Abstract
Objectives
Toxoplasma gondii is an obligate intracellular coccidian pathogen, with domestic cats and other members of the Felidae family serving as its definitive hosts. The aim of the study was to identify risk factors for positive test results.
Methods
A laboratory database was screened for T gondii PCR results from faecal samples and serology results (IgM, IgG) from serum/plasma taken from cats in Europe between January 2008 and December 2022. Logistic regression analysis was performed to identify risk factors associated with positive T gondii results, such as breed, age, sex, neuter status, regionality, seasonality, feline leukaemia virus (FeLV) and feline immunodeficiency virus (FIV) status. Odds ratios (ORs) were calculated.
Results
A total of 45,523 cats were included: 14,500 (31.9%) tested positive by direct and/or indirect detection methods for T gondii (PCR: 126/7896 [1.6%], IgG: 14,148/37,882 [37.3%], IgM: 1539/37,882 [4.1%]). Age >5 years (IgG: OR 2.591, P <0.001; IgM: OR 1.954, P <0.001), European domestic shorthair cats/cross breeds (IgG: OR 3.848, P <0.001; IgM: OR 2.152, P <0.001), male sex (IgG: OR 1.134, P <0.001), neuter status in male (IgG: OR 0.536, P <0.001) and female cats (IgG: OR 0.577, P <0.001), FeLV antigen positivity (IgG: OR 1.358, P = 0.030) and FIV antibody positivity (IgG: OR 2.350, P <0.001; IgM: OR 2.650, P <0.001) significantly impacted the serological results. In PCR testing, neuter status had a significant impact in male (OR 2.455, P = 0.002) and female cats (OR 2.988, P <0.001). Serological and PCR results were significantly influenced by regionality for IgG (central: OR 1.454, P <0.001; north: OR 0.768, P <0.001; south: OR 0.526, P <0.001; east: OR 0.768, P <0.001; west: OR 0.709, P <0.001), IgM (central: OR 0.616, P <0.001; north: OR 1.456, P <0.001; south: OR 1.767, P <0.001; east: OR 1.456, P <0.001) and PCR testing (central: OR 0.460, P <0.001; north: OR 3.020, P = 0.002; east: OR 3.020, P = 0.002). Seasonality had a statistically significant impact on IgM (summer: OR 1.402, P <0.001; winter: OR 0.732, P <0.001) and PCR testing (autumn: OR 1.473, P = 0.038).
Conclusions and relevance
Breed, age, sex, neuter status, seasonality and regionality significantly impacted serological results. Neuter status, seasonality and regionality significantly impacted the PCR results. Immunosuppression (FeLV/FIV) had a significant impact on serological results. PCR-positive cats shed oocysts and spread infection to other susceptible hosts, including humans. Surveillance is therefore recommended, taking into consideration the associated risk factors.
Keywords
Introduction
Toxoplasma gondii is an obligate intracellular coccidian parasite that infects a wide range of warm-blooded animals, including humans. Domestic cats (Felis catus) are natural definitive and intermediate hosts, whereas non-feline species serve as intermediate hosts.1,2 Sporozoites in oocysts are excreted in faeces and can develop exclusively in the intestinal epithelium of felids. Tachyzoites are the actively multiplying stage included in tissues and milk, while bradyzoites are the slowly multiplying stage enclosed in tissue cysts. 2
In cats, the transmission modes of T gondii include congenital infection, ingestion of infected tissue and ingestion of food or water contaminated with oocysts.2,3 More rarely, blood transfusions and organ transplantations have also been reported as transmission routes in both veterinary 2 and human medicine.4,5 As T gondii has been detected in the milk of experimentally infected queens, lactogenic transmission of the pathogen is also suspected. 6 Rodents serve as intermediate hosts infected with tissue cysts and cats are predominantly infected by ingesting these cysts. The natural cycle of pathogen development is typically completed within 3–10 days after ingestion of T gondii in 97% of naive cats. 2 In rare cases, oocysts and/or tachyzoites are ingested by cats, which leads to a delay in the formation of new oocysts and shedding for up to 18 days. 2 In contrast, the ingestion of oocysts leads to shedding in only 20% of infected cats. 2
