Abstract
Objectives
The objective of this prospective study was to compare the nociceptive blunting and analgesic efficacy of two different local anesthetic solutions (lidocaine and a mixture of lidocaine plus bupivacaine) and a placebo for cat castrations in a high-quality, high-volume spay/neuter (HQHVSN) clinic as an adjunct to standard HQHVSN anesthetic protocols.
Methods
A total of 88 healthy, shelter-owned male cats presenting to an HQHVSN clinic in the northeastern USA between August 2019 and October 2020 were enrolled in this blinded, randomized, placebo-controlled trial. Cats were randomized to receive an intratesticular injection of saline placebo (SP group; n = 32), lidocaine 1 mg/kg (L group; n = 34) or a mixture of lidocaine 1 mg/kg and bupivacaine 1 mg/kg (LB group; n = 22) before routine castration. Other than the use of randomized local anesthetic blocks, all standard HQHVSN protocols for anesthesia and surgery were followed. Measured variables included perioperative heart rates, surgeon satisfaction with anesthetic depth, surgeon satisfaction with tissue quality, postoperative pain scores and the rate of incisional complications.
Results
Treatment groups did not differ in demographic characteristics or baseline heart rate. Percent increase in intraoperative heart rate was significantly larger in cats in the SP group than in the L (P <0.001) and LB groups (P <0.001); the L and LB groups did not differ from each other (P = 0.457). Surgeon-reported unsatisfactory anesthetic depth occurred more frequently in the SP group (z = 2.68, P = 0.044). Surgeon satisfaction with tissue quality, postoperative pain scores and incision complication rates were statistically similar between groups.
Conclusions and relevance
This study is the first to assess the efficacy of local anesthesia for feline castration in an HQHVSN environment, where sustainable operations require delivering humane care with optimal efficiency and resource management. Despite the brief interval between intratesticular anesthetic administration and auto-ligation of the second testicle (median 5 mins, interquartile range 3–7), local anesthesia significantly reduced nociception compared with placebo and did not increase the incidence of incisional complications. These perioperative advantages support incorporating local blocks into multimodal anesthesia and analgesia protocols in HQHVSN clinics.
Plain language summary
The objective of this study was to compare the effectiveness of two different local anesthetic solutions (lidocaine and a mixture of lidocaine plus bupivacaine) and a placebo for cat castrations in a high-quality, high-volume spay/neuter (HQHVSN) clinic. A total of 88 healthy, shelter-owned male cats presenting to an HQHVSN clinic in the northeastern USA between August 2019 and October 2020 were enrolled. Cats were randomized to receive an intratesticular injection of saline placebo, lidocaine 1 mg/kg or mixture of lidocaine 1 mg/kg and bupivacaine 1 mg/kg before being neutered. Perioperative heart rates, surgeon satisfaction with anesthetic depth, surgeon satisfaction with tissue quality, postoperative pain scores and the rate of incisional complications were measured. The heart rate of cats in the placebo group increased significantly compared with those that received lidocaine or lidocaine/bupivacaine. Surgeons also reported unsatisfactory anesthetic depth more frequently in the placebo group. Surgeon satisfaction with tissue quality, postoperative pain scores and incision complication rates were statistically similar between groups. This is the first study to assess the value of local anesthesia for cat neuters in the HQHVSN environment where sustainable operations require delivering humane care with optimal efficiency and resource management. Local anesthesia was more effective than placebo and did not increase the incidence of incisional complications. Measured benefits support the use of local blocks in HQHVSN clinics.
