Abstract
Objectives
The incidence of calcium oxalate (CaOx) urolithiasis in cats is rising globally, while magnesium ammonium phosphate (MAP) uroliths remain prevalent. MAP uroliths are commonly associated with urease bacterial infection in dogs. Urinary tract infections (UTIs) could increase the risk of urolithiasis, but there have only been limited studies to date in cats in developing countries. This study evaluates the association between UTI, sex, age and the occurrence of MAP uroliths compared with CaOx uroliths in cats treated for urolithiasis at Kasetsart University Veterinary Teaching Hospital, Thailand.
Methods
A retrospective study was conducted of medical records from cats undergoing surgery for urolithiasis at Kasetsart University Veterinary Teaching Hospital, Thailand between 2016 and 2021. Data retrieval included mineral type, age, sex, breed and culture results. Uroliths were analysed via Fourier-transform infrared spectroscopy. Odds ratios (ORs) and 95% confidence intervals (CIs) assessed risk factors for MAP urolith formation.
Results
Data from 264 cats (168 males, median age 5 years; 95 females, median age 5.1 years; one unidentified) were included. Calcium oxalate (CaOx) was the most common urolith (170/264, 64.4%), followed by MAP urolithiasis (80/264, 30.3%). A total of 176 urine samples were cultured, of which 36/58 MAP cases, 32/107 CaOx cases and 5/11 other urolith cases tested positive. The most common bacteria were Staphylococcus species (21/73, 28.8%), Escherichia coli (16/73, 21.9%) and Enterococcus species (9/73, 12.3%). Multiple logistic regression indicated strong urease-producing bacteria increased MAP risk by 11.93 times (OR 11.93, 95% CI 4.28–33.23) and other bacteria (Enterococcus species, Pasteurella species, Acinetobacter species) increased risk by 3.84 times (OR 3.84, 95% CI 1.32–11.12). Age and sex had no significant effect.
Conclusions and relevance
The findings of this study unveiled an association between strong urease-producing bacteria and MAP in cats with urolithiasis.
Introduction
Urolithiasis is a common disease in cats worldwide and can result in clinically significant morbidity and mortality rates. 1 The two most common types of feline urolith are magnesium ammonium phosphate (MAP) and calcium oxalate (CaOx).2,3 The type of urolith is influenced by age, breed, sex and geographic location. 4 MAP uroliths commonly recur, but this can be prevented by optimal management. Risk factors for MAP urolithiasis in dogs have been identified, including urinary tract infection (UTI), mineral composition of diets, urine alkalinising metabolites in diets, urine pH and concentrated urine.5,6 Studies in dogs indicated that the occurrence of MAP uroliths is significantly associated with urease-producing bacteria in the urinary tract. 7 Dogs with strong urease-producing bacterial UTIs (Staphylococcus species and Proteus species) were observed to have an increased risk of developing MAP uroliths vs CaOx uroliths.7,8 In cats, urolithiasis accounts for 15–23% of feline lower urinary tract signs and only 5% of cases are associated with a UTI.6,9 Sterile struvite uroliths have been regularly reported in feline urolithiasis. The two most common types of uroliths found in cats in Thailand are MAP and CaOx.10,11 However, the association between UTI and uroliths in cats in this region remains unclear. Thus, the aim of this retrospective study was to investigate the association between UTI, sex, age and the proportion of MAP uroliths compared with CaOx uroliths in cats treated for urolithiasis at Kasetsart University Veterinary Teaching Hospital, Thailand, between 2016 and 2021. This study was approved by the Kasetsart University Institutional Animal Care and Use Committee (ID ACKU67-VET-031).
Materials and methods
Urolithiasis data collection
Urolithiasis data were obtained from the medical records of cats diagnosed with urolithiasis, presenting with lower urinary tract clinical signs and undergoing routine surgery and treatment at Kasetsart University Veterinary Teaching Hospital, Thailand, between 1 April 2016 and 31 December 2021. Cats were included if they had one or more of the following clinical signs: haematuria, stranguria, pollakiuria, dysuria, periuria, urinary incontinence or urethral obstruction. Consent was obtained from the owners for the collection of uroliths. The cats’ age (0–2 years, >2–4 years, >4–7 years, >7–10 years or >10 years), sex (intact female, spayed female, intact male or castrated male) and type of urolith (CaOx, MAP, purine, cystine, calcium phosphate [CaP] or mixed) were recorded.