Antibody prevalence varies widely across different geographical areas, with 30–40% of cats infected worldwide. 2 For example, 24% of cats in Portugal 7 and 16–40% of cats in the USA 2 had positive serological results. A higher likelihood for positive serological testing was seen in cats fed raw meat, 8 in cats living among animals farmed for meat, 9 adult cats, cats with outdoor access, cats from rural areas and hunting cats.10–12 Some studies have identified feral and stray cats as being more likely to test positive serologically.13–15 Birmans, Ocicats, Norwegian Forest Cats and Persians were found to be 4–7 times more likely to test positive serologically compared with Burmese and Korat cats, most likely as a result of their different lifestyles.8,16
The frequency of shedding of T gondii is not affected by age; however, it is affected by seasons, with peaks occurring between July and December. 17 The shedding of T gondii oocysts in faeces is considered rare, with detection rates in 1/252 (0.4%) samples in Switzerland 18 and in 0.25% of German cats. 17 The annual burden in the environment is variable, with 90–5000 oocysts/m2. 19
Feline leukaemia virus (FeLV) and feline immunodeficiency virus (FIV) infections,20,21 along with the application of immunosuppressive drugs such as glucocorticoids or cyclosporin, predispose cats to the reactivation of infections.22,23 Immunity to T gondii infections in cats is still poorly understood. All infected cats develop IgG antibodies and approximately 80% show IgM antibodies, with IgG being positive 4–6 weeks after infection and the highest titres achieved within 2 or 3 weeks. 2 In clinically sick cats suspected of having toxoplasmosis, diagnosis is ideally established through the detection of T gondii via PCR from muscle biopsies, bronchoalveolar lavage, cerebrospinal fluid or aqueous humour, depending on the clinical signs. A tentative diagnosis is based on serological IgM testing and a favourable clinical response to treatment. 2 IgG and IgM antibodies are frequently detected in both healthy and sick cats and may persist over long time frames; therefore, positive titres do not necessarily prove clinical toxoplasmosis. 24
The aim of the present study was to evaluate the incidence of T gondii in cats in Europe that underwent serological and/or PCR testing. The secondary aim was to identify potential risk factors associated with positive test results.
Materials and methods
This study included both direct and indirect detection assays for T gondii in cats, with samples provided by veterinarians working across Europe to the LABOKLIN laboratory (Bad Kissingen, Germany) between January 2008 and December 2022. Samples were sent to the laboratory by the veterinarians using an overnight courier and data were analysed retrospectively. The laboratory database was electronically screened for molecular and serological testing results for T gondii in the specified time frame.
Molecular testing
Direct detection methods included PCR testing faecal samples. Before nucleic acid extraction, faecal samples were incubated in 1 ml STAR buffer in MagNA Lyser Green Bead tubes (both from Roche Diagnostics). Mechanical disruption was performed for 40 s at 6500 g in a MagNA Lyser instrument (Roche Diagnostics). A total of 200 µl of the resulting supernatant was utilised for automated total nucleic acid extraction using a commercially available kit (MagNA Pure 96 DNA and Viral NA Small Volume Kit; Roche Diagnostics) according to the manufacturer’s instructions. The resulting nucleic acids were eluted in a volume of 100 μl.
In samples submitted in the time frames between January 2008 and June 2016 and between February 2020 and December 2022, a commercially available PCR kit (ADIAVET TOXO REALTIME Kit; BioX Diagnostics) was used for the direct detection of T gondii from faecal samples, according to the manufacturer’s instructions. An in-house PCR test was performed for samples in the remaining time frame between June 2016 and February 2020 using the DNA Process Control Detection Kit (Roche Diagnostics) and primer and probe according to Lin et al. 25 All tests complied with the national requirements for testing epizootic diseases.
Each PCR run included a negative and a positive control. An extraction control for each sample was included for the in-house PCR test according to Lin et al 25 to check for nucleic acid extraction and PCR inhibition (DNA Process Control Detection Kit; Roche Diagnostics). A LightCycler 96 instrument (Roche Diagnostics) or Rotor-Gene (Qiagen) was used for PCR testing. PCR tests were conducted as qualitative assays (negative/positive), with cycle threshold (Ct) values <35 considered positive.
