Abstract
Objectives
The goal of this study was to determine whether a drug combination using nalbuphine with dexmedetomidine and tiletamine/zolazepam is non-inferior to one that uses butorphanol.
Methods
All healthy cats presenting solely for gonadectomy to two trap–neuter–return mobile clinic days were randomly assigned to induction with a combination of tiletamine/zolazepam 3 mg/kg, dexmedetomidine 7.5 µg/kg and either butorphanol or nalbuphine at 0.15 mg/kg. All participants were blinded to the identity of the combinations. The primary endpoint was clinician satisfaction, comprised of the mean of four satisfaction ratings on a 7-point Likert scale (highly dissatisfied through to highly satisfied) recorded for induction, maintenance of anesthesia, surgery and recovery. Exploratory endpoints included each individual score, number of injections, duration of induction, duration of recovery and need for reversal agent. To assess non-inferiority for the primary endpoint and individual scores, the difference and 95% confidence intervals (CIs) of the difference between the mean clinical scores for the nalbuphine and butorphanol-based combinations were calculated and compared with a prespecified non-inferiority margin of 20% (1.4 points).
Results
Seventy-two cats were enrolled, 36 in each group. The mean ± SD composite score for the combination with nalbuphine was 6.06 ± 0.59 (95% CI 5.86–6.25) points, while the combination with butorphanol was 6.22 ± 0.62 (95% CI 6.01–6.43). The difference between mean scores was 0.17 (–0.12 to 0.45), which did not exceed the prespecified boundary of 1.4, establishing the non-inferiority of nalbuphine. No individual clinical score for nalbuphine was inferior to butorphanol, and there were no significant differences for any secondary endpoints.
Conclusions and relevance
The clinical experience of the nalbuphine-based combination was non-inferior to the butorphanol-based combination. Nalbuphine is an effective substitute for butorphanol, providing another option if butorphanol is unavailable due to shortage, controlled status or cost, without requiring a change in anesthetic workflow.
Keywords
Introduction
Pre-mixed combinations of an opioid, dissociative and alpha2 agonist (popularly known as ‘kitty magic’) are commonly used for intramuscular (IM) induction of anesthesia, particularly in facilities that process large numbers of animals, including: animal shelters; high-quality, high-volume spay–neuter (HQHVSN) clinics; field clinics; and facilities that perform trap–neuter–return (TNR) of community cats.1–6 A commonly used combination (coined ‘TTD’ based on the trade names of the drugs [Telazol, Torbugestic and Dexdomitor; Zoetis]) uses the α2 agonist dexmedetomidine, the dissociative and benzodiazepine tiletamine/zolazepam, and the opioid butorphanol. The combination has a wide range of safety, good efficacy, small volume of administration and consistent duration of onset and recovery. 1 However, two of the three components, butorphanol (schedule IV) and tiletamine/zolazepam (schedule III), are controlled substances, which have enhanced requirements for secure storage and logging. There are also restrictions applied to international transport that affect the ability to stock anesthetic drugs for international field clinics.
In addition to the restrictions surrounding controlled substances, many medications, particularly opioids, have experienced shortages and dramatic price swings in the past few years. For example, fentanyl, hydromorphone and morphine are opioids recently seen on the US Food and Drugs Administration’s drug shortages list. 7 Opioids are an important part of perioperative pain management and surgery, as they allow veterinarians to decrease the amount of inhalant anesthetic used and potentiate the action of other anesthetic drugs, thereby decreasing the amount of other drugs required. As supply decreases, practices and shelters must pay more for opioids, which can undergo extreme price increases. 8 Owing to the volume of surgery performed at facilities that commonly employ these protocols, even relatively small increases in the price of medications have a significant effect on costs when multiplied by the number of patients. While there are many drug protocols that are appropriate for the shelter or field setting, not all share the advantages of TTD. This may be particularly true for environments that use standardized protocols to safely process large volumes of animals.