The uroliths were sent to Kasetsart Urolith Center, Faculty of Veterinary Medicine, Kasetsart University for analysis. The urolith composition was classified by Fourier-transform infrared (FTIR) spectroscopy using a Tensor 27 FT-IR spectrometer (Bruker). The FTIR spectra of each sample were analysed in duplicate and the data were compared using Bruker’s RENAL 1 and 2 spectral libraries to determine the chemical composition of the uroliths. Mineral amounts ⩾70% and one layer were classified as the corresponding mineral types. Mineral amounts <70% were classified as mixed uroliths. Uroliths with at least two layers containing different minerals, where each layer comprised ⩾70% of the urolith’s composition, were classified as compound uroliths.
UTI data
Culture data were extracted from the medical records database. Cultures were performed on urine collected via cystocentesis or bladder mucosal swabs obtained during cystotomy and submitted to the Veterinary Diagnostic Center, Faculty of Veterinary Medicine, Kasetsart University. When cats had multiple cultures performed, only data from the first visit were entered into the study. All specimens were refrigerated at 4°C until processed, typically within 12 h. Cats that received antibiotics for up to 2 weeks or that were catheterised before bacterial urine culture were excluded. The urine was cultured on blood and MacConkey agar before incubation at 37°C for a maximum of 72 h. Cats with multiple bacteria identified on culture were excluded. Microbial isolates were identified using routine biochemical and automated systems (VITEK 2 COMPACT; Biomérieux). Antibacterial susceptibility testing was performed on Mueller–Hinton agar with antimicrobial discs (MASTDISCS AST; Mast Group).
Bacterial UTI was classified into four groups as follows: group 1, strong urease-producing bacteria (Staphylococcus species, Proteus species); group 2, weak urease bacteria (Streptococcus species, Klebsiella species, Escherichia coli, Pseudomonas species); group 3, other bacteria (Enterococcus species, Pasteurella species, Acinetobacter species); and group 4, negative bacterial cultures (reference group).12,13
UTI is defined as significant bacteriuria on culture in combination with clinical signs of infection. Quantification of colony-forming units (CFUs) was not performed and any growth was considered significant bacteriuria.
Statistical analysis
The ages of cats are reported as mean ± SD. We estimated odds ratios ( ORs) and 95% confidence intervals (CIs) to determine the association between the type of bacteria, cats’ age and sex, and the development of MAP compared with CaOx using univariate logistic regression. Statistical analyses were performed using a commercial statistical package (NCSS 11 Statistical Software; NCSS). To determine the association between age and sex and MAP or CaOx risk, we used the cats in the >10 year group and castrated males as references. P <0.05 was considered statistically significant.
Results
This study included 264 cats with urolithiasis (168 males, 95 females and one unidentified). The 264 uroliths were categorised into six urolith types: CaOx (n = 170, 64.39%), MAP (n = 80, 30.3%), purine (n = 8, 3.03%), cystine (n = 3, 1.14%), CaP (n = 1, 0.38%) and mixed (n = 2, 0.76%) (Figure 1). The mean age of cats with each urolith type is presented in Table 1.