Serological testing
Serological testing using an immunofluorescence antibody test (IFAT) and ELISA was carried out from serum or plasma. Between January 2008 and March 2022, the MegaFLUO TOXOPLASMA IFAT (⩾1:50 classified as positive; MEGACOR Veterinary Diagnostics) was used to detect IgG and IgM antibodies, as described by Verhofstede et al. 26 From April 2022 onwards, the VetLine Toxoplasma ELISA for the detection of IgG (>55 IU/ml classified as positive) and the VetLine Toxoplasma IgM ELISA for the detection of IgM antibodies (>11 NTU classified as positive; NovaTec Immundiagnostica) were used according to the manufacturer’s guidelines, with a reported diagnostic sensitivity and specificity of >98% each (https://www.goldstandarddiagnostics.com/vetline-toxoplasma-96-tests.html), using the automated DSX ELISA system (DYNEX Technologies) and including negative and positive controls. In general, initial samples sent in for each cat were included. If any cat initially tested negative and later tested positive in a follow-up examination, the positive results were included in the statistical analysis. Therefore, to avoid multiple testing results in each cat, only the initial or first positive results were included.
FeLV/FIV testing
Further test results, such as for FeLV antigen and FIV antibodies (NovaTec VetLine Feline Immunodeficiency Virus ELISA and NovaTec VetLine Feline Leukemia Virus Antigen ELISA; NovaTec Immundiagnostica) were also evaluated, if available. For the NovaTec VetLine Feline Immunodeficiency Virus ELISA, diagnostic sensitivity and specificity were reported as 95.5% and 96.3% (https://www.goldstandarddiagnostics.com/vetline-feline-immunodeficiency-virus-fiv.html) and as 100% and 98.5% for the NovaTec VetLine Feline Leukemia Virus Antigen ELISA (https://www.goldstandarddiagnostics.com/vetline-feline-leukemia-virus-felv-antigen-96-tests.html), respectively, according to the manufacturer’s information.
Statistical analysis
Fisher’s exact test was used for categorical parameters to calculate the statistical impact of age (split into age categories of <5 years and ⩾5 years owing to the median of the study population), sex, season and regionality, and FeLV/FIV status on PCR and serological results. Binary bivariate (often also called ‘simple binary bivariate’) logistic regression analysis was performed, and odds ratios (ORs) were calculated. A descriptive statistical analysis was carried out using SPSS for Windows version 29.0 (IBM) and P <0.05 was considered to indicate statistical significance. The 95% confidence intervals (CIs) for the proportions of cats that tested positive were calculated using the Wilson procedure, including correction for continuity.
Results
Signalment
A total of 45,523 cats were included, of which 14,500 (31.9%) tested positive using direct and/or indirect detection methods for T gondii (PCR: 126/7896 [1.6%]; IgG: 14,148/37,882 [37.3%]; IgM: 1539/37,882 [4.1%]) (see Table 1 in the supplementary material). PCR and serological testing were performed concurrently in 255 cats.
Of these 255 cats, 70 (27.5%) tested positive for IgG antibodies. Of those 70 cats, one (1.4%) tested serologically positive and additionally tested positive on PCR, while the remainder of the cats (n = 69, 98.6%) were negative on PCR. Out of 255 cats, 10 (3.9%) tested solely positive for IgM. One cat tested positive on PCR and negative on IgM testing, with the remaining 254 cats testing negative on PCR.
The breed was known in 36,345/45,523 (79.8%) cats (see Table 1 in the supplementary material). By far the largest proportion was European domestic shorthair cats (n = 26,450/36,345, 72.8%), followed by purebred cats (n = 7503/36,345, 20.6%), most often British Shorthairs (n = 2029), Maine Coons (n = 1315), Persians (n = 787), Siamese (n = 435) and Birman (n = 414), and crossbreeds (n = 2392/36,345, 6.6%). A statistically significant impact of breed was found for IgG and IgM (both P <0.001) when dividing cats into purebreds, European domestic shorthair cats and crossbreeds; however, this was not the case for PCR testing (P = 0.123).
The age was known in 35,512/45,523 (78.0%) cats, with a median of 5.0 years (mean [± SD] 5.8 ± 4.8, range 0.2–23.0). A statistically significant impact of age was detected for IgG (P <0.001), with 4428/16,072 (27.6%) cats aged ⩽5 years and 6855/13,811 (49.6%) cats aged >5 years testing positive; and for IgM (P <0.001), with 447/16,072 (2.8%) cats aged ⩽5 years and 731/13,811 (5.3%) testing positive. However, this was not the case for PCR testing (P = 0.292), with 53/3606 (1.5%) cats aged ⩽5 years and 25/2240 (1.1%) cats aged >5 years testing positive.
