Abstract
Objectives
The aim of the present study was to evaluate health-related quality of life (HRQoL) in cats with chronic kidney disease (CKD) and integrate these results with clinical data to identify factors associated with decreased HRQoL.
Methods
Caregivers completed a validated HRQoL tool (VetMetrica) and clinical data (history, laboratory work, physical examination findings, etc) were collected. Vitality, comfort and emotional wellbeing (EWB) scores were compared between groups using non-parametric analyses as appropriate.
Results
Data from 24 healthy cats and 68 cats with CKD were collected. Cats with CKD had significantly lower median vitality, comfort and EWB scores (vitality: 41.7, range 4.3–64; comfort: 36.1, range 21.2–59.6; EWB: 44.6, range 1.6–58.8) than healthy cats (vitality: 56.5, range 40.7–64; comfort: 59.6, range 34.2–59.6; EWB: 58.7, range 45.1–58.8) (P <0.0001 for all analyses). Cats with International Renal Interest Society (IRIS) stage 3 and 4 CKD had lower median EWB scores (30.8, range 1.6–54.4) than IRIS stage 1 and 2 CKD (47.3, range 11.2–58.8) (P = 0.04) and lower vitality scores (38.2, range 4.3–54.0) in comparison with IRIS stage 1 and 2 CKD (43.1, range 12.8–64.0) (P = 0.04). Cats with anaemia had lower median EWB scores (25.9, range 10.4–54.4) than those without (46.6, range 1.6–58.8) (P = 0.005), and haematocrit was positively correlated with EWB (P = 0.005, r = 0.33). Cats with abnormal appetite had lower median EWB scores (29.1, range 1.6–56.7) than cats with normal appetite (48.1, range 11.2–58.8) (P = 0.001). Cats with constipation had lower median comfort scores 29.5 (range 21.2–59.5) (P = 0.0003) and lower median EWB scores (22, range 1.6–58.8) (P = 0.008) than those without (comfort: 37.1, range 25.3–59.6; EWB: 47.9, range 11.2–58.8).
Conclusions and relevance
Cats with CKD have decreased HRQoL. Several modifiable clinical factors are associated with this decreased HRQoL.
Introduction
Quality of life (QoL) is a broad concept that is impacted by many features of an individual’s life, including physical health, psychological state, social relationships and salient features of their environment. Health-related quality of life (HRQoL) is concerned with the effects of illness and the impact treatment may have on QoL.1,2 In human medicine, assessment of HRQoL in addition to biomedical outcomes has become an integral part of the understanding of disease management. In the veterinary setting, the assessment of HRQoL is determined by caregiver observation and report of specific behaviours that indicate how the animal might ‘feel’ about its circumstances. The importance of HRQoL to the caregiver is likely a significant component in decisions made regarding the sick pet. For example, in a study of the perception of HRQoL in cats with heart disease, 93% of caregivers were willing to trade survival time for comfortable QoL. 3
In human patients with chronic kidney disease (CKD), HRQoL is decreased in comparison with healthy individuals and also decreases according to severity and progression of disease.4 –8 Children with CKD have lower HRQoL scores in comparison with healthy children, and children who have more severe clinical symptoms have lower HRQoL scores in comparison with those who do not.9,10 In cats with CKD, QoL scores related to eating (eg, liking food and appetite) and management aspects, such as administration of medication and accessing veterinary care, are lower in comparison with healthy cats. 11 The potential impact of disease stage and specific clinical and physiologic abnormalities associated with CKD has not been studied in cats.
Conditions such as hypertension, anaemia, electrolyte abnormalities, dysrexia, dehydration and constipation commonly occur as sequelae of CKD and should be identified and treated. 12 These pathologic changes may have a negative effect on the patient and thus alter HRQoL. 11 Administration of medications or treatments in cats that are not amenable to these interactions could adversely affect the human–animal bond and detract from HRQoL.3,11 Identification of modifiable factors that might be associated with a decreased HRQoL may aid clinicians in tailoring treatment recommendations for cats with CKD. Improving our understanding of HRQoL has the potential to better address the emotional health of our patients, understand the impact of disease on the patient and caretaker, and provide a tool to help assess the efficacy and impact of treatments.
