Abstract
Objectives
The aim of this study was to evaluate the safety and efficacy of cabergoline to control hypersomatotropism (HST) and diabetes mellitus (DM) in cats.
Methods
This was a prospective cohort study. Twenty-three cats with HST and concurrent DM were enrolled. Cats received a dose of 10 μg/kg cabergoline q48h PO for 6 months. Serum insulin-like growth factor 1 (IGF-1) and fructosamine concentrations, insulin dose and Insulin Resistance Index (IRI) were measured at the time of diagnosis of HST and at the start of cabergoline treatment (t0), and 3 months (t1) and 6 months (t2) during cabergoline treatment.
Results
A decrease and normalization of serum IGF-1 concentration was observed in 35% and 26% of cats, respectively. Median IGF-1 (t0: 1350 ng/ml [range 832–1501]; t1: 1284 ng/ml [range 365–1501]; t2: 1240 ng/ml [range 263–1501]; P = 0.016) decreased significantly. Twelve cats underwent diagnostic imaging of the pituitary area. The median pituitary height at t0 of cats that experienced an IGF-1 reduction (n = 5/12) was significantly lower compared with those that did not experience an IGF-1 reduction (n = 7/12) (3.2 mm [range 3.1–3.7] vs 6 mm [range 3.5–9.5]; P = 0.011). Median fructosamine (t0: 628 µmol/l [range 400–963]; t1: 404 µmol/l [range 249–780]; t2: 400 µmol/l [range 260–815]; P <0.0001), insulin dose (t0: 1.3 IU/kg [range 0.5–4.6]; t0: 0.5 IU/kg [range 0–2.3]; t2: 0.4 IU/kg [range 0–2.1]; P <0.0001) and IRI (t0: 800 µmolIU/kgl [range 257–2700]; t1: 300 µmolIU/kgl [range 0–1498]; t2: 250 µmolIU/kgl [range 0–1498]; P <0.0001) decreased significantly during cabergoline treatment. Eight cats achieved diabetic remission between months 1 and 6 of cabergoline treatment (median time to achieve remission: 3 months [range 1–6]). Three cats experienced asymptomatic hypoglycemia.
Conclusions and relevance
Cabergoline was effective in normalizing IGF-1 concentration in 26% of cats. Cabergoline improved diabetes control and was associated with remission of DM in 35% of cases. Cabergoline could be a treatment option for cats with HST and DM, especially in those cases with a relatively small pituitary tumor.
Introduction
Hypersomatotropism (HST), or acromegaly, is a chronic disease caused by hypersecretion of growth hormone (GH) and occurs in both humans and companion animals. 1 In the domestic cat (Felis catus), it is caused by a functioning pituitary adenoma (somatotrophinoma) or hyperplasia of the pars distalis of the anterior pituitary gland. 2 HST is often characterized by the presence of diabetes mellitus (DM), and it is estimated that 17.8–24.8% of diabetic cats in Europe (Switzerland/the Netherlands and the UK) have an excess of GH.3,4
Therapeutic options for acromegaly in humans are surgery, mainly via a transphenoidal approach, followed by medical treatment and radiotherapy. Pharmacological treatment includes somatostatin receptor ligands (SRLs), dopamine agonists (DAs) and GH receptor antagonists. 5 Cabergoline is a long-acting DA, with a high affinity for dopamine receptor 2 (D2R). International consensus is that it can be used in patients with mild disease (insulin-like growth factor 1 [IGF-1] <2 ng/ml). 6 Used as monotherapy, cabergoline is effective in normalizing serum IGF-1 concentrations and in reducing tumor size in one-third of patients. When a first-generation SRL fails to control acromegaly, combined treatment with cabergoline normalizes serum IGF-1 concentrations in 50% of cases in a subset of patients without biochemical control and slightly elevated IGF-1 levels.6,7
Expression of D2R has been demonstrated in the pituitary gland of cats with HST. In addition, a moderately negative correlation between D2R expression and pituitary size has been described. 2 Studies assessing the use of cabergoline for the treatment of feline HST are lacking, and the results on the effects on blood glucose and the improvement of DM are discordant.8–10 In a pilot study carried out in the UK, cabergoline did not normalize IGF-1 concentration nor improve diabetic control. 8 Conversely, we reported, for the first time, the remission of DM in three cats with HST treated with cabergoline. Nevertheless, in this report, cabergoline did not normalize IGF-1 concentration, despite the improvement of diabetic control. 9
Considering that the effectiveness of feline HST treatment with surgery and radiotherapy involves invasive modalities, which require specialized centers, and that pasireotide is an expensive drug,11–13 it is necessary to evaluate new effective and accessible treatments. Therefore, the aim of this study was to evaluate the safety and efficacy of cabergoline in controlling glycemic disorder and IGF-1 concentration in cats with HST and DM.
