Abstract
Objectives
The aim of the present study was to evaluate the feasibility and safety of percutaneous ultrasound- and fluoroscopy-guided cholecystostomy drain placement.
Methods
An experimental cadaveric study was conducted on 16 cat cadavers weighing between 2.5 and 6.4 kg. Two drain systems were tested for percutaneous ultrasound- and fluoroscopy-guided placement: the nephrostomy component of a subcutaneous urethral bypass system (SUB-nephrostomy drain) and a paediatric percutaneous access set (paediatric-nephrostomy drain). Ultrasound-guided cholecystocentesis was performed via the 8th–12th intercostal space. Using a Seldinger technique, a guidewire was advanced into the gallbladder over which the drains were then passed under fluoroscopic control. Protocol modification was required mid experiment. Gallbladders were filled via catheterisation of the common bile duct before cholecystocentesis. After fluoroscopy-confirmed cholecystostomy drain placement, CT scans were performed to assess drain position, iatrogenic organ injuries and leakage. Leak pressure testing was performed followed by anatomic dissection. Organ injuries were recorded and classified as minor, moderate or severe.
Results
SUB-nephrostomy drain placement was performed in 15 cats and placement into the gallbladder was feasible in two: one was passed before and one after technique modification. Paediatric-nephrostomy drain placement was tested in one cat. The gallbladder could not accommodate the drain size, placement was not feasible and the device was not further tested. A CT scan of the two cats with drain placement showed a moderate amount of free peritoneal contrast, no pleural space penetration and one liver injury. Leakage occurred at a pressure of 4.5 cm H2O. For all drains, injuries recorded during anatomic dissection were to the liver, pleural space and gallbladder. The majority of injuries were classified as minor.
Conclusions and relevance
Percutaneous placement of cholecystostomy drains was not feasible with the method and devices tested. Further studies are needed to investigate alternative techniques in cats.
Keywords
Introduction
Extrahepatic biliary obstruction in cats is a complex condition often associated with a range of serious underlying diseases. Inflammatory, infectious or neoplastic conditions – among others – can impair bile flow from the liver to the bowel. Notably, feline anatomy may predispose cats to biliary obstruction, as the common bile duct merges with the major pancreatic duct before entering duodenum at the major duodenal papilla.1 –3 The shared opening of both ducts into the duodenum highlights how pathologies in any of these three organs affects the others’ function. Although a separate pancreatic ductal opening could mitigate this risk, only approximately 20% of cats have an accessory pancreatic duct opening at the minor duodenal papilla.4,5 Surgical procedures to relieve extrahepatic biliary obstruction include cholecystectomy, choledochotomy, choledochal stenting and rerouting procedures, such as cholecystoduodenostomy or cholecystojejunostomy. 6
Cholelithiasis, cholangitis, cholecystitis or triaditis, which involves inflammation of the pancreas, hepatobiliary system and small intestine, are common non-neoplastic causes of extrahepatic biliary obstruction in cats.2,7 –11 There are three recognised forms of cholangitis in cats: neutrophilic (suppurative), lymphocytic (non-suppurative) and chronic cholangitis (liver fluke infestation).10,12,13 Although the aetiology of neutrophilic cholangitis remains unclear, two pathophysiological mechanisms have been proposed. 2 First, mechanical or functional obstruction of the biliary tree may lead to bile stagnation and increased intraductal pressure, creating bacterial proliferation and invasion into the ductal epithelium.14 –18 Second, primary bacterial infiltration of bile is suggested, as positive bacterial cultures from bile, liver or both are frequently seen in cats with neutrophilic cholangitis.8,19 –22 Immunohistochemstry 23 and fluorescence in situ hybridisation (FISH) 24 have demonstrated that they detect bacterial infections more frequently than standard culture methods. Inflammatory bowel disease, in particular, appears to be a main driver for the development of lymphocytic cholangitis and pancreatitis in cats. 25 This is believed to result from the migration of lymphocytes and plasma cells from the small intestine into the choledochal duct.13,26
