Abstract
Objectives
The aim of the study was to analyze the association of the administration of liposomal bupivacaine (Nocita) with postoperative opioid use in cats undergoing gastrointestinal surgery, as well as to analyze the administration of antiemetics postoperatively in this study population to qualify the degree of nausea related to opioid administration.
Methods
Records of 187 cats undergoing gastrointestinal surgery across two institutions, the Matthew J Ryan Veterinary Hospital at the University of Pennsylvania and the University of Florida Small Animal Hospital, were analyzed for multilayer liposomal bupivacaine infiltration at the time of abdominal closure, postoperative injectable opioid use and postoperative injectable antiemetic use.
Results
Cats that were administered liposomal bupivacaine at the time of abdominal closure received significantly lower doses of full µ-opioid agonists postoperatively. Significantly more cats that received liposomal bupivacaine were managed with full µ + partial µ-opioid agonists postoperatively than any other opioid profile. Cats that received liposomal bupivacaine received lower doses of ondansetron in hospital compared with cats that did not receive liposomal bupivacaine.
Conclusions and relevance
This study suggests that in cats requiring analgesia postoperatively, the use of liposomal bupivacaine at closure reduces the use of full µ-opioid agonists postoperatively. In addition, more patients that received liposomal bupivacaine were de-escalated from full µ-opioid agonists to partial µ-opioid agonists during their hospital stay, suggesting that less analgesia was required throughout their hospitalization. Lastly, lower doses of ondansetron, a typical second-line antinausea medication after gastrointestinal surgery, were used in cats that received liposomal bupivacaine, which may suggest less postoperative nausea.
Plain language summary
This study analyzed the association between the long-acting local anesthetic, liposomal bupivacaine (Nocita) and the use of opioid pain medication postoperatively for cats undergoing gastrointestinal surgery. Records of 187 cats undergoing gastrointestinal surgery across two institutions, the Matthew J Ryan Veterinary Hospital at the University of Pennsylvania and the University of Florida Small Animal Hospital, were analyzed for liposomal bupivacaine administration, postoperative injectable opioid use, postoperative injectable antiemetic use and length of hospitalization. In cats that required pain medications, those that were administered liposomal bupivacaine at the time of abdominal closure received significantly lower amounts of first-line injectable opioid pain medications postoperatively. Moreover, those that received liposomal bupivacaine were de-escalated from first- to second-line opioid pain medications postoperatively more commonly. In addition, cats that received liposomal bupivacaine received less second-line antinausea medications compared with cats that did not receive liposomal bupivacaine. There was no significant difference in the length of hospitalization between cats that did and did not receive liposomal bupivacaine. This study suggests that in cats requiring pain medications postoperatively, the use of liposomal bupivacaine at closure reduces the use of first-line injectable opioids postoperatively.
Keywords
Introduction
Gastrointestinal surgery in cats is extremely common. Postoperative care after gastrointestinal surgery focuses on analgesia and support of the gastrointestinal tract, including the use of antiemetics and other medications to decrease ileus and regurgitation. Analgesia after gastrointestinal surgery is inherently complex. Non-steroidal anti-inflammatory drugs (NSAIDs) and corticosteroids both have inherent gastrointestinal side effects, most notably gastrointestinal ulceration risk and delayed wound healing. Opioids are commonly used in the immediate postoperative period; however, they have been shown to delay gastric motility and cause nausea. 1 Delayed gastric motility and postoperative nausea and regurgitation correlate to an extended hospital stay and increased financial burden for the client. One study demonstrated that of dogs that received methadone after stifle surgery, 33% were reported to experience nausea. 2 Another study found that the incidence of regurgitation and vomiting in dogs was significantly higher in dogs undergoing soft tissue surgery, specifically gastrointestinal surgery, and higher in dogs receiving opioids postoperatively. 3 In addition, the opioid crisis in the USA has led to more cautious use of opioids in hospital settings, as well as a decreased availability of these medications.4,5 It is particularly important, then, to research potentially opioid-sparing medications in the postoperative period of animals undergoing gastrointestinal surgery.
