Abstract
Objectives
The purpose of this study was to objectively measure defecation frequency in cats with and without chronic kidney disease (CKD). Cats with CKD are at higher risk for presenting with constipation. It was hypothesized that cats with CKD would defecate less frequently than healthy cats.
Methods
A prospective observational study was conducted of healthy cats (n = 9) and cats with CKD (n = 9). Serum biochemistry, complete blood count, total thyroxine measurement, urinalysis and blood pressure measurement were performed in all cats to confirm their health status. The Petivity Smart Litter Box Monitor System was utilized to collect real-time defecation data. Data were collected via the Petivity app for 30 days, during which medical management and husbandry remained consistent. A Mann–Whitney U-test was performed to compare the total number of days without defecation between CKD cats and healthy cats over 14, 21 and 30 days.
Results
Cats with CKD had significantly more total days without defecation than healthy cats over 14 days (P = 0.001), 21 days (P = 0.0004) and 30 days (P = 0.0003). The median total days without defecation over 14, 21 and 30 days was 5 (range 1–8), 7 (range 3–9) and 10 (range 5–13), respectively, for cats with CKD, compared with 0 (range 0–4), 2 (range 0–4) and 2 (range 0–7) for healthy cats. Cats with CKD had a lower mean number of defecation events per day over the 30-day study period (0.86 ± 0.22 defecations/study day) compared with the healthy cats (1.07 ± 0.13 defecations/study day) (P = 0.02).
Conclusions and relevance
Cats with CKD defecate less frequently than healthy cats. The use of litter box monitors could allow for earlier interventions and therapies, possibly preventing episodes of overt constipation.
Introduction
Chronic kidney disease (CKD) is a highly prevalent disease in cats that results in multiple medical complications, including constipation, commonly defined as infrequent or difficult evacuation of feces.1,2 The association between CKD and constipation is also recognized in humans, and is likely multifactorial in etiology in both species, being attributed to hydration status, electrolyte disturbances (specifically hypokalemia), gastrointestinal physiologic alterations, pharmacologic management, such as phosphate binders, and changes in diet.2 –7 In feline patients, early recognition of decreased bowel movement frequency is important as constipation is a common presenting complaint in emergency medicine. 2 Therefore, there is a critical need for monitoring of defecation habits in cats, particularly in disease processes such as CKD where patients are predisposed to constipation. 2 Unfortunately, because of the typically private litter box habits of cats and variable litter box hygiene practiced by caregivers, a lack of awareness of abnormalities in urination and defecation may be present.1,8 A previous study identified a decreased defecation frequency in cats with CKD in comparison with healthy cats, but this study relied on prospective owner observation and survey responses for data collection. 1 Limitations therefore included potential sampling and response bias, and verification of these findings with more objective monitoring is necessary.
The Petivity Smart Litter Box Monitor System (Nestlé Purina Petcare) uses artificial intelligence (AI) to collect real-time urination and defecation data each time cats use their litter box.9,10 These data are then immediately transferred to the smartphone application to allow monitoring of litter box habits for clinical and research use. By utilizing an objective monitoring system to characterize defecation frequency, this technology addresses and circumvents many of the limitations of the previous survey study assessing defecation frequency in cats with CKD. 1 The aim of this study was to use the smart litter box monitoring system to evaluate the frequency of defecation in apparently healthy cats and those with CKD. The hypothesis of the study was that cats with CKD would have decreased defecation frequency compared with apparently healthy cats.
Materials and methods
A prospective observational study design was utilized to evaluate differences in defecation frequency between healthy cats and cats with diagnosed CKD. The study was approved by the Institutional Animal Care and Use Committee (IACUC-2023A00000032) at The Ohio State University. Client- and staff-owned cats were screened for enrollment in one of two groups: healthy cats or cats with CKD. All cats were housed indoors only and did not have a history of house-soiling. All participating households had at least one litter box per cat. All cats enrolled in the study were required to have informed consent signed by the animals’ caregiver.