The overall prognosis for cats with neutrophilic cholangitis depends on the severity of the condition and aggressiveness of treatment. 2 A mean survival time of 29.3 months was reported for cats with neutrophilic cholangitis, with 47% surviving less than 1 year. 27 Cholangitis in the presence of extrahepatic biliary obstruction is associated with a worse prognosis as cholestasis progressively debilitates physiological processes leading to multi-organ failure, coagulopathies, pancreatitis and often sepsis. Cats with surgically treatable conditions and those undergoing cholecystectomy showed superior survival rates than those undergoing conservative management alone or surgery after initial medical management. 28 Mortality rates for cats undergoing extrahepatic biliary surgery for non-neoplastic conditions remain high, with a range of 21–40%.7,8 In humans, cholangitis may respond to conservative therapy; however, in most cases, decompression of the biliary tree is necessary. 29 Mortality rates for the surgical management of cholangitis in humans is in the range of 10–40% and is associated with severity of disease.30 –32 Endoscopic vs percutaneous treatment methods, ideal timing of intervention (<48 h vs >48 h) and extent of initial intervention (comprehensive vs decompression) have long been debated when treating humans with cholangitis. In 2021, the American Society of Gastroenterology published guidelines recommending endoscopic decompression within 48 h, with concurrent removal of choleliths and sphincterotomy, rather than decompression alone, unless the patient is unable to tolerate more extensive endoscopic treatment. 29 Similarly, early definitive surgical intervention has been advocated to improve survival in cats with extrahepatic biliary obstruction.6,33 As in humans, not all cats are suitable candidates for early definitive surgery, particularly those with significant physiological derangement. In human medicine, the implementation of preoperative biliary decompression has been associated with reduced mortality and complication rates after cholecystectomy or biliary rerouting procedures.34 –38
Malignant extrahepatic biliary obstruction in cats is associated with a significantly worse prognosis than non-neoplastic causes. Biliary adenocarcinoma, lymphoma, exocrine pancreatic carcinoma and squamous cell carcinoma of the duodenum and pancreas are malignancies associated with cholestasis.8,33 More than half of the cats with neoplasia in one study were also found to have concurrent inflammatory hepatobiliary disease. 33 Perioperative mortality rates for cats with underlying neoplasia reportedly range from 44% 33 to 100%. 8 Cardiopulmonary arrest, refractory hypotension, coagulopathies, acute kidney failure, hepatic failure and septic peritonitis are the reported reasons for euthanasia or cause of death.8,33,39,40
Endoscopic procedures of the bile duct in veterinary medicine, however, are limited as a result of patient size and availability of specialised equipment. To the authors’ knowledge, there are no published reports of endoscopic procedures for biliary obstruction in cats. Before the introduction of endoscopic biliary decompression methods, 37 cholecystostomy drain placement allowed biliary diversion and patient stabilisation in humans before cholecystectomy surgery. The clinical benefits of cholecystostomy drains for perioperative or palliative therapy in small animals have been published. The majority of reported methods rely on either an open approach via ventral midline laparotomy or laparoscopic-assisted methods through which either a pig-tail catheter, Foley catheter or percutaneous endoscopic gastrostomy (PEG) tube is placed into the gallbladder.41 –43 It has been suggested that cholecystostomy drains should only be placed into functionally healthy gallbladders. 41 However, it could be argued that cats with extrahepatic cholestasis due to cholelithiasis, cholecystitis, pancreatitis or neoplasia of the biliary tree could benefit from cholecystostomy drain stabilisation. In the recent literature, the use of temporary cholecystostomy drains with the specific intent of patient stabilisation before definitive surgery is reported in only a handful of cases, namely five cats21,43,44 and eight dogs.42 –46 In these cases, total bilirubin levels dropped rapidly after drain placement. Although these drains show promise, the need for two abdominal surgeries and associated anaesthesia may deter owners and clinicians from implementing them. It is disheartening that despite the great strides taken in advancing interventional and minimally invasive procedures in veterinary medicine, a percutaneous technique for placing these drains has not been established. There is a clear paucity of data on percutaneous cholecystostomy drain placement methods, particularly in cats.
There is currently no minimally invasive, commonly available, safe technique for the placement of percutaneous cholecystostomy drains in cats. It was therefore the purpose of this study to investigate the feasibility and safety of ultrasound and fluoroscopically guided, percutaneous cholecystostomy drain placement via the Seldinger technique with two commercially available nephrostomy drains in a cadaveric model. The hypothesis was that the tested technique is feasible and safe.