The use of injectable liposomal bupivacaine has increased in veterinary medicine since its development in 2011 and approval by the US Food and Drug Administration (FDA). Similar medications are used in human medicine and the opioid-sparing effect of liposomal bupivacaine in the postoperative period is well documented.6–9 The FDA-approved product, Nocita (Aratana Therapeutics), has been approved for a single dose by infiltration injection at the time of incisional closure of cranial cruciate ligament surgery and feline onychectomy.10–12 Single dose infiltration via injection of Nocita is widely used off-label in veterinary surgery in both elective and emergency surgery. The efficacy of Nocita, particularly as it pertains to its opioid-sparing effect, in the immediate postoperative period is debated. Multiple clinical trials have been performed to assess the efficacy of Nocita based on the need for rescue analgesia in the form of opioids compared with both placebos and other bupivacaine formulations. The need for rescue analgesia in these studies was determined on the basis of either the Colorado State University Feline Acute Pain Scale (CSU-FAPS) or Glasgow Composite Measure Pain Scale-Short Form (CMPS-SF).13,14 One study concluded that in dogs undergoing a lateral retinacular suture placement for a cranial cruciate tear, the use of rescue analgesia (based on CSU-CAPS) was significantly lower in dogs that had received Nocita. 15 Another study, using CSU-CAPS and CMPS-SF, found rescue analgesia and opioid consumption were lower in dogs receiving Nocita after a tibial plateau leveling osteotomy (TPLO). 15 A third study found that dogs that had received Nocita, when compared with dogs that had received injectable 0.5% bupivacaine hydrochloride, had a decreased incidence of requiring rescue analgesia and received less opioids overall postoperatively after a TPLO. 16 On the contrary, one study found that Nocita did not provide a decrease in opioid consumption and decreased pain scores (CMPS-SF) in dogs undergoing a TPLO when compared with a group of dogs receiving a saline placebo. 12 A more recent study described an opioid-sparing effect of Nocita in the context of gastrointestinal surgery in dogs, publishing that postoperative hourly fentanyl use in dogs after foreign body surgery was significantly lower in dogs that received Nocita. 15 However, another study found that there was no difference in pain scores, electric algometer readings and need for rescue analgesia between dogs undergoing abdominal surgery that had received Nocita and those dogs that had received a saline placebo. 17 No studies have reported the opioid-sparing property of Nocita in the postoperative period in cats undergoing gastrointestinal surgery.
The aims of this study were, first, to analyze postoperative opioid use in cats receiving Nocita after gastrointestinal surgery; and second, to analyze the use of antiemetic medications in animals receiving Nocita after gastrointestinal surgery. We hypothesized that patients that received Nocita would receive fewer opioids and antiemetics. The objective of this study was to allow for further analysis of the efficacy of off-label use of Nocita after gastrointestinal surgery in cats.
Materials and methods
Data collection
The medical records of cats that underwent gastrointestinal surgery at the Matthew J Ryan Veterinary Hospital of the University of Pennsylvania and the University of Florida Small Animal Hospital between 1 July 2020 and 1 April 2023 were evaluated. Cats were included if they underwent any procedure in which a full-thickness incision was made into the stomach, small intestine or large intestine. Surgeries were performed by surgical residents with or without supervision of board-certified surgeons, as well as by board-certified surgeons. Patients recovered postoperatively in patient care wards or the intensive care unit. Postoperative treatment orders were determined by the primary surgeon (either a house officer under the supervision of a board-certified surgeon or a board-certified surgeon). Based on the treatment orders at both institutions, rescue analgesia was given if there was a pain score greater than 2 (based on the CSU-CAPS).
Subjects were divided into two groups: those that received Nocita at the time of surgery and those that did not. Indication for surgery, surgery performed, average injectable postoperative use of opioids (in total mg/kg) administered during hospitalization, average injectable maropitant and ondansetron use, and postoperative use of opioids (in total mg/kg) administered during hospitalization were recorded.
Statistical analysis
All descriptive data were generated, and statistical tests were performed using Python 3 (version 3.9.6) with the Pandas (version 1.4.1), SciPy (version 1.11.1) and Statsmodels (version 0.14.0) libraries. The variables of interest were assessed and found to be non-normally distributed using a combination of the Shapiro–Wilk test and quantile-quantile plots. Patient groups were stratified based on surgery indication, procedure, drug profiles and antiemetic profiles, and relationships between Nocita usage and stratification were interrogated for significance using χ2 tests. Length of hospitalization and dose distribution comparisons between different groups were evaluated using the Mann–Whitney U-test, with dose considered both in terms of cumulative dose administered over entire hospitalization (ie, total mg/kg) as well as dose normalized to length of hospital stay (ie, mg/kg/day) to account for varying hospitalization periods. Statistical significance was defined using a level of α = 0.05.