Healthy cat group
Cats eligible for enrollment as apparently healthy were screened with a comprehensive clinical history and physical examination, blood pressure (BP) measurement (Doppler), serum biochemistry, complete blood count (CBC), serum total thyroxine (TT4) measurement and urinalysis. If all clinical, physical examination and diagnostic information was within normal limits, the cats were enrolled as healthy control cats. Specifically, healthy cats had to show adequate urine-concentrating ability (urine specific gravity >1.035) and serum creatinine concentrations ⩽1.6 mg/dl at the time of enrollment. Nutrition and general husbandry, including the litter box maintenance schedule, were required to remain consistent throughout the data collection period. Deviations from nutrition or husbandry reported at baseline during the study period would result in removal from the study. Apparently healthy cats were permitted to receive monthly heartworm, flea and tick preventatives but no other medications or therapeutics other than pre-appointment gabapentin.
CKD cat group
Cats with stable International Renal Interest Society (IRIS) stage 2–4 CKD were screened for enrollment in the CKD group. Clinical history, physical examination, BP (Doppler), serum biochemistry, CBC, TT4 measurement and urinalysis were performed to verify the diagnosis at the time of enrollment according to the 2019 IRIS guidelines for staging CKD. Disease was considered stable in cats with CKD if the serum creatinine concentration had not changed by more than 20% on at least two measurements taken at least 2 weeks apart. Exclusion criteria included other uncontrolled systemic illnesses, known or suspected gastrointestinal disease, hyperthyroidism, obesity (body condition score 8 or 9) and complications of CKD such as pyelonephritis or decompensation of CKD requiring hospitalization. Concurrent therapies for CKD, such as dietary management, potassium supplementation, phosphate binders, antihypertensive medications and subcutaneous fluids, were acceptable as long as they remained consistent throughout the data collection period. Cats receiving medications such as laxatives, promotility agents, gabapentin (chronic administration) and selective serotonin reuptake inhibitors were excluded from the study.
Litter box monitoring system
The Petivity Smart Litter Box Monitor System was used to collect real-time body weight, defecation and urination data. Once cats met the inclusion criteria, caregivers were given one litter box monitor for each litter box in the home. Monitors were set up and linked with the product app and the caregivers’ home Wi-Fi network according to the manufacturer’s instructions. 11 When multiple cats were present in the home, caregivers manually verified events belonging to each specific cat in the household during the optimization period (eight events are required per cat) to enable the AI system to distinguish between the cats correctly while using the litter box. Monitor setup and correct usage were confirmed by study personnel during a 7–14-day optimization period before the start of data collection (Figure 1). Households with more than five co-housed cats were considered ineligible for enrollment because of additional limitations of the Petivity system in such higher population-density housing. 11
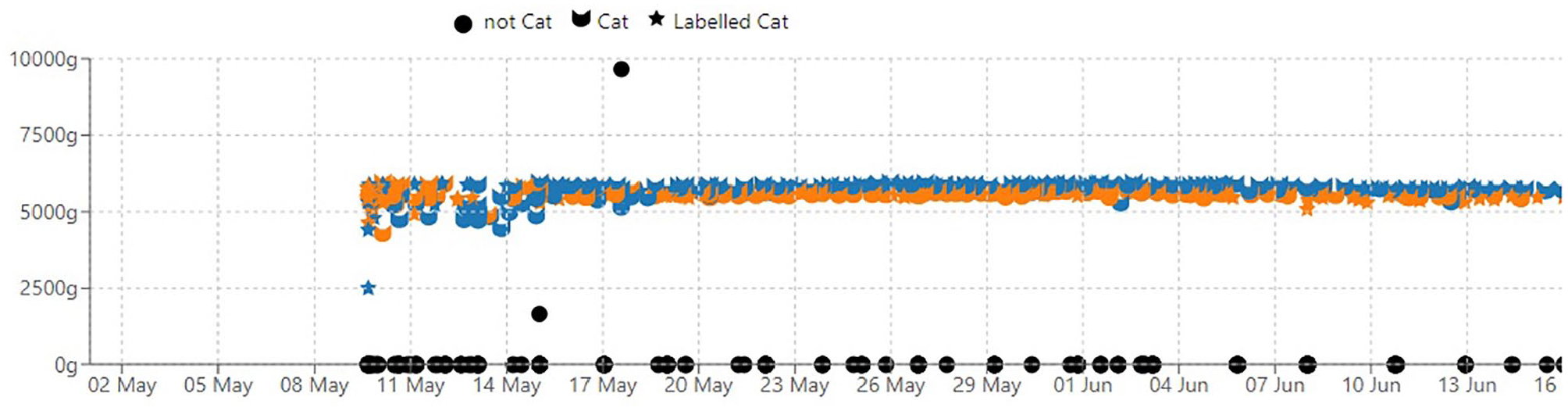