Keywords
Introduction
Elective gonadectomy is the most common surgical procedure performed in veterinary medicine. 1 In the USA, approximately 53 million dogs and 47 million cats (69% and 80% of total populations, respectively) were reported to be spayed or castrated in 2016. 2 In many American states, gonadectomy is required before adoption of dog and cats from animal shelters. According to 24Pet ShelterWatch, between 2021 and 2024, approximately 91% of dogs and 94% of cats in reporting animal shelters in the USA were neutered before adoption. 3
The high-quality, high-volume spay/neuter (HQHVSN) model emerged to serve the surgical capacity of animal shelters, address pet overpopulation and provide affordable preventive care in the community.4 –6 This model depends on maximizing efficiency and minimizing costs, while ensuring humane care and animal wellbeing. 5 HQHVSN clinics rely heavily on protocoled anesthetic and surgical care for young healthy patients and routinely perform 35–50 surgeries per day per surgeon. 5
Multimodal pain management is the most effective means of controlling surgical pain. 7 Locoregional anesthetic blockade is an advantageous component of multimodal anesthesia/analgesia plans as these techniques interrupt transduction and transmission of nociception, have an extended duration of effect and are relatively low cost and easy to administer.8 –12 Local blocks are safe and effective in dogs13 –21 and cats22 –32 undergoing general anesthesia for elective gonadectomy. However, with few exceptions,23,24 these studies were performed in university or primary care settings, and it is unclear how findings apply to cat castrations performed in the HQHVSN environment.
HQHVSN settings commonly utilize combinations of injectable agents to provide multimodal anesthesia and analgesia. Protocols used across HQHVSN operations33 –35 often differ significantly from those in primary care settings. For cat castrations, injectable mixtures frequently serve as both induction and maintenance agents, sometimes supplemented by volatile anesthetics.5,6,33 Although opioids and non-steroidal anti-inflammatory drugs are frequently used for analgesia, local anesthesia is not often part of standard HQHVSN protocols.5,6 Adding local anesthetic blocks to a protocol increases patient preparation time, materials and labor costs as well as introducing the additional opportunity for error. 36 Considering the speed at which induction and patient preparation occur and cat castrations are performed, 5 the impact of local anesthesia via intratesticular block on patient care in the HQHVSN setting remains an important question.
Another question is which local anesthetic agent could be most effective for cat castrations in the HQHVSN context. In other settings, solo agents are commonly employed based on timing and duration of procedures: lidocaine has a faster onset and shorter duration of action than other local anesthetics (eg, bupivacaine and carbocaine).8 –10 Reports in the human37 –47 and veterinary24,25,48 –50 literature include combining lidocaine with other agents to gain the benefits of both (ie, a fast onset with a longer duration of action). Although recommendations vary as to the efficacy of this approach,10 –12 there is some evidence demonstrating a benefit to combining agents.25,46 –48
The objective of this prospective study was to compare nociceptive blunting and analgesic efficacy of two different local anesthetic solutions (lidocaine and a mixture of lidocaine plus bupivacaine) and a placebo for cat castrations in an HQHVSN clinic as an adjunct to standard HQHVSN anesthesia protocols. Comparative measures included perioperative heart rates, 30 surgeon satisfaction with anesthetic depth and tissue quality, and postoperative pain scores and incisional complications. 13
Materials and methods
All procedures were reviewed and approved by Cornell University’s Institutional Animal Care and Use Committee (#2019-0074).
Study population
Male cats presenting for elective castration at an HQHVSN clinic in the northeastern USA were assessed for enrollment in this blinded, randomized, placebo-controlled clinical trial. Eligibility criteria included cats that were: (1) shelter-owned and available for adoption or part of the trap–neuter–return (TNR) program between August 2019 and October 2020; (2) estimated age ⩾8 weeks and ⩽8 years as determined by veterinary visual examination of dentition; (3) body weight ⩾1.36 kg; (4) on no current medications other than preventatives; and (5) considered American Society of Anesthesiologists status I, with heart rate, respiratory rate and rectal temperature within normal intervals, a Purina Body Condition System score of 4–7/9 and no visual evidence of scrotal/testicular disease or coagulopathy on the basis of a pre-anesthetic physical examination by a staff veterinarian. Cats were not excluded from enrollment if they required an ancillary procedure after castration (eg, enucleation, aural polyp removal or dental) or if one inguinal cryptorchid testis was present.
Treatment groups and blinding
Cats were assigned via simple randomization to receive one of three intratesticular anesthetic treatments (A, B or C). One of two licensed veterinary technicians manually drew a letter from a bag to determine the treatment group for each cat, documented the assignment in the medical record and returned the letter to the bag to ensure a 1/3 probability for each cat being assigned to each group.
