Abstract
Case series summary
Three skeletally immature cats were treated for pectus excavatum using a modified video-assisted thoracoscopic surgery (VATS) technique that involved using a thoracoscopic cannula to intraoperatively ventrally displace affected sternebrae, thereby assisting the placement of circumsternal sutures while simultaneously protecting underlying viscera. All cases recovered well from their procedure, and the described technique was successful in allowing good radiographic improvement of pectus excavatum in all cases. No intraoperative complications were encountered and all cases were judged as having an excellent outcome.
Relevance and novel information
Routine VATS-assisted correction of pectus excavatum is possible in cats but may not confer any obvious advantage over traditional ‘blind’ placement of sutures owing to the difficulty in visualising the target sternebrae during surgery. The modification described here, in which the sternum is pushed ventrally, makes subsequent placement of percutaneous sutures easier while simultaneously reducing the risk of viscus penetration. It may therefore reduce the risks inherent in this procedure and has potential to be the treatment of choice for this condition.
Plain language summary
Funnel chest can be treated by using a splint to realign the breastbone. This can be done either ‘blind’ or by using keyhole surgery to guide the passage of sutures (stitches) around the breastbone. Keyhole surgery can be very tricky in such small, young patients, so we have modified the technique and now use one of the instruments to help ‘push’ the breastbone into a normal position. This then makes it easier and potentially safer to pass the suture around. We report using this new technique in three kittens, all of which went on to do very well.
Introduction
Pectus excavatum (PE) is an uncommon congenital condition that has been widely reported in humans, dogs and cats.1 –4
The severity of PE has been traditionally assessed using radiographic indices such as the vertebral (VI) and frontosagittal (FSI) indices; 1 however, this may not correlate well with the severity of clinical signs. Further assessment of the nature of the defect using CT has been reported, and this can give information regarding lateralisation/asymmetry of the displaced sternebrae, which may provide more useful information concerning the likelihood of direct cardiac compression and associated diastolic restriction than reliance on VI and FSI alone.5,6
Clinical signs of PE typically include coughing, cyanosis, anorexia, weight loss, exercise intolerance, cardiac murmur and respiratory distress/dyspnoea, the severity of which is believed to correlate with the degree of cardiopulmonary compromise caused by the abnormality.5,7 –9
The corrective surgery most commonly performed in skeletally immature cats is placement of a sternal cast secured to the sternebrae by placement of circumsternal sutures that apply ventral traction to the displaced sternebrae as they are fastened. The cast is typically maintained for 4–5 weeks, during which continued growth occurs resulting in a long-term correction of the PE deformity, alleviation of clinical signs and prevention of future complications such as pulmonary hypertension.
Placement of circumsternal sutures during cast application is usually performed blindly and may therefore be associated with a risk of vascular or cardiac puncture, given the proximity of these structures to the sternebrae. Video-assisted thoracoscopic surgery (VATS) is now used in humans to decrease the risk of intrathoracic complication associated with PE correction.10,11 VATS may therefore provide an opportunity to decrease these risks in feline patients. VATS-assisted PE repair has been reported in a dog 12 as well as in two cats.13,14
It can be challenging to perform VATS in juvenile feline PE patients because of their smaller size and the further decreased thoracic volume seen in severely affected kittens. We have used VATS to assist PE correction in several cases at our institution; we have found that it can be very challenging to maintain visualisation of the sternebrae and to directly guide suture placement in these patients. This has led to significantly prolonged surgical times than when routine, blind placement of sutures has been performed for similar cases that typically take <30 mins (unpublished data). This has led to us modifying our technique of VATS-assisted PE correction in cats so that the procedure remains safe and potentially advantageous compared with routine circumsternal suture placement, even when the circumsternal sutures cannot be directly visualised with the thoracoscope. We present a case series of three skeletally immature cats that underwent correction of PE using this modified technique in which the thoracoscopic cannula is used to directly push displaced sternebrae ventrally.