Example of a training and optimization period (depicted here as 9–16 May) required at the start of litter box monitor setup to ensure accurate collection of data. Body weight is displayed on the y-axis and time on the x-axis. The blue cat head icon represents one cat in the household and the orange cat head icon represents the second cat as identified by the device. Blue and orange stars represent cat identification labels manually chosen by the caregiver during the training period. During training and optimization of the monitor location and setup (eg, foot position), body weight may not be consistent. After training and optimization, body weight appears consistent, and cats are consistently identified by the AI model
Litter box data, including body weight and defecation and urination events, were then collected via the app for 30 consecutive days. During the optimization period and the 30-day data collection period, caregivers were asked to perform daily litter box husbandry and monitor the application closely to ensure the device’s AI model correctly identified events as a backup data collection throughout the study period. Caregivers were also asked to inform study personnel if defecation or urination occurred outside the litter box. They were also asked at the end of the 30-day observation period to both provide an average score for their cats’ feces based on the Purina Fecal Scoring system as well as provide a photograph of a typical bowel movement for verification and scoring by investigators. Enrolled cats were disqualified if monitors could not distinguish between cats in the household.
Statistical analysis
Data were assessed for normality using the D’Agostino and Pearson test and were subsequently reported according to data type and results of the statistical testing. Descriptive data were presented as median (range) or mean ± SD. Defecation and urination events were summed and averaged per study day for each cat across the 30-day study period. The average number of defecation and urination events per study day was compared across the entire 30-day study period between groups using an unpaired t-test. The number of total study days without defecation at 14, 21 and 30 days was compared between groups using a Mann–Whitney U-test. Age, serum creatinine concentration and fecal score were also compared between groups using a Mann–Whitney U-test. Statistics were performed with the commercial statistics software Prism 10 (GraphPad Software). Individual body weight measurements provided for each cat during every elimination by the smart litter box device were averaged at baseline (the first 5 days after validation) and study conclusion (the final 5 days of the study period). Averaged data were compared between baseline and study conclusion using Stata statistical software (StataCorp). Significance was set at P = 0.05 for all analyses.
Results
A total of 31 cats were screened for enrollment in this study. During the screening evaluation, four cats failed to meet the criteria for study inclusion or failed to complete the initial study requirements (Figure 2). The remaining 27 cats (14 healthy controls, 13 cats with CKD) were enrolled into the clinical trial. An additional five cats in the healthy group and four cats in the CKD group were removed during the study based on various pre-set criteria. The specific reason for withdrawal for each of these cats is detailed in Figure 2. In total, nine healthy cats and nine cats with CKD met all the study criteria, were enrolled with informed consent and successfully completed the study. No clinical, physical or clinicopathologic abnormalities were identified in the healthy screened cats. There were no cats in either group for which caregivers expressed concerns regarding defecation frequency or constipation at the time of enrollment.

