Abstract
Objectives
The purpose of this study was to describe B-mode and colour Doppler ultrasound (US) features of gastric lymphoma in cats.
Methods
Cats with histopathological or cytological confirmation of gastric lymphoma that underwent abdominal US were included in this retrospective study. Clinical signs were recorded and radiographs reviewed when available. Gastric lesions were ultrasonographically evaluated for location, distribution, wall layering, echogenicity and thickness. Colour Doppler assessment was based on vessels, location and direction.
Results
Forty-five cats were included. All presented with vomiting, 7/45 (15.5%) with haematemesis. Radiographs were available in 27/45 cases, and demonstrated a gastric mass in 19/27 (70.4%) cases. The most common US appearance was transmural wall thickening with absent layering 42/45 (93.3%) and hypoechogenicity 39/45 (86.7%). Median maximum thickness was 2.1 cm. Ulceration or erosion was suspected in 23/45 (51%) cases, two of which perforated. Colour Doppler images were available in 33/45 cases. The most common Doppler finding was transmural vessels coursing perpendicularly to the lumen in 14/33 (42.4%). Abdominal effusion was uncommon (n = 11/45; 24.4%). Regional lymphadenopathy was frequent, observed in 44/54 lymph nodes reviewed on US, and US suspicion of splenic, hepatic and intestinal lymphomatous infiltration was reported in 24/45 (53.3%) cases.
Conclusions and relevance
The most common US appearance of gastric lymphoma was transmural hypoechoic segmental wall thickening with layering loss. Location and distribution were variable. Colour Doppler displayed increased blood supply with a predominant pattern of perpendicular vessels, possibly representing aberrant arteries originating from subserosal and submucosal plexus. Lymphomatous infiltration of lymph nodes and other abdominal organs was frequent, confirming local and distant aggressiveness of gastric lymphoma in cats. Vomiting and haematemesis were frequently associated with the suspicion of ulcer on US. Perforation was a rare occurrence.
Introduction
Gastrointestinal lymphoma (GIL) is the most common form of lymphoma in cats.1–3 It can involve the gastrointestinal (GI) tract at any site with a focal or multifocal distribution, among which an exclusive involvement of the stomach is relatively uncommon, representing only 18% of feline GIL presentations. 4
Histopathological classification of GIL includes different subtypes based on immunophenotype (T cell or B cell), cell size (small or large) and histological grade (low, intermediate, high).1,5–8 Regarding immunophenotype, B-cell phenotype predominance has been observed in gastric lymphoma vs the more common T-cell phenotype of small intestinal lymphoma. 4
Abdominal ultrasound (AUS) is a well-established diagnostic modality to assess feline GI tract. Several studies have described the ultrasound (US) features of GIL and have reported a variety of US signs associated with GIL.9–14 Hypoechoic circumferential wall thickening with loss of layering, reduced motility and regional lymph node enlargement are the most frequently described US findings.9–11 The ultrasonographic appearance of GIL can differ significantly, depending on the anatomical location and distribution of neoplastic lesions, as well as histological grade. The disappearance of wall architecture, severe thickening and masses are usually representative of high-grade alimentary lymphoma (HGAL) or intermediate-grade alimentary lymphoma.9,12 Intestinal low-grade alimentary lymphoma (LGAL) is usually associated with less aggressive US findings such as diffuse wall thickening with preserved layering and relative thickening of the muscularis propria.13,14
Overall, descriptions of feline primary gastric lymphoma (FGL) are scarce in comparison with the extensive literature investigating the US characteristics of intestinal lymphoma. In addition, none of published reports provided detailed information on the vascular assessment of gastric lymphomatous lesions using Doppler US.
Doppler US is a safe and rapid imaging modality to assess macroscopic perfusion of organs and lesions. Assessment of peripheral lymphomatous lymph node perfusion through combined power Doppler and contrast-enhanced US (CEUS) has been proven effective in accurately depicting lymph node angioarchitecture and identifying criteria of malignancy in dogs. 15 Specifically, Doppler US was superior to CEUS in distinguishing pericapsular vessels malignant pattern. 15 We hypothesised that Doppler examination may be of value for the characterisation of perfusion patterns in gastric lymphomatous lesions.