Nalbuphine, an opioid analgesic that is not controlled, can be used as a direct substitute for butorphanol.1,9 Nalbuphine and butorphanol are both agonists of the kappa opioid receptor and are mu receptor antagonists. In humans, their pharmacokinetics are considered to be similar, with a rapid onset of <20 mins, peak effects within 30–60 mins and a duration of action of 3–6 h. 10 However, the one prior study in cats reported nalbuphine to have a shorter duration of action than butorphanol. 11
Butorphanol has been extensively investigated as a safe and effective anesthetic component in cats.12–17 To our knowledge, only one study has previously examined nalbuphine as a single-agent analgesic in cats, with no studies having been performed using it as part of multimodal anesthesia. 11 In wildlife species, nalbuphine has been shown to be as effective as butorphanol in premixed anesthetic combinations.18,19 However, a recent study in dogs found nalbuphine to cause less sedation than butorphanol, both alone and when combined with acepromazine. 20
While both nalbuphine and butorphanol exhibit a ceiling effect for respiratory depression, only butorphanol’s duration of respiratory depression correlates with dose. 21 In dogs, when combined with dexmedetomidine, there was no significant difference in respiratory rate, mean arterial blood pressure or heart rate with butorphanol as compared with nalbuphine. In addition, both drugs provided antinociceptive effects equivalent to morphine and methadone. 22
Ideally, no change in clinician satisfaction with the anesthetic protocol or workflow should occur if nalbuphine is used to replace butorphanol. The primary goal of this study was to determine whether an anesthetic protocol that uses nalbuphine in combination with dexmedetomidine and tiletamine/zolazepam is as good as one that uses butorphanol from the clinical experience in an environment where this protocol is commonly employed, with non-inferiority being declared if the mean difference between satisfaction scores did not exceed the margin of 1.4 points (20%). As there are no prior studies to rely upon for determining a threshold, the margin was defined at 20% based on clinical judgement, as has been accepted before for comparing groups when there is no prior evidence.23,24
The secondary goal of this study was to provide more detailed information (individual score, the number of injections required to render animals non-responsive to stimuli, time from induction of anesthesia to recording of post-induction vitals, the time from end of surgery to return to trap and the need for reversal agent during recovery) about the clinical experience during critical steps of the anesthetic workflow using these combinations.
Materials and methods
Animal and study design
This study was a randomized non-inferiority trial approved by the Midwestern University Institutional Animal Care and Use Committee. A power analysis that evaluated a one-sided two-sample comparison of means with α set to 0.025, power to 0.9, clinically meaningful difference of 20% (1.4 points on a 7-point scale) and an assumed SD of 1.5 points on a 7-point scale determined that a minimum of 50 cats, 25 in each group, would be required. This study is reported in accordance with the revised CONSORT statement. 25
Free-roaming community cats presented for TNR over the course of 2 days at a mobile clinic were recruited for the study. Cats were eligible for inclusion if they were intact, older than 6 weeks, required no procedure under anesthesia other than gonadectomy and appeared healthy for surgery. Prior to induction, cats were given a visual examination to assess their health status, which included an estimate of their weight by an experienced technician.
The primary endpoint was clinician satisfaction, comprised of the mean of four satisfaction ratings on a 7-point Likert scale (highly dissatisfied through to highly satisfied) recorded by the technician who induced anesthesia, the surgeon who performed the surgery, the anesthetist who monitored anesthesia and the technician who recovered the animal. Specifically, the scoring was 1 (highly dissatisfied), 2 (dissatisfied), 3 (somewhat dissatisfied), 4 (undecided), 5 (somewhat satisfied), 6 (satisfied) and 7 (highly satisfied). Scoring was guided by a rubric (Table 1). All raters remained the same for the course of the study. Secondary exploratory endpoints included each individual score, the number of injections required to render animals non-responsive to stimuli (injections), time from injection of anesthesia drugs to recording of post-induction vitals (duration of induction), the time from end of surgery to return to trap (duration of recovery), and the need for reversal agent during recovery (reversal).