Number of cats screened for the study and those that successfully completed the study
Signalment and relevant clinicopathologic data for both groups are presented in Table 1. Dietary information is presented in Table 1 in the supplementary material. Breeds represented in the healthy cat group were domestic shorthair (n = 6), domestic longhair (n = 2) and Siamese (n = 1). Breeds represented in the CKD group were domestic shorthair (n = 7) and domestic longhair (n = 2). The median age of the CKD group was significantly higher than that of the healthy group (P = 0.0002). The serum creatinine concentration in the CKD group was significantly higher than that in the healthy group (P <0.0001). Cats in the CKD group were classified as IRIS stage 2 (n = 4), stage 3 (n = 3) and stage 4 (n = 2).
Baseline demographic and selected screening data for cats with CKD and healthy cats
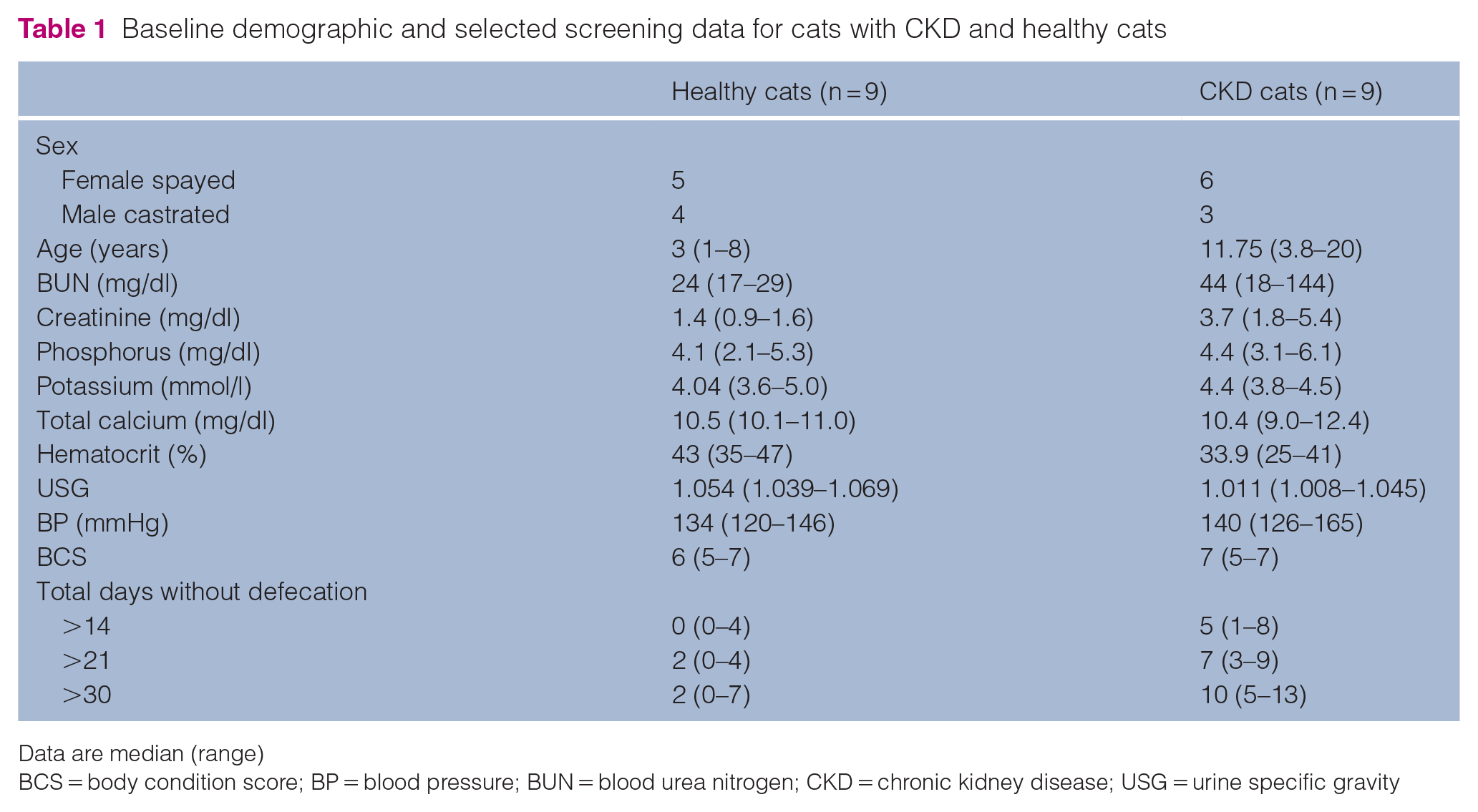
Data are median (range)
BCS = body condition score; BP = blood pressure; BUN = blood urea nitrogen; CKD = chronic kidney disease; USG = urine specific gravity
Body weight of individual cats did not differ between the beginning and end of the study in either the CKD group (P = 0.57) or the healthy control group (P = 0.54). The median percentage change in body weight over the study period in the healthy control group was 0.005% (range −0.02 to 0.05). The median percentage change in body weight over the study period in the CKD group was 0.002% (range −0.03 to 0.03). No cats in either the CKD group or the healthy control group had laboratory or physical examination findings consistent with clinically significant dehydration at study enrollment.
CKD cats received the following medications during the study period, without changes in dosing or administration: transdermal mirtazapine (n = 8), pre-visit gabapentin (n = 6), subcutaneous fluids (n = 4), amlodipine (n = 3), potassium gluconate (n = 3), aluminum hydroxide (n = 3), Renaltec (n = 2), frunevetmab (n = 2), darbepoetin alfa (n = 2), prednisolone (n = 1), maropitant (n = 1), oral hydration supplements (n = 1) and glucosamine/chondroitin sulfate (n = 1). No healthy cats received medication other than three cats that received gabapentin before screening evaluation. Diets fed were variable, including kidney diets for cats with CKD and over-the-counter maintenance diets for the healthy control cats (see Table 1 in supplementary material). Individual diet histories in the CKD group typically comprised rotations of different diets, which were kept consistent throughout the study period (no new dietary elements were added).
