. In 28 cats with direct insertion, a second branch of the LGV entered the SV. In eight cats, a single LGV entered the SV, as is typically seen in dogs. In the remaining two cats, the LGV was not identified. In 34/52 cats, the gastroduodenal vein entered the EHPV, as described in dogs, whereas in the remaining 18 cats, the right gastric vein and cranial pancreaticoduodenal vein entered the EHPV together.
Conclusions and relevance
Various anatomical variations of the inserting veins in the EHPV were identified. Knowledge about the variation in normal anatomy of the EHPV and its tributaries is important, especially to evaluate complex pathologies of the EHPV, such as vascular anomalies and EHPV thromboses.
Plain language summary
It is often assumed that the network of blood vessels leading to the liver is the same in cats and dogs, but some studies have shown that this might not always be true. In this study, researchers looked closely at detailed three-dimensional scans of cats to see how certain blood vessels outside the liver are connected. Previous scans taken between 2020 and 2024 were reviewed, and only the ones that clearly showed these blood vessels were selected. In total, 52 scans were analysed. It was found that although some blood vessels were present in all cats, others showed different patterns. For example, one vein called the left gastric vein connected in different ways in different cats: sometimes directly, sometimes through another vein and occasionally it was not seen at all. Understanding these normal differences is important. It helps veterinarians and specialists better spot unusual or serious issues in the liver’s blood flow, such as blockages or abnormal blood vessel formations.
Keywords
Introduction
The portal vein (PV) is anatomically divided into an extrahepatic and an intrahepatic portion. Although the intrahepatic portion of the PV in cats has been extensively documented in the literature, there is considerable variation in the description of the feline extrahepatic PV (EHPV) and its main tributaries.1 –5 Nonetheless, it is crucial for the understanding of pathological conditions affecting the EHPV and its tributaries to have a good knowledge of normal anatomy, particularly to correctly evaluate complex diseases, such as EHPV thrombosis, and vascular anomalies, such as portosystemic shunts, portal vein aplasia and arteriovenous communications.6 –9
In dogs, the EHPV is formed by four primary tributaries: the caudal mesenteric vein, cranial mesenteric vein, splenic vein (SV) and gastroduodenal vein (GdV) (Figure 1).3,10 –12 The caudal and cranial mesenteric veins join dorsally to the transverse colon to form the EHPV; more cranially on the left dorsal side, the SV enters the EHPV. 10 The canine left gastric vein (LGV) is a consistent tributary of the SV.11,12 Just before the PV dives into the liver parenchyma, the GdV enters the EHPV ventrally, as the confluence of the right gastric vein (RGV) and the cranial pancreaticoduodenal vein (CrPDV). 10 In dogs, the anatomical configuration of the EHPV is regarded as consistent across the literature. 3 Conversely, in cats, considerable anatomical variability and conflicting descriptions have been documented regarding the tributaries contributing to the EHPV.1,3,4,5
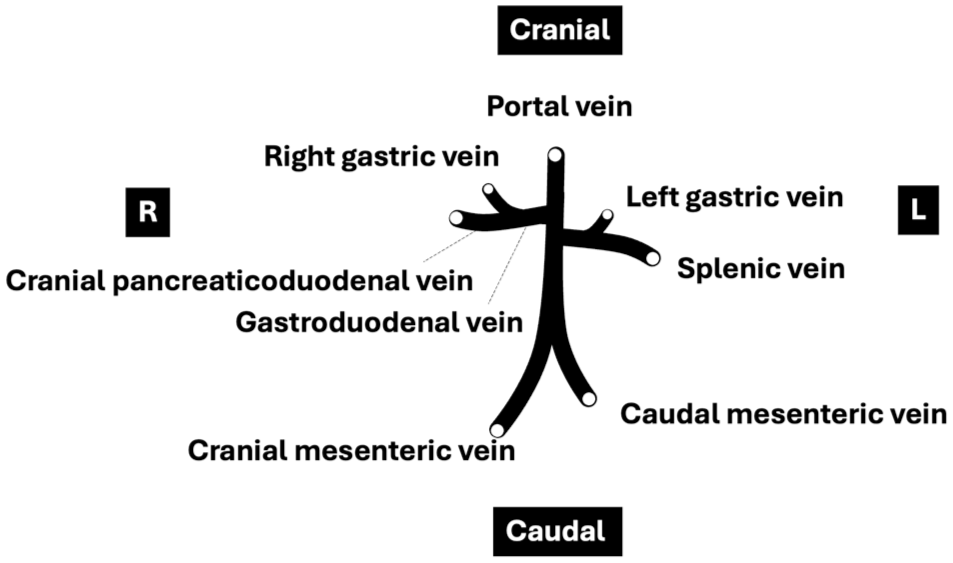
Schematic representation of portal vein tributaries in dogs
The aim of the present study was to describe the anatomy of the main tributaries of the EHPV based on CT angiography (CTA) in a cohort of cats without evidence of EHPV pathology.