Results
In total, 187 cats underwent gastrointestinal surgery within this study period. Surgery for foreign body obstruction and gastrostomy represented the most common indication and procedure carried out, respectively (see Tables 1 and 2). Tables 3 and 4 show the cumulative average use (mg/kg) during hospitalization across all opioid profiles of injectable full µ-opioid agonists in the form of intermittent boluses (methadone), partial µ-opioid agonists (buprenorphine), full µ-opioid agonists in the form of a constant rate infusion (fentanyl) and antinausea medications (maropitant and ondansetron) for cats that did and did not receive Nocita after gastrointestinal surgery. Cats that received Nocita received significantly higher cumulative doses (mg/kg) of partial µ-opioid agonists (buprenorphine) (see Tables 3 and 4). Cats that did not receive Nocita most frequently received full µ-opioid agonists (methadone) alone postoperatively, while those that had received Nocita most frequently received both full µ-opioid agonists (methadone) and partial µ-opioid agonists (buprenorphine) postoperatively (see Table 5). Different classes of opioids received by cats that did and did not receive Nocita after gastrointestinal surgery are shown in Table 5. Cats that received Nocita and were given intermittent boluses of full µ-opioid agonists postoperatively received significantly lower doses (mg/kg) of these (see Table 6).
Indications for surgery for cats undergoing gastrointestinal surgery

Data are presented as n (%)
Surgical procedures for cats undergoing gastrointestinal surgery

Data are presented as n (%)
Comparison of average postoperative injectable opioid and antiemetic use in cats undergoing gastrointestinal surgery that did and did not receive Nocita at the time of closure

Statistically significant result. A cut-off value of P <0.05 was used to determine statistical significance
†Bolused full µ-opioid: P = 0.0004
‡Partial µ-opioid: P = 5.71 × 10–8
§Maropitant: P = 0.004
CRI = constant rate infusion
Comparison of average postoperative injectable opioid and antiemetic use in cats undergoing gastrointestinal surgery that did and did not receive Nocita at the time of closure, excluding patients that did not receive opioids postoperatively
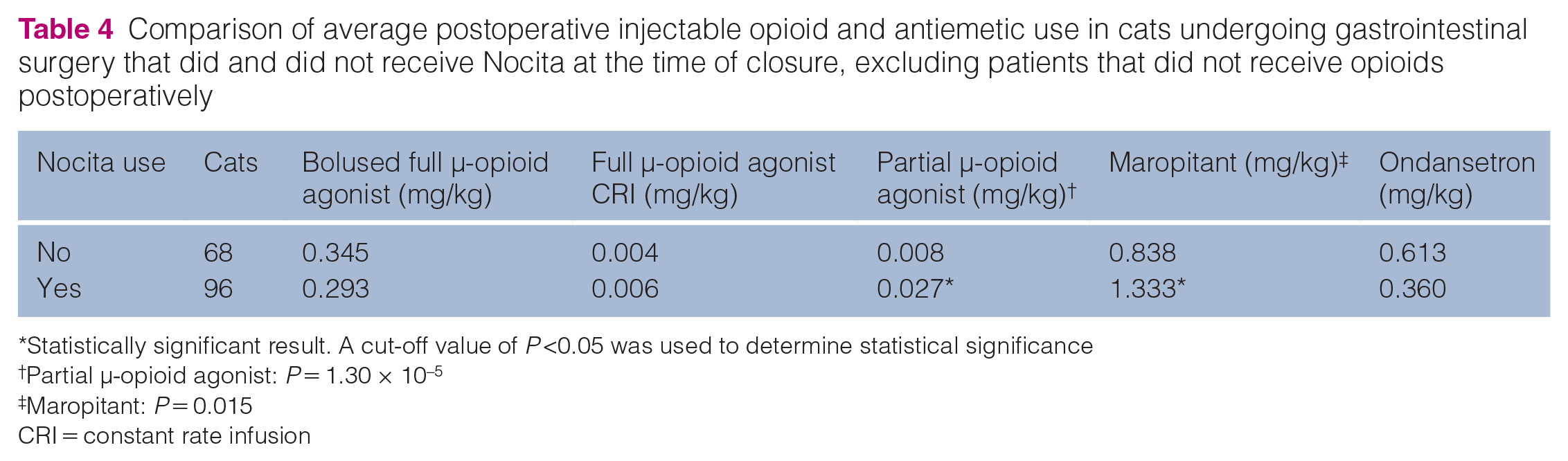
Statistically significant result. A cut-off value of P <0.05 was used to determine statistical significance
†Partial µ-opioid agonist: P = 1.30 × 10–5
‡Maropitant: P = 0.015
CRI = constant rate infusion
Types of opioids received by cats after gastrointestinal surgery

Data are presented as n (%)
CRI = constant rate infusion
Comparison of average postoperative dose of intermittent bolused full µ-opioid agonists in cats only receiving full µ-opioid agonists postoperatively

Statistically significant result. A cut-off value of P <0.05 was used to determine statistical significance
†Bolused full µ-opioid: P = 0.01
Compared with cats that did not receive Nocita, a significantly greater proportion of cats that received Nocita were initially managed on full µ-opioid agonists (methadone) and subsequently de-escalated to partial µ-opioid agonists (buprenorphine) during their hospital stay (P <0.001). In only the cats that received Nocita and were de-escalated from full µ-opioid agonists to partial µ-opioid agonists, the dose of full µ-opioid agonists (mg/kg) administered was significantly lower compared with cats that were only on full µ-opioid agonists postoperatively (see Table 7). Similarly, in only the cats that received Nocita and were de-escalated from full µ-opioid agonists to partial µ-opioid agonists, the dose of partial µ-opioid agonists (mg/kg) was significantly lower compared with cats that were only on partial µ-opioid agonists postoperatively (see Table 8).
