Abstract
Objectives
Feline idiopathic cystitis (FIC) and urethral obstruction (UO) are commonly linked to increased stress. The influence of human movement restrictions on their incidence remains undetermined. FIC with or without UO is associated with environmental stress factors. The severe acute respiratory syndrome coronavirus 2 (COVID-19) pandemic restricted human movement and working behaviours. It is unknown if these restrictions increased the risk of FIC or UO in cats.
Methods
Total cat emergency accessions and transfers between 8 February 2019 and 8 February 2021 at two private hospitals were retrospectively reviewed. Cats were included in the FIC group if they presented with lower urinary tract signs and supporting urinalysis, and were included in the UO group if they presented with UO. Cats with current urinary tract infection, or previous FIC or UO, were excluded. Groups were considered ‘pre-COVID-19’ between February 2019 and 2020 and ‘COVID-19’ between February 2020 and 2021. Cases of FIC and UO were compared between COVID-19 and pre-COVID-19 using Fisher’s exact test and relative risk (RR) calculations.
Results
The pre-COVID-19 incidence of FIC was 4.3% (63/1477, 95% confidence interval [CI] 0.0332–0.053), non-obstructive FIC was 1.4% (20/1477, 95% CI 0.008–0.020) and UO was 2.9% (43/1477, 95% CI 0.020–0.038). One cat was excluded as obstruction occurred during hospitalisation. The COVID-19 incidence of FIC was 5.4% (113/2081, 95% CI 0.044–0.64), non-obstructive FIC was 2.1% (70/2081, 95% CI 0.014–0.027) and UO was 3.4% (70/2081, 95% CI 0.026–0.042). The risk of non-obstructive FIC (P = 0.122; RR 0.652, 95% CI 0.387–1.096), UO (P = 0.382; RR 0.839, 95% CI 0.577–1.22) or either (P = 0.098; RR 0.773, 95% CI 0.572–1.044) was not significantly higher in the COVID-19 period than the pre-COVID-19 period.
Conclusions and relevance
No clear association between COVID-19 movement restrictions and the incidence of UO or non-obstructive FIC was found within this retrospective population.
Keywords
Introduction
Feline lower urinary tract disease (FLUTD) is a common syndrome in cats encompassing bacterial cystitis, urolithiasis, crystalluria, urethral plugs, neoplasia, trauma and anatomical defects. The prevalence of FLUTD is estimated to be in the range of 1.5–2.2% of the domestic feline population.1 –3 Feline idiopathic cystitis (FIC) is the most common condition, accounting for 54–69% of FLUTD cases, and is diagnosed when other causes of FLUTD are excluded.1,3 –9 FIC is a poorly understood but complex condition that is associated with husbandry practices and environmental stressors, including abrupt changes to the environment, the addition of new pets or people to the home environment, and owner stress.3,4,6,7,10 –13 Neuroendocrine abnormalities in combination with stressors are suggested to be a primary causal factor in the development of FIC.1 –3 A possible mechanism of stress-induced FIC is by central dysregulation of autonomic neurons that either regulate bladder contraction or directly compromise the urothelium. 3
Urethral obstruction (UO) is a common complication of FLUTD, due to mechanical obstruction from struvite crystals or mucous plugs, or functional obstruction due to urethral spasm from inflammation. 14 UO is more common in male cats due to their relatively long and narrow urethra. 15
The severe acute respiratory syndrome coronavirus 2 (COVID-19) pandemic caused a change in the home environment on a global scale. From early 2020, the pandemic restricted human movement and working behaviours, fundamentally changing the environment of indoor and indoor/outdoor cats. 16 Lifestyle changes that have impacted home environments include human local movement restrictions (‘lockdowns’), self-isolation, travel restrictions, work-from-home practices, homeschooling, and increased emotional stress and declining mental health for owners.17–19 Literature is emerging that links COVID-19 with increased stress and illness in companion animals globally.19,20 Stress behaviours were increased in cats and dogs in Spain, particularly those with pre-existing behavioural conditions. 20 An increased prevalence of UO during COVID-19 has been documented in cats at two university teaching hospitals in the USA. 19 Anecdotally, the authors felt more cats were presenting with FIC and UO during COVID-19 to the Australian study centres than previously, and case numbers felt higher during periods of stricter government-mandated movement restrictions. The primary objective of this study was to compare the incidence of FIC and UO during lockdowns compared with before the pandemic. It was hypothesised that due to the increase in the risk factors for FIC during COVID-19 (including an abrupt change in the home environment), cases of FIC and UO would increase during lockdown periods compared with the pre-COVID-19 period.