To maintain blinding of HQHVSN and study personnel, a university-based veterinary pharmacy repackaged and diluted each solution of local anesthetic to ensure equal and appropriate dosage of total solution per body weight. Solutions were drawn up aseptically and added to an empty sterile multi-use vial (Greer) in an ISO-7 class hood. The beyond use date was calculated by using 2019’s proposed US Pharmacopeia <797> guidelines for category 2 preparations made under sterile conditions (www.usp.org/compounding/general-chapter-797): 4 days at room temperature and 10 days under refrigeration. Solutions were constituted and labeled as follows: vial A (L group) contained one part lidocaine (20 mg/ml; Auromedics) to four parts 0.9% bacteriostatic sodium chloride (Hospira); vial B (SP group) contained 0.9% bacteriostatic sodium chloride (Hospira); vial C (LB group) contained one part lidocaine (20 mg/ml; Hospira) to four parts bupivacaine (5 mg/ml; Hospira). Unblinding to treatment group identity occurred after the completion of all data collection and analysis.
Anesthetic and surgical protocols
A brief physical examination was performed by a staff veterinarian up to 24 h before anesthesia. Body weight (in kg) and estimated age group (pediatric: aged <6 months; adult: aged ⩾6 months) were recorded. Anesthesia was induced and maintained following standard HQHVSN protocols. 6 Cats were induced with an intramuscular injection of a pre-mixed combination of tiletamine-zolazepam (2–3 mg/kg, Telazol; Zoetis), butorphanol (0.1–0.15 mg/kg, Torbugesic; Zoetis) and dexmedetomidine (0.005–0.007 mg/kg, Dexdomitor; Zoetis), commonly referred to as TTD. 34 The total amount of the pre-mixed combination was based on a protocolled table of dose volume per body weight rounded down to the nearest 0.5 kg. After induction to anesthesia, all cats received buprenorphine (0.02 mg/kg IM) and cats considered older than 4 months of age also received a subcutaneous injection of meloxicam (0.05–0.1 mg/kg, Metacam; Boehringer Ingelheim) for analgesia. Oxygen and isoflurane were available by mask.
The cat was placed in dorsal recumbency, and the surgical site was shaved and prepared with dilute chlorhexidine scrub and solution. The intratesticular injection was performed by one of two licensed veterinary technicians. The total dose volume per body weight (0.25 ml/kg) was drawn in a single sterile syringe from the assigned vial. Half of the volume was injected into each testis using a 25 G needle. The needle was directed to the caudal aspect of the testicle, aspirated to ensure seating and slowly retracted as solution was injected so that the subcutaneous portion of the caudal scrotum would also receive local anesthetic.15,30,51 The total volume of intratesticular injection received by each cat was recorded. Anesthetic depth, heart rate, mucous membrane color and blood oxygen saturation were monitored throughout the procedure. Standard closed castration was performed by an experienced HQHVSN staff veterinarian or a fourth-year veterinary student under direct supervision of a staff veterinarian. The time from intratesticular injection to the exteriorization and clamping of the second testis, and whether the castration was performed by a veterinarian or student, were recorded. Castration and data collection were completed before any ancillary procedures, with the exception of postoperative pain scores.
Perioperative evaluation of nociception and surgeon satisfaction
Heart rate was measured using a pulse oximeter with tongue probe (Surgivet V130) at baseline once the cat was under general anesthesia but before intratesticular injection (T0), and again during the surgical stimulus of the exteriorization of the second testis (T1).25,30,31 Surgeon satisfaction with anesthetic depth at the time of castration was assessed as yes/no: yes, anesthetic depth was effective for performing castration, or no, physical response to stimulus was observed. Surgeon satisfaction with tissue quality was assessed as yes/no: yes, neither edematous tissue nor hematoma was observed, or no, edematous tissue and/or hematoma was observed.
Assessment of postoperative pain and incisional complications
Postoperative data were collected by one of two trained student investigators. Postoperative pain was assessed with the Colorado State University Feline Acute Pain Scale (CSU-FAPS) 52 at 1, 3 and 24 h after castration. The CSU-FAPS provides behavioral, positional and palpation criteria to assess pain on a scale of 0 (minimal) to 4 (moderate to severe). Cats assessed with a score of 2 received evaluation and those with a score above 2 received rescue analgesia per standard protocol (buprenorphine 0.02 mg/kg IM). Data were dichotomized: below 2 and 2 or above. Incisions were evaluated for swelling and bruising using a previously published scale 13 at 1, 3 and 5 days after castration and were similarly dichotomized to present or absent.