The sex was known in 38,835/45,523 (85.3%) cats (Table 1). Of these 38,835 cats, 20,326 (52.3%) were male and 18,509 (47.7%) were female. When comparing male and female cats, a statistically significant impact was shown for IgG results (P <0.001), but not for IgM (P = 0.543) and PCR testing (P = 0.291). Neuter status had a statistically significant impact on PCR results (male: P = 0.002; female: P = 0.001) and IgG results (male and female: P <0.001, each); however, this was not the case for IgM (male: P = 0.415; female: P = 1.000).
Sex of cats tested for Toxoplasma gondii using molecular and serological detection methods between January 2008 and December 2022

Data are presented as n/n (%) (95% confidence interval)
Fisher’s exact test
FeLV/FIV status
FeLV antigen testing was positive in 210/5913 (3.6%) cats and FIV antibodies were detected in 344/5760 (6.0%) cats. A statistically significant impact of FeLV was seen on IgG testing (P = 0.032) with 2282/5690 (40.1%) FeLV negative cats testing positive compared with 100/210 (47.6%) FeLV-positive cats. A significant impact was also found on IgM testing (P <0.001), with 202/5405 (3.7%) FIV-negative cats testing positive vs 32/343 (9.3%) FIV-positive cats. In addition, for IgG testing (P <0.001), 2109/5405 (39.0%) FIV-negative cats were positive compared with 206/343 (60.1%) FIV-positive cats. No statistically significant impact was demonstrated for FeLV in IgM testing (P = 0.715; 220/5690 [3.9%] FeLV-negative cats vs 9/210 [4.3%] FeLV-positive cats). As all cats with FeLV and FIV test results were negative on PCR for T gondii, it was not possible to further evaluate the statistical impact of FeLV and FIV infections on the results of molecular testing for T gondii.
Regionality and seasonality
Data regarding regionality and seasonality were available for all 45,523 cats included in the study (Tables 2 and 3). Monthly distribution is demonstrated in Table 2 in the supplementary material. Of the 45,523 cats included in the study, 33,673 (74.0%) were from central Europe, 3609 (7.9%) from southern Europe, 3143 (6.9%) from northern Europe, 2779 (6.1%) from eastern Europe and 2319 (5.1%) from western Europe, spanning 32 countries (Table 3).
Seasonal distribution of molecular and serological test results for Toxoplasma gondii in cats between January 2008 and December 2022

Data are presented as n/n (%) (95% confidence interval)
March to May
June to August
September to November
December to February
Fisher’s exact test
Regional distribution of molecular and serological test results for Toxoplasma gondii in cats between January 2008 and December 2022

Data are presented as n/n (%) (95% confidence interval)
Austria (n = 2696), Switzerland (n = 1049), Germany (n = 29,655)
Czech Republic (n = 1088), Hungary (n = 5), Poland (n = 1120), Slovakia (n = 561)
Denmark (n = 39), Estonia (n = 541), Finland (n = 338), Great Britain (n = 1481), Iceland (n = 3), Ireland (n = 23), Latvia (n = 84), Lithuania (n = 107), Norway (n = 327), Sweden (n = 200)
Bulgaria (n = 194), Croatia (n = 218), Cyprus (n = 18), Greece (n = 52), Italy (n = 1004), Malta (n = 9), Portugal (n = 438), Romania (n = 1150), Serbia (n = 25), Slovenia (n = 166), Spain (n = 340)
Belgium (n = 1851), France (n = 41), Luxembourg (n = 309), Netherlands (n = 118)
Fisher’s exact test
Logistic regression analysis
The results of the simple binary bivariate logistic regression analysis are demonstrated in Table 4, and Table 3 in the supplementary material.
