Abstract
Objectives
The aim of this study was to assess laser ablation-inductively coupled plasma-mass spectrometry (LA-ICP-MS) as a tool for measuring concentrations and determining accumulation of copper in frozen liver specimens from cats.
Methods
Six frozen liver specimens were evaluated by qualitative copper staining and quantitative flame atomic absorption spectroscopy. Tissue specimens were cryo-sectioned and quantitative bioimaging of copper was performed using LA-ICP-MS. Results were compared with those obtained using conventional methods.
Results
Of the six specimens, only one showed positive staining for copper with rhodanine. Using flame atomic absorption spectroscopy (FAAS), one specimen showed a deficient copper level (<100 µg/g dry weight), two specimens had copper within the reference interval (RI; 150–180 µg/g) and three specimens had copper concentrations above the RI. Bioimaging from LA-ICP-MS showed inhomogeneous distribution of hepatic copper. The areas with dense copper accumulation were represented as hotspots in the liver specimens. Hepatic copper quantification by LA-ICP-MS correlated well with copper quantified by FAAS (r = 0.96, P = 0.002).
Conclusions and relevance
Our findings suggest that quantitative bioimaging by LA-ICP-MS could be used to demonstrate the distribution and concentration of copper in frozen liver specimens from cats. The distribution of copper in these specimens was inhomogeneous with dense accumulation represented as hotspots on tissue sections. A positive correlation of hepatic copper concentrations determined by LA-ICP-MS and FAAS was found. Further studies to establish an RI for hepatic copper using this technique and to further determine its clinical utility are warranted.
Keywords
Introduction
Copper is a trace element that is essential for the growth, development and maintenance of living cells. 1 Copper metabolism occurs primarily within the liver where it is stored in hepatocytes before being exported to other tissues or excreted through the biliary system. 2 Alterations of copper excretion can induce excessive hepatic copper accumulation, which can promote oxidative stress via the Fenton reaction. 3 Hepatic injury induced by excessive copper is known as Wilson’s disease (WD) in humans and copper-associated chronic hepatitis (CuCH) in dogs.4–6 This disease is well-studied in humans and dogs. Genetic defects of hepatic copper transporters in people with WD and in dogs have been shown to be the cause of excessive hepatic copper accumulation.7–12 In addition, excess dietary copper intake is also suggested to contribute to this disease in dogs.13–15 CuCH has also been reported in cats and the characteristics of copper accumulation are similar to those found in dogs.16–19 Recent studies have suggested that hepatic copper accumulation occurs more commonly in cats than previously documented, and copper concentrations above the upper limit of previously established RIs are commonly found;20,21 nonetheless, the significance of increased concentrations and its association with the disease have not been clarified.
In veterinary medicine, a diagnosis of CuCH is based on histologic evaluation as well as conventional qualitative and quantitative copper assessments.6,12,22 Hepatic copper accumulation can be determined using copper-specific histologic staining with rhodanine or rubeanic acid.23,24 Copper granules in the liver are qualitatively scored, in the range of 0–5 (none to panlobular accumulation). 25 Hepatic copper concentration can be measured using flame atomic absorption spectroscopy (FAAS) or inductively coupled plasma-mass spectrometry (ICP-MS). In dogs, qualitative assessment of copper-specific histological stains showed a positive correlation with hepatic copper quantification. 13 A similar but weak correlation was demonstrated between these two techniques in cats. 21
Recently, new techniques for hepatic copper evaluation, such as digital image analysis based on histologic copper staining 26 and LA-ICP-MS, have become available. 27 Digital image analysis provides copper concentrations on copper-stained tissue sections. The distribution of copper is determined by copper staining. The stained tissue section is digitally scanned, and copper pigments are quantified with a positive pixel algorithm. This technique has been used to assess hepatic copper in dogs, cats and ferrets.18,26,28 Studies in humans with WD and rats suggested that LA-ICP-MS can be a useful tool for visualizing copper in tissue specimens.29–31 LA-ICP-MS is the technique where a laser unit is coupled with ICP-MS. The unstained tissue sections are scanned with a digital microscope and then processed in the LA-ICP-MS instrument. At the laser unit, the laser beam ablates across the tissue section and the ablated particles are transported by the carrier gas to the ICP-MS system. The elements of interest are then determined based on their mass-to-charge ratio and the concentrations are calculated. These data are converted to pixels and the image is reconstructed using in-house software. LA-ICP-MS enables the visualization of elements distributed in the tissue section and determines their concentrations. However, to our knowledge, none of the studies of copper accumulation in cats have previously been performed using this analytical method. The aim of the present study was to determine the feasibility of using LA-ICP-MS for copper assessment in frozen liver specimens from cats.