Materials and methods
This study was approved by the Ethics Committee of the Veterinary Science Center (Institutional Committee on the Care and Use of Experimental Animals [CICUAE]), in accordance with the laws on animal testing in Argentina and the recommendations of the World Health Organization. Informed written consent was obtained from all study cat owners before enrolment. Client-owned cats admitted to the Veterinary Science Center, Maimonides University, were enrolled in the study between 1 May 2018 and 30 April 2021. The diagnosis of HST was made according to clinical signs (acromegalic features: enlargement of the head, abdomen and paws; prognathia inferior; respiratory stridor; and degenerative arthropathy), serum IGF-1 concentration and pituitary CT and MRI images (n = 12). Serum IGF-1 was measured by a commercially available radioimmunoassay (Immuno-Biological Laboratories). 9 The CT studies were performed with a Siemens SOMATOM Perspective 128 slice. The MRI studies were performed with a Hitachi 0.23 Tesla open magnetic resonator.
Inclusion criteria were diabetic cats with a serum IGF-1 concentration >1000 ng/ml 3 or IGF-1 >800 ng/ml with pituitary enlargement (>4 mm dorsoventral height on CT, 14 or >3.3 mm transverse height on MRI 15 ). Considering the results of Niessen et al, 3 where it was shown that serum IGF-1 concentrations >1000 ng/ml have a positive predictive value for HST in 95% of UK diabetic cats, the necessity of pituitary imaging in these cats was not deemed essential.3,8 Likewise, inclusion criteria were insulin treatment for at least 6 weeks prior to enrolment, 16 indoor lifestyle and good owner compliance. Exclusion criteria were long-term insulin treatment, 17 glucocorticoid or progestagen therapy prior to enrolment and concurrent severe illness with poor prognosis.
Cats received a dose of 10 μg/kg cabergoline (Dostinex; Pfizer) q48h PO for 6 months.9,10 All cats received insulin glargine (Lantus; Sanofi-Aventis) q12h and were fed a commercial diet for diabetic cats (Royal Canin Diabetic for cats).
Serum IGF-1 and fructosamine concentrations were measured at the time of diagnosis of HST and at the start of cabergoline treatment (t0), and 3 months (t1) and 6 months (t2) during cabergoline treatment. Periodic clinical examinations and home blood glucose curves were carried out to determine insulin requirements and to prevent hypoglycemia. The blood glucose curves were undertaken on days 1, 7, 14, 21 and 30 from the start of cabergoline treatment and then monthly thereafter. The monitoring of clinical signs, as well as home blood glucose, was performed by the owners and the veterinary team to make appropriate adjustments to the insulin dose. The cats were evaluated monthly in the hospital for clinical control, for insulin dose adjustment and for possible side effects of cabergoline. Intensive blood glucose monitoring was performed by all owners at home (glucose measured with a FreeStyle Optium glucose meter [Abbott Laboratories]), and the owner of one cat used a FLASH glucose monitor system (FreeStyle Libre; Abbott Laboratories). Cats that presented with normal blood glucose concentrations (3.3–6.6 mmol/l) without insulin or any medical antidiabetic therapy (with the exception of diet) for a minimum of 28 days were classified as being in DM remission. The product of serum fructosamine concentrations and insulin doses (IU/kg) was calculated as an indicator of insulin resistance (Insulin Resistance Index [IRI]), previously evaluated in diabetic cats with HST. 13
Statistical analysis
Statistical analysis was performed with GraphPad Prism 6. Data were analyzed for normal distribution (Shapiro–Wilk tests). Values were expressed as mean ± SD, median and range, as applicable, with a significance level set at P <0.05. The pretreatment (t0) and during treatment (t1 and t2) results were analyzed using the Friedman and Wilcoxon tests. The Mann–Whitney U-test was used to compare pituitary size between groups. The IGF-1 test was calibrated with an upper limit of quantification of 1500 ng/ml, and any serum IGF-1 concentration >1500 ng/ml was analyzed as being 1501 ng/ml.