Materials and methods
Data acquisition
Abdominal CTA studies of cats performed between January 2020 and July 2024 in the veterinary clinic of Ghent University were retrospectively reviewed. All CTA studies had been performed with a 320-slice Aquillon ONE TSX-301C CT scanner (Toshiba). Intravenous contrast consisted of a non-ionic iodinated contrast agent (600–700 mg I/kg body weight). Because of the retrospective nature of this study, the iodinated contrast agent was not standardised; iohexol (350 mg I/ml), iopromide (300 mg I/ml) and iomeprol (300 mg I/ml) had been used.
All CTA scans were reviewed to verify whether the inclusion criteria were met. Cats had to be positioned in ventral recumbency, the entire abdomen had to be included in the scan and sufficient contrast had to be present in the portal system. Studies were excluded if foreign objects caused artifacts that prevented assessment, in cases of large abdominal masses impairing visualisation of the EHPV and/or its tributaries, when motion artifacts were present, when images had a low signal:noise ratio, or when PV thrombosis or vascular anomalies were present. For cats that underwent multiple CTA scans within the study period, only one scan providing optimal visualisation of the veins of interest was included in the study.
For all included cats, data were collected on breed, sex, age and body weight at the time of the CTA scan as well as the indication for the scan.
Given the retrospective nature of the study, different anaesthetic protocols were used; however, all cats were consistently maintained under general anaesthesia with isoflurane vaporised in oxygen. As is current practice, before each scan, cats were hyperventilated to induce a period of apnoea, minimising motion artifacts.
Image evaluation
All CTA scans were systematically reviewed by three evaluators simultaneously: a Diplomate of the European College of Veterinary Diagnostic Imaging (ECVDI) (ES), a Diplomate of the European College of Veterinary Surgery (ND) and a third-year ECVDI resident (MD). Images were reviewed on a dedicated workstation using Osirix software with a soft tissue window, preferably in the portal phase.
The EHPV was systematically examined from the caudal to the cranial aspect of the abdomen, starting at the confluence of the cranial and caudal mesenteric veins. All veins expected to contribute to the EHPV – namely, the SV, LGV, CrPDV and RGV (with the latter two merging to form the GdV) – were identified, and their points of insertion were documented. The orientation of each vein as it entered the EHPV, as well as the location and order of insertions, were recorded. In addition, the diameters of the EHPV and the aorta at the liver hilum were measured to calculate the EHPV:aorta ratio. Finally, the course of the EHPV at the liver hilum was described. Consensus was reached for all obtained results.
Data analyse
Numerical data (age, body weight and EHPV:aorta ratios) were tested for normality using the Shapiro–Wilk test with SPSS Statistics version 29 (IBM).
Results
Study population
A total of 128 CTA scans were initially reviewed. Of them, 76 scans were excluded for various reasons: presence of an intra-abdominal mass (n = 22), presence of a vascular anomaly (n = 15), absence or suboptimal contrast in the portal system (n = 8), low signal:noise ratio (n = 8), blurring by motion artifacts (n = 6), CTA performed in lateral recumbency (n = 5), presence of a thrombus in the EHPV (n = 5) and presence of foreign objects causing artifacts making assessment impossible (n = 4), Finally, three CTA scans were excluded as another CTA scan of the same cat during the study period had already been included.
All cats were client-owned and included breeds were domestic shorthair (n = 40), British Shorthair (n = 5), Persian (n = 2), Austrian Shorthair, Bengal, domestic longhair, Maine Coon and Scottish Fold (n = 1 each). Half of the cats were male (24/26 castrated) and half were female (23/26 spayed). The mean age was 110.3 ± 56.6 months; the age of one cat was unknown. The median body weight was 4.2 kg (2.0–8.3 kg). Cats underwent CTA for various reasons: (suspected) neoplasia (n = 18), trauma (n = 10), neurological signs (n = 7), respiratory signs (n = 6), exclusion of a portosystemic shunt (n = 2), behavioural changes, chronic hyporexia and weight loss, chylothorax, colonic stricture, lameness, intrathoracic abscess, pleural effusion and multiple fistulae (n = 1 each). In one cat, the reason for CTA was unknown, as it was referred for imaging without prior consultation.