Materials and methods
Samples
Six frozen liver specimens from cats were used for this study. Post-mortem specimens were collected from feral cats that were euthanized for non-study-related reasons in Malaysia in 2017. The liver specimens (approximately 2 × 1 × 1 cm in size) were immediately placed on ice for 1–2 h. Then, the specimens were transferred to and stored at −80°C. The tissue specimens were sectioned into portions. Each specimen was used for conventional qualitative and quantitative assessments and for quantitative bioimaging. One set of tissue sections was used for qualitative copper assessment using rhodanine staining. Tissue sections were fixed in formalin and evaluated by routine histologic evaluation. All rhodanine-stained tissue sections were evaluated by the board-certified veterinary anatomic pathologist (boarded by the American College of Veterinary Pathology; RMG). Copper accumulation was scored from 0 to 5 (no accumulation to panlobular presence) based on the copper scoring system adapted from the scoring criteria used in liver biopsies in dogs. 26
Another set of liver tissue sections was used for copper quantification using FAAS. Based on the RI for FAAS, copper concentrations <100 µg/g dw were considered deficient and concentrations of 150–180 µg/g dw were considered normal. 32 The remainder of the specimens were kept at −80°C until further analysis was performed. These tissue sections (approximately 1 × 1 × 1 cm in size) were later embedded in an OCT medium and were shipped to Germany on dry ice to perform quantitative bioimaging of copper using LA-ICP-MS.
Quantitative bioimaging using LA-ICP-MS
The frozen liver tissue specimens were sectioned at 10 µm thickness at the Institute of Pathology at the Technical University Munich. The unstained cryosections were then evaluated by quantitative bioimaging of copper by LA-ICP-MS at the Institute for Inorganic and Analytical Chemistry at the University of Münster.
For the LA-ICP-MS process, the microscopic image of each tissue section (10 µm thick) was scanned with a digital microscope (BZ-9000; Keyence) before ablation. For the ablation, a laser ablation instrument (imageBIO266; Elemental Scientific Lasers) was used. A scan speed of 100 µm/s was applied and the laser fluence was set to 2.2 J/cm2 during the ablation. All samples were ablated with a spot size of 25 µm. The ablated particles were flushed with a constant helium flow of 1 l/min and combined with an additional argon gas flow behind the ablation cell. After laser ablation, an ICP-MS system (iCAP TQ; Thermo Fisher Scientific) was used to determine copper. In essence, with this analytical method, the copper in the tissue was atomized and ionized using the plasma source and passed into the mass spectrometer. The copper ions were then separated based on their mass-to-charge ratio in the triple quadrupole system.
Matrix-matched gelatin standards were used for external calibration. In total, 11 gelatin standards covering the range of elemental concentrations (0–100 µg/g) were used. The final concentrations of the standard aliquots were validated using ICP-MS analysis. The external calibration by LA-ICP-MS was performed under the same conditions as the cryosections of liver specimens. To generate the calibration curve, 11 lines of each standard were ablated, and the first line was dismissed for further data processing due to reasons of consistency. The average signal intensity of 10 lines was calculated and plotted against the elemental concentrations. For the pixels that showed a higher concentration than 100 µg/g, the calibration line was extrapolated. All data were processed with in-house developed software (ImaJar 3.64, written by Robin Schmid). The mean elemental concentration was determined by averaging the signal of all pixels within the tissue sample. A phosphorus signal was used to depict the tissue structure. The concentration was reported as µg/g wet weight (ww). The quantitative distribution of copper was presented as a heat map with the rainbow color scale.