Materials and methods
A retrospective observational study was performed at two private referral and emergency and hospitals in southeast Queensland between 8 February 2019 and 8 February 2021. All cats presenting as either primary accessions or transfers from a primary care veterinarian were included. Groups were considered ‘pre-COVID-19’ between 8 February 2019 and 7 February 2020, and ‘COVID-19’ between 8 February 2020 and 8 February 2021. Specific periods of extreme movement restriction in southeast Queensland (March–April 2020 and 8–11 January 2021) were also compared with their contemporaneous points before COVID-19.
Data collection
The electronic medical record (EMR) database was searched for the invoice item buprenorphine. This item was chosen because buprenorphine is a routine analgesic in the management of inpatient and outpatient cases of FLUTD at this institution, and the EMR permits search only by invoice items. Records were then individually assessed for FLUTD. Records were reviewed for patient age (months), sex (male entire, male neutered, male unknown, female entire, female neutered, female unknown), breed (domestic shorthair, domestic mediumhair, domestic longhair, purebred), outdoor access (yes/no) and presence of other cats in the household (yes/no). Total cat accessions for each period were retrieved by searching for all consultations where the patient was registered as a cat.
Group definition
Cats were assigned to the FIC group if they had at least one compatible clinical sign, and their diagnostic evaluation was sufficient to exclude other causes of FLUTD. Compatible clinical signs included dysuria, haematuria, stranguria, pollakiuria or periuria. Diagnostic evaluation consisted of a complete physical examination, urinalysis, stained urine sediment examination, and/or urine culture and susceptibility where available. Cats were assigned to the UO group if UO was diagnosed on physical examination and documented in the medical record. Only cats presenting with their first incidence of FIC were included in the FIC group.
Exclusion criteria
Cats with a known history of FLUTD (including FIC and/or UO) or that did not have urinalysis performed at the study hospital were excluded. Cats with urinalysis performed only with their primary care veterinarian were therefore excluded. Cats with urinary tract infection (UTI) were excluded, as this disease was considered unlikely to be influenced by COVID-19 factors.
Statistical analysis
A statistical analysis was conducted using SPSS version 27 (IBM Corp) and Prism version 9.2 (GraphPad). Normality was assessed histographically. Non-parametric data were expressed as median (range). Due to small case numbers in categories, Fisher’s exact test was performed to compare signalment and history factors (such as sex, multi-cat household, outdoor access) between the COVID-19 and pre-COVID-19 periods, and the occurrence of FIC ± UO. Mann–Whitney U tests compared age between periods and occurrence of FIC and/or UO. Relative risk ratios with 95% confidence intervals (CIs) were calculated for both time groups to compare the incidence of non-obstructive FIC, UO or both. A P value <0.05 was considered significant.
Results
For the pre-COVID-19 period, 11,493 patients presented to the two hospitals, of which 1477 (12.8%) were cats. A total of 1414 cats did not have FLUTD signs or were not assigned to the FIC group due to the presence of UTI (n = 20, 1.4%), lack of urinalysis (n = 56, 3.9%) or previous occurrence of FLUTD (n = 104). In total, 63 cases (4.3%, 95% CI 0.033–0.053) were diagnosed with FIC, of which 20 (31.7%, 95% CI 0.199–0.436) were diagnosed with non-obstructive FIC and 43 (68.3%, 95% CI 0.564–0.801) with UO. The incidence of FIC, non-obstructive FIC or UO was 4.3% (95% CI 0.0332–0.053), 1.4% (95% CI 0.008–0.020) and 2.9% (95% CI 0.020–0.038), respectively.