Statistical analysis
A power analysis 53 was performed a priori to determine the minimum sample size to be enrolled in each treatment group to identify a clinically significant difference in intraoperative nociception as measured by heart rate. Using a significance level of alpha (α) = 0.05 and a medium effect size (d = 0.5), a sample size of 22 cats per group was calculated to achieve a power of 0.95. Data normality was evaluated using the Shapiro–Wilk test. Percent change in heart rate was calculated for each individual and averaged across treatment group: [(T1 – T0)/T0] × 100. Heart rate data were analyzed using ANOVA, followed by Bonferroni-corrected post-hoc tests for multiple comparisons. Kruskal–Wallis tests were used to determine if the treatment groups differed by body weight, time from intratesticular injection to the exteriorization of the second testis and total intratesticular injection volume received. Whether treatment groups differed by binary estimated age group classification (<6 months vs ⩾6 months) was assessed with a χ2 test. The Freeman–Halton extension of Fisher’s exact test (two-tailed) for 3 × 2 contingency tables, followed by Bonferroni-corrected post-hoc comparisons of adjusted residuals, was used to determine if treatment group was associated with outcome, ancillary procedures received, surgeon, additional anesthetic requirements, surgeon satisfaction scores, and postoperative pain and incision scores. P <0.05 was considered significant. Statistical analyses were performed using commercially available software (Prism 10; GraphPad Software).
Results
A total of 88 shelter-owned male cats presenting for elective castration at an HQHVSN clinic were enrolled in the study. Of the study population, age was estimated at less than 6 months for 49 (56%) cats and 6 months or older for 39 (44%) cats. Median body weight was 2.3 kg (interquartile range [IQR] 1.6–3.7). Staff veterinarians performed 79 (90%) castrations, while supervised fourth-year veterinary students performed nine (10%) castrations. In total, 85 (97%) cats were adopted and three (3%) were part of the TNR program. Eight (9%) cats received ancillary procedures: two unilateral inguinal cryptorchid neuters, two unilateral enucleations, three dental procedures and one aural polyp were performed. The median total volume of intratesticular injection received was 0.5 ml (IQR 0.4–1). The median time from intratesticular injection to exteriorization of the second testis was 5 mins (IQR 3–7).
Cats were randomized to receive an intratesticular injection of saline placebo (SP group; n = 32), lidocaine (L group; n = 34) or lidocaine plus bupivacaine mixture (LB group; n = 22) before routine castration (Table 1; Figure 1). Treatment groups did not differ significantly based on body weight or age group composition (all P >0.05) (Table 1).
Descriptive characteristics of cats administered intratesticular saline placebo (SP group), lidocaine (L group) or lidocaine plus bupivacaine mixture (LB group) before routine castration in a high-quality, high-volume spay/neuter clinic

Data are n (%) or median (interquartile range). P values were calculated via Kruskal–Wallis test or χ2 test

Flow diagram of subject enrollment, allocation and data collection/analysis in a randomized clinical trial of local anesthetic blocks and a placebo in cat castrations in a high-quality, high-volume spay/neuter setting. CSU = Colorado State University; group L = lidocaine; group LB = lidocaine plus bupivacaine mixture; group SP = saline placebo; TNR = trap–neuter–return
Treatment groups also did not differ significantly based on total volume of intratesticular injection received, time from intratesticular injection to the exteriorization of the second testis, surgeon type, outcome type or frequency of cats receiving ancillary procedures at baseline (all P >0.05) (Table 2).