A recent study described the vascular features of gastric inflammatory and neoplastic diseases in cats by means of CEUS, 16 whereas no studies have described Doppler vascular patterns of FGL.
The purpose of this study was to describe B-mode US and colour Doppler features of FGL cases in order to optimise imaging diagnosis and to provide an overview of clinical presentation, diagnostic imaging findings, and local and distant spread of the disease at time of diagnosis.
Materials and methods
The medical records of cats diagnosed with gastric lymphoma at Tufts Cummings School of Veterinary Medicine, USA, and the Veterinary Teaching Hospital – University of Bologna, Italy, between 2006 and 2020, were reviewed.
Inclusion criteria were complete AUS with access to archived images and video clips of the stomach, written US report, and cytological or histological reports confirming FGL diagnosis.
Signalment, clinical history and physical examination findings were recorded. When available, staging tests, including complete blood count, serum biochemical analysis, retroviral testing and thoracic radiographs, were evaluated. Treatment protocols and survival time were recorded, when available.
Original cytological and histopathological reports were reviewed. In order to standardise FGL characterisation based on two different diagnostic methods, a simplified classification system based on cell size and tumoral grading was adopted. FGL was classified as small cell (nuclei approximately 1× the diameter of erythrocytes), intermediate (nuclei 1.5× the diameter of erythrocytes) or large cell (nuclei 2–2.5× the diameter of erythrocytes). Tumoral grading and immunophenotyping were recorded, when available.
The original US reports and the archived DICOM images and videos were reviewed by board-certified radiologists (ADJ and DP) and a third-year resident (MQ) at their respective institutions. Before image review, a consensus was made between the readers to address the following criteria: lesion location (gastric fundus, body or antrum); distribution (transmural symmetric or asymmetric, focal or multifocal); wall layering (normal, altered or absent); and echogenicity (hypoechogenicity, hyperechogenicity, homogeneous or heterogeneous). Wall thickness was measured with electronic callipers at the thickest point of the lesion from the mucosal–luminal interface to the outer hyperechoic serosal layer. Lesion extension was measured, when possible, in millimetres, using electronic callipers. If multifocal lesions were present, the one with the greatest thickness and extent was measured and recorded. Other relevant findings were noted, including intramural gas suggesting ulcers, hyperechogenicity of adipose tissue adjacent to the stomach, presence hypoechoic tissue plaques throughout peritoneal fat consistent with lymphomatosis, and the presence of abdominal effusion. 17
Colour Doppler assessment of gastric lesions was based on visualisation of a single or multiple vessels, their location within the lesion (peripheral or transmural) and vessel direction compared with the luminal surface (parallel, perpendicular or mixed tortuous).
When archived images of regional lymph nodes were available, their size, shape and echotexture were recorded, as well as perinodal hyperechoic fat. Lastly, abnormalities of the remainder abdominal organs were recorded.
Basic descriptive statistics were used to characterise the study population. Continuous variables were expressed as median and range.
Results
Forty-five cats met the inclusion criteria: 24 spayed females, 20 neutered males and one intact male. Median age was 9.5 years (range 9.5). Breeds included domestic shorthair (n = 29; 64.4%), domestic longhair (n = 8; 17.8%), Siamese (n = 2; 4.4%), Siberian (n = 2; 4.4%) and one each (2.2%) of Maine Coon, Persian, Chartreux and Bengal.
All cats presented with vomiting. Seven cats (15.5%) had haematemesis, five of which had suspected ulcer (n = 3; 6.7%) or erosion (n = 2; 4.4%) based on US findings. Other common clinical signs at presentation included weight loss (n = 18; 40%), decreased appetite (n = 26; 57.8%) and lethargy (n = 13; 28.9%). A cranial abdominal mass was suspected on physical examination or initial radiographs in eight cats (17.8%). Laboratory findings were non-specific and included anaemia (n = 10; 22.2%), neutrophilic leukocytosis (n = 9; 20%), hypoalbuminemia (n = 5; 11.1%) and azotaemia (n = 7; 15.5%). Feline leukaemia virus and feline immunodeficiency virus (FIV) testing was performed in 14 cats (31.1%) cats, three (6.7%) of which were positive for FIV. Radiographic studies, either thoracic or abdominal, were available in 27 cases (60%) and demonstrated gastric thickening or mass in 19/27 (70.4%) cases.