Materials and methods
Percutaneous ultrasound- and fluoroscopy-guided placement of two drain systems was evaluated in feline cadavers. Cats included in the study had died or were euthanased for reasons unrelated to the investigation. The exclusion criteria included a history of hepatobiliary or pancreatic disease, perforating abdominal injuries, abdominal neoplasia or the presence of free peritoneal fluid. Cadavers were refrigerated and then frozen within 24–36 h post mortem. Bodies were stored at −18°C for 2–12 months. Before drain placement, bodies were thawed at 20°C over a period of 36–48 h. 47
The drain systems tested were the 6.5 French (Fr) locking loop nephrostomy component of the subcutaneous ureteral bypass (SUB) system (SUB-nephrostomy drain, Locking Loop Catheter with Stiffening Cannula; Norfolk Vet Products) and an 8 Fr paediatric nephrostomy percutaneous access set (PNPAS) (paediatric-nephrostomy drain; Boston Scientific).
Percutaneous drain placement was performed with cats positioned in left lateral recumbency. The fur over the right lateral thorax and entire abdomen was clipped. The gallbladder was identified using an ultrasound device (HM70 EVO; Samsung) for percutaneous access between the eighth and 11th intercostal space.43,45 A small skin incision was made, through which either a 12 G over-the-needle venous catheter (MILA Catheter over needle; 1211) or a 20 G spinal needle (BD) was introduced for ultrasound-guided cholecystocentesis via either a transhepatic or transcholecystic approach (Table 1). Aspiration of bile confirmed successful placement followed by an injection of 2 ml of a 50:50 solution of iobitridol (Xenetix 350) and water. Then, either a SUB-nephrostomy drain or a paediatric-nephrostomy drain was placed using the Seldinger technique. The procedure time was defined as the interval from successful cholecystocentesis to the point at which the drain was secured to the body.
Summary of cats in chronological order with details for to drain placement, cholecystocentesis and organ damage

Organ damage severity scores: 1 = minor; 2 = moderate; 3 = major
NA = not applicable; Paediatric = paediatric-nephrostomy drain; SUB = subcutaneous ureteral bypass-nephrostomy drain
Placement of SUB-nephrostomy drain
A 0.035-inch J-tip guidewire (Infiniti Medical) was passed under ultrasound and fluoroscopic guidance through the catheter into the gallbladder to form a complete coil within the gallbladder. Then the spinal needle or catheter was removed and a 5 Fr dilator (Medical Instruments for Animals ZVK Set; Mila International) was inserted through the body wall, but not through the gallbladder wall. The dilator was subsequently removed and the SUB-nephrostomy drain was advanced over the guidewire into the gallbladder under fluoroscopic guidance, where it was secured using its locking loop. Additional contrast solution was then injected into the gallbladder via the SUB-nephrostomy drain to confirm correct drain placement via fluoroscopic imaging. The drain was then secured to the body wall using a finger-trap suture technique. A subcutaneous vascular access port (PleuralPort; NorfolkVet Products) was then attached to the end of the SUB-nephrostomy drain.
Placement of paediatric-nephrostomy drain
A 0.38-inch guidewire (Infiniti Medical) was passed through the spinal needle or catheter into the gallbladder. Once the spinal needle or catheter was removed, a series of increasing sized dilators (6, 8 and 10 Fr) as provided by the manufacturer, was passed over the wire through the body wall, again avoiding the gallbladder wall. The paediatric-nephrostomy drain was then advanced over the wire into the gallbladder under fluoroscopic control, the wire was removed and again contrast solution injected to confirm correct drain placement. The drain was then secured to the body wall.
Modification of the method
After completing the procedure on the first six cats, the study design was modified to a catheterisation approach. Cholecystocentesis was not possible in the cadavers, as the gallbladders were frequently empty and therefore too small for ultrasound-guided cholecystocentesis. Even when moderately filled, the gallbladder wall often pushed away from the needle and could not be punctured. The method was revised to include a small ventral midline laparotomy. The proximal duodenum was gently retracted and opened along its anti-mesenteric border at the level of the duodenal papilla. A 5 Fr feeding tube (Vygon) was then passed retrograde into the neck of the gallbladder. The cystic duct was ligated around the drain using 3-0 polyglactin 910 (Vicryl; Ethicon, MedTech; Johnson & Johnson). The gallbladder was then filled with water via the feeding tube until it reached an average ultrasonographic size of 2.0 × 1.5 cm. Once adequate distension was achieved, drain placement was carried out as previously described (Figure 1).