Statistically significant impact of risk factors on molecular and serological test results for Toxoplasma gondii in cats between January 2008 and December 2022 using simple binary bivariate logistic regression analysis (P <0.05)
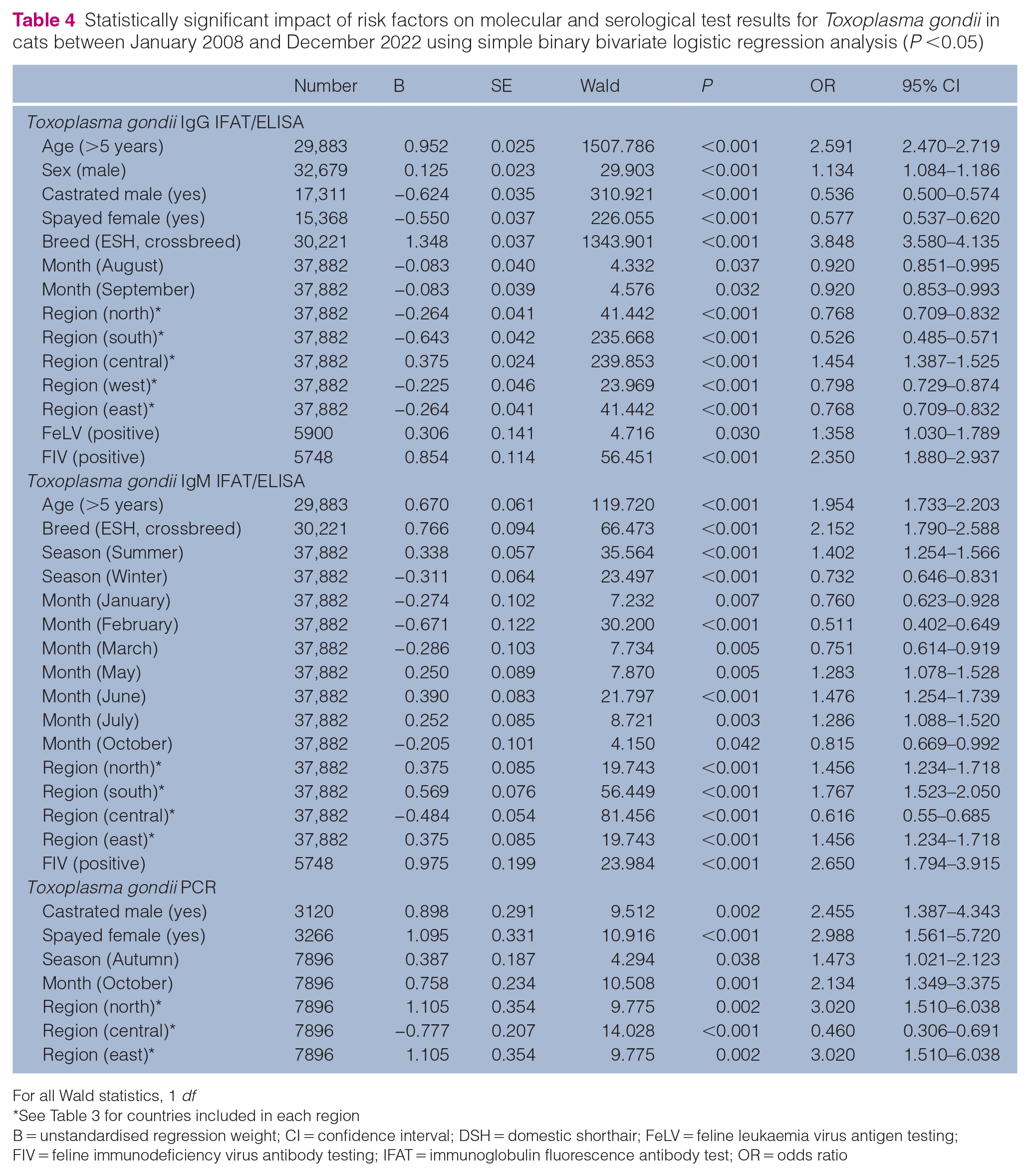
For all Wald statistics, 1 df
See Table 3 for countries included in each region
B = unstandardised regression weight; CI = confidence interval; DSH = domestic shorthair; FeLV = feline leukaemia virus antigen testing; FIV = feline immunodeficiency virus antibody testing; IFAT = immunoglobulin fluorescence antibody test; OR = odds ratio
Discussion
This study represents one of the largest evaluations of molecular and serological detection of T gondii in cats in Europe, including an analysis of several risk factors. The global serological prevalence in domestic cats was reported to be 35% (95% CI 32–38) 27 and 37.5% (95% CI 35–40). 28 This is in accordance with the 37% of cats that tested IgG positive (95% CI 37–38) in our study. The global pooled prevalence of oocysts in the faeces of domestic cats was reported to be 2.6% (95% CI 2–3%), 28 which was slightly higher when compared with our data of 1.6% (95% CI 1–2). However, when compared with other reports from Germany and Switzerland, our data show relatively high rates of positive molecular test results (0.4% in Switzerland 18 and 0.25% in Germany 17 ). Positive serological test results indicate previous contact with T gondii, whereas the positive molecular detection of T gondii in the faeces correlates with the shedding of oocysts. Therefore, the high rates of cats that tested serologically positive indicate zoonotic potential, while the number of cats shedding oocysts directly represents an important threat to animal and public health. Almost 30% of the global human population is serologically positive for T gondii, indicating exposure to the pathogen. In human medicine, the prevalence of T gondii varies across different countries and regions, 27 which is reflected in this study’s molecular and serological results for cats as well. The seroprevalence of T gondii was reported to be highest in Eastern Europe, followed by the Southeast and Southwest regions, while the Northern and Western regions show the lowest rates. 29 This is in accordance with our study, in which the lowest percentages of cats that tested IgG positive were detected in northern, southern and western Europe (Table 3). In general, toxoplasmosis is more prevalent in moist, warm regions at low altitudes, 30 which might be consistent with the countries in central and eastern Europe. In our study, the regional analysis was based on the postal code of the veterinarian submitting the faecal and/or serum/plasma samples. We assumed that these reflect the areas where the cats were kept, as more precise data on the cats’ origins were not available. The regional analysis was most likely not affected as the data were clustered based on countries in Europe and therefore on a larger spatial scale than the postal codes.