Data regarding treatment and outcome were unavailable in seven cats (15.5%). Of the remaining 38 cases, 16 (42.1%) did not receive chemotherapy (declined by owners), or owners elected euthanasia due to their cats’ poor quality of life and 22 (57.9%) received chemotherapy. Information on chemotherapy protocols was sparse and inconsistent, and lead to inconclusive data. Of the cats that received treatment, 8/22 (36.4%) were lost to follow-up. Survival time from diagnosis was retrievable in 14/22 (63.6%) cases and ranged from 7 to 1685 days (median 95.5).
Samples from gastric lesions, lymph nodes and other abdominal organs that lead to diagnosis of lymphoma were obtained prior to treatment commencing in all cats (n = 22/22; 100%) that received chemotherapy.
FGL diagnosis was achieved by histopathological analysis in 14/45 cases. Samples were obtained by endoscopic biopsy (n = 10; 22.2%), surgical biopsy (n = 1; 2.2%), US-guided core biopsy (n = 1; 2.2%) and necropsy (n = 2; 4.4%). Immunohistochemical analysis was performed in 4/14 cases, of which two were diagnosed as small B-cell LGAL and two as large B-cell HGAL.
The remaining 31/45 cases were diagnosed by cytology alone following US-guided fine-needle aspiration. A full list of diagnoses is provided in Table 1. A final diagnosis of large-cell lymphoma was obtained in 31/45 cases (68.9%) and medium-cell lymphoma was diagnosed in 4/45 cases (8.9%). Small-cell lymphoma was confirmed in 3/45 cases (6.7%).
Diagnosis and relative diagnostic methods of the 45 cats included in the study

Cytological samples of other abdominal organs were available in seven cases (15.5%) and confirmed concurrent lymphomatous infiltration of gastric and hepatic lymph nodes (n = 1; 2.2%); kidneys, spleen and liver (n = 1; 2.2%); kidneys (n = 1; 2.2%); and spleen (n = 2; 4.4%). In one case, FNA of the gastric lymph node was inconclusive. In another, FNAs of the small intestine and regional lymph nodes were suggestive of inflammation. In one of the two cases diagnosed by necropsy, samples collected for histopathology proved lymphomatous infiltration of the diaphragm, jejunum, kidneys and peritoneal fat.
AUS was performed with three imaging systems (iU22, Epiq 5G and Epiq 7G ultrasound system [Philips Healthcare]) at the two institutions, each equipped with linear (5–18 MHz, 7–15 MHz, 5–12 MHz) and curvilinear (5–8 MHz) transducers.
Location, US appearance, and the size and extent of the gastric lesions are summarised in Table 2. The most common US appearance was transmural wall thickening with absent layering (n = 42; 93.3%) and hypoechogenicity (n = 39; 86.7%) (Figure 1).