Anatomical variations of tributaries entering the EHPV
A total of 10 different variations were identified in this study population (Figure 2).

Schematic representation of all described variations in portal vein tributaries in 52 cats observed with CT angiography: (a) one branch of the left gastric vein entering the splenic vein (8/52); (b) one branch of the left gastric vein entering the portal vein (14/52); (c) two branches of the left gastric vein, one entering the splenic vein and one entering the portal vein (28/52); and (d) inserted left gastric vein not identified (2/52)
The SV was consistently identified in all feline specimens, representing the most caudally positioned tributary entering the EHPV.
In 28/52 (53.8%) cats, two branches of the LGV were identified, of which one entered the SV and the other directly entered the EHPV. In 22/52 (42.3%) cats, one branch of the LGV was identified: this branch entered the SV in 8/52 (15.4%) cats, as is typically seen in dogs, whereas it entered the EHPV directly in 14/52 (26.9%) cats. CT images showing direct entrance of the LGV into the EHPV can be found in Figure 3. No LGV was identified in 2/52 (3.8%) cats.
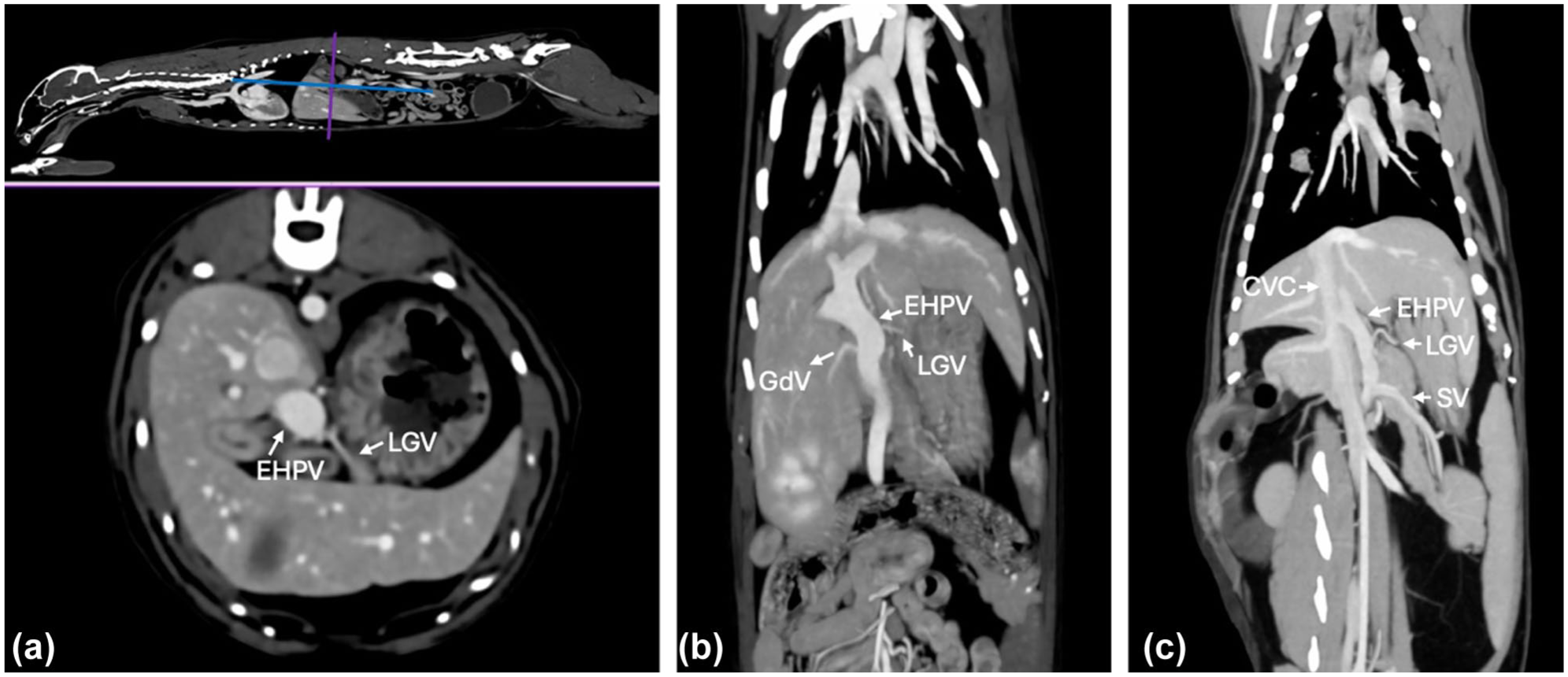
CT angiography images showing three different cats with separate entrances of the left gastric vein (LGV) into the extrahepatic portal vein (EHPV), with reference slices on the left top sagittal image: (a) transverse CT image; (b) dorsal maximum intensity projection (MIP) with the LGV entrance in the EHPV cranial to the entrance of the gastroduodenal vein (GdV); and (c) dorsal MIP. CVC = caudal vena cava; SV = splenic vein
A GdV, as described in dogs, was present in 38/52 (73.1%) cats, whereas the RGV and CrPDV merged at the level of the insertion in the EHPV in 14/52 (26.9%) cats, implying that these cats do not have a GdV. Figure 4 shows two CT images of the described merging pattern of the RGV.