Subcutaneous urethral bypass-nephrostomy drain placed into the gallbladder. The drain is observed traversing the liver parenchyma (injury score 1) before entering the gallbladder. This type of placement corresponds with a previously described transhepatic drain placement technique. a = drain; b = liver; c = feeding tube; d = stomach; GB = gallbladder
CT
Cats with successful drain placement underwent CT imaging (IQon Spectral CT; Philips) (Figure 2). They were maintained in left lateral recumbency, and 2 ml of 50:50 iobitridol (Xenetix 350) to water solution was administered into the gallbladder via the cholecystostomy drain before scanning. CT images were evaluated for drain position, evidence of leakage and organ damage in the thorax and abdomen. Leakage was subjectively classified as minor, medium and major.

Three-dimensional reconstruction of the CT scan. a = drain; GB = gallbladder
Pressure measurement
In cases with successful drain placement, leak pressures were tested using a water manometer (Medifix Messleiste; B Braun [4279913], Medifix Infusionssysteme zur Messung des zentralen Venendrucks [Artieklnummer 4276116]). The manometer was attached to the table at the level of the gallbladder while the cats remained in left lateral recumbency. It was connected to the 5 Fr feeding tube (Vygon) that had been passed retrograde into the gallbladder via a duodenotomy. If this step had not been taken before drain placement, it was performed according to the modified catheterisation method. Water was then injected into the gallbladder through the cholecystostomy drain in increments of 5 ml. Pressure measurements were recorded before leakage occurred and at the point of leakage, along with the total volume of water injected.
Anatomic dissection
Cats were placed in dorsal recumbency and a ventral midline laparotomy was performed. The thoracic cavity was accessed via a transdiaphragmatic approach. Organs within both cavities were examined for iatrogenic injures. Injuries were categorised as minor, moderate or major. Minor injuries were defined as subtle lesions identifiable only upon close inspection and unlikely to necessitate intervention in a clinical patient, such as peripheral liver penetration (eg, transhepatic) or traversal of the caudoventral pleural recess by the drain. Moderate injuries were clearly visible and would require treatment, although they were not considered life-threatening. Major injuries were defined as those that would be life threatening for a clinical patient.
Results
A total of 16 cats were included in this study, with a median weight of 4.1 kg (range 2.5–6.4) (Table 1). The SUB-nephrostomy drain was evaluated in 15 cats, while the paediatric-nephrostomy drain was tested in one cat. Cholecystostomy drain placement using the SUB-nephrostomy drain was achieved in two cats – one with and one without catheterisation of the common bile duct. Placement of the paediatric-nephrostomy drain was unsuccessful, as the gallbladder could not accommodate the size of the drain.
Percutaneous cholecystocentesis was not possible in three of the first six tested cats without catheterisation of the common bile duct. The empty gallbladder shifted away as the needle indented the wall rather than puncturing it. Multiple attempts were made with varying degrees of force, angles and locations. After approximately 15 mins, the gallbladders in most cats emptied physiologically and were no longer visible on ultrasound. In one cat (cat 1), cholecystocentesis was successful, and the guidewire was advanced and coiled within the gallbladder; however, the SUB-nephrostomy drain could not be advanced through the gallbladder wall. In cat 3, both cholecystocentesis and drain placement were successful. Fluoroscopy and CT imaging confirmed correct position of the drain within the gallbladder. Moderate leakage was observed on imaging, with no organ damage identified on anatomical dissection. The procedure time was 12 mins.
In all 10 cats with catheterisation of the common bile duct, cholecystocentesis and insertion of the guidewire were successful. The SUB-nephrostomy drain was successfully advanced into the gallbladder of one cat (cat 12) confirmed fluoroscopically and on CT scan. The procedure time was 7 mins. The CT scan showed moderate amounts of free contrast in the abdomen. In cat 14, the gallbladder emptied through the cholecystocentesis site once the guidewire was coiled within the gallbladder and the venous catheter was removed. Despite repeated attempts to re-distend the gallbladder via the catheter, it failed to maintain sufficient distention, and a SUB-nephrostomy drain placement was not possible. In cats 11, 13, 15 and 16, the SUB-nephrostomy drain could not be advanced into the gallbladder, as the drain tip was unable to penetrate the gallbladder wall.