Comparison of average postoperative dose of intermittent bolused µ-opioid agonists for cats receiving only full µ-opioid agonists and full µ and partial µ-opioid agonists postoperatively

Statistically significant result
A cut-off value of P <0.05 was used to determine statistical significance
Full µ-opioid agonist use for full and partial µ-opioid: P = 0.04
Comparison of average postoperative dose of partial µ-opioid agonists for cats receiving only full and partial µ-opioid agonists and full µ and partial µ-opioid agonists postoperatively

Statistically significant result
A cut-off value of P <0.05 was used to determine statistical significance
Partial µ-opioid use for partial and full agonists: P = 0.03
Of the 89 cats that did not receive Nocita, 31 (35%) received maropitant only, 13 (14%) received maropitant and ondansetron, nine (10%) received ondansetron only and 36 (40%) received neither. Of the 98 cats that received Nocita, 50 (51%) received maropitant only, 20 (20%) received maropitant and ondansetron, four (4%) received ondansetron only and 24 (24%) received neither. Patients that were administered Nocita received significantly more maropitant in mg/kg postoperatively than those that did not receive Nocita (P <0.001). Excluding patients that received no opioids postoperatively from both groups, patients receiving Nocita received significantly more maropitant than those that did not receive Nocita (P = 0.015).
Discussion
This retrospective study found that full µ-opioid agonist requirements may be lower postoperatively in cats that received Nocita at the time of abdominal closure. The information presented in this manuscript is novel in feline medicine and surgery and builds upon previously published work in dogs. This is the only study to date evaluating postoperative opioid requirements in cats receiving Nocita after gastrointestinal surgery.
Recent studies have published conflicting results on the opioid-sparing effect of Nocita for dogs undergoing foreign body and abdominal surgery. In one retrospective study, an opioid-sparing effect of fentanyl use only for dogs undergoing foreign body surgery was demonstrated. 18 In contrast, no opioid-sparing effect or difference in pain scores postoperatively was demonstrated for dogs undergoing abdominal surgery receiving methadone depending on pain score in a prospective study. 17 No studies have analyzed an opioid-sparing effect, if present, in cats. This study demonstrated that when intermittent doses of full µ-opioid agonists were used exclusively, the average dose administered (mg/kg) for cats that underwent gastrointestinal surgery and received Nocita was significantly lower than cats that did not receive Nocita. The majority of cats during this time frame, both those that did and did not receive Nocita, received only intermittent boluses of full µ-opioid agonists postoperatively. The lack of any significant findings within other opioid profiles may be related to sample size and type II error, which emphasizes the importance of future prospective studies with more subjects receiving consistent dosages across multiple opioid profiles.