Cats with CKD had a lower mean number of defecation events per day over the 30-day study period (0.86 ± 0.22 defecations/study day) compared with the healthy cats (1.07 ± 0.13 defecations/study day) (P = 0.02). In addition, cats with CKD had a greater total number of study days without defecation than healthy cats over the 14-day (P = 0.001), 21-day (P = 0.0004) and 30-day (P = 0.0003) observation periods (Figure 3). Healthy cats had a cumulative total of 0, 2 and 2 days without defecating over the 14-, 21- and 30-day periods, respectively. Cats with CKD had a cumulative total of 5, 7 and 10 days without defecating over the 14-, 21- and 30-day timespans, respectively (Table 1). Photographs of bowel movements were available for confirmation of fecal scoring by investigators in eight healthy cats and six cats with CKD. Caregivers did not submit verification of typical fecal appearance in the remaining cats from each group. The typical fecal score of the cats with CKD (median 2, range 1–4) was not statistically different from that of the healthy cats (median 2, range 2–2) (P = 0.32). Cats with CKD had a higher mean number of urination events per day over the study period (3.86 ± 1.04 urinations/study day) compared with the healthy cats (2.47 ± 0.54 urinations/study day) (P = 0.002).

When litter box monitors were used to assess defecation frequency in cats with and without CKD, cats with CKD had significantly more cumulative days without defecating than healthy cats after (a) 14 days (**P = 0.001), (b) 21 days (***P = 0.0004) and (c) 30 days (***P = 0.0003). CKD = chronic kidney disease
Discussion
This study confirmed previously reported differences in voiding habits between cats with CKD and apparently healthy cats, utilizing litter box monitoring technology to provide an objective assessment. 1 Specifically, the cats with CKD had a decreased defecation frequency over the course of the study period and more days without defecation events compared with healthy cats.