For the COVID-19 period, 15,727 patients presented to the two hospitals, of which 2081 (13.2%) were cats. Of these cases, 1968 cats did not have FLUTD signs or were not assigned to the FIC/UO group due to the presence of UTI (n = 31, 1.5%), lack of urinalysis (n = 94, 4.7%) or a previous occurrence of FLUTD (n = 165, 8.3%). In total, 113 cases (5.4%, 95% CI 0.044–0.64) were diagnosed with FIC, of which 43 (38.1%, 95% CI −1.606–2.367) were diagnosed with non-obstructive FIC and 70 (61.9%, 95% CI −1.363–2.602) with UO. The incidence of FIC, non-obstructive FIC or UO during this period was 5.4% (95% CI 0.044–0.64), 2.1% (95% CI 0.014–0.027) and 3.4% (95% CI 0.026–0.042), respectively. A cumulative bar graph of percentage of FIC, non-obstructive FIC or UO for both periods is presented in Figure 1.

Broken-scale cumulative bar graph depicting the total number of feline cases presented during the pre-COVID-19 period (February 2019–2020) and the COVID-19 period (February 2020–2021). FIC = feline interstitial cystitis; non-FIC = general cat accessions and exclusions due to FIC recurrence, other urinary condition, lack of urinalysis; UO = urethral obstruction
Cats seen during the two periods were not significantly different in terms of age, sex, outdoor access nor for presence of other cats in the household (Table 1). The median age for cats in the pre-COVID-19 group was 48 months (range 12–174 months) and in the COVID-19 group was 55 months (range 8–168 months). Within this study, the majority of cats presenting with FIC for either group were male neutered, with 55/63 (87.3%) in the pre-COVID-19 group and 91/113 (80.5%) in the COVID-19 group, followed by male entire, with 5/63 (7.9%) in the pre-COVID-19 group and 10/113 (88%) in the COVID-19 group. Female entire (n = 0/63, 0%) and neutered (n = 3/63, 4.8%) presented within the pre-COVID-19 group, compared with 2/113 (1.7%) and 10/113 (8.8%) for the COVID-19 group. Domestic shorthair was the predominant breed for both groups, with 41/63 (65.1%) cats in the pre-COVID-19 group and 82/113 (72.5%) cats in the COVID-19 group. The remaining breeds are presented in Table 2.
Demographic information (age, sex, indoor status, multi-cat household status) for cats with feline idiopathic cystitis or urethral obstruction during the pre-COVID-19 period (February 2019–2020) and the COVID-19 period (February 2020–2021)
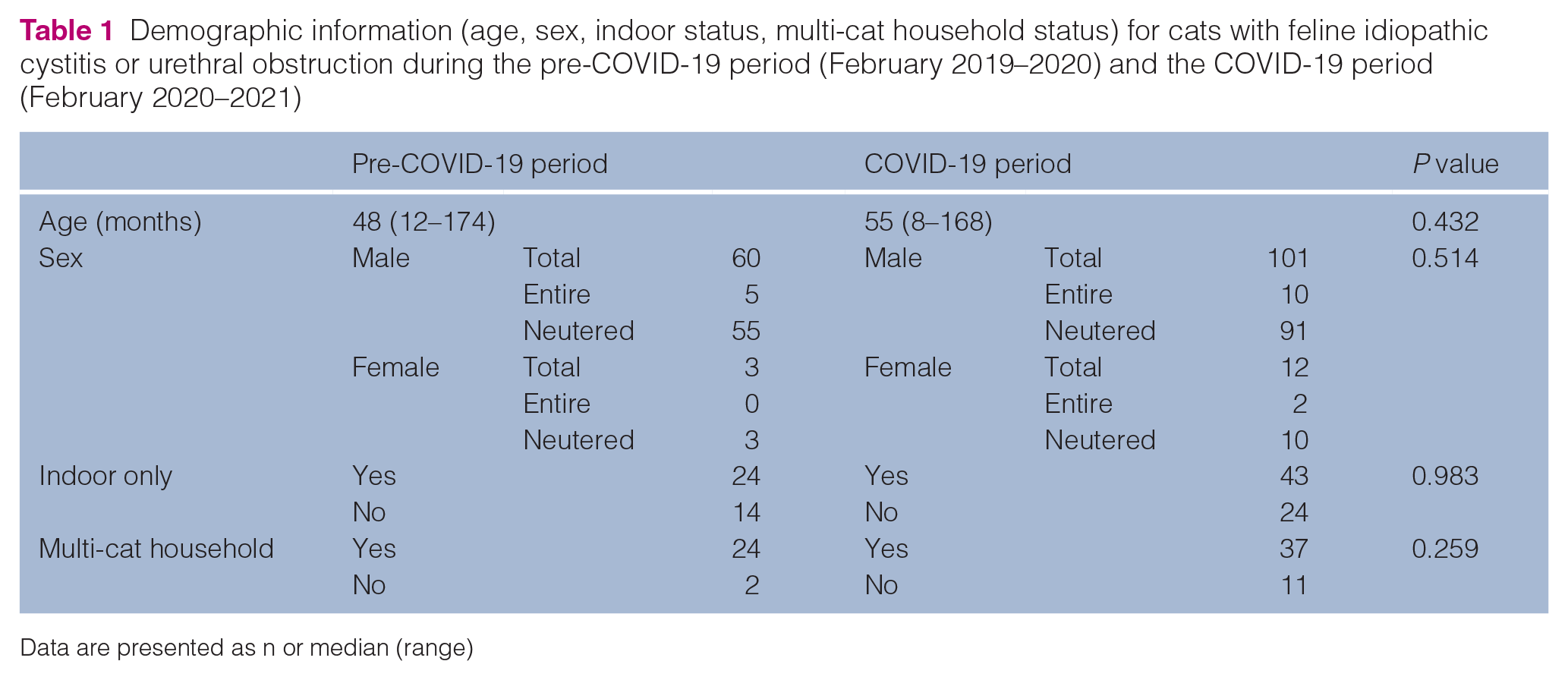
Data are presented as n or median (range)
Breed distribution in the pre-COVID-19 and COVID-19 groups

Data are presented as n (%)
The risk of developing non-obstructive FIC (P = 0.122; RR 0.652, 95% CI 0.387–1.096), UO (P = 0.382; RR 0.839, 95% CI 0.577–1.22) or either (P = 0.098; RR 0.773, 95% CI 0.572–1.044) was not significantly higher in the COVID-19 group. There was no significant difference in developing non-obstructive FIC, UO or both during COVID-19 relative to pre-COVID-19.