Results
Twenty-five cats with HST and DM were recruited during the study period; of these 25, the owners of two declined to participate. Twenty-three cats were therefore enrolled in the study: 22 completed the study period, and only one cat was withdrawn at month 3, at the request of the owner. Nineteen cats were domestic shorthairs, three were Siamese and there was one Burmese. Sixteen were male and seven were female; mean age was 10.6 ± 3 years; mean body weight was 5.7 ± 2.1 kg. Intracranial imaging was performed in 12 cats; nine with MRI and three with CT. The median pituitary dorsoventral height at enrolment was 4.6 mm (range 3.1–9.5). All cats had at least 6 weeks of insulin therapy and none exceeded 4 months of insulin at the time of enrolment. No cats changed insulin type during the study period. Fourteen (60%) had clinical signs of poor diabetic control. Twelve cats (52%) had phenotypic changes consistent with acromegaly at the time of diagnosis of HST. No owners reported any difficulties with the oral administration of cabergoline.
Serum IGF-1 concentration
Eleven of 23 cats (48%) had a serum IGF-1 concentration >1500 ng/dl at t0, seven (30%) had an IGF-1 level between 1000 and 1500 ng/dl at t0, and five (22%) had an IGF-1 concentration of between 800 and 1000 ng/dl at t0, and diagnosis of HST was confirmed with intracranial imaging. The median pituitary height at t0 of cats that had an IGF-1 concentration <1500 ng/dl at t0 (n = 4) was significantly lower compared with those that had an IGF-1 concentration >1500 ng/dl at t0 (n = 8; 3.4 ± 0.3 vs 5.8 ± 1.9 mm [P = 0.05]).
Serum IGF-1 concentrations showed significant differences when comparing t0 and t2 (P = 0.021; Table 1 and Figure 1), showing a decrease in 35% of cases. No significant differences were observed when comparing t0 and t1 (P = 0.052), or t1 and t2 (P = 0.052). Cabergoline administration resulted in normalization of IGF-1 concentrations (values within the reference interval [200–800 ng/dl]) in 4/23 cats (17%) at t1 and 6/23 cats (26%) at t2. The median pituitary height at t0 of cats that experienced a reduction in IGF-1 (n = 5) was significantly lower compared with those that did not experience a reduction (n = 7; 3.2 mm [range 3.1–3.7] vs 6 mm [range 3.5–9.5]; P = 0.011).
Serum insulin-like growth factor 1 (IGF-1), fructosamine, insulin dose and Insulin Resistance Index (IRI) before and during cabergoline treatment

All values are expressed as median (range)
t0 = diagnosis of hypersomatotropism and start of cabergoline treatment; t1 = month 3 of cabergoline treatment; t2 = month 6 of cabergoline treatment
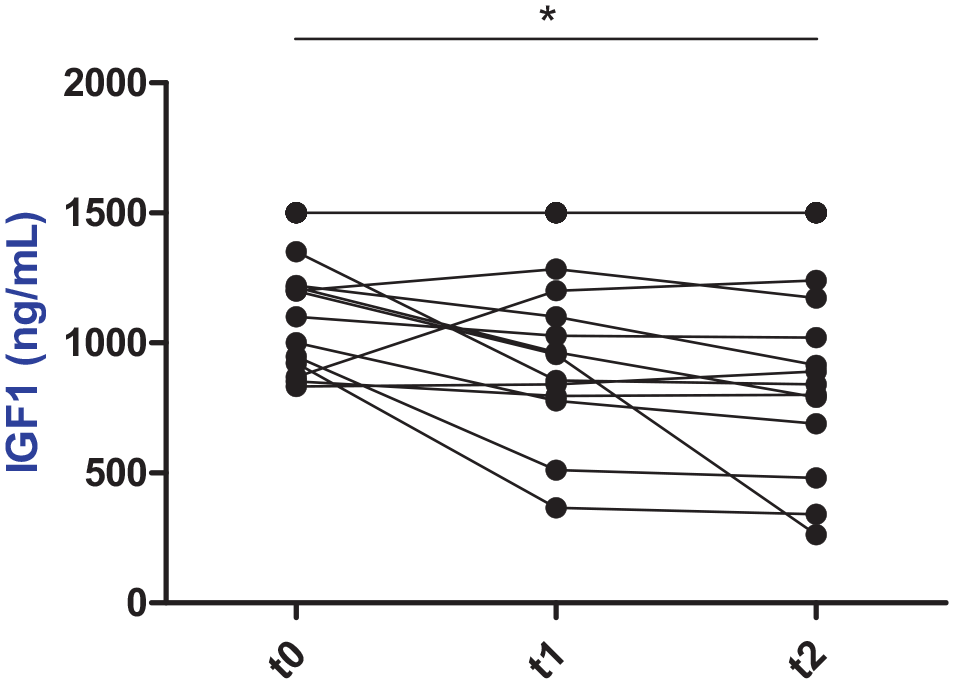
Serum insulin-like growth factor 1 (IGF-1) concentrations before and during cabergoline treatment.