Statistical analysis
Normality of the data was determined using the Shapiro–Wilk test. Pearson’s correlation test was used to determine the relationship between hepatic concentrations assessed by LA-ICP-MS and those measured by FAAS. Data were analyzed with a commercially available statistical software package (Prism 9; GraphPad Software). Values of P <0.05 were considered statistically significant.
Results
Six liver specimens were collected from three male and three female cats. The estimated ages of the cats were in the range of 1–15 years. The data on histopathological findings and the results from traditional qualitative and quantitative copper assessments are summarized in Table 1. Four specimens were reported as having no significant histopathological hepatic changes and the other two were reported as having hepatic steatosis. Only one of six specimens showed positive rhodanine staining with a copper score of 2/5. The copper granules accumulated in the centrilobular and midzonal areas. Based on the RI for FAAS, hepatic copper concentrations were below the level considered deficient (<100 µg/g dry weight [dw]) in one specimen, two specimens had concentrations within the RI (150–180 µg/g dw) and three specimens had concentrations above the upper limit of the RI.
Summary of histopathological analysis of the liver specimens with the results for qualitative copper staining, hepatic copper concentrations quantified by FAAS and LA-ICP-MS

Reference interval 150–180 µg/g dry weight
FAAS = flame atomic absorption spectroscopy; LA-ICP-MS = laser ablation-inductively coupled plasma-mass spectrometry
Quantitative bioimaging of copper in the tissue specimens using LA-ICP-MS was depicted as heat maps with a rainbow scale (Figure 1). The highest copper concentration of the tissue section is represented by the red color, while the lowest concentration is represented by purple. The hepatic copper concentrations ranged from below the detection limit (0.9 µg/g ww) to above 1000 µg/g ww in the case of ID5.

Quantitative bioimaging of copper in hepatic tissue specimens from six cats. The rainbow scale represents the elemental copper concentrations as determined by laser ablation-inductively coupled plasma-mass spectrometry. (a–f) Sample IDs 1–6, respectively
The mean hepatic copper concentrations determined by LA-ICP-MS are shown in Table 1. The data of copper concentrations from both quantitative techniques were normally distributed. Pearson’s correlation test showed that the copper concentrations measured by LA-ICP-MS and FAAS were significantly correlated (r = 0.96, P = 0.002) (Figure 2).
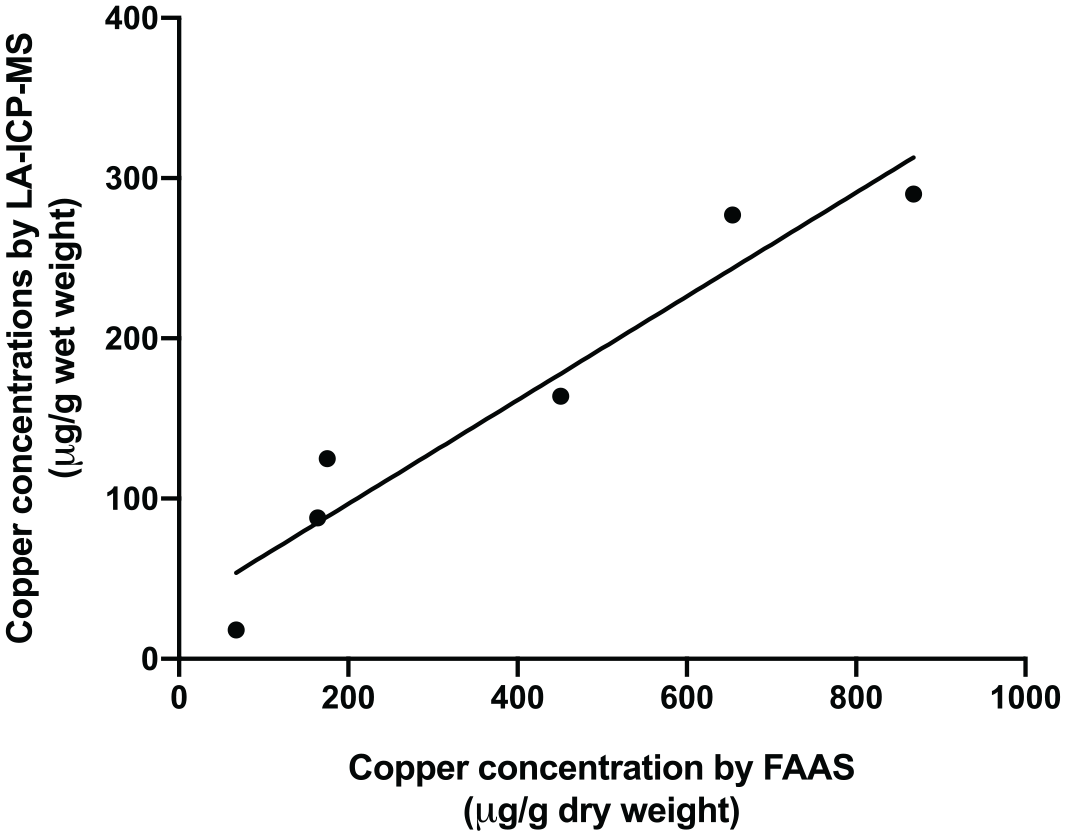
Correlation between hepatic copper concentrations determined by laser ablation-inductively coupled plasma-mass spectrometry and those determined by flame atomic absorption spectroscopy (r = 0.96, P = 0.002)
Discussion
This is the first study to describe LA-ICP-MS-based quantitative bioimaging of copper in liver specimens from cats. We assessed hepatic copper concentrations and its distribution in frozen liver specimens from six cats using LA-ICP-MS. We compared these results with those by rhodanine staining and FAAS.