Pressure test
Leak pressure testing was successfully performed in 1/2 cats with a SUB-nephrostomy drain placed (cat 12). Leakage occurred at the drain entry point at a pressure of 4.5 cm H2O after the injection of 15 ml of fluid. In the other cat (cat 3), the SUB-nephrostomy drain was inadvertently severed during the abdominal dissection, preventing leak pressure assessment.
Anatomic dissection
Anatomic dissection was performed on all 16 cats (Table 1). In five cats, the drain traversed liver parenchyma. These injuries were classified as minor. In four cats with liver injury, additional pleural (n = 3) and/or gallbladder (n = 2) injuries were recorded. In one cat, pleural space penetration was found without additional organ injury. In all but one cat, pleural space injury was classified as minor, with the drains located in the caudoventral region of the pleural space, between the thoracic wall and diaphragm (Figure 3a). In one cat, pleural space injury was classified as moderate, as the drain traversed the thorax in an area where avoiding lung tissue would be impossible (Figure 3b). Pulmonary parenchyma remained uninjured in all cats. Gallbladder rupture occurred in two cats during the procedure, with a defect observed on the wall opposite the drain entry point, resulting in bile leakage into the abdomen (Figure 4). The remaining abdominal structures were uninjured.

Pleural space penetration: (a) moderate penetration; (b) mild penetration of the pleural space. a = drain; b = pleural space; c = diaphragm; d = abdomen
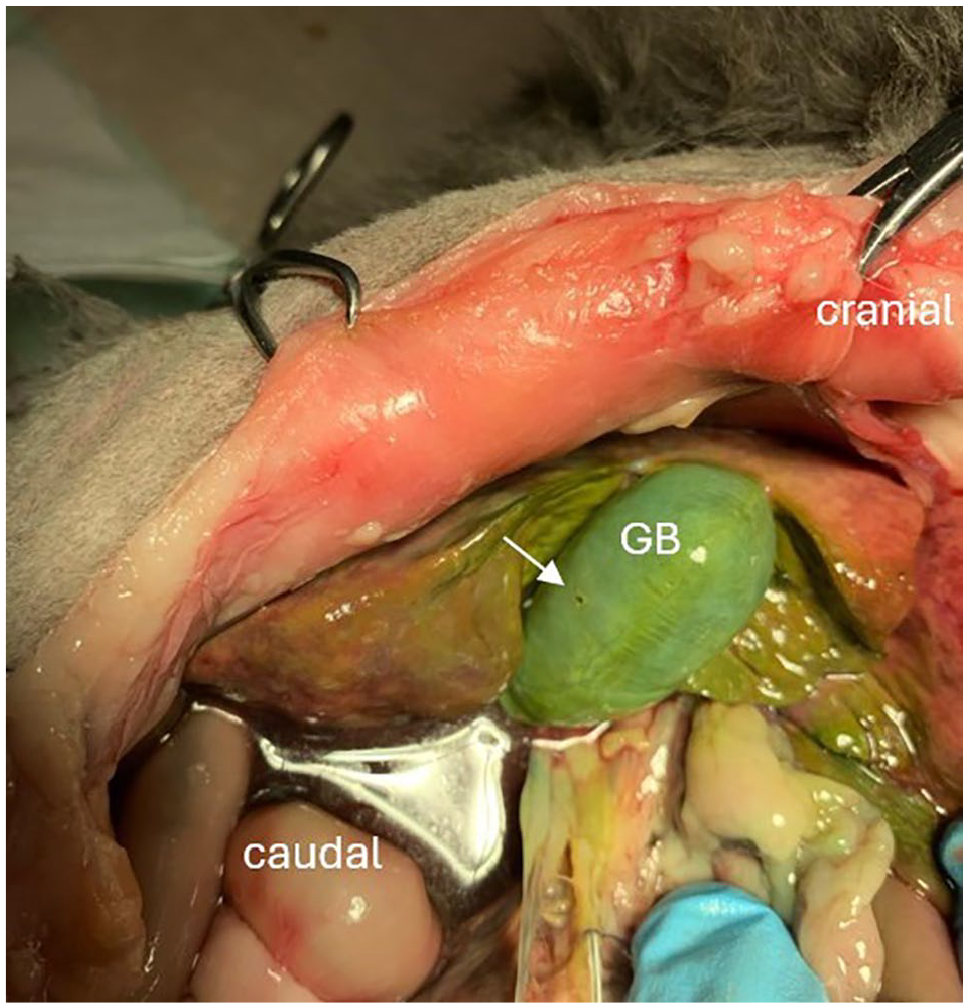
Gallbladder injury showing the puncture site on the wall opposite the gallbladder (white arrow). GB = gallbladder
Discussion
The placement of percutaneous cholecystostomy drains using the described approach was not feasible in most cats (14/16, 87.5%). Organ injuries were identified in nearly half (n = 7/16) of the cats; although they were mostly considered minor, the technique poses some safety concerns.