Distribution of 264 feline uroliths by type from cats seen at Kasetsart University Veterinary Teaching Hospital, 2016–2021. CaOx = calcium oxalate; CaP = calcium phosphate; MAP = magnesium ammonium phosphate
Number of cats with each urolith type and their corresponding mean ages
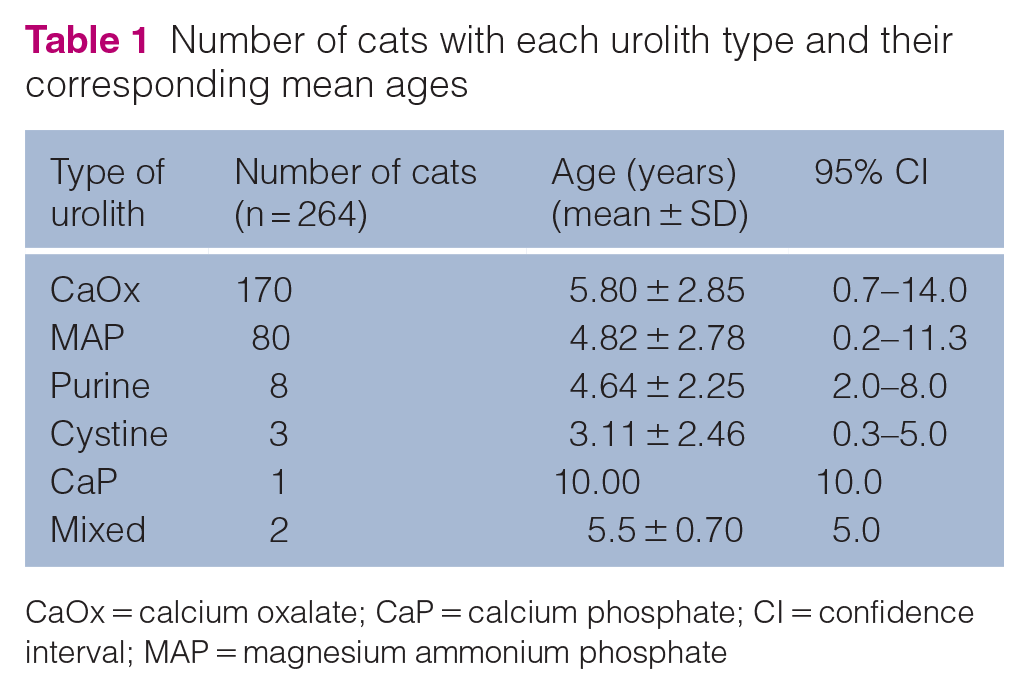
CaOx = calcium oxalate; CaP = calcium phosphate; CI = confidence interval; MAP = magnesium ammonium phosphate
This study obtained 263 data points for cat breeds; one cat was unidentified. A total of 10 breeds of cats presented with urolithiasis, with the top three breeds as follows: domestic shorthair (n = 162, 61.6%), Persian (n = 70, 26.6%) and Scottish Fold (n = 23, 8.8%). The remaining breeds included in the study, along with their corresponding case numbers, were American Shorthair (n = 1), Bengal (n = 1), British Shorthair (n = 1), European Shorthair (n = 1), Exotic Shorthair (n = 1), Thai (n = 1) and Maine Coon (n = 2).
A total of 176 cats were tested with bacterial urine culture, of which 73 were culture positive. Bacterial culture were performed on 154 urine samples obtained via cystocentesis and 22 bladder mucosal swabs. The bacteria identified from the UTI cases were as follows: 21 (28.8%) cases of Staphylococcus species; 16 (21.9%) cases of E coli; nine (12.3%) cases of Enterococcus species; seven (9.6%) cases of Klebsiella species; six (8.2%) cases of Proteus species; four (5.5%) cases of Pseudomonas species; four (5.5%) cases of Pasteurella species; three (4.1%) cases of Streptococcus species; and three (4.1%) cases of Acinetobacter species.
Of the 80 cats with MAP uroliths, bacterial culture testing was performed on 58 samples. In total, 22 (37.9%) cases yielded negative bacterial culture results. UTI cases (bacteriuria by culture in combination with clinical signs) accounted for 36 (62.1%). Bacterial species identified included Staphylococcus species (n = 18, 50%), Enterococcus species (n = 5, 13.8%), Proteus species (n = 3, 8.3%), Pasteurella species (n = 3, 8.3%), E coli (n = 2, 5.6%), Pseudomonas species (n = 2, 5.6%), Klebsiella species (n = 1, 2.8%), Streptococcus species (n = 1, 2.8%) and Acinetobacter species (n = 1, 2.8%). Based on bacterial identification, strong urease-producing bacteria were detected in 21 samples, weak urease bacteria in six samples and other types of bacteria in nine samples (Table 2).