Case series description
Three client-owned kittens were referred for investigation and treatment of PE and underwent modified VATS-assisted correction between 2023 and 2025. The initial diagnosis was made based on physical examination, including the presence of a palpable dorsal deviation of the sternebrae. This was further characterised by thoracic radiography, which was used to determine the VI and FSI, as previously reported. The severity of clinical signs was graded using a previously published scheme 9 and this, together with the VI/FSI, was used to determine whether surgical intervention was warranted. Further diagnostics, including serum biochemistry/haematology, testing for feline immunodeficiency virus/feline leukaemia virus, CT and echocardiography, were performed based on the results of clinical examination and clinician discretion.
Once anaesthetised, the kittens were placed into dorsal recumbency and a sternal cast was then fashioned using Delta-cast Elite (BSN Medical), taking care so that the caudal portion of the cast matched the desired, not the actual, conformation of the sternum. The ventral thorax and cranial abdomen were then clipped and prepared for surgery using chlorhexidine gluconate 4% (antimicrobial skin cleanser, Hibiscrub) and the final skin preparation was performed using chlorhexidine gluconate and isopropyl alcohol (Chloraprep 3 ml applicator; Becton Dickinson) once the kitten had been moved into theatre.
A 5 mm incision was made dorsal to the costochondral junction over the right sixth intercostal space. A rigid, 70 mm, 5 mm ribbed plastic optical trocar/cannula (VersaOne; Medtronic) was then used to enter the thoracic cavity with a 5 mm 30° oblique, 29 cm telescope (Karl Storz) loaded within the assembly with the valve open to establish pneumothorax. Cases were ventilated with respiratory function closely monitored using capnography, pulse oximetry, spirometry and blood gas analyses when indicated. The trocar was removed once access had been achieved and the thorax assessed using the telescope. The mediastinum was fenestrated using the end of the telescope as previously described. 