Serum fructosamine concentration, insulin dose and IRI
Serum fructosamine concentrations significantly decreased at t1 (P <0.0001) and t2 (P = 0.0001), compared with t0 (Table 1 and Figure 2) during cabergoline treatment. No significant differences were observed when comparing t1 and t2 (P = 0.57). Insulin dose was significantly decreased at t1 (P = 0.0002) and t2 (P = 0.0001), compared with t0 (Table 1 and Figure 3), and also when comparing t1 and t2 (P = 0.001). IRI was significantly decreased at t1 (P <0.001) and t2 (P <0.001) compared with t0 (Table 1 and Figure 4); no significant differences were observed when comparing t1 and t2 (P = 0.13).
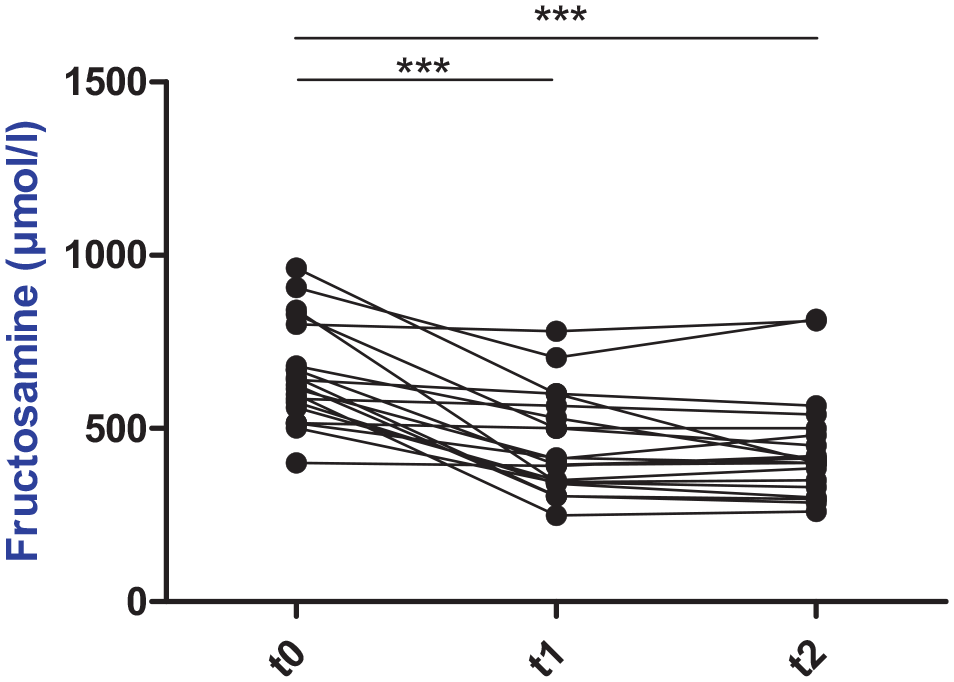
Serum fructosamine concentrations before and during cabergoline treatment. ***P <0.001. t0 = diagnosis of hypersomatotropism and start of cabergoline treatment; t1 = month 3 of cabergoline treatment; t2 = month 6 of cabergoline treatment
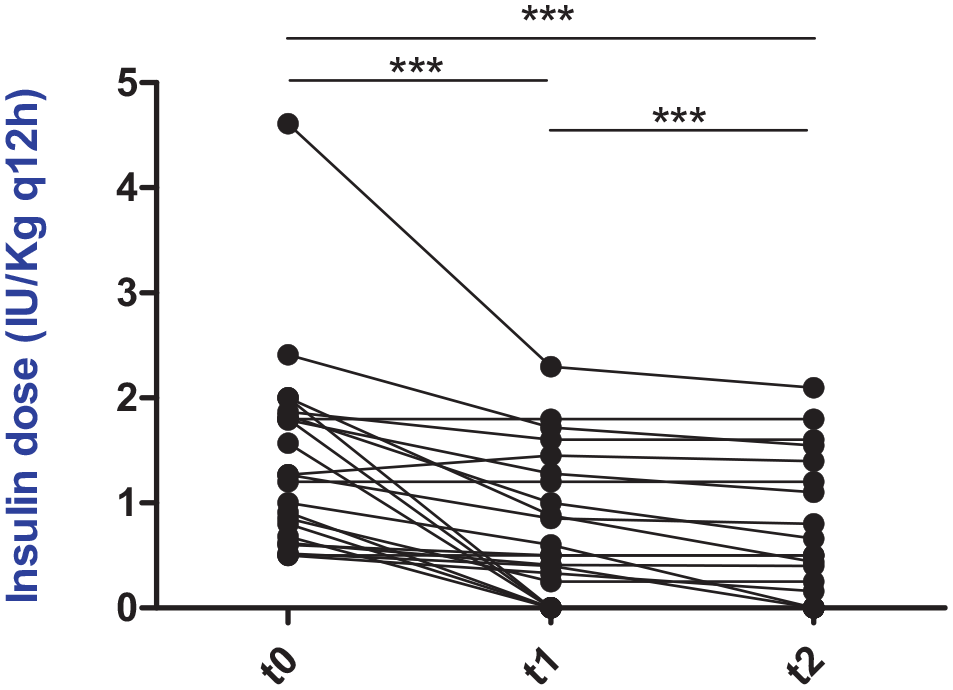
Individual insulin dose (IU/kg q12h) before and during cabergoline treatment. ***P <0.001. t0 = diagnosis of hypersomatotropism and start of cabergoline treatment; t1 = month 3 of cabergoline treatment; t2 = month 6 of cabergoline treatment

Insulin Resistance Index (fructosamine q12h insulin dose [U/kg]) before and during cabergoline treatment. ***P <0.001. t0 = diagnosis of hypersomatotropism and start of cabergoline treatment; t1 = month 3 of cabergoline treatment; t2 = month 6 of cabergoline treatment
DM remission