The purpose of this study was to assess HRQoL in cats with CKD using a validated HRQoL instrument (VetMetrica)13 –15 completed online by the caregiver, and integrate these results with medical information from the patient in order to identify factors associated with decreased HRQoL. We hypothesised that cats with CKD would have decreased HRQoL scores, and specific clinical factors (eg, anaemia, poor appetite) would negatively impact their score. The above represents an example of ‘known-groups’ construct validation, and assessing this in the feline CKD patient population was a secondary aim of the study.
Materials and methods
Cats
The study was approved by the Institutional Animal Care and Use Committee (IACUC#2019A00000128) at The Ohio State University. Cats were screened for prospective enrolment in 1/2 groups: healthy cats or cats with CKD. All cats enrolled in the study were required to have informed consent signed by the animal’s caregiver.
Healthy cats Client-owned healthy cats with normal systemic blood pressure, serum biochemistry profile, complete blood count (CBC), urinalysis and serum total thyroxine measurement were enrolled. Healthy control cats were defined as those with no clinical abnormalities, normal physical examination, a serum creatinine concentration less than 1.6 mg/dl and a urine specific gravity greater than 1.035.
Cats with CKD Cats with stable International Renal Interest Society (IRIS) stage 1–4 CKD were enrolled. Diagnostic tests required before enrolment included systemic blood pressure, serum biochemistry profile, CBC, urinalysis and serum total thyroxine measurement. The diagnosis of CKD was confirmed, and the affected cats were staged by a board-certified small animal internal medicine specialist according to IRIS guidelines. In cats with stage 1 CKD and early stage 2 CKD, diagnosis was based on either ultrasound findings consistent with degenerative renal disease and/or persistent inadequate urinary concentrating ability without an identifiable non-renal cause. Based on the clinical experience of the investigators, cats with CKD were considered to have stable CKD if serum creatinine had not changed by more than 20% on at least two measurements taken at least 2 weeks apart. Exclusion criteria included other uncontrolled systemic illnesses, pyelonephritis, ureteral obstruction, decompensation of CKD (eg, sudden negative change in clinical status associated with an increase in azotaemia) or any condition requiring hospitalisation and intravenous fluid therapy. Patients receiving dietary management or various therapies, such as potassium supplementation, antihypertensive medications and subcutaneous fluids, for CKD management were not excluded.
HRQoL instrument and clinical data collection
A previously published web-based, generic HRQoL instrument (VetMetrica) was used to measure the impact of chronic disease in cats (see Appendix in the supplementary material).13 –15 Caregivers were asked to participate in the study at the time of the veterinary visit and to complete the survey within 1 week. This instrument consists of 20 behaviour-based items to which the cat caregiver responds using a 7-point Likert-type scale (0 = could not be less to 6 = could not be more), resulting in the generation of scores in three HRQoL domains: vitality, comfort and emotional wellbeing (EWB). A summary score for physical wellbeing (PWB) was obtained by averaging vitality and comfort scores. To aid interpretability of the tool, raw domain scores (0–6) are normalised such that 50 on a scale of 0–100 represents the score for the average healthy cat, and 70% of healthy cats will score 44.8 or higher.13,14 The VetMetrica instrument was supplemented with additional questions (see Appendix in the supplementary material) to gather information about the patient’s clinical history, disease status and management regimen, but these did not impact the manner in which the HRQoL scores were calculated.
Clinical parameters collected for analysis included the following: anaemia (haematocrit or packed cell volume <27%; yes/no); muscle condition score (normal/mild vs moderate/severe); body condition score (BCS; 1–3, 4–5, 6–9); adequate hydration on physical examination (yes/no); decreased or picky appetite since last visit (yes/no); history of hypertension based on IRIS guidelines (yes/no); constipation in the past 6 months (difficulty defecating, smaller/drier stools or decreased frequency of bowel movements (yes/no); active urinary tract infection (UTI) in the past year (clinical signs of lower urinary tract disease plus positive urine culture; yes/no); and number of medications administered (0–1, 2–4, 5–7). Continuous variables collected for analysis included patient age, body weight, serum creatinine concentration, serum potassium concentration, serum phosphorus concentration, haematocrit, number of medications and systolic blood pressure.