Perioperative measures, procedural information and outcomes for cats administered intratesticular saline placebo (SP group), lidocaine (L group) or lidocaine plus bupivacaine mixture (LB group) before routine castration in a high-quality, high-volume spay/neuter clinic
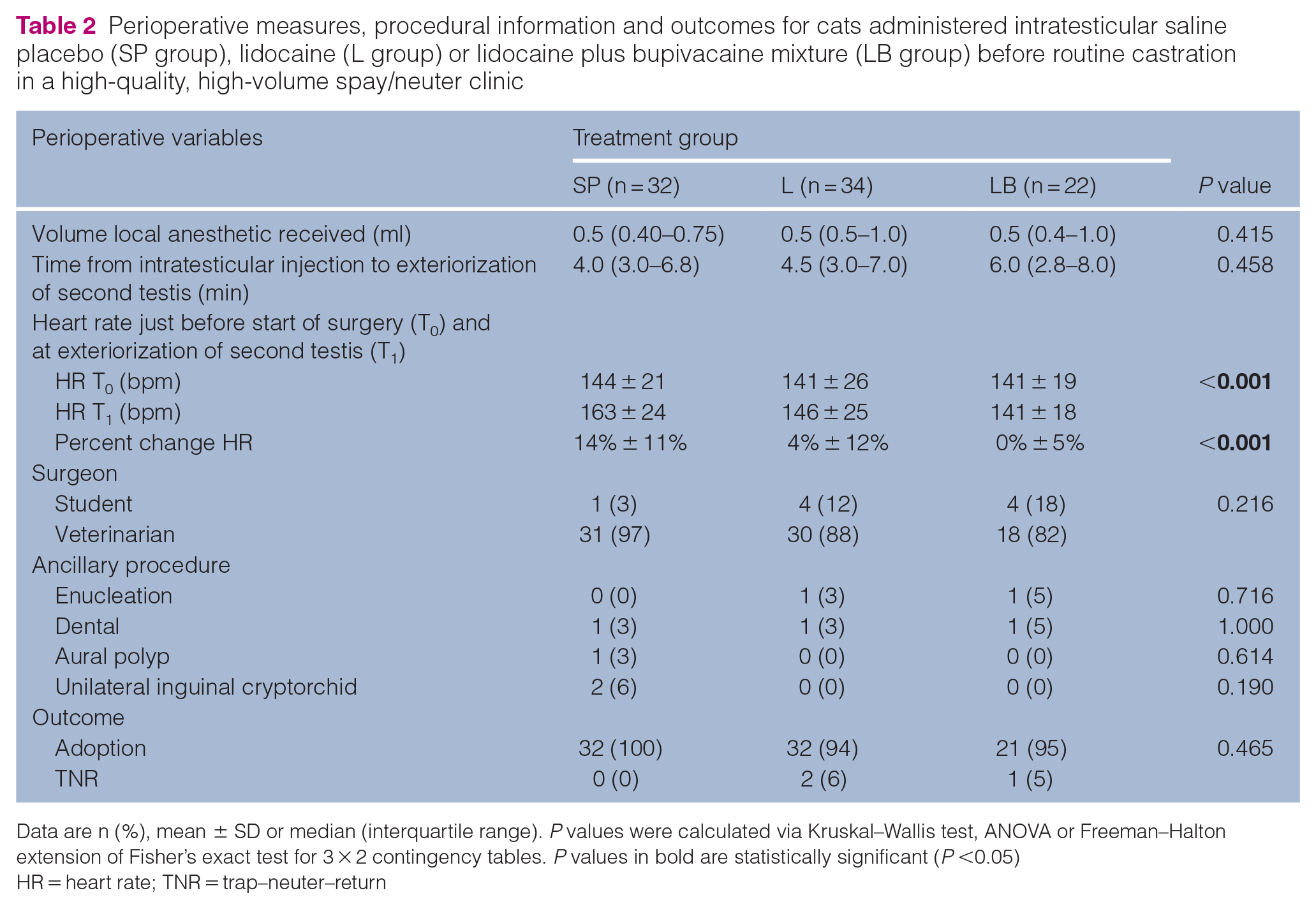
Data are n (%), mean ± SD or median (interquartile range). P values were calculated via Kruskal–Wallis test, ANOVA or Freeman–Halton extension of Fisher’s exact test for 3 × 2 contingency tables. P values in bold are statistically significant (P <0.05)
HR = heart rate; TNR = trap–neuter–return
Heart rate was measured at baseline (T0) and again during surgical stimulus (T1) (Table 2). Two cats from the LB group were excluded from analysis (Figure 1). The percent change in intraoperative heart rate was calculated for each cat and averaged across the treatment group (Table 2). A one-way ANOVA revealed a significant effect of the treatment group (F [2, 83] = 12.59; P <0.001). Bonferroni-corrected post-hoc comparisons showed that the percent increase in intraoperative heart rate was significantly larger in cats in the SP group than the L (P<0.001) and LB groups (P<0.001), while the L and LB groups did not differ (P = 0.457). This difference was consistent even when analysis was restricted to only cats with adoption as their outcome, neutered by a veterinarian and requiring no ancillary procedures (SP group: n = 27; L group: n = 26; LB group: n = 14). A one-way ANOVA showed a significant effect of the treatment group (F [2, 64] = 8.31; P <0.001). Bonferroni-correct post-hoc comparisons confirmed that the SP group (mean ± SD: 13% ± 11%) exhibited a significantly larger increase in intraoperative heart rate compared with the L (mean ± SD: 5% ± 14%; P = 0.024) and LB groups (mean ± SD: –1% ± 5%; P <0.001); the L and LB groups did not differ (P = 0.238).