Cats that received Nocita were significantly more likely to receive both full µ-opioid agonists and partial µ-opioid agonists during their hospitalization. This is likely the reason that cats receiving Nocita received higher cumulative doses of partial µ-opioid agonists across all opioid profiles. This population of patients were also administered a statistically significantly lower dose of full µ-opioid agonists. The introduction of partial µ-opioid agonists in this study represents a de-escalation of analgesic requirements, especially when considering the relative efficacy and analgesia provided by a partial µ-opioid agonist compared with a full µ-opioid agonist.19,20
Cats receiving Nocita received significantly more maropitant but received less ondansetron postoperatively. Maropitant was used more commonly at these institutions and therefore may represent a first-line antiemetic, while ondansetron may be more commonly used as a second-line antiemetic in cases of breakthrough nausea. Although opioids like methadone and buprenorphine are less associated than others with nausea and hypersalivation in cats, the addition of ondansetron may represent either breakthrough nausea from these drugs or be associated with the surgery or disease process itself that necessitated surgery. In addition, it was noted that maropitant was used empirically in a large number of patients at both institutions, whereas ondansetron may have been used as an adjunct. Therefore, it is hard to elucidate whether there is a direct association between the postoperative use of antiemetics and Nocita, as it is likely there was a large amount of bias owing to clinician preference in this data set. These findings, however, may prompt clinicians to critically evaluate patients postoperatively and ensure they demonstrate a need for antiemetics before empiric administration.
The limitations of this study are inherent in its retrospective nature. The data obtained and discussed here were reliant on accurate and consistent pain scoring of patients in a clinical setting resulting in appropriate administration of postoperative analgesics. Although treatment sheets in both facilities included the instruction to only administer an injectable opioid if there was a pain score greater than 2 (based on the CSU-CAPS), the retrospective nature of this study prohibits assurance of adequate training of staff members to make that assessment. In addition, the choice to give Nocita in this study was at the clinician’s discretion, as patient selection was not blinded, such that certain case-specific factors may have made it more likely that one patient would receive Nocita compared with another. Further, treatment orders were at the discretion of multiple clinicians, leading to possible inconsistencies in dosages set and frequency of rescue analgesia.
Anxiety and fear can often manifest similarly to pain and nausea (eg, lip licking) in cats and misidentification of these behaviors can result in over- or under-medication of a clinical patient and could have artificially altered the differences observed between the patients receiving Nocita and those that did not. Postoperative housing may have also played a role in the misidentification of behaviors, as some cats, depending on their clinical status, may have been housed in areas with higher traffic or noise, potentially leading to additional stressors. Another limitation was that preoperative pain scoring and perioperative analgesia (including regional nerve blocks and pre-medications) were not specifically analyzed for each patient, as both preoperative pain and multimodal analgesia during surgery may have impacted pain postoperatively in this study population. There were no specific, consistent criteria delineated on the treatment sheet to determine when to administer antinausea medications, meaning that the administration of these medications was a result of clinician preference. A prospective study in which the criteria are better standardized for not only both postoperative analgesic and anti-nausea medication administration, but also peri- and intraoperative analgesia, is needed to draw specific conclusions about the influence of Nocita on postoperative nausea and the overall opioid-sparing effect. Other future studies should evaluate whether incision length and dilution impact the effectiveness of bupivacaine liposomal injectable suspension, the impact of surgical time and if the relative surgical invasiveness plays a role in the opioid-sparing effect of Nocita.21,22
Conclusions
This study demonstrated a potential opioid-sparing effect of Nocita in cats needing full µ-opioid agonists in the postoperative period after gastrointestinal surgery. In addition, a combination of full µ-opioid agonists and partial µ-opioid agonists were used in a significantly higher proportion of animals that received Nocita, suggesting a de-escalation of analgesia in these cases.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken. No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