Ultrasound distribution and appearance of feline gastric lymphoma (FGL) lesions in 45 cats

Data are presented as n (%) unless otherwise indicated
Lesions were too extensive to measure their length
NA = not applicable
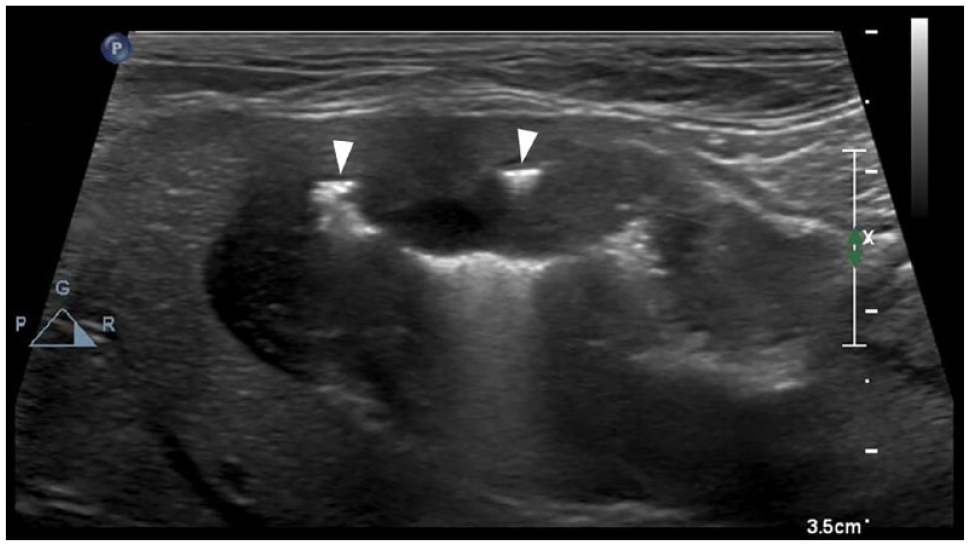
Ultrasound image of a cat diagnosed with high-grade feline gastric lymphoma showing severe circumferential transmural thickening, loss of layering and hypoechogenicity. The hyperechoic reverberating foci within the gastric wall (arrowheads) are consistent with ulceration
Lesions with altered wall layering included a single nodule within the mucosal or submucosal layer (n = 2; 4.4%), and loss of mucosal and submucosal detail, while the muscular and serosal layers were spared (n = 1; 2.2%) (Figure 2).

Ultrasound image of the gastric fundus in a cat with intermediate to high-grade feline gastric lymphoma. An ovoid hypoechoic nodule is observed in the submucosa (arrowhead). Wall layering is otherwise normal. The same patient had a segmental hypoechoic thickening with loss of layering in the region of the gastric body
In the remaining cases, the lesions were too extensive or spanned from lesser to greater curvature of the stomach, making their length unmeasurable.
Ulceration or erosion were suspected on US images in 23 cases (51.1%). Only two cats with US features of ulcers had pneumoperitoneum, close to the stomach, consistent with gastric perforation. In four cases (8.9%), multiple hypoechoic nodules were observed within the hyperechoic fat surrounding the lesions, one of which was confirmed as lymphomatosis on cytology.
Colour Doppler videoclips or still images were available in 33 cases (73.3%), of which all but one demonstrated multiple vessels within the lesions.
Transmural vessels perpendicular to the gastric lumen were seen in 14/33 cases (42.4%; Figure 3a). Eight lesions (n = 8/33; 24.2%) demonstrated a combination of transmural perpendicular vessels and a large peripheral vessel paralleling the lumen (Figure 3b). Other colour Doppler patterns observed included multiple transmural tortuous signals (n = 3/33; 9%), a single large peripheral vessel (n = 2/33; 6%) and a single transmural vessel parallel to the lumen between the mucosa and submucosal layers (n = 1/33; 3%). In 5/33 cases, Doppler examination was limited to still images, making colour Doppler pattern unclear.

Examples of Doppler patterns associated with feline gastric lymphoma. (a) Multiple vessels perpendicular to the gastric lumen and (b) combination of perpendicular vessels and a large peripheral vessel paralleling the lumen, resembling a comb-teeth like pattern
Regional lymph nodes were imaged in 38 cases (84.4%), including gastric (n = 31; 68.9%), pancreaticoduodenal (n = 17; 37.8%), hepatic (n = 3; 6.7%) and splenic (n = 3; 6.7%). Of these, 44/54 lymph nodes (81.5%) were enlarged, and demonstrated abnormal features such as heterogeneous parenchyma, decreased echogenicity and hyperechoic perinodal fat. Mild-to-moderate abdominal effusion was observed in 11/45 cases (24.4%). Concomitant abnormalities of other abdominal organs included diffuse thickening of the small intestinal muscularis layer (n = 6 [13.3%]: 3/6 with overall thickened wall and 3/6 with normal wall thickness) and segmental hypoechoic thickening of the small intestine with loss of layering (n = 2; 4.4%). The spleen was enlarged and/or heterogeneous in four (8.9%). Hepatomegaly with altered echotexture and echogenicity of the hepatic parenchyma was observed in 11 (24.4%) cases. Bilateral renal lymphomatous infiltration was suspected on US in one (2.2%) case, and confirmed on cytology.