12 The cannula was then positioned directly under the point of maximum sternal deformity, the telescope was withdrawn within the proximal 25% of the cannula and the cannula itself was then used to push the deviated sternebrae dorsally, taking care to avoid any lung/tissue becoming entrapped between the cannula and the sternebrae (Figures 1 and 2). This was achieved by retracting the external portion of the cannula ventrally, taking care to avoid using the telescope itself to push against the sternebrae. 3M polypropylene (Prolene; Ethicon) circumsternal sutures were then placed percutaneously using the underlying cannula as a barrier to inadvertent visceral trauma. Visualisation through the transparent cannula was felt to be adequate to ensure that the sternebrae were being adequately retracted away from the underlying viscera. The cannula was withdrawn and the telescope was advanced after each suture had been placed to confirm correct placement of sutures. Not all sutures were visible and many were thought to be trans-sternal/subpleural; however, traction on these sutures after placement confirmed adequate purchase had been achieved.

Schematic clarifying surgical technique. (a) The scope is within the cannula underlying the dorsally (downwards) displaced sternebra. The cannula is advanced forwards (arrow) and the telescope is retracted. (b) The cannula is then pushed ventrally (upwards) to push the sternebra into a more normal position and also act as a guard for the underlying viscera. (c) Circumsternal sutures can now be more easily placed as the sternum will resist dorsal displacement and the underlying viscera are protected from iatrogenic damage
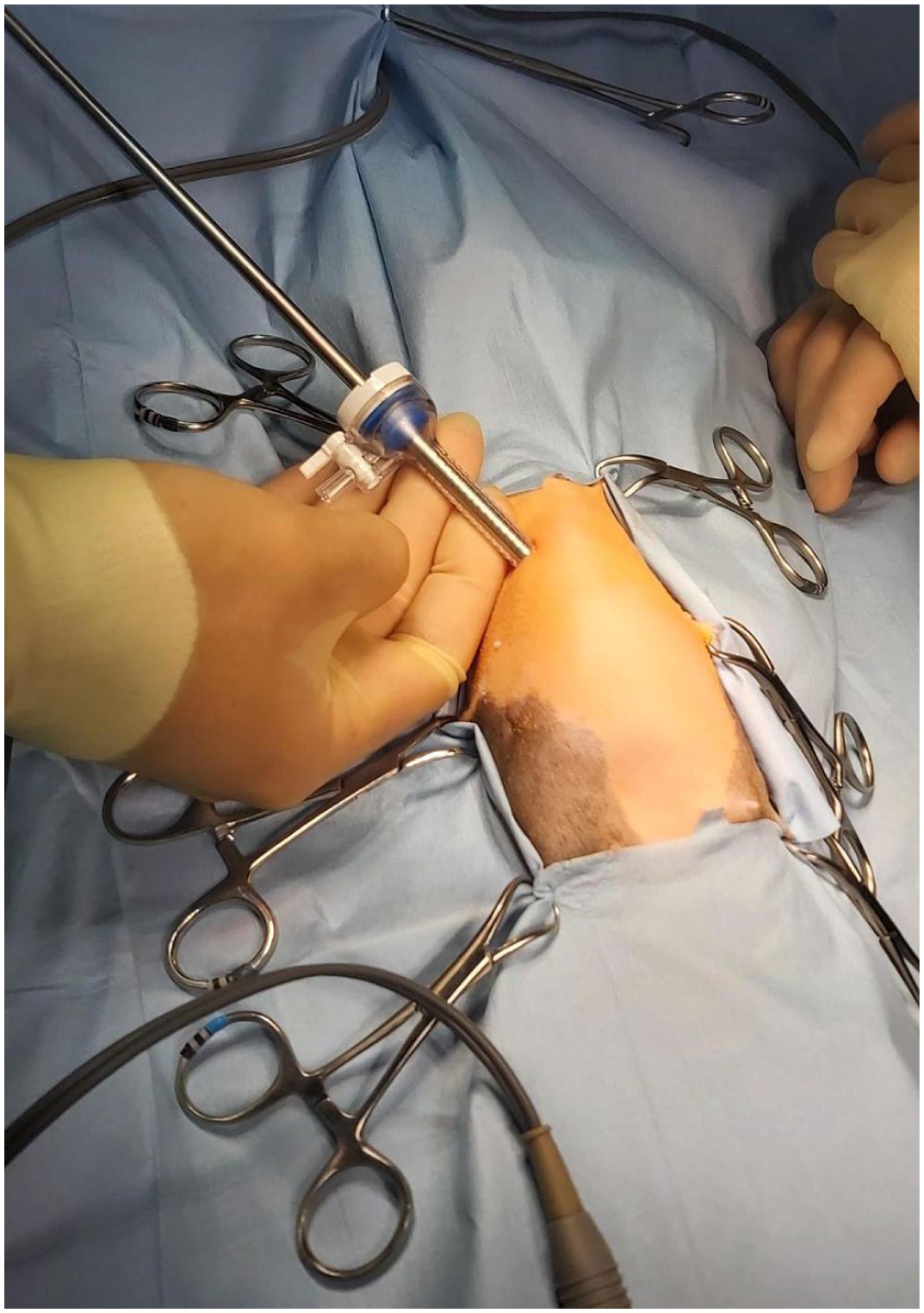
The cannula being used to ventrally deviate the abnormal sternebrae (cranial is to the top of the picture)
Once six or seven sutures had been placed, the cannula was then advanced until it was in contact with the contralateral (left) thoracic wall where it could be palpated. The telescope was again withdrawn to the proximal 25% and a percutaneous catheter advanced into the end of the cannula to enable safe introduction of a Seldinger chest drain (14 ga, 20 cm, Mila; DMS) (Figures 3 and 4). Gradual withdrawal of the cannula allowed release of the guidewire into the pleural cavity and subsequent drain advancement.