The percentage of accessions per month for both groups are summarised in Figure 2. When looking specifically at times associated with strict regional lockdown restrictions, March–April 2020 and 8–11 January 2021, there was no increase in cases compared with the prior year.20,21 From 8 to 11 January in 2020, there were 0/63 cases (0%, 95% CI −0.016–0.048), and there was only 1/113 cases (0.8%, 95% CI −1.982–2.000) in 2021. Between March and April 2020, there were 9/63 cases (14.3%, 95% CI 0.054-0.232) and there were 11/113 cases (9.7%, 95% CI −1.892–2.088) in 2021. Further analysis was not performed on these time frames due to the small sample sizes.

Monthly percentage of cats presenting with feline idiopathic cystitis (pre-COVID-19 February 2019–2020, n = 63; COVID-19 February 2020–2021, n = 113) from total cats presenting in the pre-COVID-19 period (n = 1414) and COVID-19 period (n = 1968)
Discussion
This retrospective study has shown no significant association between the COVID-19 movement restrictions and the incidence of FIC or UO in cats presenting to private referral and emergency practice in southeast Queensland. Although the number of cats presenting to hospital for all causes increased during the COVID-19 period, there was no increase in the relative risk of developing FIC with or without UO. Increased accessions may represent business growth, increased pet ownership per capita or early recognition of clinical signs by owners spending more time with their pets.12,22 Alternatively, they may reflect an overall increase in morbidity due to unusual external events leading to increased stress. Stressors have been associated with physiological changes (such as an increase in pro-inflammatory cytokines) and increased sickness behaviours. Sickness behaviours are a group of non-specific clinical and behavioural signs. 13 Commonly reported sickness behaviours exhibited in response to stressors are vomiting, hyporexia and urination outside of the litter tray. 13
Change in activity (such as ability to roam) has previously been noted to influence stress and the development of cystitis.10,23 It is unknown if changes to human movement influenced the activity levels of cats. This study is unable to directly assess the extent and nature of environmental changes to individual cats in the COVID-19 period. A Spanish study assessing the effects of confinement during COVID-19 restrictions noted that while 46.3% of cats had no behavioural changes, a range of different behaviours were observed, from more relaxed (21.7%) and calmer (9.7%), to more demanding (36.4%), attention seeking (36.4%) and more nervous (7%). 20 Social preference can vary between individual cats and can be influenced by their genetics, early rearing and life experiences. 3 Some cats may have been affected positively by increased owner interaction and environmental enrichment. Studies have shown reduced frequency of feline lower urinary tract signs when multimodal environmental modification and environmental enrichment are utilised.12,24,25 Other cats may have experienced increased stress due to unfamiliarity, change in routine or intrinsic unpleasantness (increased ambient noise or auditory and visual stimuli).16,20,22,26 For cats owned by essential workers who left the house for work, movement restrictions may not have altered routine as much as for those with furloughed owners.
