Abstract
Practical relevance:
Despite substantial advances in assisted reproductive techniques having been recently reported in cats, the use of these is limited and routine application is still far from being a reality in veterinary clinics. Nevertheless, there is an increasing demand from domestic cat breeders for artificial insemination (AI) techniques that are already commonly used in dogs. Where natural breeding is not possible in tom cats and queens of high breeding value, AI could offer a solution. Clinical challenges: AI in cats is more difficult than in other species – both in terms of semen collection/handling and oestrous cycle management given that ovulation must be induced.
Aim:
For practitioners wishing to perform AI in queens, there are challenges to overcome, and a good understanding of the techniques and procedures involved is pivotal. This review aims to contribute to improved knowledge by providing an overview of AI protocols, encompassing choice of breeding animals, procedures for semen collection, oestrus and ovulation induction, AI techniques and equipment.
Equipment and technical skills:
Depending on the animals involved and the specific AI technique chosen, essential equipment may include an artificial vagina, electroejaculator, endoscope (sialendoscope, which can be fairly expensive) and special catheters for transcervical insemination. Other instrumentation and materials needed are typically readily available in a veterinary clinic. In general, no particular skills are needed to perform the procedures described in this review, with the exception of endoscopic transcervical catheterisation, where the ability to use an endoscope is required.
Evidence base:
The information and advice/recommendations provided are based on specific feline research and reviews published in scientific peer-reviewed journals, animal reproduction textbooks, and presentations at national and international congresses. The authors also drew on their own clinical experience with regard to the choice of protocols and procedures presented in this review.

Introduction
Although artificial insemination (AI) in the cat was first described in 1970s 1 and, during the following five decades, substantial advances in feline-assisted reproduction were reported, its use in cats is limited, particularly in comparison with dogs where this practice is very common. AI in cats is more difficult than in other species, not only in terms of semen collection and handling, but also oestrous cycle management given that ovulation must be induced. 2 Nevertheless, there is growing demand for semen collection, evaluation and subsequent use by AI as a means of preserving important or valuable genetic material, 3 for disease control (eg, respiratory infections) and in order to bypass psychological or physical breeding problems. AI is additionally frequently used in a research setting; for example, to develop practical and reliable domestic animal models to improve techniques for the reproduction of endangered wild felids, and to breed experimental cats used as models for human disease.
Choice and evaluation of breeding animals
In the authors’ opinion, AI should be considered only for animals in which natural mating is not achievable due to acquired (and not heritable) conditions; in particular, for problems associated with copulation (fear-fulness, inexperience, distraction, mate preference), painful (eg, acquired orthopaedic or oral) conditions, and genital disorders in the tom cat and/or queen (eg, phimosis, persistent penile frenulum or acquired pathologies). Even if a tom has a history and/or evidence of infertility (eg, poor semen quality due to an acquired condition), natural mating should be encouraged where possible. Reproductive performance is enhanced by the multiple matings that are a feature of normal feline reproduction, and the natural induction of ovulation stimulated by coitus.
As for natural breeding, the choice of tom and queen for AI should be based on a prebreeding examination. A complete history (including vaccination status, viral diseases, etc) should be obtained and a thorough general physical examination performed to assess for any non-reproductive abnormalities. The recommended minimum database includes a complete blood count and serum biochemistry, urinalysis, serology for feline leukaemia virus, feline immunodeficiency virus and feline infectious peritonitis virus, and blood typing to ensure compatible mat-ings. 4 As discussed in accompanying reviews on breeding and pregnancy management, 5 neonatology 6 and fading kitten syndrome, 7 a kitten with type A or AB blood born to a mother with type B blood is at risk of developing neonatal isoerythrolysis after maternal colostrum is ingested, with sequelae including pigmenturia, icterus, anaemia, tail tip necrosis and sudden death. 8
Before moving on to examination of the reproductive system, pertinent aspects of the breeding management, general husbandry and physical examination findings should be evaluated and recorded. 8 In the queen, examination of the reproductive tract should include inspection of the vulva and transabdominal palpation of the uterus to rule of the presence of abnormal uterine enlargement. Uterine condition and ovarian morphology can be evaluated using two-dimensional ultrasonography (see accompanying review on infertility in queens 9 ), although this technique does not provide information about organ function, such as vascular perfusion. 10
A thorough reproductive examination in the tom should consist of manual palpation of the testicles, assessing for position, size, texture and symmetry, followed by ultrasound examination to assess any irregularities within the testicular parenchyma. Unfortunately, there is scant information in the literature concerning imaging of the normal feline testes;11–13 although age-related histological changes in the testes have been reported, there is no description of diagnostic ultrasound. 14 The penis should be evaluated for any discolouration or discharge, and the presence of spines. Exteriorisation of the penis may be difficult without sedation, so this portion of the examination might best be accomplished just before sperm collection when the tom is sedated or anaesthetised. 3
Oestrus management and ovulation induction
AI can be performed in queens in natural or induced oestrus, with induction of ovulation on the second or third day of oestrus (assessed on the basis of oestrus behaviour and vaginal cytology); this corresponds to ovulation induced by coitus, which is normally within 24-48 h.15,16 Natural oestrus is preferable because treatment with some exogenous gonadotropins, a regimen typically used in AI, has been reported to produce an inappropriate maternal endocrine response. 17 Oestrous cyclicity can be induced in normal queens in anoestrus by artificially modifying the photoperiod with a supplemental lighting programme or by various therapeutic protocols described in the literature.17–26
Follicular development and ovulation are induced in queens by treatment with exogenous gonadotropins. Administration of equine chorionic gonadotropin (eCG) is widely used due to its follicle-stimulating hormone (FSH)-like action, which triggers ovarian follicular activity. Commonly used protocols consisting of 100-150 IU of eCG administered intramuscularly (IM), followed by 75-100 IU of human chorionic gonadotropin (hCG) IM 80-90 h later,17–24 are known to produce a high number of follicles and related corpora lutea. However, eCG treatment has some disadvantages, such as the necessity to repeat the injections in some protocols (which can cause immune reactions) and the high risk (particularly in primiparous queens) of inducing ovarian superstimulation or superovulation (Figure 1). Also, eCG has luteinising hormone (LH)-like activity, which can lead to the development of follicular cysts or premature luteinisation. 27