Statistical analysis
Statistical analysis was performed using Prism version 9 (GraphPad Software) and Minitab version 20 (Minitab). Data were assessed for normality using the D’Agostino – Pearson test and visual inspection of quantile–quantile plots, and analysed using non-parametric methods accordingly. A Mann–Whitney U-test was used to compare each of the HRQoL domains between healthy cats and those with CKD aged 10 years or younger. All other analyses were performed within the CKD cat group. A Mann–Whitney U-test was used to compare each of the HRQoL domains between IRIS stages (1 and 2 vs 3 and 4), anaemia, muscle condition score, hydration, decreased or picky appetite, hypertension, constipation and active UTI. The Kruskal–Wallis test was used to compare each of the HRQoL domains between healthy cats, those with IRIS stage 1 and 2 CKD and those with IRIS stage 3 and 4 CKD, between BCS categories and between the number of medications administered. Spearman’s rank analysis was used to assess the correlation between each of the HRQoL domains and age, serum creatinine concentration, haematocrit, body weight, number of medications, serum potassium concentration, serum phosphorus concentration, systolic blood pressure and serum creatinine vs haematocrit. Significance was determined by P <0.05 for all comparisons.
Results
Cats
The 68 cats with CKD had a median age of 14 years (range 0.8–20) and consisted of 47 domestic shorthairs (DSHs), eight domestic longhairs (DLHs), seven domestic medium hairs (DMHs), three Maine Coons, two Siamese and one Tonkinese. Of them, 31 cats were castrated males and 37 cats were spayed females. The 24 healthy cats had a median age of 6 years (range 2–10), which was significantly different from the CKD cats (P <0.0001). They consisted of 18 DSHs, five DLHs and one DMH. Of them, 14 cats were castrated males and 10 cats were spayed females. CKD cats had a median serum creatinine concentration of 2.2 mg/dl (range 1.1–6.3). Distribution of disease stage was as follows: IRIS stage 1, n = 8, IRIS stage 2, n = 41, IRIS stage 3, n = 15 and IRIS stage 4, n = 4. Healthy cats had a median serum creatinine concentration of 1.3 mg/dl (range 0.9–1.5).
HRQoL scores
A summary of median (range) HRQoL scores is presented in Table 1. Cats with IRIS stage 1 and 2 CKD and those with IRIS stage 3 and 4 CKD had significantly lower vitality, comfort, EWB and PWB scores than healthy cats (P <0.0001 for all analyses) (Figure 1). When only cats with CKD were considered, cats with IRIS stage 3 and 4 CKD had lower EWB scores (P = 0.04) and lower vitality scores (P = 0.04) than cats with IRIS stage 1 and 2 CKD (Figure 2).
Normalised health-related quality of life (HRQoL) domain score results and physical domain summary score (physical wellbeing) for cats in each study population and clinical sign, treatment or clinicopathologic category
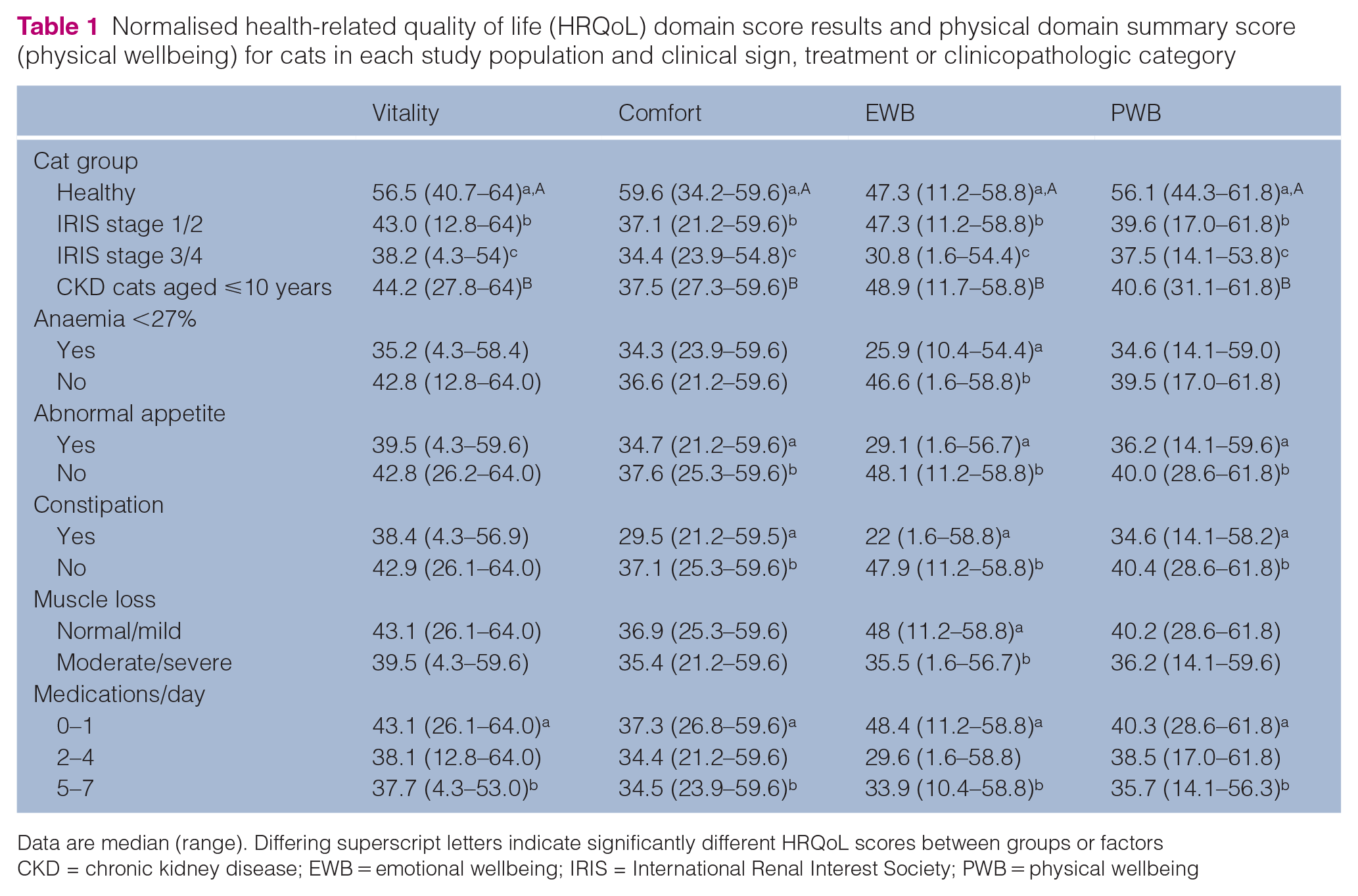
Data are median (range). Differing superscript letters indicate significantly different HRQoL scores between groups or factors
CKD = chronic kidney disease; EWB = emotional wellbeing; IRIS = International Renal Interest Society; PWB = physical wellbeing