Treatment group was not significantly associated with surgeon-reported unsatisfactory tissue quality (all P >0.05); however, the association between treatment group and surgeon-reported satisfaction with anesthetic depth at the time of castration was significant (P = 0.017) (Table 3). To identify specific differences, adjusted residuals were examined for each cell. Bonferroni-corrected post-hoc comparisons of adjusted residuals revealed that the frequency of cats in the SP group reported to have ‘unsatisfactory’ anesthetic depth was significantly greater than expected by chance (z = 2.68, P = 0.04) if the null hypothesis of no difference across treatment groups was true.
Results of surgeon satisfaction survey of cats administered intratesticular saline placebo (SP group), lidocaine (L group) or lidocaine plus bupivacaine mixture (LB group) before routine castration in a high-quality, high-volume spay/neuter clinic

Data are n (%). P values were calculated via Freeman–Halton extension of Fisher’s exact test for 3 × 2 contingency tables. P values in bold are statistically significant (P <0.05)
The results of postoperative pain and incision assessments revealed no significant associations between treatment group and pain score of 2 and above or swelling or bruising scores of 2 and above (all P >0.05) (Table 4). Cats were excluded from this analysis if they received an ancillary procedure or if they had been adopted or returned to field at the time of assessment (Figure 1). The CSU-FAPS was administered 1, 3 and 24 h after castration, and incision sites were evaluated for swelling and bruising 1, 3 and 5 days after castration.
Postoperative Colorado State University Feline Acute Pain Scale (CSU-FAPS) and incision site assessments in cats administered intratesticular saline placebo (SP group), lidocaine (L group) or lidocaine plus bupivacaine mixture (LB group) before routine castration in a high-quality, high-volume spay/neuter clinic

Data are n (%). Denominators differ as subjects were lost to follow-up postoperatively. P values were calculated via Freeman–Halton extension of Fisher’s exact test for 3 × 2 contingency tables
Discussion
Previous research in university and private practice settings has demonstrated benefits to incorporating local anesthesia into a multimodal analgesia plan for castrations, including decreased nociception,15,30 reduced requirements for inhalants 18 and intra- and postoperative analgesics, 21 and decreased postoperative pain.18,21 The present findings extend the benefits of intratesticular block into the HQHVSN environment. Despite the short duration between intratesticular injection to near completion of castration (5 mins, IQR 3–7), local anesthesia was effective at decreasing nociception and increasing surgeon satisfaction with anesthetic depth compared with saline placebo. Lidocaine infiltrates the spermatic cord within 3 mins of intratesticular injection in piglets, 54 but this speed is not known in cats. Here, both local anesthetic solutions blocked the heart rate response to surgical stimulus still exhibited by the placebo group, which suggests the onset of action of both lidocaine and lidocaine plus bupivacaine mixtures were fast enough to be efficacious in the HQHVSN setting.