Transverse maximum intensity projection CT angiography images of two different cats, accompanied by corresponding reference slices on the sagittal images above. Both displayed cases demonstrate a merging pattern of the right gastric vein (RGV). CrPaV = cranial pancreaticoduodenal vein; CVC = caudal vena cava; EHPV = extrahepatic portal vein; LGV = left gastric vein; RGV = right gastric vein
Differing order of entrance of the LGV, GDV, RGV and CrPDV into the EHPV was identified. In 42/50 cats with an LGV, the LGV was the most cranial tributary in 36/42 (85.7%) cats, ending just cranial to either the GdV or the merged RGV and CrPDV. In 2/42 (4.8%) cats, the LGV entered at the same level as the GdV or the merged RGV and CrPDV, and in 4/42 (9.5%) cats the LGV entered just caudal to the GdV or the merged RGV and CrPDV.
Orientation of insertion of tributaries into the EHPV
The orientation of venous insertion into the EHPV was also variable (Table 1). In 45/52 (86.5%) cats, the SV entered the EHPV on the left side, including left dorsolateral, left lateral and left ventrolateral positions. Direct insertion of the LGV into the EHPV occurred in 39/42 (92.9%) cases at the left ventral side. In cases where only one LGV branch inserted directly into the EHPV, insertion occurred at the left ventrolateral side in 9/14 (64.3%) cats and at the ventral side in 5/14 (35.7%) cats. Both the GdV and the combined RGV and CrPDV most often inserted at the right ventral side of the EHPV (29/38, 76.3% and 11/14, 78.6%, respectively). Notably, in one cat, the GdV inserted into the EHPV at the left ventrolateral position, and in another, the RGV and CrPDV entered jointly at the left lateral aspect. In the 14/52 (26.9%) cats where the RGV and CrPDV entered the EHPV together, the RGV was consistently positioned ventrally relative to the EHPV and to the left of the CrPDV.