Health-related quality of life (HRQoL) scores in healthy cats vs cats with International Renal Interest Society stage 1/2 and 3/4 chronic kidney disease (CKD). Cats with both early- and late-stage CKD have significantly lower HRQoL scores in all three domains than healthy cats. The dot and whisker plot portrays median and range, with dots representing individual cats for each domain. The dotted line represents the mean score for healthy cats. ****P < 0.0001. IRIS = International Renal Interest Society
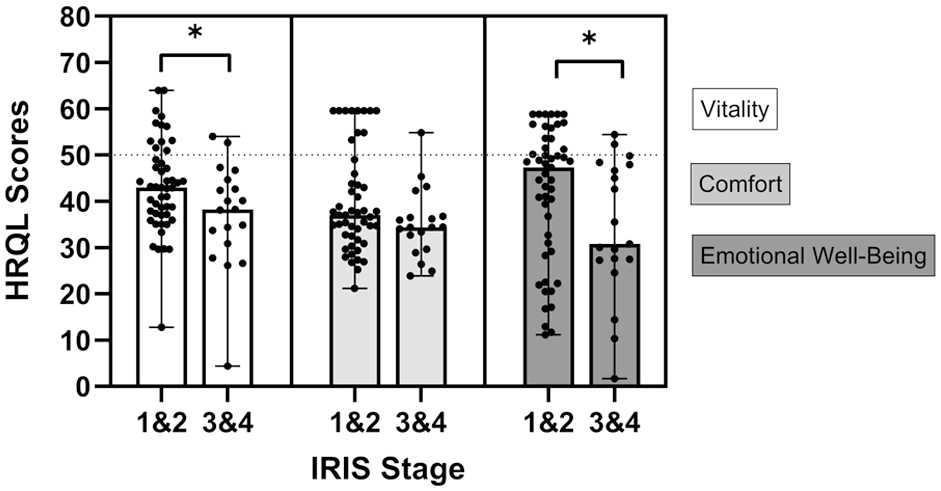
Health-related quality of life (HRQoL) scores in early and late International Renal Interest Society (IRIS) stage chronic kidney disease (CKD). Cats with IRIS stage 3 and 4 CKD (n = 19) had lower vitality and emotional wellbeing scores than IRIS stage 1 and 2 (n = 49) (*P = 0.04 for both comparisons). The dot and whisker plot portrays median and range, with dots representing individual cats for each domain. The dotted line represents the mean score for healthy cats
When only cats with CKD aged 10 years or younger (n = 18) were compared with healthy cats, those with CKD still had significantly lower vitality, comfort, EWB and PWB scores (P = 0.0008, P <0.0001, P = 0.0001, P <0.0001, respectively). Age within the CKD population was negatively correlated with vitality (P = 0.0006, r = –0.40), comfort (P <0.0001, r = −0.47) and EWB (P = 0.004, r = −0.47).
Cats with anaemia <27% had lower EWB scores than those with haematocrit of 27% or greater (P = 0.005) (Figure 3). Haematocrit was positively correlated with EWB (P = 0.005, r = 0.33). Cats with abnormal appetite had lower EWB (P = 0.001), lower comfort (P = 0.04) and lower PWB scores (P = 0.03) than cats with normal appetite (Figure 4). Cats with constipation had lower comfort (P = 0.0003), lower EWB (P = 0.008) and lower PWB scores (P = 0.003) than those with reportedly normal stools (Figure 5). Cats with moderate or severe muscle loss had lower EWB scores than those with normal to mild muscle loss (P = 0.008). Cats that were receiving five to seven medications daily had significant lower vitality, comfort and EWB and PWB scores than cats receiving zero to one medications daily (P = 0.04, P = 0.01, P = 0.005 and P = 0.01, respectively). The number of medications administered was negatively correlated with EWB (P = 0.03, r = −0.25). No cat was reported to be receiving more than seven medications.

Health-related quality of life (HRQoL) scores in cats with chronic kidney disease with and without anaemia (haematocrit <27%). Cats with anaemia had lower emotional wellbeing scores than those without (**P = 0.005). The dot and whisker plot portrays median and range, with dots representing individual cats for each domain. The dotted line represents the mean score for healthy cats

Health-related quality of life (HRQoL) scores in cats with chronic kidney disease with normal vs decreased appetite. Cats with decreased appetite since the last examination had lower comfort and emotional wellbeing scores than cats with normal appetite (*P = 0.04 and **P = 0.001, respectively). The dot and whisker plot portrays median and range, with dots representing individual cats for each domain. The dotted line represents the mean score for healthy cats
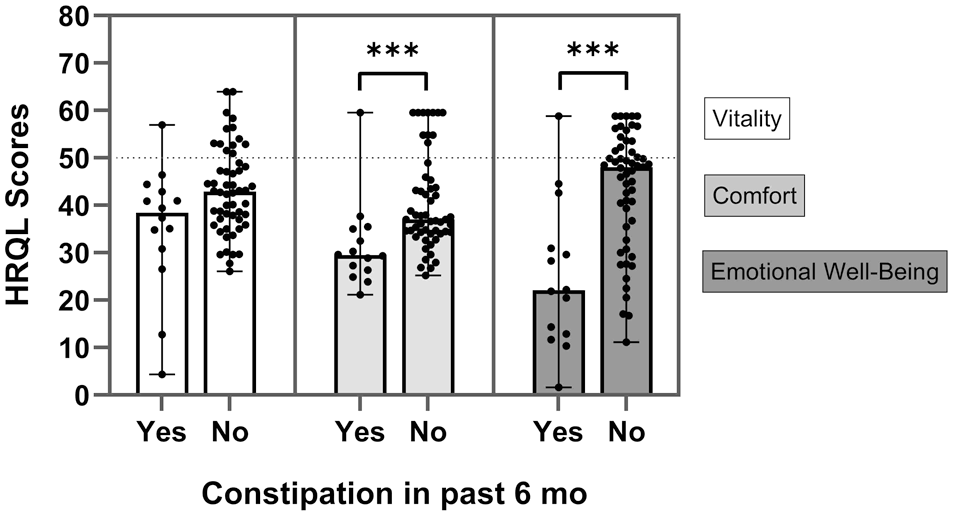