The method used in this study was a modified version of those previously described.43,45 Three ultrasound-guided cholecystostomy techniques were tested on canine cadavers by Murphy et al, 43 with an overall success rate of 20%. In comparison, 2/16 (12.5%) drains were successfully placed in the present study. The two techniques differ in several aspects, including the needle size for cholecystocentesis, nephrostomy drain size, drain delivery system, transhepatic access to the gallbladder and intercostal access point. Murphy et al 43 tested 8 Fr and 10 Fr locking pigtail nephrostomy catheters in dogs, accessing the gallbladder via the ninth to 11th intercostal space using 21 G and 22 G needles. Similar difficulties with drain passage were reported in that canine model, with the authors stating that the drains would not pass through the cholecystocentesis stoma and the gallbladder deflected away from the drain tip. 43 Comparable issues were observed in the present study using the smaller 6.5 Fr SUB-nephrostomy drain in cat cadavers. To address this, gallbladders were punctured with either a 12 G catheter or 20 G needle to create comparably large holes in the gallbladder wall to accommodate a 6.5 Fr drain; however, the drains would still not advance through the created stoma. A 5 Fr dilator was used in both methods; however, its insertion into the gallbladder was not specified in the canine study. In the present study, the dilator was not introduced into the gallbladder to minimise bile leakage and reduce the risk of gallbladder rupture. The conclusions from this feline study align with those of the canine study, that the percutaneous, imaging-guided method for cholecystostomy drain placements were feasible. 43
The method reported by Chmelovski et al, 45 however, successfully placed a SUB-nephrostomy drain into the gallbladder of a Rottweiler with extrahepatic biliary obstruction secondary to pancreatitis. Using an ultrasound- and fluoroscopy-guided Seldinger technique, the intrahepatic bile duct was accessed via the 11th intercostal space using a 14 G over-the-needle catheter. A guidewire was passed from the duct into the gallbladder, over which a SUB-nephrostomy drain was then advanced. 45 Difficulties accessing the biliary tree, use of a dilator and complicated drain placement were not reported in this case. Compared with the current study, the SUB-nephrostomy drain was placed transhepatically into a bile duct rather than directly into the gallbladder. In the absence of bile duct distension, this method could not be tested in the current cadaver model and further studies are needed to test this method in cats. The difference in ease of passing the SUB-nephrostomy drain in the clinical case compared with the cadaveric model could be attributed to the post-mortem change in soft tissue elasticity. 12
The frequency and type of iatrogenic organ injuries in this study are comparable with those previously reported. The liver was the most frequently injured organ in six cats, followed by pleural space penetration in four cats, with three cats having both liver and pleura injury. A transhepatic approach to the gallbladder was not specified with the tested method, as there is no clear evidence to show lower risk for bile leakage with this approach. 48 Access to the gallbladder was gained via the safest and most feasible route based on ultrasound without specifically avoiding the liver. The liver lesions were classified as minor and could be considered as transhepatic drain placement; however, as this was not specified in the method, these were recorded as iatrogenic organ damage.