Discussion
Over a period of 14 years, a relatively small number of FGL cases were documented at both institutions participating in this study. Accordingly, previous studies reported a primary gastric location to be a rare occurrence of GIL, which is most often localised to the small intestine. 4 In the present study, cats with a single gastric location accounted for 88.9% of the population, whereas concomitant small intestinal involvement was confirmed on necroscopy in only one case, and suspected in another based on ultrasonographic features suggestive of neoplasia. The affected small intestinal segments demonstrated focal circumferential wall thickening with decreased echogenicity and loss of layering, corresponding to the most commonly reported US appearance of large-cell GIL.9,11,12 In agreement with literature, the histopathological findings of confirmed cases in our study population were consistent with a dense population of large neoplastic lymphocytes infiltrating the gastric and small intestinal wall.
Diffuse thickening of the muscularis layer was observed in six cases; however, none of these cats underwent full-thickness biopsy to confirm aetiology. As previously reported, diffuse muscularis thickening with otherwise-preserved wall layering is a US feature commonly found in cats with small T-cell lymphoma, inflammatory bowel disease and eosinophilic enteritis; discriminating between these conditions solely with US is not feasible.13,14,18 In our study, all but one of the cats with small intestine muscularis thickening were diagnosed with large or medium-to-large cell FGL. In our opinion, it is unlikely that those cats had concurrent large-cell FGL and small T-cell lymphoma of the small intestine, although this could not be ruled out without performing intestinal biopsies.
The majority of cats in this study were adults or older. The median age (9.5 years) was similar to previously reported cats with primary gastric lymphoma or GIL.2–4,9,19
Cats with gastric neoplasia often present with chronic vomiting, anorexia and weight loss.19,20 In our study population, vomiting and haematemesis were the most common complaint at the time of diagnosis. Haematemesis was frequently associated with suspicion of gastric erosion and ulcers on US. Gastric ulceration results from damage to gastric mucous membrane through impairment of bicarbonate buffer, gastroprotective prostaglandins, mucous layer secretion and decreased blood flow. 22 Predisposing factors to erosion and ulceration include neoplasia, gastritis, ischaemia, steroidal and non-steroidal anti-inflammatory drugs, hypergastrinaemia, and renal and hepatic failure. 21 When cytoprotective mechanisms are overwhelmed, mucosal erosion and bleeding occur leading to clinical signs of vomiting, haematemesis, melaena and pale mucous membrane. 21 Based on pathophysiology, the frequent occurrence of haematemesis in FGL cats with suspicion of gastric erosion is therefore not surprising.
Interestingly, two of our FGL cats with gastric ulcers had US signs of perforation. Spontaneous perforation associated with gastrointestinal neoplasia is an uncommon presentation in cats. In a published study of 13 cats with gastrointestinal perforation, lymphoma was identified as the most common underlying cause. 22 Another report of two cats that presented with spontaneous pneumoperitoneum identified FGL as the cause of gastric perforation in one cat. 23 Our findings are similar to previous publications and reinforce the importance of a thorough US examination, possibly combined with abdominal radiographs, in cats with FGL to rule out gastric perforation that may significantly affect treatment options and prognosis.
It is worth noting that a gastric mass was often suspected on review of survey radiographs. The potential of identifying a gastric tumour on radiographs depends on multiple variables, including tumour size, discrete mass vs diffuse lesion, and the presence of luminal gas providing spontaneous contrast. 24 While radiography has been largely replaced by ultrasound, endoscopy and, more recently, CT in the diagnostic work-up of gastric neoplasia in veterinary patients, 25 our results support its value as a screening tool. Conventional radiography is inexpensive, widely available and should be considered a valid initial test when gastric neoplasia is clinically suspected, especially in clinical settings where US evaluation is not readily available.