Health-related quality of life (HRQoL) scores in cats with chronic kidney disease with and without constipation. Cats with signs of constipation in the past 6 months had lower comfort and emotional wellbeing scores than those with reportedly normal stools (***P = 0.0003 [comfort] and ***P = 0.008 [emotional wellbeing], respectively). The dot and whisker plot portrays median and range, with dots representing individual cats for each domain. The dotted line represents the mean score for healthy cats
There were no significant differences in HRQoL scores for cats with or without hypertension, active UTI in the past year, hydration status or BCS categories. There was no significant correlation between any of the three HRQoL domains and systemic blood pressure, serum creatinine, potassium or phosphorus concentrations.
Discussion
This study assessed HRQoL in cats with CKD using a validated metric and compared these data to clinical and clinicopathologic parameters. Cats with CKD have significantly decreased HRQoL in comparison with healthy cats, and cats with late-stage CKD have significantly decreased HRQoL scores in comparison with cats with earlier stage CKD. In addition, several key clinical parameters were associated with decreased HRQoL scores including anaemia, decreased appetite, decreased muscle mass and constipation. Although these results demonstrate association and not causation, they provide a foundation for clinical assessment of patients as well as a basis for interventional studies to further demonstrate the relationship between these factors and HRQoL.
In humans, multiple studies have found that CKD significantly affects HRQoL in both non-dialysis and dialysis-dependent patients, impacting primarily physical but also mental HRQoL domains.4 –8 Although many studies describe a correlation between severity and progression of disease and decreasing HRQoL,4,7 these results are inconsistent. 16 These findings are similar to those of our study, where no correlation between serum creatinine concentration and HRQoL domains was seen, but EWB scores were decreased in late-stage disease compared with early-stage disease. Similarly, in the study by Bijsmans et al, 11 HRQoL scores were subjectively decreased in cats with IRIS stage 3 and 4 CKD, in comparison with IRIS stage 2, but failed to reach statistical significance. Alternatively, recent studies on human CKD hypothesise that symptom burden associated with CKD likely has a more impactful role on HRQoL than stage of disease.8,17 –19 Symptoms such as fatigue, decreased appetite, anaemia and constipation are commonly mentioned specifically.6,8 Importantly, many of these CKD symptoms are considered modifiable factors, which, if targeted with management, have the potential to improve HRQoL.6,19