Quantitative assessment by FAAS showed that 3/6 specimens had copper concentrations above the upper limit of the RI (150–180 µg/g dw). However, based on previous studies, there is considerable doubt concerning the validity of this RI.21,33 In contrast to the quantitative findings, positive rhodanine staining was found in only one specimen. The reported copper score was 2/5 in this specimen, with accumulation in the centrilobular and midzonal areas. There were no significant histological changes in this specimen. Based on the previous criteria of CuCH and the findings of other studies in cats,18–20 primary CuCH seems unlikely in any cats included in our study.
Studies in dogs showed that hepatic copper concentration and copper staining scores were moderately correlated (r = 0.66 and rs = 0.75, all P <0.001).13,34 Our previous study in archived cat liver specimens found a weak correlation between these two methodologies (rs = 0.46, P <0.001). 21 One possible explanation for this relatively weak correlation could be that these two modalities detect different forms of copper. Rhodanine stain is suggested to detect protein-bound copper rather than detecting the copper itself since this chelating agent has a strong affinity for proteinaceous copper deposits.35,36 On the other hand, FAAS is a technique that uses a flame to disassociate the metal elements in the tissue specimens into atoms. 37 Both free and bound forms of copper would be detected by FAAS.
LA-ICP-MS is an advanced technique that has been suggested to be a useful tool for the quantitative bioimaging of copper in tissue specimens. 27 It combines technologies of optical imaging and elemental quantification. The two-dimensional mapping of elements is visualized, and the concentrations are quantified by their mass ratio. LA-ICP-MS can provide information concerning both copper concentration and elemental distribution of copper by assessing a single tissue sample. In contrast, conventional qualitative and quantitative methods require submission of two separate tissue samples. Thus, LA-ICP-MS could be advantageous when the number of samples is limited. LA-ICP-MS has not only been suggested to be an add-on for WD diagnosis, it could also be used to monitor the disease progression or treatment efficacy.31,38–40 LA-ICP-MS can be performed on either fresh cryosections or formalin-fixed paraffin-embedded tissue sections. 27 However, the tissue types selected should be considered in accordance with the elements of interest. For instance, a study of elemental imaging in humans found a decrease in the distribution pattern of copper and zinc in formalin-fixed tissue sections of the brain. 41 These investigators suggested that fresh tissue is required for the determination of these two elements. Another study found that metal elements could be affected by the formalin-fixed paraffin-embedded tissue process, especially alkaline metals compared with the transition metals. 42 In comparison, the use of fresh-frozen cryosections in this study minimized any tissue wash-out of the trace elements. LA-ICP-MS allows direct analysis of 5–100 µm-thick cryosections. 27 In addition, using fresh cryosections requires no additional sample preparation. This also helps minimize the contamination of tissue samples.