This study reports an FIC incidence of 4.27% (pre-COVID-19 group) and 5.43% (COVID-19 group), which is higher than previously published rates of 1.5–3.4%.1 –3,27 A cross-sectional study of 52 private veterinary practices evaluating 15,225 cats in the USA noted a FLUTD prevalence of 1.5%, compared with 3.4% in a 2001 study of 221,447 cats in North America.27,28 Another more recent study in 2016–2017 in Chiang Mai, Thailand diagnosed FLUTD in 2.24% of cats presenting to their teaching hospital.2,29 Neither incidence nor prevalence of FLUTD in Brisbane have been reported previously.
Risk factors for FLUTD include location, weather, diet and lifestyle.2,11 It is possible that the incidence of FIC is higher in Brisbane due to geographic variables. Predominantly young adult castrated male cats were presented with FIC/UO in this study, which is similar to the existing literature and consistent with the syndrome’s pathophysiology.2,30
In this study population, domestic shorthair cats were the predominant breed for both FIC with and without UO groups, similar to the existing literature.7,8 This is in contrast to other studies in Indonesia and New Zealand, which noted an association between Persian and other longhaired cats with FIC.9,23,30 It is unknown if this variability is related to the increased popularity of particular breeds in the area where they are affected.
Seasonal variation may contribute to the variation in cases of FIC. Southeast Queensland has a subtropical climate, with little temperature variation but seasonal differences in rainfall. Previous studies have noted an increased incidence of rainfall in New Zealand, as well as a peak in cases during Spring in northeast USA, which was not noted in the northwest.3,11,23 By including a whole 12-month period in both groups (Figure 2), this may mitigate for any seasonal variation that may exist; however, seasonal effect and monthly relative risk was not measured within this study.
Numerous limitations exist in this study, largely pertaining to its retrospective nature. The relatively small sample sizes may lead to a type II error, which may explain the non-significance of the results in this study. A post-hoc analysis was performed to evaluate the likelihood of type II error. Using the existing literature, the incidence of FIC was estimated at 0.81% (54% prevalence of FIC within a 1.5% prevalence of FLUTD). To identify an incidence of 3% with an alpha value of 0.05 and 80% power, 213 cats would be required, more than the current study population. However, both time periods demonstrated a higher incidence of FIC and UO; therefore, it is possible that this calculation is inaccurate for the regional population evaluated here. Pre-existing risk factors in cases were often unknown or medical records were incomplete. Many records did not include environmental information, such as outdoor access or multi-cat households, which could have changed and specifically contributed to stress. Some cats may have had existing stress factors to precipitate FIC independent of the pandemic. To minimise the influence of these unknown factors, only cats with their first occurrence of FIC or UO were included. However, the strict inclusion criteria may have prevented inclusion of a sub-population susceptible to a recurrence of FIC during COVID-19. In addition, the exclusion of 94 cases without a urinalysis in the COVID-19 group may have contributed to substantial selection bias.