Abnormalities in appetite are common in cats with CKD, and appetite dysregulation may play a role.20,21 Poor appetite and concomitantly low body weight are associated with a poorer prognosis.22,23 In our study, cats with decreased appetite had lower EWB scores than cats with normal appetite. In the study by Bijsmans et al, 11 which assessed cats with CKD using a different instrument, QoL scores related to eating, including ‘liking food’ and ‘appetite’, were also decreased in comparison with young and old healthy cats. Very similar findings regarding the relationship between appetite and HRQoL are present in human medicine. Children with conservatively managed predialysis CKD with ‘good’ or ‘very good’ appetite scores have higher PedsQL (both self and parent-proxy scores) in comparison with children who have an appetite described as ‘poor’ or ‘very poor’. 24 CKD patients receiving haemodialysis who self-reported ‘poor’ or ‘very poor’ appetite have lower kidney disease-specific HRQoL scores in a range of domains, including physical and mental/social, implying that a reduction in appetite has an effect on patients’ lives that goes beyond the impact on food intake and nutritional status. 25 In addition, appetite management in elderly haemodialysis patients has been associated with improvements in both mental and physical domain kidney disease-specific HRQoL scores after treatment with an appetite stimulant. 26 These changes were accompanied by increased appetite, caloric intake, weight and body mass index. 26 Addressing appetite therefore appears to be an important, modifiable HRQoL factor in CKD across species. 6
Anaemia is a common clinicopathologic finding in cats with CKD. Its prevalence increases with disease severity, with up to 53–65% of cats affected in late-stage disease.23,27,28 It is associated with disease progression and may potentially contribute to progression.28 –30 Clinical signs of anaemia, such as lethargy, weakness and poor appetite, may be vague and easily confused with other aspects of disease, and as such have a very plausible potential to effect HRQoL as evidenced by the significant decrease in EWB scores in our study. In humans, multiple studies have documented a relationship between anaemia and HRQoL.6,31 In a cross-sectional study of humans with CKD, patients with anaemia had lower HRQoL scores than those without anaemia. 31 This effect was more apparent in non-dialysis patients with IRIS stage 3 and 4 CKD. In addition, tiredness and impaired activity were both greater in patients with anaemia. Findings were true regardless of the HRQoL scoring system used and were significant in both mental and physical domains. 31 In a study evaluating daprodustat, a hypoxia-inducible factor prolyl hydroxylase inhibitor, treatment resulted in improvements in anaemia, associated fatigue and HRQoL compared with placebo. 32 Importantly, a study assessing caregiver burden in partners of patients with CKD found that patient anaemia was associated with lower HRQoL and significantly higher caregiver burden. 33 Taken together, these findings identify anaemia as a key modifiable factor for improving HRQoL in human CKD patients.6,19 Given the prevalence of anaemia in feline CKD, this may represent an important target for tailored management.