Categories of bacterial urinary tract infection and occurrence of MAP and CaOx uroliths, showing ORs and 95% CIs for cats with MAP uroliths, with CaOx as a reference

CaOx = calcium oxalate; CI = confidence interval; MAP = magnesium ammonium phosphate; NA = not applicable; OR = odds ratio
Of the 170 cats with CaOx uroliths, bacterial urine cultures were performed in 107 (62.9%) cases. Among these, UTI was diagnosed in 32 (29.0%) cases, while 75 (71.0%) cases tested negative for bacterial culture. The bacterial species identified included Staphylococcus species (3/32 cases, 9.4%), Enterococcus species (3/32 cases, 9.4%), Proteus species (3/32 cases, 9.4%), Pasteurella species (1/32 cases, 3.1%), E coli (11/32 cases, 34.3%), Pseudomonas species (2/32 cases, 6.3%), Klebsiella species (5/32 cases, 15.5%), Streptococcus species (2/32 cases, 6.3%) and Acinetobacter species (2/32 cases, 6.3%). Based on bacterial identification, strong urease-producing bacteria were detected in six samples, weak urease bacteria in 18 samples and other types of bacteria in eight samples (Table 2).
Cats with strong urease-producing bacteria had increased odds of MAP (OR 11.93, 95% CI 4.28–33.23) compared with CaOx uroliths. Those with ‘other’ bacterial infections also had increased odds of MAP (OR 3.84, 95% CI 1.32–11.12). Weak urease bacteria were not associated with an increased risk of MAP urolithiasis (OR 1.14, 95% CI 0.40–3.21) (Table 2). Comparing the 56 cats with culture-positive MAP uroliths and 105 cats with CaOx (note that age data were missing for two cats with each urolith), age was not a statistically significant factor (Table 3).
Association between age range of cats and occurrence of MAP or CaOx uroliths

CaOx = calcium oxalate; CI = confidence interval; MAP = magnesium ammonium phosphate; NA = not applicable; OR = odds ratio
There were 165 cases of MAP or CaOx uroliths submitted for bacterial urine culture, and sex data were available for 164 cats. The distribution among sexes was as follows: 50 (30.5%) intact females, 110 (67.1%) intact males and four (2.4%) castrated males. As a result of the low number of castrated males, this study compared female and male cats. The analysis showed no statistically significant influence of sex on the type of urolith observed (Table 4).
Association between sex of cats and occurrence of MAP or CaOx uroliths

CaOx = calcium oxalate; CI = confidence interval; MAP = magnesium ammonium phosphate; NA = not applicable; OR = odds ratio
For cases of CaOx urolithiasis, the mean age of female cats (29/50, 58%) was 5.6 years, while the mean age of male cats (77/114, 67.5%) was 6.3 years. For MAP urolithiasis, the mean age of female cats (21/50, 42%) was 4.5 years, while the mean age of male cats (37/114, 32.5%) was 5.2 years. The proportions of female and male cats with UTIs, along with their median ages, are presented in Table 5
Association between sex of cats and occurrence of MAP or CaOx uroliths with and without UTI

Data are n (%) or median (range)
CaOx = calcium oxalate; MAP = magnesium ammonium phosphate; UTI = urinary tract infection
Discussion
Most MAP uroliths in cats are formed in sterile urine, with only approximately 5–11% of MAP uroliths associated with UTI according to prior studies.4,9,14,15 In contrast, our study found urinary bacteria in up to 62.1% (36/58) of cats with MAP urolithiasis, of which strong urease-producing bacteria represented 58.3% (21/36) of cases. The association between UTI and MAP formation has not previously been reported in cats, 16 and it is possible that the small number of cats included in our study overestimates the association between bacteriuria and MAP uroliths. However, other studies have found that MAP uroliths were more likely to have positive culture results compared with other mineral types, and that Staphylococcus species was the most common pathogen in MAP uroliths. 1 Though the causal relationship between uroliths and bacteriuria in our study population is unknown, the predominance of Staphylococcus pseudintermedius and Proteus species suggests a potential role for urease production as part of MAP urolith pathogenesis.