These findings may have important implications, as the development of constipation can destabilize a feline patient with CKD, requiring emergency treatment and hospitalization. 2 Caregivers may struggle to identify when decreased defecation frequency has progressed to constipation. Definitions of constipation are quite variable in the literature, but it is commonly characterized as the infrequent (less than three times per week) or difficult evacuation of hard feces.7,12,13 The link between constipation and CKD is well recognized in human medicine, where constipation is associated with increased risk of CKD and progression of disease.3,4,14 In addition, the presence and severity of constipation is associated with worsening health-related quality of life (HRQL) and thus is identified as a modifiable factor that can improve HRQL in humans with CKD.6,7,15 If the associations observed in human medicine are also present in cats, constipation represents an important clinical target in cats with CKD.3 –6,14 The litter box monitoring technology used in this study to confirm decreased defecation frequency in cats with CKD may, with further research, assist in constipation surveillance and recognition, optimized patient management and potentially improved clinical outcome.
The increased risk of constipation in cats with CKD is multifactorial and may be attributed to hydration status, electrolyte disturbances (specifically hypokalemia), gastrointestinal physiologic alterations, accumulation of uremic toxins, pharmacologic management of the condition (eg, phosphate binders) and changes in diet.2 –4 More specifically, poor hydration status likely contributes to harder feces and hypokalemia contributes to decreased intestinal motility.2,4,16,17 Based on the inclusion criteria and physical examinations used to screen cats in this study, it was less likely that clinically detectable dehydration or hypokalemia were the primary effectors of the change in defecation frequency in the current study.
Use of medications used to alter gastrointestinal motility (such as promotility agents and laxatives) was an exclusion criterion to minimize their impact on study outcomes. However, many of the enrolled cats with CKD received medications that may affect gastrointestinal motility but were also deemed necessary by attending clinicians for the management of CKD. These include medications that may decrease gastrointestinal motility (eg, phosphate binders) as well as medications that may increase gastrointestinal motility (eg, mirtazapine).4,18,19 This study was not large enough to evaluate whether there was an association between bowel movement frequency and individual medications administered.
Dietary fiber content and hydration obtained from eating are major factors impacting defecation frequency in humans. 13 The diet histories provided in this study by caregivers (see Table 1 in supplementary material) did not provide sufficient detail to definitively determine how much the water or fiber content differed between the healthy cats and those with CKD, as many of the cats with CKD ate a variety of diets in rotation over the course of the study. As this may be a factor affecting defecation in these patients, future studies should ideally evaluate the impact of diet on defecation frequency. Theoretically, decreased oral caloric intake could lead to decreased defecation frequency, as appetite dysregulation is reported in cats with CKD.17,20 As daily diaries of caloric intake were not part of the study design, diet histories provided by caregivers did not allow caloric intake in individual animals to be calculated over the duration of the study. However, as a surrogate, each cat was monitored throughout the study for changes in body weight, and there was no significant difference between baseline body weight and completion body weights in enrolled cats. Therefore, it is unlikely that decreased caloric intake was a primary driver of the reduction in bowel movement frequency observed in this study.
A limitation of our study is that the CKD group was significantly older than the healthy group. A healthy age-matched group of cats was originally part of the study design but, because of the challenge of identifying such animals, this could not be carried out. Changes in gastrointestinal function occur in older cats, including decreased motility and nutrient assimilation.21,22 The presence of osteoarthritis may contribute to changes in motility in the older cat, and three of the cats with CKD were receiving therapeutics for osteoarthritis. Studies describing the clinical presentation of feline CKD include lethargy as a commonly reported clinical sign. 17 In humans, changes in physical activity are correlated with defecation frequency, where by decreased physical activity is linked to the development of constipation. 13 In addition, the link between kidney disease and decreased physical activity is well recognized in human patients with CKD, and is compounded by age-related decreases in physical activity.23,24 These factors have not been specifically evaluated in feline medicine, but there is a possibility that a similar correlation exists. Thus, both age and comorbidities such as osteoarthritis may potentially contribute to the defecation frequency reported.