The identification of cases was limited by the search options of the medical record database. The database allows searches by invoice item only. Buprenorphine was chosen as it is the most commonly used analgesic for the management of FLUTD at the authors’ hospital. Records were then searched and included if they were consistent with FIC. Cats with FIC that were untreated, prescribed alternative analgesics or euthanased may not have been identified. As a referral and emergency hospital, case presentations may not be reflective of the general population.
Cats with a history of FLUTD may be susceptible to a recurrence of FIC or UO in the presence of an unusual external event, such as COVID-19 lockdowns. Finstad et al 19 identified an increased prevalence of urethral obstruction in cats during COVID-19, which is different from the findings of this study. It is possible that cats with a history of FIC were more likely to develop recurrent FIC or UO during human movement restrictions. The recurrence rate for UO varies drastically in studies, with rates reported in the range of 16.7–36.0%, days after treatment.31,32,33 When including cases of FIC with or without UO, the recurrence rate increases to 51.0–65.0%.33,34 This study did not investigate if there was an increase in UO or non-obstructive FIC presentations in cats that had a history of FLUTD. The true incidence of FIC may be underestimated in this study.
The clinical signs of cystitis and the timing of its occurrence may differ among individual cats after experiencing stress. A study conducted by Westropp et al 35 in 2006 observed clinical signs, changes in bladder wall permeability and elevated levels of plasma catecholamine during the initial onset of stress. The potential delay in the onset of clinical signs may have affected the incidence when comparing the strict regional lockdown periods; however, it is unlikely to have affected comparisons between the two 12-month COVID-19 and pre-COVID-19 periods.
In this population, cases with positive bacterial urine sediment examinations were excluded. Previous literature evaluating urinalysis in cats has identified a low sensitivity (75.9%) and specificity (56.7%) for bacteria on wet unstained sediment.5,36 This increased with Wright staining to a sensitivity of 82.8% and specificity of 98.7%. 36 It is possible that the exclusion of cats with cytological bacteriuria has overestimated the incidence of UTI in this population and may have affected our estimate of the incidence of FIC during both periods.
Queensland had comparatively minimal social isolation practices and movement restrictions during COVID-19 compared with other Australian states and other countries.19,20 A similar American study found a significant increase in the prevalence of UO during the first 20 weeks of the COVID-19 pandemic compared with the prior 2 years. 19 Studies conducted in other geographical regions with longer periods of stricter movement restrictions during COVID-19 may yield different findings. In addition, the identification of ‘lockdown’ and ‘border restrictions’ were not clear in the first stages of the pandemic in Queensland. The nature of movement restrictions varied within the evaluated time frame and included the following: shutdown of public areas only; state border closure with free movement within the state; and full lockdown (isolation for all but essential services).21,37 COVID-19-related human movement behaviours were inconsistent throughout this period and therefore may have had a varied influence on cats.
Within the COVID-19 dataset, there were only three periods of total movement restrictions; these include 23 March to April 2020, 8–10 January 2021 and 29–31 March 2021. On 26 March 2020, restricted entry into Queensland was introduced, with permits being required for entry into the state.21,37 From 2 April 2020, people could not leave their principal place of residence except for work, food, medical requirements or exercise, which expanded on 9 April 2020 to the closure of all ‘non-essential’ businesses.21,37 Additional lockdowns occurred in the Brisbane region outside of the study dates, and different geographical regions (eg, Melbourne, Australia) had stricter movement restrictions for longer periods of time.21,37,38 Global variations in human movement restrictions and behaviours may explain the difference in findings between this study and the prevalence of UO reported in the USA. 19 An analysis of broader populations over a longer time frame may be required to assess the true influence of human movement restrictions on the greater cat population.
More research is required to further determine the significance of human movement restrictions on the risk of UO and non-obstructive FIC in cats. This study suggests that cats are not at higher risk of FIC from the implementation of movement restrictions on the human population, but this finding may reflect the reduced human movement restrictions of the pandemic in the geographical region evaluated. 19
Conclusions
In this study population, there is no evidence to suggest that the human movement restrictions imposed during the COVID-19 pandemic increased the presentation of FIC with or without UO in domestic cats. More research is required to be able to generalise these findings on a national or global scale.
Footnotes
Author note
The preliminary results of this study were presented at the Australian and New Zealand College of Veterinary Scientists’ virtual conference in 2021.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