Cats with CKD are more likely to experience constipation than healthy cats and are overrepresented in the population of feline patients presenting to the emergency room for constipation.34,35 The association between CKD and constipation is also recognised in humans, and is likely multifactorial in aetiology in both species, attributed to hydration status, electrolyte disturbances (specifically hypokalaemia), gastrointestinal physiologic alterations and pharmacologic management such as phosphate binders.35 –40 When the effect on HRQoL was considered, cats with constipation in the past 6 months – defined as difficulty defecating, smaller or drier stools, or decreased frequency of bowel movements – had lower EWB and PWB HRQoL scores than those without constipation. These findings are similar to human data demonstrating that the presence and severity of constipation are associated with poorer HRQoL in both physical and mental domains.39,40 Constipation is therefore recognised as a modifiable factor for improving HRQoL in human CKD patients, 6 and our results suggest a similar approach may be beneficial for feline patients.
Administering five or more medications to cats with CKD was associated with lowered HRQoL scores in all categories. Earlier literature demonstrates the difficulty many cat caregivers experience in administering medication to their pets,3,41,42 a phenomenon that over half of caregivers felt altered their relationship with their cat. 41 Difficulty can be experienced with both short-term and long-term medication administration, with caregivers experiencing worry about the interaction and a percentage finding themselves unable to complete a course of medication. 41 These factors are logically compounded with chronic administration of multiple medications. In human patients with CKD, polypharmacy is associated with decreased HRQoL. 43 When hyperpolypharmacy (⩾10 classes of medications) and major polypharmacy (five to nine classes of medications) were compared with minor polypharmacy (four or fewer classes of medications), patients with hyper- and major polypharmacy had significantly lower HRQoL scores in both mental and physical domains. 43 Therefore, pill burden is likely an important part of developing a treatment plan for feline patients with CKD.
Given the propensity for disease in elderly cats, enrolling a population of healthy cats that were age-matched with CKD cats was not possible, and the CKD cat group that was enrolled was older in age than the healthy cat group. The impracticality of age-matching to CKD patients was also present in a previous study assessing HRQoL in cats with and without CKD. 11 In that study, HRQoL was assessed in young healthy (n = 105), old healthy (n = 36) and CKD cats (n = 70); it was concluded that young cats had significantly better HRQoL than older cats and those with CKD. 11 This was particularly true in domains including mobility, stools, playing and hunting. 11 This may reflect other age-related changes, such as osteoarthritis and increased incidence of concurrent disease. However, in that study, the health of the young cats was not confirmed by examination or laboratory work. In the present study, when only cats with CKD under the age of 10 years were considered in comparison with healthy control cats, there was still a statistically significant decrease in HRQoL scores in the cats affected by CKD. In addition, within the CKD cat group, there was a negative correlation with age for all three HRQoL domains. Overall, it can be concluded that age plays a role in HRQoL, but based on the data in the present study, changes in scores in CKD cats are not just attributable to ageing.