Although a variety of factors may contribute, decreased defecation frequency was observed in cats with CKD, and this may have implications for gut–kidney health. The gut–kidney axis and associated dysbiosis noted in CKD is a rapidly expanding focus in both human and veterinary medicine.14,25,26 Experimental models of CKD and clinical studies in human patients with CKD link gut dysbiosis and uremic toxins (eg, indoxyl sulfate) to decreased gastrointestinal motility and constipation.27 –30 Reduced defecation frequency may exacerbate the production of uremic toxins because of prolonged protein fermentation and absorption.29,30 Uremic toxins, which accumulate in kidney disease, have multiple deleterious effects, including impaired gastrointestinal motility.27,28,31
Interestingly, caregivers of cats with CKD did not report any abnormalities with litter box habits or constipation at the time of enrollment. This is in contrast to previous research, which indicated that caregivers of cats with CKD were more aware of the frequency of their cats’ defecations than the caregivers of healthy cats. 1 The current study was not designed to determine whether this finding represents caregivers being unaware of a decreased defecation frequency or unaware of what constitutes a deviation from the normal frequency of bowel movements in healthy cats. 1 This underscores the importance of litter box monitoring and caregiver education regarding defecation frequency in cats with CKD.
Uncommonly, the litter box monitors had trouble distinguishing between cats of very similar body weights within the same household, a phenomenon that was observed in two households in our study. In addition, there was one cat with unusual litter box behaviors (jumping immediately out of the box after the elimination event and reaching back into the cover while standing outside the box) for which the AI technology appeared to have trouble classifying events. If implementing Petivity technology in either clinical practice or research, verification of events as recommended by the manufacturer is necessary for confidence in the results of the data output.
This study had several limitations. First, it was not possible to individually evaluate variables associated with CKD to better elucidate the mechanisms underlying constipation in affected cats. The study population was small, and additional information may be obtained from larger epidemiological studies on the association between CKD and constipation in cats. This study also relied upon caregiver observations to establish agreement between the Petivity technology and the cats’ real-time behaviors and elimination events. As such, there is always potential for human error during that process. However, this was minimized through standardized setup and verification processes. Although all cats were screened before inclusion, the screening was limited and did not rule out all secondary diseases in either population and their respective potential to influence results. Finally, one large, well-muscled 3-year-old male cat in the healthy group had a serum creatinine concentration of 1.6 mg/dl with adequately concentrated urine, so early CKD cannot be ruled out.
Conclusions
The litter box monitoring devices proved to be a reliable tool for tracking defecation and urination events in the majority of cats in this study. As such, the devices identified that cats with CKD defecated less frequently than healthy cats. Thus, this new technology may serve as a useful tool for caregivers to monitor their pets. The benefits of its use in cats with CKD include potential early detection, enabling timely intervention to help prevent overt constipation. In addition, this technology may facilitate further research into therapeutic interventions aimed at improving bowel movement frequency in affected cats.
Supplemental Material
Table 1
Type of diets fed to enrolled cats.
Footnotes
Acknowledgements
The authors wish to thank the caregivers and cats who participated in this study.
Author note
The results of this study were presented in part, as a research report, at the 2024 American College of Veterinary Internal Medicine Annual Forum.
Supplementary material
The following files are available as supplementary material:
Table 1: Type of diets fed to enrolled cats.
Conflict of interest
Jessica Quimby is an advisory board member for Nestlé Purina Petcare Company.
Funding
The study was supported in part by Buttons Fund for Feline CKD Research at The Ohio State University and by a NIH T35 training grant (T35OD010977). Petivity monitors were provided by Nestlé Purina Petcare Company.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals and procedures that differed from established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient. The study therefore had prior ethical approval from an established (or ad hoc) committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