Rubric for scoring each station

Scores correspond to a 7-point Likert scale, where 1 is highly dissatisfied and 7 is highly satisfied
Randomization and blinding procedures
Two anesthetic drug combinations were created by reconstituting tiletamine/zolazepam to a concentration of 5 mg/ml using 2.5 ml dexmedetomidine at 0.5 mg/ml and 2.5 ml of either nalbuphine (‘A’) or butorphanol (‘B’) at 10 mg/ml. Cats in traps that were covered with trap covers were randomly assigned to protocol A or B using a two-block block randomization plan generated by using a website (www.randomization.com). The technician administering the medication, surgeon, anesthetist and technician assisting with recovery were all blinded to the identity of combination A and B. The technicians, surgeon and anesthetist remained consistent throughout the study. The drug combinations were indistinguishable based on appearance, dose, volume and other grossly apparent properties.
Induction workflow
Cats were given the protocol via the IM route through the trap using an insulin syringe at a dose of 3 mg/kg tiletamine/zolazepam, 7.5 µg/kg dexmedetomidine and either butorphanol or nalbuphine at 0.15 mg/kg. The dose was determined via a pre-filled chart common to both combinations and was based on the estimated weight. The dose, time of injection, route and protocol were recorded. After induction, traps were partially covered to reduce stimulation while allowing for visual monitoring of the patient.
Once cats were unresponsive to stimuli, they were removed from the trap for a full physical examination, measurement of their actual weight, estimate of their age, placement of a supraglottic airway control device in the oropharynx (V-Gel docsinnovent), eye lubrication and shaving of the surgical site. The bladders of female cats were expressed.
Cats that remained responsive after 10 mins were administered another injection at one-half to one-quarter of the original dose either intramuscularly or intravenously, depending on the level of sedation achieved from the first dose, as determined by the supervising clinician. This was repeated up to a maximum of four injections. The technician who induced anesthesia scored the induction experience based on their satisfaction with the efficiency and smoothness of the induction process.
Surgical and anesthesia maintenance workflow
Cats were transferred to the surgical table and connected to a non-rebreathing circuit. Oxygen was set at 2 l/min. Male cats were maintained solely on oxygen unless they required isoflurane inhalant anesthesia to remain at a surgical plane. Female cats were maintained at 1.5% isoflurane unless the patient required more or less based on anesthetic plane as measured by the anesthetist using common measures of anesthetic depth such as palpebral reflex, eye position, jaw tone and monitored cardiopulmonary parameters. Patients were monitored via capnography (EMMA Capnograph; Masimo) and pulse oximetry (Rad-87 pulse oximeter; Masimo), with heart and respiratory rates recorded every 5 mins until the patient was moved to recovery. The start and end times of surgery were recorded. The anesthetist scored their satisfaction with the maintenance of anesthesia based on the amount of effort required to safely maintain the patient at a surgical plane of anesthesia.
Surgical sites were aseptically prepared. Orchiectomies were performed closed using auto-ligation of the testicular pedicle and with the scrotal incision left open to heal by second intention. Ovariohysterectomies (OVHs) of non-gravid females were performed with auto-ligation of the ovarian pedicle, a single miller’s knot on the uterine body, and closure of the body wall with a cruciate and purse string intradermal suture. The procedure was modified for gravid females to close the body wall with simple continuous pattern, stick tie of the uterine arteries and use of a modified Colorado pattern in the subcutaneous tissue and skin. The surgeon scored their satisfaction based on their ability to perform surgery without compromise due to response of the patient to surgical stimulation. Robenacoxib at 2 mg/kg (cats >4 months of age) or buprenorphine at 0.02 mg/kg (cats 4 months of age and younger) to control postoperative pain, ear tipping of the left ear, tattoo and vaccines were administered prior to transfer to recovery.
Postoperative recovery workflow
The cat’s temperature was continually monitored during the recovery process and a patient warming system was used for temperature support (Hot Dog Patient Warmer; Augustine Biomedical). Cats were stimulated by flipping every 3 mins. Supraglottic devices were removed once cats were able to swallow, and the time of removal recorded. Once cats were able to pick up their head they were moved back to their cleaned trap, the time recorded, and transferred off the mobile clinic for further monitoring, which was beyond the scope of the study.