Eight cats (35%) achieved diabetic remission between months 1 and 6 of cabergoline treatment; six achieved diabetic remission between months 1 and 3 of cabergoline treatment, while the other two cats achieved remission between months 3 and 6 (median time to achieve remission 3 months [range 1–6]). All cats remained in remission at the time of writing and were free of insulin, only being under cabergoline treatment for between 5 and 45 months (median length of remission 22 months [range 5–45]). Before cabergoline treatment, three cats (37.5%) had serum IGF-1 concentrations between 800 and 1000 ng/dl, three cats (37.5%) had IGF-1 concentrations between 1000 and 1500 ng/dl and two cats (25%) had IGF-1 concentrations >1500 ng/dl. Four cats (50%) had normalized serum IGF-1 concentrations, while the remaining four cats persisted with an IGF-1 concentration >1000 ng/dl, despite having achieved DM remission. Of the four cats that had normalized IGF-1 concentrations, two achieved normalization 3 months after DM remission, while the other two achieved normalization of IGF-1 and DM remission at the same time (at t1). Serum IGF-1 concentrations showed significant differences at t2 when comparing cats with diabetic remission (840 ng/ml [range 236–1501]) and those without (1501 ng/ml [range 689–1501]; P = 0.04). No significant differences were observed when comparing IGF-1 at t0 in cats with diabetic remission (1200 ng/ml [range 832–1501]) and cats without diabetic remission (1501 ng/ml [range 852–1501]; P = 0.1).
The median pituitary height was 3.5 mm (range 3.1–3.8) (normal transverse height on MRI 3.09 ± 0.26 mm) in 5/8 cats that had intracranial imaging. Two did not show pituitary enlargement. Likewise, none had evidence of pituitary macroadenoma. The median pituitary height at t0 of cats with diabetic remission (n = 5) was significantly lower compared with those cats without diabetic remission (n = 7; 6 mm [range 3.2–9.5]; P = 0.03).