Another highly unexpected finding in our study was the proportion of intact male cats with bacteriuria. Bacteriuria is more prevalent in female cats compared with male cats,13,17 and testosterone is considered a protective factor against UTI in intact males in other species. There is no obvious biological rationale to explain the high proportion of intact male cats with bacteriuria in our study population. However, intact male cats are poorly described in earlier studies of feline UTI and feline urolithiasis, as most male cats participating in prior studies were castrated. In Thailand, positive cultures are common in cats with lower urinary tract disease17 and a prior study from this region 18 found a prevalence of UTI of 11% in a small cat population that was predominantly male, of which 50% were intact. The exact number of male cats with UTI was not described in that study but underlying abnormalities were identified in multiple cats with UTI. It is possible that male cats with UTI had unidentified underlying disease in our study. Lastly, bacteriuria could have been overestimated in our population because of lack of quantification of colonies on agar plates. However, animals were sampled with cystocentesis, and the authors consider it unlikely that contamination would explain the high numbers of cats with bacteriuria.
As in other studies based on surgical urolith removal, our study population was biased towards male cats and we included very few female cats.2,19,20
We found that the proportion of MAP uroliths in male cats was higher than in female cats. This concords with findings in other, larger studies. 1 In our study, the proportion of MAP was low relative to CaOx, which was unexpected, but may be explained by the low number of female cats (n = 50) in our study population.
The results of this study showed no significant difference between age groups. The proportion of CaOx uroliths was higher in cats aged >7 years (30/41, 73.2%) compared with cats aged ⩽7 years (75/120, 62.5%), similar to previously published studies, where CaOx uroliths were most commonly found in cats aged >7 years.1,15 The proportion of MAP uroliths in cats aged ⩽7 years (45/120, 37.5%) was higher than in cats aged >7 years (11/41, 26.8%), particularly among cats aged 0–2 years. This suggests that younger cats may be more likely to develop MAP than other types of uroliths, which is consistent with other studies.1,2 Approximately three-quarters of MAP uroliths (45/56, 80.4%) and CaOx uroliths (75/105, 71.4%) were found in cats aged ⩽7 years. Therefore, greater emphasis should be placed on the assessment of urolithiasis in cats aged <7 years with clinical signs of lower urinary tract disease.
Although other studies have reported that UTIs are more common in cats aged >10 years, 16 only 4.3% of cats in this age group were represented in our study. This may be explained by our study population having been biased towards cats aged <10 years.
In our study, strong urease-producing bacteria and other bacteria increased the odds of MAP uroliths by 11.93 and 3.84 times, respectively, which previously has only been reported in dogs.7,8 Therefore, these associations should be considered as potential underlying causes of MAP in cats with urolithiasis.
Urinary bacterial cultures and antimicrobial susceptibility testing should be performed before antimicrobial treatment. Medical dissolution could be considered as a treatment strategy in cats with urolithiasis and positive urine culture with strong urease-producing bacteria, provided there are no contraindications, such as obstructions. Although the dissolution of feline MAP uroliths is possible with a therapeutic diet alone, medical dissolution would include a course of antimicrobial therapy if there is an associated infection. 21 Therefore, bacterial urine culture and antimicrobial drug susceptibility testing are key diagnostic tools in cats with urolithiasis.
This study has some limitations. First, the data for this study were obtained from cats treated exclusively at Kasetsart University Veterinary Teaching Hospital, which may not represent the broader feline population. Second, although urine samples were collected via cystocentesis, where contamination of samples is rare, lack of CFU data prevents complete differentiation between contamination and actual bladder colonisation. 22 Therefore, the positive cultures are somewhat overestimated; contamination can also occur from the skin or the laboratory. Finally, some cats may have been previously treated with antibiotics, potentially influencing an association with UTIs.
Conclusions
The identification of strong urease-producing bacteria significantly Association associated with increased odds of MAP urolithiasis underscores the importance of tailored treatment strategies based on bacterial urine culture. This approach may improve medical urolithiasis management in clinical practice. Longitudinal studies evaluating long-term outcomes and recurrence rates after targeted antimicrobial therapy are warranted to validate the clinical utility of bacterial urine culture in managing MAP urolithiasis in cats.
Footnotes
Acknowledgements
The authors would like to thank Dr Chalermpol Lekcharoensuk and Dr Gunn Kaewmongkol for their valuable comments during manuscript preparation. Technical writing assistance was provided by Royal Canin Thailand.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