If the patient had a decreasing body temperature, and/or lack of palpebral response by 10 mins after movement to recovery, atipamezole 5 mg/ml was administered intramuscularly via an insulin syringe at one-third the total volume of the anesthetic combination to reverse the dexmedetomidine. The recovery technician scored the cat based on the efficiency of the recovery process, presence of dysphoria and whether they required reversal.
The analgesic rescue protocol consisted of two steps. In the case of a cat showing postoperative signs of distress upon resumption of consciousness, cats would be given an IM injection of low-dose dexmedetomidine (0.003 mg/kg). If distress resumed after 15 mins (distress would be interpreted as postoperative pain), rescue analgesia of buprenorphine (0.03 mg/kg) would be administered intramuscularly.
Statistical analysis
To assess non-inferiority for the primary endpoint and individual satisfaction scores, the mean for each combination was determined, and then the difference between the means and 95% confidence interval (CI) around the difference was calculated. The 95% CI of the difference was compared to a prespecified non-inferiority margin (estimated clinically meaningful difference) of 20% (1.4 points) to determine if the CI crossed the non-inferiority margin. Continuous and ordinal secondary exploratory endpoints and baseline demographics were assessed using the Wilcoxon rank-sum test. Normality was determined using the Shapiro–Wilk test for normality. The proportions of binary secondary exploratory endpoints and baseline demographics were assessed using a two-sample z-test, while non-normal continuous variables were assessed using a two-sample Wilcoxon rank-sum test. The difference between the number of induction injections was assessed using Poisson regression and the influence of the difference in weight estimation vs actual weight on the composite satisfaction score assessed using linear regression. Significance was set at P <0.05 for all tests.
Results
Baseline and demographic data
Of the 78 cats presented to the two TNR days, 72 (butorphanol n = 36; nalbuphine n = 36) met the inclusion criteria. Two were too young, one required an enucleation in addition to OVH and three were found to be previously sterilized. There were no significant differences between groups for the baseline data (Table 2). The difference in weight between the estimate and the actual was not significantly different between groups. One cat in the nalbuphine group was found to be pregnant. No cats required rescue analgesia.
Summary of demographic and baseline data and their statistical comparison via the Wilcoxon rank-sum test
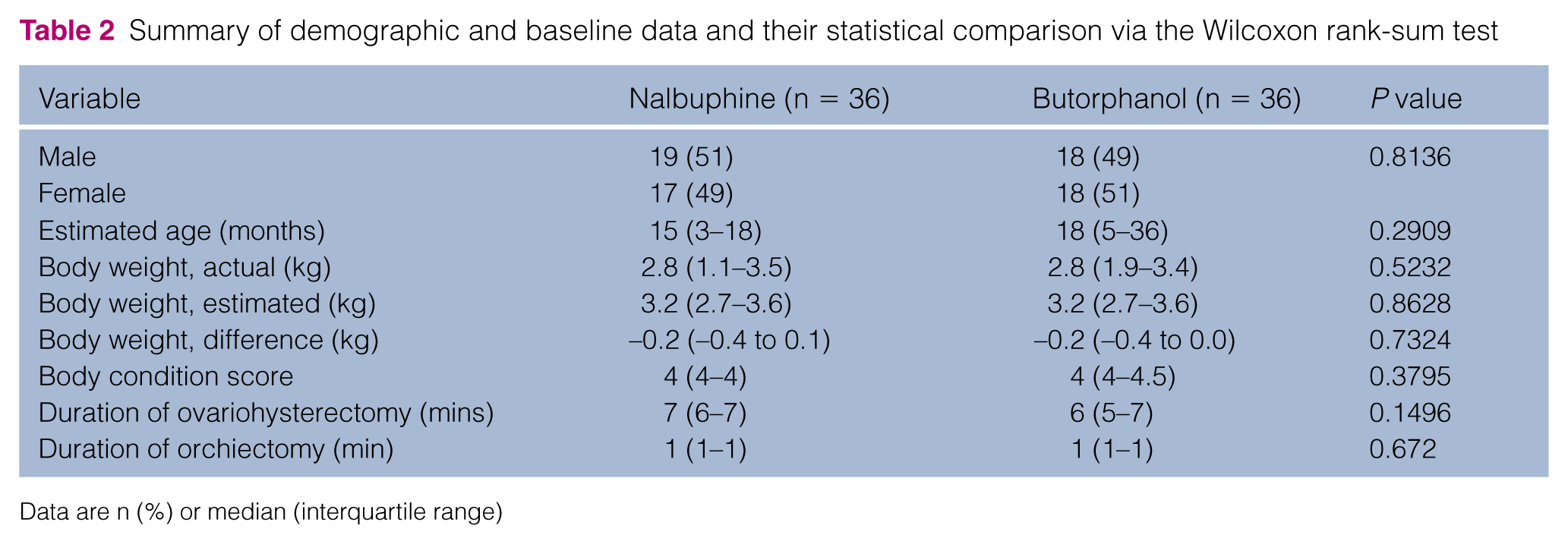
Data are n (%) or median (interquartile range)
Endpoints