Possible adverse events
Three cats experienced biochemical hypoglycemia (fasting glycemia between 2.8 and 3.3 mmol/l) without clinical signs. One of them achieved diabetic remission after hypoglycemia, another cat achieved remission 1 month after hypoglycemia and the third cat had a lowered insulin requirement without achieving diabetic remission. In one cat a change in hair color was observed at month 3 on cabergoline treatment. No additional adverse events were observed during the study.
Discussion
This is the largest study to date on the use of cabergoline (Dostinex; Pfizer) in cats with HST and concurrent DM, demonstrated by a considerable number of cases with a long follow-up. This study provides evidence that the use of cabergoline in cats with HST is associated with an improvement in glycemic control with a reduction in IGF-1 concentrations in some cases. Cabergoline is associated with a reduction in fructosamine concentrations and insulin requirements, which allows diabetic remission in approximately one-third of cats with HST.
Studies evaluating pharmacological treatment in cats with HST are scarce, with a small number of cases and no successful results in the case of long-acting octreotide and L-deprenyl.13,18,19 Pasireotide long-acting release is the first drug to show potential benefits as a long-term management option for diabetic cats with HST, 13 but its high costs reduce the availability of its clinical use. Previous studies on cabergoline are also scarce and with a small number of cases: two case reports with three cats in each study,9,10 and a pilot study that included nine cats. 8 We have previously seen that cabergoline can be associated with an improvement in the metabolic control of DM, regardless of reducing IGF-1 concentrations. 9 In contrast, a UK pilot study concluded that cabergoline did not appear to control HST as the cats did not experience improved diabetic control or achieve IGF-1 control. 8 In the current study, the results suggest that cabergoline can be another option, apart from pasireotide, as a treatment for cats with HST and DM. We have previously formulated several hypotheses that could explain the discordant results between the studies. 9 First, different subpopulations of somatotrophinomas or regional variants of HST exist (in this case, in Argentina and the UK). In human medicine, different histological subtypes of somatotropinoma, densely vs sparsely granulated adenomas, have been described as having a different response to pharmacological treatment. 20 Likewise, response to cabergoline can vary according to the expression of D2R isoforms, agonist affinity and signal transduction, as has been described in humans with pituitary tumors. 21 A second possibility is the different dosage regimens and commercial preparations of cabergoline. A third possible reason is the severity of HST in the cats evaluated in both studies appears to be different. In the UK pilot study, only 22% (n = 2/9) of the cats had an IGF-1 concentration <1500 ng/dl and mean pituitary height was 6.3 ± 1.6 mm. In the present study, 52% (n = 12/23) of cats had an IGF-1 concentration of <1500 ng/dl and the median pituitary height (n = 12) was 4.6 mm (range 3.1–9.5). It appears that the best effect of cabergoline is seen in cats with with a relatively small pituitary tumor and a serum IGF-1 concentration <1500 ng/dl. In human medicine, the benefits of cabergoline are limited to patients with mildly elevated levels of IGF-1 at baseline. According to the 11th Acromegaly Consensus Conference, the greatest benefit is seen in those patients with IGF-1 levels ⩽1.5 times the upper limit of normal. 6 This phenomenon can be applied to cats with HST and DM treated with cabergoline. Fourth, a greater number of cats with HST were evaluated in this study than in the UK pilot study, increasing the statistical power of the data analysis.
It is important to highlight that most of cats (65%) in the present study did not show a reduction in the concentration of IGF-1. However, 35% of cats experienced reduced IGF-1 concentrations, and 26% achieved normalization of IGF-1 with cabergoline treatment at month 6. In cases where IGF-1 concentrations do not normalize, cats receiving cabergoline are likely still under threat from the complications of excess GH.
The effect of cabergoline on glycemic control in cats with HST and DM is promising; cabergoline treatment was associated with reduction in insulin requirements and fructosamine concentration, and diabetic remission in 35% of cases. Insulin requirements were progressively reduced in most cats, allowing appropriate adjustment of insulin doses. Nevertheless, cabergoline can have a rapid effect on glycemia and promote hypoglycemia. This phenomenon was observed in three cats in this study and also in a cat with hyperadrenocorticism. 22 Likewise, the IRI showed improvement in insulin sensitivity in cats with HST and DM treated with cabergoline; this method was previously used in cats with HST treated with pasireotide.13,23
Probably the most relevant finding of cabergoline treatment was the high rate of diabetic remission observed in this study. Feline diabetic remission has previously been described in cats with HST treated with hypophysectomy, radiotherapy, pasireotide and cabergoline.9,11–13,24,25 In this study, eight cats achieved diabetic remission, which rarely occurs when diabetic cats with GH excess are only managed with insulin. 16 However, it is important to highlight that intensive blood glucose control was performed in this study. Therefore, better diabetic control may also have resulted in diabetic remission and a decrease in serum fructosamine concentration and IRI.