Overview of the orientation of insertion into the extrahepatic portal vein by the different tributaries
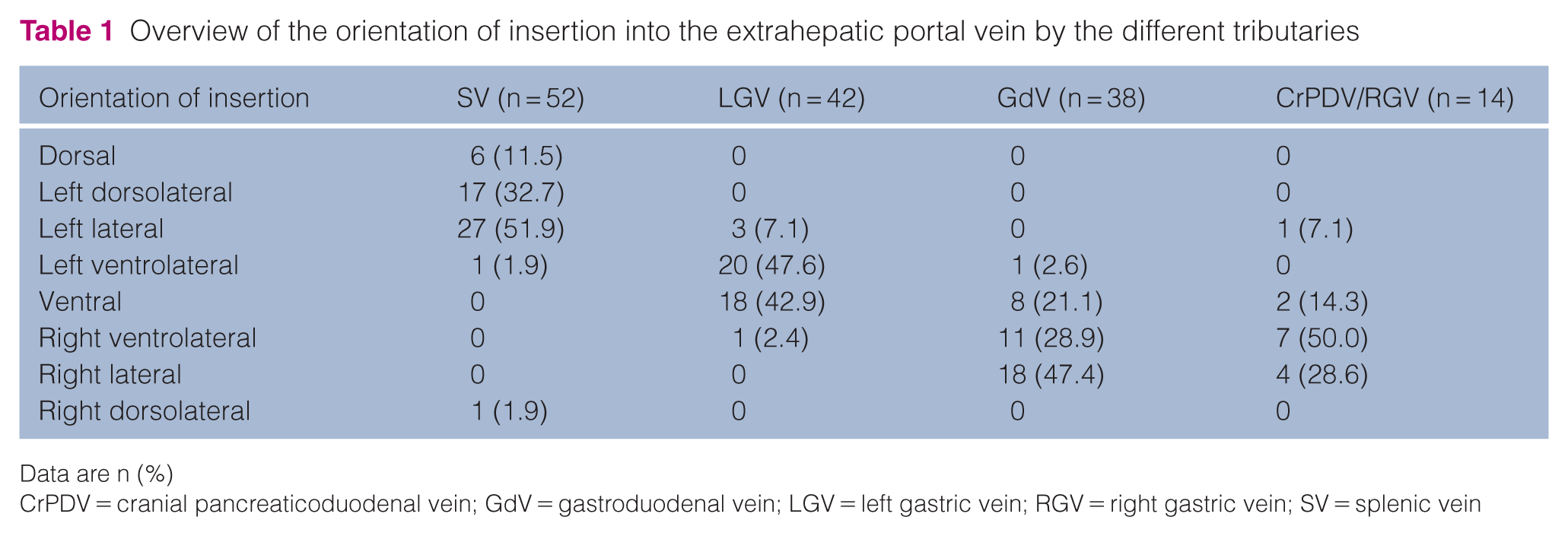
Data are n (%)
CrPDV = cranial pancreaticoduodenal vein; GdV = gastroduodenal vein; LGV = left gastric vein; RGV = right gastric vein; SV = splenic vein
EHPV:aorta ratio and course of extrahepatic portal vein
At the liver hilum, the mean EHPV diameter was 6.0 ± 1.0 mm and the mean diameter of the aorta was 4.0 ± 0.6 mm. The associated mean EHPV:aorta ratio was 1.5 ± 0.3.
The course of the EHPV at the porta hepatis was straight in 18/52 (34.6%) cats and 360° tortuous in an anticlockwise direction in 34/52 (65.4%) cats.
Discussion
This study provides novel insights into the anatomical variability of venous tributaries entering the EHPV in cats, identifying multiple deviations from previously reported descriptions. These findings enhance the understanding of species-specific anatomical characteristics and underscore the importance of avoiding erroneous extrapolations between canine and feline vascular anatomy. A notably high degree of variability was observed in the insertion pattern of the LGV, with a separate branch directly entering the EHPV in over 80% of cats. Interestingly, only 15.4% of the feline specimens exhibited the traditionally described vascular anatomy of the LGV entering the SV, as is observed in dogs.3,11,12 These findings contrast with previous descriptions in the veterinary anatomy literature, which state that the LGV is a consistent tributary of the SV in both dogs and cats.4,11,12
The independent insertion of the LGV into the EHPV has never been reported before and may have significant clinical implications. Given that the LGV is implicated as the most frequent origin of extrahepatic portosystemic shunts, in approximately three-quarters of feline cases, anatomical variations in this vessel could potentially be relevant to both the development and clinical management of portosystemic anomalies.5,13 For example, an additional direct LGV branch might serve as an alternative drainage route in case of venous obstruction or thrombosis, thereby influencing the presentation and severity of clinical signs. In addition, the LGV could not be identified in almost 4% of cats; however, the reason for this remains unclear because of the retrospective nature of the study. It is possible that the vessels were too small to be visualised, although a true anatomical absence, albeit unlikely, cannot be entirely excluded.
Anatomical variability was also noted in the presence of the GdV, which was absent in 26.9% of cats. This partially aligns with earlier injection studies that confirmed the GdV as a consistent tributary of the EHPV.1,4 However, this observation contradicts a leading surgical textbook that states that the GdV is absent in all cats, while no information regarding the possible separate insertions of the RGV or CrPDV in the EHPV is provided. 3 Moreover, the discrepancy in insertion patterns of the RGV and CrPDV, which appeared to enter the EHPV at the same level in almost 27% of cases, further complicates the anatomical interpretation. In contrast to a previous anatomical study that used latex injection into the cranial mesenteric vein followed by dissection in eight Van cats – where the RGV was reported to insert approximately 3 mm cranial to the CrPDV 4 – our CTA findings in a larger feline cohort demonstrated that these two vessels converge at a common insertion point. The inability to definitively confirm separate insertions on CTA is likely due to blooming artifacts, highlighting the limitations of CTA alone for closely situated vascular structures. Therefore, a multimodal approach combining CTA with corrosion casting and gross anatomical dissection would provide a more comprehensive assessment and allow fine-tuning of potential variations in cats.