The mean ± SD composite score for the protocol with nalbuphine was 6.06 ± 0.59 points (95%; 5.86–6.25), while the score for the protocol with butorphanol was 6.22 ± 0.62 points (95% CI; 6.01–6.43). The difference between mean scores was 0.17 (95% CI; 0.12–0.45), which did not cross the specified boundary of 1.4 (Figure 1). Mean scores were 5.72 ± 1.70 vs 6.31 ± 1.12 for induction, 6.72 ± 0.74 vs 6.83 ± 0.56 for surgery, 5.97 ± 1.08 vs 6.25 ± 0.91 for anesthesia and 5.81 ± 1.55 vs 5.50 ± 1.76 for recovery for nalbuphine and butorphanol, respectively. The difference between the mean for each of these individual satisfaction scores also did not cross the specified boundary of 1.4 (Figure 2).
The mean total volume per kg of actual weight for cats induced with either combination was 0.03 ± 0.01 ml. Of the cats administered nalbuphine, 42% (95% CI 26, 59) required more than one injection (median 1, interquartile range [IQR] 2–1), and 31% (95% CI 16, 48) required reversal. Of the cats administered butorphanol, 28% (95% CI 14, 45) required more than one injection (median = 1, IQR 2–1) and 42% (95% CI 26, 59) required reversal. Number of injections, total dose and the proportion of cats requiring reversal were not significantly different between groups.
Mean duration was 10.2 ± 6.8 vs 8.8 ± 5.6 mins for induction and 16.1 ± 5.1 vs 17.1 ± 6.7 mins for recovery for nalbuphine and butorphanol, respectively. Buprenorphine was not significantly associated with the duration of recovery. Neither the duration of induction nor the duration of recovery were significantly different between groups.

Graph of the 95% confidence interval for the difference in the mean of the composite satisfaction score (mean of the 7-point induction, anesthesia, surgery and recovery scores) of the nalbuphine-based combination as compared with the butorphanol-based combination. Scores correspond to a 7-point Likert scale, where 1 is highly dissatisfied and 7 is highly satisfied. The predefined clinically meaningful difference was set at −1.4 points (20%)

Graph of the 95% confidence interval for the differences of the means of the 7-point satisfaction scores of a nalbuphine-based combination as compared with a butorphanol-based combination for the secondary endpoints. (a) Induction score; (b) anesthesia score; (c) surgery score; (d) recovery score. Scores correspond to a 7-point Likert scale, where 1 is highly dissatisfied and 7 is highly satisfied. The predefined clinically meaningful difference was set at −1.4 points (20%)
Discussion
The principal finding of the present study was that use of nalbuphine as a substitute for butorphanol in combination with dexmedetomidine and tiletamine/zolazepam was a non-inferior clinical experience. Non-inferiority held true for all aspects of the perioperative period (induction, surgery, anesthesia, recovery) and thus nalbuphine is a reasonable substitute for butorphanol in this common anesthetic protocol. Additionally, the time to induction, the number of injections required, time to recovery, and number of cats requiring reversal were all found to be non-significant between the two groups.