Infections in humans and cats are usually asymptomatic. In congenital cases or in immunosuppressed individuals, clinically evident and harmful effects are reported,31,32 which is consistent with the higher odds for positive serology in cats with FeLV and FIV infection in our study. Therefore, from a One Health perspective, surveillance of toxoplasmosis is recommended to reduce animal and human infections. 28
Climate changes influencing the ecology of (transport) hosts are also considered to affect the seroprevalence of T gondii. 30 In a 10-year study of an urban population of domestic cats in France, the prevalence of antibodies against T gondii was related to the interaction between temperature and rain. 33 Although we were not able to correlate our data to climate factors, the existing impact of regionality on molecular and serological test results in our study opens doors for further investigation. Shedding in cats is more common between July and December. 17 In our study, there was a 46% increase in the odds of positive molecular detection of T gondii in autumn and a 40% increase in the odds of detecting IgM antibodies in the summer, which correlates with the findings reported in the literature.
Our data should not be misinterpreted as prevalence data for Europe, as the samples were submitted to a commercial laboratory without any accompany anamnesis. The study population was therefore preselected by the submitting veterinarians for various reasons, including clinical suspicion of toxoplasmosis and the need for clarification of zoonotic potential. Nevertheless, it is highly probable that the serological testing in our study was requested for screening purposes, while PCR testing aimed to identify cats shedding oocysts. This explains the concordance of our serological data with findings from two systematic global reviews.27,28
According to federal law in Germany, feline toxoplasmosis is classified as a notifiable disease. Consequently, if the diagnostic assay is approved by the authority and available, a PCR test kit that is approved by the Friedrich-Loeffler-Institut must be used. Alternatively, it is permitted to use a PCR protocol based on prior scientific publications. In-house validation showed a comparable performance between the approved test kits and the in-house PCR tests.
The two different diagnostic assays used for the detection of anti-T gondii antibodies in our study showed comparably good diagnostic performances. The IFAT used in the present study has the same relative sensitivity (94%) and specificity (100%) compared with ELISA testing, as well as the same positive (100%) and negative (98%) predictive value, with an accuracy of 99% each. 34 In addition, the short time frame of 9 months using ELISA testing most likely did not significantly influence the rates of positive serology in our study.