Bioimaging of copper revealed an inhomogeneous distribution of the copper, even in specimens where copper was not visualized using rhodanine staining. Most of the samples had red-colored hotspots throughout the tissue sections. The hotspots represent the highest concentration of copper for each liver sample. It is known that there is a variation in copper distribution between liver lobes in humans and rats.43,44 The uneven metal distribution was also found within the liver tissue of dogs.34,45 Our findings of inhomogeneous distribution emphasized the importance of the collection of multiple liver specimens, which would ideally be collected from multiple areas.
With FAAS copper quantification, the specimen is desiccated during tissue preparation, followed by analysis. The total tissue specimen would have its constant weight after the removal of moisture and water content. After the atomization and the detection of the absorbance of copper atoms, the concentration is calculated and reported based on the dry weight of liver tissue, which is approximately four times that of the wet-weight concentration. 32 On the other hand, copper quantification by LA-ICP-MS was performed on liver cryosections. The cryosections were prepared without drying and, thus, represented as wet-weight samples, the results of which were calculated and reported based on wet-weight tissue. Therefore, the RI for FAAS, which is based on dry-weight tissue, could not be applied to the data reported by LA-ICP-MS. The interpretation of copper concentrations, such as deficient, normal or excessive, could not be determined. However, a strong correlation between the concentrations determined by these two quantitative methods was found. This suggests that LA-ICP-MS can be used to assess copper concentrations in cat liver specimens and would provide similar results to FAAS. Although there was a correlation between these methods, there might be factors that may cause variation (eg, moisture content, inhomogeneous distribution) and further investigations would be needed. In addition, investigations to establish species-specific RIs for hepatic copper in both cats and dogs for LA-ICP-MS are warranted. Moreover, the cryosection would represent in vivo conditions but it also has a tissue heterogeneity characteristic, which would make it difficult to estimate the precise constant factor. Therefore, to establish the RIs using cryosections, the condition of tissue samples, such as sample size and storage duration, would have to be standardized to minimize the variations among the tissues. 46
The present study has some limitations. The aim of the study was to show that LA-ICP-MS is feasible in cats. Therefore, the sample size was small, which could limit the power of the statistical analysis and lead to a type II error. Moreover, freezing artefacts were found in the cryosections. This could have been caused by the slow freezing in the tissue specimen collection procedure and the instability of temperature during the tissue delivery process. The artefacts could cause tissue damage and affect the achievable spatial resolution of the analysis. Thus, small copper granules in the lower range might have been difficult to identify. In addition, this technique requires user expertise. It might thus limit the use or the availability of the LA-ICP-MS to only well-established or well-equipped laboratories.
Conclusions
LA-ICP-MS was successfully used for the assessment of copper concentration and spatial distribution in liver specimens in cats. This technique allows for bioimaging and the determination of copper concentrations at the same time. Inhomogeneous distribution of copper was found with dense hotspots of copper accumulation in liver specimens from the cats evaluated, even though most stained negatively for copper using conventional rhodanine staining. The hepatic copper concentrations determined by LA-ICP-MS were strongly correlated with those measured by conventional copper quantification (FAAS). Determination of an RI for hepatic copper concentrations using this technique in cats is warranted. Further studies assessing the utility of LA-ICP-MS for the investigation of CuCH in cats and dogs are warranted.
Footnotes
Acknowledgements
The authors would like to thank Dr Sue Yee Lim and Dr Mazlina Mazlan from Universiti Putra Malaysia for providing liver specimens for this study. We would also like to thank Olga Seelbach and Ulrike Mühlthaler from Comparative Experimental Pathology, TUM for their technical assistance in tissue cryosectioning.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