The scoring system used for the current study was based on clinician satisfaction and did not directly measure patient satisfaction between the two anesthetic combinations. In humans, it is common to directly assess patient satisfaction; however, in veterinary medicine, this is a limitation, in which direct assessment of patient satisfaction is not possible. Patient experience in the present study was indirectly assessed by characterizing the number of injections required, which can be a stressful experience for the patient. The supervising clinician dictated the route of the injections, which could introduce potential bias, although this method most closely mimics clinical practice and was largely influenced by the level of sedation obtained from the initial injection. Additionally, patient experience was assessed by the outward expression of the quality of recovery (dysphoria or need for rescue analgesia). As none of these metrics was significantly different between the two groups, the patient experience indirectly appears to be similar.
While very limited study with nalbuphine has been performed in cats, at least in humans, it has been found that both nalbuphine and butorphanol are rapidly absorbed from the IM route and have a similar pharmacokinetic profile.9,10 These are likely the factors as to why the time to induction and number of injections required were not found to be significantly different between the two groups of cats in the present study. Additionally, the sedative and adverse effects of nalbuphine and butorphanol are similar in humans, 9 which may explain the non-significance of the time to recovery and number of cats requiring reversal between the two groups. However, one study in cats showed no sedative or behavioral effects with nalbuphine, while butorphanol administration had occasional sedative or behavioral effects. 11 These negative effects were primarily only seen at higher doses of butorphanol (0.4–0.8 mg/kg) and not at lower doses (0.1–0.2 mg/kg) that more closely mimic those in the present study, which is likely why it was not noted here.
While standard practice with feral cats and wildlife, one potential limitation of the study is that the anesthetic dose was based on estimated weights due to the inability to safely handle the patient prior to induction. This necessitates the use of a lower dose as it must be safe if inadvertently administered intravenously. However, despite the need for weight estimation, the difference between the estimated and actual weights had no relationship with the composite clinical satisfaction score.
Analgesic studies in companion animals are the most commonly identified non-inferiority trials in veterinary medicine, likely owing to the ethical concerns regarding withholding pain medication with a placebo when many effective analgesics currently exist. 26 One of the most challenging aspects of non-inferiority trials is the appropriate designation of the non-inferiority margin with most studies having no justification of their margin selection. 25 The inferiority margin in the present study of 20% was determined based on clinical judgement, as no established margin exists. To our knowledge, no veterinary study has employed a concept of acceptability/smoothness of anesthesia to compare protocols before and thus the rubric used in this study has not been previously validated. However, the use of ratings scales for assessing provider satisfaction with drug protocols has previously been employed in human medicine.27,28 The rubric was designed to mimic the typical HQHVSN clinic, in which the level of vigilance is relative to the anticipated clinical scenario where multiple surgical tables may be monitored by a single technician or a single technician in the same room tasked with other duties in concert with audible monitoring equipment. However, secondary endpoints were provided so practitioners can make their own determination as to what is clinically relevant for their workflow, as all clinics have unique resources and rate-limiting factors.
Conclusions
The clinical satisfaction throughout the perioperative period (induction and maintenance of anesthesia, surgery and recovery) when using dexmedetomidine and tiletamine/zolazepam combined with nalbuphine was non-inferior to that of the butorphanol-based combination. Nalbuphine is an effective substitute for butorphanol in this combination for routine gonadectomy in cats. This provides another option in the event that butorphanol is unavailable owing to shortage, controlled status or cost, without requiring a change in anesthetic workflow.
Footnotes
Acknowledgements
We thank Jamie Thomas for her assistance with generating the induction score.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding provided by Midwestern University startup funds (Kreisler).