The SV consistently acted as the most caudal contributing vessel to the EHPV in all cats, which aligns with the literature.1 –4 This consistency reinforces the SV’s role as a reliable anatomical landmark for both surgical planning and diagnostic imaging.
Regarding the spatial orientation of vessel insertions, this study provides the first detailed characterisation in cats. The SV most commonly entered from a left lateral or dorsolateral direction, comparable to findings in dogs. 10 The LGV, when present as a single branch, inserted from a left ventrolateral direction, whereas a potential second branch more often entered from a ventral position. Interestingly, the GdV, when present, entered the EHPV in nearly half of the cats from a right lateral position, and in approximately one-fifth from a ventral position, contrasting with a previous study in eight cats, which reported ventral insertion as dominant. 1 Variability was also observed in the insertion of the RGV and CrPDV, particularly when they entered the EHPV at a shared point; in these cases, the approach was most often from the right ventrolateral aspect. As noted in the results, several unexpected variations in vascular orientation were identified. For example, in one cat, the GdV entered the EHPV from a left ventrolateral position, while in another the RGV and CrPDV simultaneously inserted at the left lateral aspect. These observations highlight the presence of highly atypical anatomical configurations in certain individuals. Such heterogeneity in insertion orientation may have practical implications for interpreting diagnostic imaging modalities, including ultrasonography, CTA and intraoperative portography. A better understanding of these patterns could enhance accuracy in identifying vascular structures during both diagnostic and surgical procedures, particularly in cases involving vascular anomalies or shunt surgeries.
The EHPV:aorta diameter was in line with previously published data of CTA in cats. 14 Similarly, the observation of the course of the EHPV at the liver hilum, characterised as either straight or exhibiting a 360° anticlockwise curvature, as observed in two-thirds of cats, is likely an anatomical variant arising during embryogenesis. The absence of any known pathological associations renders its clinical relevance unclear at present. Nonetheless, its recognition is important to avoid misinterpretation during imaging or surgical procedures.
The present study has some limitations. The retrospective design of this study and the inclusion of cats undergoing CTA for clinical indications inherently introduce selection bias. Despite the application of strict exclusion criteria (resulting in the inclusion of only 40% of the initially selected CTA scans to rule out identifiable EHPV pathology) the study population cannot be fully regarded as representative of a healthy cohort. Furthermore, because of the limited breed diversity and sample size, subtle breed-specific vascular patterns may have been missed. Future studies should aim to include larger, prospectively enrolled feline populations with known health status, possibly using cadaveric specimens for complementary dissection-based confirmation of imaging findings.
As mentioned earlier, the reason why the LGV could not be identified in nearly 4% of cats remains unknown. Although variation in contrast phases can present a challenge in imaging studies, this factor was carefully controlled by applying stringent inclusion criteria, ensuring that only high-quality CTA scans with optimal contrast enhancement were included in the analysis. Moreover, all other tribu-taries of the EHPV in these cases were clearly visualised.
CTA, although highly informative, remains constrained by imaging artifacts and resolution limitations. Blooming artifacts and overlapping vascular structures complicate the delineation of individual vessel insertions. As such, the integration of multiple imaging modalities, including high-resolution micro-CT, corrosion casting, and histological validation, is essential to deepen our anatomical understanding.
Conclusions
Significant anatomical variation exists in cats concerning the presence and orientation of tributaries entering the EHPV. The findings of this study indicate that the anatomy of the EHPV in cats differs from that described in dogs, thereby confirming that anatomical characteristics observed in dogs cannot be directly extrapolated to cats.
Footnotes
Acknowledgements
The authors would like to thank Arnout Beirnaert, who started this research as part of his Master thesis. Figures used in this article are based on figures he made for his thesis.
Author note
This paper was presented in part at the 2024 European College of Veterinary Diagnostic Imaging conference.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animals described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedures undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