It has been reported that the age of the cat does not have a significant impact on T gondii shedding. 17 Our data confirm this finding. However, a statistically significant correlation between antibody detection and increasing age was demonstrated in cats in Poland. 35 In our study, cats aged >5 years had a more than doubled odds (OR 2.591) for positive IgG antibody detection compared with younger cats. This is in accordance with the reported data from Poland and can be explained by an increasing possibility for pathogen contact during the life of the cat and the prolonged persistence of IgG antibodies. 24
One study reported that sex does not affect the serological prevalence of T gondii. 35 In contrast, our study found that male cats had a 13% higher likelihood for testing positive for IgG compared with females, which may be explained by the higher activity levels in male cats. However, the impact of neuter status on the detection of IgG antibodies and PCR results in male and female cats remains unclear, as the findings were contradictory.
The risk of acquiring T gondii infection is higher in animals kept outdoors and the difference in T gondii prevalence between animals kept indoors or outdoors has been well documented in various species.36–38 A recent literature review detected a higher seroprevalence of T gondii in European felids with outdoor access (54.1%) compared with felids kept indoors (31.9%). 29 Cats that are kept outdoors experience a higher infectious pressure, mostly caused by environmental contamination with oocysts and the presence of potentially infected rodents and birds that act as prey.39–41 The higher odds seen in European domestic shorthair cats and crossbreeds in our study compared with purebreds may be explained by the fact that free roaming is less common in purebred cats. The same can be observed for larger cities, where cats tend to be kept indoors more than in rural areas, resulting in a lower risk of pathogen exposure in cities compared with rural areas. As a result of the retrospective study design, it was not known if the cats included in this study were privately owned, stray cats or living in shelters, which may have especially impacted the percentages of cats that tested serologically positive for T gondii as well as the results for FeLV and FIV testing.
Clinical toxoplasmosis has been documented in cats infected with FIV or FeLV, 20 similar to findings in people with human immunodeficiency virus (HIV). 31 No significant effect on shedding was demonstrated in our study, likely due to the relatively low numbers of cats that tested PCR positive. Since none of the cats that tested positive by PCR were also positive for FeLV and/or FIV in our study, we were not able to draw any conclusions regarding the outcome of these infections on shedding. FeLV and FIV can predispose cats to reactivate infections,20,21 which may explain the 2.7-fold higher odds of detecting IgM antibodies in cats with FIV.
Conclusions
More than one-third of the domestic cats in our study were exposed to T gondii and 1.6% shed oocysts. In their role as definitive and final hosts of T gondii, felids are of major importance in the epidemiology of toxoplasmosis. Studies on the prevalence of feline toxoplasmosis are important to estimate the ongoing threat to animal and public health in the context of the One Health approach. Several risk factors, such as breed, age, sex, neuter status, seasonality and regionality were identified for serological testing (cats that tested positive had been exposed to the pathogen). Risk factors, such as neuter status, seasonality and regionality for PCR testing from faeces (cats that tested positive were shedding oocysts), were described further, all of which potentially contribute to a better understanding of the pathogenesis of feline toxoplasmosis. The role of FeLV and FIV infection in the context of epidemiology as well as clinical disease should be further investigated.
Supplemental Material
sj-docx-1-jfm-10.1177_1098612X241281223 – Supplemental material for Serological and molecular detection of Toxoplasma gondii in cats in Europe with evaluation of associated risk factors for pathogen contact/infection
Supplementary Table 1: Molecular and serological test results for Toxoplasma gondii in purebred cats (n ⩾10) between January 2008 and December 2022 (n/N (%) [95% CI lower bound, 95% CI upper bound])
Supplementary Table 2: Molecular and serological test results for Toxoplasma gondii in cats between January 2008 and December 2022 sorted by months (n/N (%) [95% CI lower bound, 95% CI upper bound])
Supplementary Table 3: Binary bivariate logistic regression analysis of molecular and serological test results for Toxoplasma gondii in cats between January 2008 and December 2022
Footnotes
Supplementary material
The following files are available as supplementary material:
Supplementary Table 1: Molecular and serological test results for Toxoplasma gondii in purebred cats (n ⩾10) between January 2008 and December 2022 (n/N (%) [95% CI lower bound, 95% CI upper bound])
Supplementary Table 2: Molecular and serological test results for Toxoplasma gondii in cats between January 2008 and December 2022 sorted by months (n/N (%) [95% CI lower bound, 95% CI upper bound])
Supplementary Table 3: Binary bivariate logistic regression analysis of molecular and serological test results for Toxoplasma gondii in cats between January 2008 and December 2022
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