Previously published data showed that cats diagnosed with FGL undergoing chemotherapy had a median survival time of 171 days. 19 Furthermore, survival was significantly higher in cats that achieved complete remission compared with those that achieved partial remission. 19 Median survival time in the present study population was shorter (95.5 days). However, significance of this result was hindered by limitations inherent to retrospective study design, including the small number of cases that received treatment, lack of complete tumoral staging in some cases and inconsistent information on chemotherapy protocols.
The most common US appearance of FGL was transmural hypoechoic segmental wall thickening with loss of layering, similar to previous reports of HGAL.9,16,26 The location and distribution of the lesions were variable, but overall the body and fundus seemed more frequently affected than the pyloric antrum. Masses associated with GIL have been reported as single or multifocal along the gastrointestinal tract,3,9–12,19 although no specific distribution pattern within the gastric wall was described. Owing to the paucity of studies describing primary FGL, information regarding distribution of the lesions within the gastric wall is lacking and prevents any comparison. Further studies are needed to confirm the distribution trend in gastric fundus and body described in the present study.
All lesions were highly vascular on colour Doppler. The most common Doppler feature was the presence of numerous parallel vessels oriented perpendicularly towards the gastric lumen. This Doppler pattern resembles the comb-teeth like vessels described on CEUS in human patients with gastritis, 27 and in a recent study of cats with gastric lymphoma. 17 In human medicine, comb-teeth branches on CEUS are believed to represent the normal arterial branches from the gastric wall. Their absence is considered to be an expression of gastric cancer in humans, as tumour invasion disrupts the natural vascular architecture, while tortuous irregular vessels are formed by pathological angiogenesis. 16 The significance of a comb-teeth pattern in cats with FGL is unclear. We hypothesised that, in FGL, the natural vascular architecture of the gastric wall is preserved, owing to the infiltrative nature of lymphoma compared with neoplastic tissue growth of epithelial and stromal tumours. Hence, the vessels seen on colour Doppler interrogation may represent hyperaemic arterial branches originating from subserosal and submucosal plexuses.
Large-cell lymphoma was over-represented in the present study, in agreement with previous studies that have shown a predominance of large B-cell lymphoma originating in the stomach.6,19 Presumed lymphomatous infiltration of the regional lymph nodes (81.5%) and perigastric fat (8.9%) were rather common findings in the present study, whereas multiorgan involvement (either presumed or confirmed on pathology) was present in 53% of the cases. These results confirmed the local and distant aggressiveness of FGL. Moreover, one could speculate that the higher percentage of regional lymph nodes compared with distant organs infiltration reflects a temporal line: FGL first spreads locally to regional lymph nodes, then distantly to other abdominal organs.
The main limitation of the present study is its retrospective design. Interpretation of FGL samples was performed by different pathologists at the two institutions over time, with a consequent lack of standardisation in diagnostic approach. In the majority of cases, a diagnosis of FGL was achieved through cytology alone, which is not the gold standard. Pathology reports were, in some instances, lacking information on phenotype or FGL grading.
In addition, the lack of a standardised US and colour Doppler protocol may have affected image quality and interpretation.
Conclusions
The findings of the current study indicated that large-cell lymphoma was the most frequently diagnosed subdivision of gastric lymphoma in cats. The aggressiveness of FGL was reflected by the US findings of severe wall thickening, complete loss of layering, regional lymphadenopathy and multiorgan involvement. On Doppler interrogation, multiple vessels arranged in a comb-teeth like pattern was a frequent feature. However, further studies are warranted for better understanding of perfusion patterns, ideally comparing the Doppler appearance of different tumour types and inflammatory conditions.
Footnotes
Author note
The study was conducted at the Department of Veterinary Medical Sciences of University of Bologna, Italy, and the Tufts Cummings School of Veterinary Medicine, USA. This paper was presented, in part, at the 2021 EVDI online meeting.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent either verbal or written was obtained from the owner or legal custodian of all animals, including cadavers) for all procedure(s) undertaken. No animals or people are identifiable within this publication, and therefore additional informed consent for this publication was not required.