Overall, study observations reflected the success of the HQHVSN anesthesia/analgesia standard protocols in ameliorating nociception and surgical pain peri- and postoperatively. Mild-to-moderate postoperative pain was rare, with CSU-FAPS scores of 2 and above observed in 3/75 (4%) individuals (L group, n = 1; LB group, n = 2). Only one (1%) cat (LB group) required postoperative rescue analgesia. Although this study was well-powered to detect the difference in perioperative heart rate between the placebo and local anesthetic groups, it was underpowered to detect treatment-related differences in the frequency of rare events.
HQHVSN operations emphasize protocol-driven anesthetic and surgical care to optimize efficiency without sacrificing animal welfare. Foregoing nociceptive and analgesic benefits, a potential concern of including local anesthetics in HQHVSN settings is the increased incidence of incision site reactions. Mild-to-moderate incisional swelling and/or bruising were observed in 4/75 (5%) cats on the first postoperative day, and seven cats in total across all assessments (SP group, n = 3; L group, n = 2; LB group, n = 2). However, interpretation of incision site data collected after postoperative day 1 is severely limited by unavoidable losses to adoption placement and the TNR program (Figure 1). Although this study demonstrated a statistical similarity in the frequency of mild-to-moderate incisional complications in cats treated with local anesthesia compared with placebo, future studies should be designed with enough power to resolve clinically relevant differences in the frequency of postoperative complications associated with intratesticular blocks.
Incorporating local anesthesia into HQHVSN protocols must take into account resource allocation as well as safety and efficacy. At the study location, prices for lidocaine and lidocaine plus bupivacaine mixture were estimated to be US$0.03 and US$0.15, respectively, for a 4 kg cat. Two minutes of a technician’s labor to prepare and administer the injection adds US$0.84 and materials costs are estimated at US$0.20. A total cost of US$1.07–$1.19 per cat is nominal, but across a busy clinic with 30 feline castrations per day, these increased materials and labor costs amount to approximately US$35.00 and require an additional hour of a technician’s time. Even small changes can impact the financial sustainability of HQHVSN clinics; therefore, benefits must balance costs.
This study was primarily designed to test the efficacy of local anesthesia in the fast-paced context of HQHVSN using standard anesthetic protocols. One limitation of this study is the teaching nature of this clinic could have contributed to a longer patient preparation time to surgical procedure than non-teaching or simply more efficient HQHVSN environments. This variable could impact applicability of the results to HQHVSN settings with faster clinic flow. Another potential limitation is this study relied upon assessments of intraoperative nociception (heart rate measured by pulse oximetry) and postoperative pain (CSU-FAPS), which were part of standard clinical practice in this shelter and many other HQHVSN programs. In a controlled research environment, other methods could be used to improve the objectivity, accuracy and reliability of these measurements, including mechanical nociceptive thresholds 55 and more frequent assessments of pain scores. Finally, the design lacked comparison to a sham group, which can be useful for determining if the intratesticular injection itself increases the risk of harm. 56 A sham treatment group for postoperative incisional evaluation could have been introduced to glean mechanical impacts of intratesticular injections vs scrotal trauma due to surgery. Although edema and hematomas are possible after intratesticular injection of local anesthetic or placebo, surgeons reported unsatisfactory tissue quality at the time of castration in just 3/88 (3%) cats. This low frequency of acute complications suggests a sham treatment group would not have added much value to this study design.
Conclusions
Local anesthesia with lidocaine or a lidocaine plus bupivacaine mixture was effective at decreasing nociception in cats castrated in an HQHVSN setting and did not increase the frequency of incisional complications. These results add to the evidence supporting the inclusion of local blocks in HQHVSN protocols. At an additional cost of US$1.07–US$1.19 per cat, intratesticular blocks are a resource-conscious adjunct to multimodal anesthesia and analgesia protocols in the HQHVSN setting.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors are grateful to PetSmart Charities for directly supporting this project through a Veterinary Scholars Award to Scarlett Lutz-Boulting in 2019. They are also grateful to Maddie’s Fund and Janet L Swanson for their ongoing support of the Maddie’s Shelter Medicine Program at Cornell and without whom none of this work would be possible. Funding for the publication of this study was provided by the American Society for the Prevention of Cruelty to Animals (ASPCA) Open-Access Publishing Fund.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals and procedures that differed from established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient. The study therefore had prior ethical approval from an established (or ad hoc) committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