The mechanism by which cabergoline controls DM is complex and not completely understood. On the one hand, this study provides evidence that IGF-1 concentrations were reduced in one-third of cases, which may suppose a parallel reduction in GH concentrations (not measured), reducing insulin resistance induced by excess GH. 16 Scudder et al have shown the expression of D2R in the pituitary gland of cats with HST and that there is a moderate negative correlation between expression of D2R and pituitary size. 2 The results of the current study agree with these observations; about half of the cats had an IGF-1 concentration <1500 ng/dl and the median pituitary height of cats that experienced an IGF-1 reduction (n = 5/12) was lower than those that did not experience an IGF-1 reduction (n = 7/12). Therefore, a small pituitary size could make the somatotrophinoma more likely to respond to cabergoline treatment. On the other hand, although most cats did not show a reduction in IGF-1 concentrations, they did show a marked improvement in DM control; four cats achieved diabetic remission without reducing IGF-1 concentrations, as has also been described in cats that receive radiotherapy. 24 IGF-1 concentrations may not correlate completely with GH concentrations, as we hypothesized in a previous report. 9 A reduction in GH could therefore occur without it showing up alongside reductions in IGF-1. Likewise, cabergoline could affect glycemic control by a mechanism independent of the somatotropic axis; it is likely that the suppression of monoamines and prolactin (PRL) are involved. 26 In animal models, dopamine regulates metabolic pathways in the central nervous system. A reduction in hepatic glucose production and adipose tissue lipolysis, with improvement in insulin sensitivity, were described after bromocriptine administration, affecting levels of adrenalin and serotonin.26–28 In addition, several studies have shown that PRL induces insulin resistance, weight gain and food intake, favoring the development of type 2 DM.29,30 In humans, cabergoline improves glycemic control, reduces glycated hemoglobin concentrations and also improves metabolic parameters in patients with prolactinoma.31–33 Finally, cabergoline could play a role in the hypothalamic–pituitary–adrenal axis in cats, as occurs in dogs with pituitary-dependent hypercortisolism, and thus could reduce the diabetogenic effect of cortisol.9,34
Considering that the hypoglycemia observed in three cats was caused by a decrease in insulin requirements, no direct side effects of cabergoline were observed during the entire study.
This study has several limitations. A first limitation was the lack of measuring pituitary hormones, such as PRL, adrenocorticotrophin hormone, alpha-melanocyte-stimulating hormone and especially GH. Another limitation was not diluting the samples to obtain the exact concentration of IGF-1 >1500 ng/dl; therefore, cabergoline responses in cats that exceeded this value could not be analyzed properly. In addition, the lack of a control group was an important limitation of this study. With regard to pituitary images, the limitations were the lack of images in half of the cohort studied due to owner decision (this could be the cause of bias in some comparisons); the low resolution of MRI images; not performing a second image (owner decision); and heterogeneity of images performed (some were studied with CT and others with MRI).
Conclusions
The results of this study suggest that cabergoline as a monotherapy was effective in reducing IGF-1 excess in one-third of diabetic cats with HST and in normalizing IGF-1 concentrations in 26% of cases. Cabergoline improved diabetes control and was associated with remission of DM in 35% of cases. This DA is relatively inexpensive and easy to administer, and should therefore be considered as a posible treatment option for diabetic cats with HST, especially in those cases with a relatively small pituitary tumor.
Footnotes
Correction (January 2023):
The affiliations for Diego D Miceli (1, 2 and 3) have been corrected online.
Author note
Preliminary data from the study were presented as an oral research communication at the 31st ECVIM-CA Congress, 2021 (online).
Conflict of interest
The authors declared no conflicts of interest with respect to the authorship, research and publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals and procedures that differed from established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient. The study therefore had prior ethical approval from an established (or ad hoc) committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