Pleural space penetration is a clear injury that should be avoided when placing cholecystostomy drains. In the four drains that entered the thorax, all but one had concurrent liver damage. Access to the gallbladder was gained through the eighth or ninth intercostal space in these cats. This is similar to the results reported in canine cadavers, where 100% of transhepatically placed drains crossed the pleura via a ninth to 11th intercostal approach. 43 In the same study, a clinical case developed intraoperative pneumothorax during laparoscopic-assisted cholecystostomy drain placement. 43 The intercostal access point was not reported; however, the drain passed across the right costomediastinal recess through the caudal-most part of the pars costalis of the diaphragm. 43 Anatomically, the canine gallbladder is located at the level of the seventh to eighth intercostal space and does not protrude beyond the liver margins, 49 whereas the feline gallbladder is located between the eighth and 10th intercostal space.6,50 The method was therefore amended to access the gallbladder between the eighth to 12th intercostal space compared with the ninth to 11th spaces reported in dogs.43,45 As the lungs were not inflated in the cadavers, it is unclear in what proportion pulmonary injury would have occurred using this insertion route. A safe corridor for percutaneous cholecystostomy drain placement could not be established with the current method and further studies are therefore warranted.
Iatrogenic gallbladder injury is a critical complication of this procedure, especially when drains are intended to be placed in already systemically debilitated patients with distended, potentially fragile gallbladders.51 –53 Bile within the abdomen results in a sterile, chemical peritonitis, in the presence of bacteria; however, a potentially more life-threatening septic bile peritonitis ensues.10,54 –57 Survival after gallbladder rupture is as low as 45% in veterinary medicine, 57 with reports in the range of 27–45% for septic and 87–100% for sterile bile peritonitis in dogs.46,57 Gallbladder injury during percutaneous cholecystostomy drain placement has not been reported in previous studies. In this study, injuries were presumed to have occurred during cholecystocentesis when the needle punctured through the opposite wall, creating a defect (Figure 5). This defect then continued to tear as the guidewire was inserted and coiled within the gallbladder. The cadaveric nature of this study may have contributed to the occurrence of this type of injury.

Gallbladder damage (severe). The arrow indicates the hole in the gallbladder. a = drain; b = liver; GB = gallbladder
The use of cadavers represents a major limitation of this study. Cadavers were refrigerated, then frozen and thawed before the procedure. Cooling protocols have been shown to affect the soft tissue mechanics in cadaveric studies,58,59 with short-term refrigeration causing the most significant reduction in tissue elasticity. 58 No biomechanical studies have been performed to determine the ideal storage conditions for gallbladders. The use of fresh cadavers may provide a more representative condition of a clinical patient, as the gallbladders in this study were friable.
The variable filling state of the gallbladders was another study limitation. Cats were selected without previous hepatobiliary or pancreatic disease, but the first six had empty or only moderately filled gallbladders, making cholecystocentesis challenging. In some cases, the gallbladder wall could not be punctured despite changes in force or angle, or the needle slipped out of the nearly empty gallbladder after successful puncture. This issue resolved with the modified method; however, opening the abdomen and retracting the duodenum may have shifted the gallbladder’s position and introduced air into the body cavity. On ultrasound, air artefacts did not interfere with imaging. A cranial shift of the gallbladder deeper into the ribcage could have affected the intercostal access point and increased the risk of pleural space penetration.
The paediatric-nephrostomy drain was tested in only one cat. The 8 Fr drain was too large for the gallbladder of this 4.8 kg cat. Coiling the guidewire within the gallbladder was complicated and fitting the entirety of the locking loop into organ was not possible. Once the gallbladder was punctured, 10 ml of water were injected to distend the lumen before attempting drain placement. A wide range of drain sizes has been reported for feline cholecystostomy, including 8 Fr or 10 Fr locking pigtail catheters, 43 16 Fr or 20 Fr PEG tubes, 44 and 6 Fr Foley catheters. 3 These were all placed either via an open or laparoscopic-assisted approach, and were placed in cats with extrahepatic biliary obstruction and distended gallbladders, which may better accommodate larger drains.
Conclusions
Percutaneous, ultrasound- and fluoroscopy-guided cholecystostomy drain placement presents several challenges in cats. Further studies are needed to establish a safe access route for a minimally invasive technique that can be performed efficiently and safely. Once such a method has been established, its clinical benefits in cats with extrahepatic biliary obstruction can be investigated.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional information consent for publication was not required.