The Seldinger catheter is placed percutaneously into the end of the cannula, which can be palpated through the thoracic wall

The catheter is visible within the cannula ensuring no viscus can be damaged during chest drain placement
The thoracoscopic cannula was then removed and the port wound closed routinely using 1.5M Glycomer 631 (Biosyn; Covidien). The circumsternal sutures were then passed through the pre-prepared cast and tightened, as previously described, 12 before the chest drain was used to evacuate the pneumothorax. Postoperative radiography was then performed and the chest drain removed once resolution of pneumothorax and satisfactory improvement of PE had been confirmed. A light chest bandage was then applied to help secure the sternal cast and prevent patient interference. Patients were recovered with oxygen supplementation and given postoperative methadone as needed, depending on pain score. Patients were discharged 1–2 days postoperatively with a course of 0.05 mg/kg meloxicam q24h (Metacam; Boehringer) for 5 days once eating well. The patients were re-examined by the referring veterinarian at 2 weeks postoperatively, at which point the chest bandage was replaced to accommodate growth. The patient was then sedated for repeat radiography and cast removal at 4–5 weeks postoperatively.
Long-term follow-up was achieved by direct communication with either the client or referring veterinarian at the time of manuscript preparation. Outcome was graded as poor (no improvement), good (clinical signs improved but still present) or excellent (clinical signs resolved). All reported cases were judged to have an excellent outcome at the time of last follow-up.
Clinical data for the three cases are presented in Table 1. Pre- and postoperative radiographic indices are given in Table 2.
Clinical data for cases 1–3 (all female)

CSS = Clinical Severity Score; DSH = domestic shorthair
Preoperative and immediate postoperative radiographic indices

FSI = frontosagittal index; VI = vertebral index
Discussion
Use of VATS to help guide suture placement has previously been suggested to improve patient safety by helping to avoid visceral trauma, as is the case in humans.11,12 In order to confer this advantage, it is crucial that the operating surgeon is able to directly visualise the passage of the suture needle into the thorax. There is one report 14 of a ‘needle scope’ being used in a kitten with PE using the previously described technique. 12 Needle scopes are not widely available, however, and because of restricted space in more severely affected kittens, we found visualisation of suture passage difficult to achieve in previous (unpublished) cases of feline PE using both 5 mm and 2.4 mm rigid telescopes and this technique.12,14 Given that there are increased time and costs associated with VATS compared with simple blind suture placement, we wanted to explore whether the previously reported VATS technique could be modified so that it provided a demonstrable benefit.
The use of the cannula to push the displaced sternebrae ventrally proved to be quick and easy to accomplish once pneumothorax had been achieved. This then made it very easy to pass the circumsternal sutures as there was no dorsal (downwards) movement of the sternebrae towards the underlying viscera. Suture passage was considered safer owing to the increased distance between viscera and sternebrae provided by the pneumothorax as well as the presence of the plastic cannula immediately dorsal to the site of suture passage. The cannula was not struck by a needle in any of the reported cases, and nor was it damaged by using it to retract the sternebrae in this way. We found this modification allowed relatively rapid surgery, although anaesthetic time was longer than simple blind-placement cases. We believe that the potential increases in patient safety offset the increased anaesthesia time; however, we fully acknowledge that a prospective trial would be required to confirm if this is the case. PE is a rarely reported condition, and it would be challenging to amass sufficient cases to perform such a study.
Our cases were older than the typical kitten undergoing surgical (splint) correction. No conscious effort was taken to select larger cats for this surgery, and these cases were seen consecutively at our institute. Many kittens undergo surgery at 10–15 weeks of age, 5 but it is unknown whether these cases have a better outcome than those that undergo surgery at an older age, providing that the thoracic wall remains compliant and that secondary cardiopulmonary effects have not occurred. We fully acknowledge that smaller kittens may lack the intercostal width to accommodate a 5 mm cannula. It is possible that a smaller diameter cannula could be used to achieve the same ventral retraction providing it is of sufficient length and rigidity; however, we did not have a suitable case to test this theory.
Conclusions
This modification of the previously reported VATS-assisted correction of PE proved to be effective and safe in this small case series of affected kittens. We believe that the ability to push the affected sternebrae ventrally confers a genuine advantage to the use of VATS with respect to both the ease of surgery and patient safety in affected kittens.
Footnotes
Acknowledgements
We thank Ines Gordo DipECVS for surgical assistance with case 2.
Author’s note
Data supporting the conclusions of this study are presented within this manuscript. Additional data are available on request from the author.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