The data collected in this study demonstrate the validity of the VetMetrica instrument for evaluating cats with CKD. Validity provides evidence that a tool measures what it is intended to measure – in this case, HRQoL. However, validity is not an intrinsic property of a scale; evidence must be gathered for its use in different purposes, defined populations and specific contexts. Previous studies have established the validity of VetMetrica in healthy cats, cats with various chronic conditions and cats with osteoarthritis;13,15 however, this is the first study to validate its use in a feline CKD population. Instrument validity is established first through construction methods, followed by testing for different types of construct validity. One such method, ‘known-groups’ testing, examines whether scores differ as hypothesised between groups – for example, healthy vs sick animals, or animals with increasing disease severity (mild < moderate < severe). In the present study, known-groups validity was supported by demonstrating different HRQoL profiles between healthy cats and those with CKD, between cats with IRIS stage 1/2 vs stage 3/4, anaemic vs non-anaemic cats, cats with abnormal vs normal appetite, cats with constipation vs normal stool and cats with varying degrees of muscle wasting.
Overlapping scores are still possible with known-group validation as complete separation is not expected and biological variation will be seen even in a ‘normal’ population. It is perhaps worth noting that there is much less variation in the healthy cats compared with the CKD cats. This is to be expected and that greater variability can be attributed to the heterogeneity of the sick population. In general, overlap could be due to the blurring of the distinction between IRIS stages. In particular, the overlap between healthy cats and those with stage 1/2 CKD is likely to be related to the fact that stage 1 cats may be asymptomatic and therefore present as healthy cats in terms of their PWB and EWB.
Limitations of this study include reliance on caretaker-reported data, which were based on observations rather than objective measurements and may have not been accurate (eg, assessment of constipation). In feline patients, a cat’s individual experience cannot be truly assessed, making caregiver perception central to HRQoL evaluation, although such perception may be influenced by unrecognised factors. However, VetMetrica is an observer-based tool in which the caregiver reports behaviours that can then be interpreted to reflect the animal’s emotional state, an approach favoured by the US Food and Drug Administration for non-verbal individuals, including infants, cognitively impaired individuals and animals. 44 In paediatric CKD patients, HRQoL assessments have been found to correlate moderately between patient self-reports and parental assessments, with stronger agreement for physical domains than mental domains. 9 Another limitation was that two cats in the healthy group with high-normal serum creatinine were not imaged to differentiate IRIS stage 1 disease from a healthy status.
Conclusions
Cats with CKD have lower HRQoL scores than healthy cats. Several modifiable clinical signs, such as reduced appetite, anaemia and constipation, are associated with decreased HRQoL, suggesting that identifying and addressing these abnormalities may improve wellbeing in affected cats.
Supplemental Material
Supplemental Material
The VetMetrica HRQoL instrument consists of 20 questions. Additional questions for the caregiver about the clinical wellbeing and management of the cat were included as an addendum to the instrument. Veterinary staff who assessed the cat also provided information on their assessment and clinicopathologic data were collected.
Footnotes
Acknowledgements
The authors wish to thank the caregivers who participated in this study and their cats.
Author note
The results of this study were presented, in part, as a research report at the American College of Veterinary Internal Medicine Annual Forum, 2022.
Supplementary material
The following file is available as supplementary material:
Appendix: The VetMetrica HRQoL instrument consists of 20 questions. Additional questions for the caregiver about the clinical wellbeing and management of the cat were included as an addendum to the instrument. Veterinary staff who assessed the cat also provided information on their assessment and clinicopathologic data were collected.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Dr Jacqueline Reid was previously the director of NewMetrica.
Funding
The study was supported by Buttons Fund for Feline CKD Research at The Ohio State University.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals and procedures that differed from established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient. The study therefore had prior ethical approval from an established (or ad hoc) committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