Ovaries of a queen after an equine chorionic gonadotropin/human chorionic gonadotropin (hCG) stimulation protocol. The queen was spayed about 60 h after hCG administration. The presence of several functional structures (follicles, corpora haemorrhagica) in both ovaries is evidence of superstimulation. Black and white arrows indicate a couple of the follicles and corpora haemorrhagica, respectively
Direct stimulation of pituitary activity with gonadotropin-releasing hormone (GnRH) has been investigated using different approach-es.28,29 Oestrus induction and pregnancy have been achieved with continuous administration or release of a GnRH analogue (lutrelin, deslorelin, leuprolide) via a subcutaneous osmotic mini-pump or implant.29,30 Among the GnRH agonists, deslorelin – classified as a superagonist – has a receptor affinity that is 100 times superior to that of endogenous GnRH.31,32 It is available commercially as a slow-release subcutaneous implant (4.7 and 9.4 mg, Suprelorin; Virbac) and its administration initially induces an acute stimulatory phase that lasts for several days, characterised by a substantial increase in LH and FSH concentrations. Prolonged exposure to these molecules subsequently leads to downregulation of GnRH receptors on the gonadotrope cells and, in turn, reduced synthesis of LH and FSH.33–35
A positive response to an oestrus induction protocol using 4.7 mg deslorelin implants has been described in 13 queens by the current authors (Zambelli et al 36 ). Oestrus was detected within an average of 5.0 ± 2.2 days of implant placement in all 13 queens, with a mean number of 4.8 ± 1.6 follicles per animal. Seven of 13 queens exhibited behavioural manifestations of oestrus; other studies, however, where oestrus was successfully induced, have reported oestrus behaviour in only 2/14 and 1/10 queens.37,38 The stimulation induced by deslorelin is considered comparable with a physiological oestrus, 27 with follicle numbers within the normal range of5± 0.5 per queen,17,28,39 and without the induction of cysts or other pathology (Figure 2). 36 The success rate of oestrus induction using deslorelin depends on the stage of the cycle: queens treated in anoestrus or interoestrus will show heat within a few days, while queens treated in dioestrus will not show heat. 40

Ultrasonographic assessment of the ovary (surrounded by arrows) of a queen after deslorelin stimulation. The number of follicles (arrowheads) present is within the normal range (5 ± 0.5/queen 17 )
Three of the 13 queens reported in the above-mentioned study by Zambelli and colleagues 36 were treated by AI following removal of the subcutaneous deslorelin implant. All became pregnant and gave birth to healthy litters.
In queens involved in AI programmes, ovulation is generally induced by administration of 100-250 IU hCG IM on days 2 and 3 of oestrus.41–43 A study by Chatdarong et al 44 supports this protocol. Based on evaluation of parameters such as cervical patency, oestrus behaviour, vaginal cornification and oestradi-ol-17 beta serum concentration, these authors recommended performing intravaginal AI on the second day of oestrus; for queens with only a short period of cervical patency in late oestrus, a second insemination 2 days later is advisable. Ovulation in queens implanted with deslorelin has also been observed after a single dose of hCG, 100 IU IM, once the peak of oestrus was identified. 36 Ovulation can be indirectly confirmed by a serum progesterone concentration of >2-3 ng/ml assayed 5-6 days after hCG administration. 36
Ovulation has additionally been achieved in queens by administering 1000 IU of porcine luteinising hormone (pLH) 85 h following eCG treatment to induce oestrus. 45 Vaginal stimulation (with a glass rod or sterile cotton swab) of queens in oestrus is another method that has been described for induction of ovulation. 46
The protocol for oestrus and ovulation induction currently used by the authors, based on the study by Zambelli et al, 36 is described in the box. For any AI programme, it is recommended that queens should preferably be in anoestrus or postoestrus (interoestrus) and showing pre-treatment basal levels of serum progesterone (^2 ng/ml). This permits the best possible stimulatory effects to be achieved by the planned AI.

Semen collection for artificial insemination
The priority with any semen collection procedure is to obtain good quality ejaculates with minimal stress for the animal. Sperm may be collected in clinical practice using a variety of different methods. The use of an artificial vagina (AV) and electroejaculation (EE), both described in studies in the 1970s, have traditionally been the techniques employed.1,47–55 More recently, urethral catheterisation after pharmacological induction (UrCaPI) has been reported as a simple method of semen collec-tion,56–65 and is now one of most widely used semen collection techniques, both in domestic cats and wild felids. 66 While researchers frequently obtain sperm for experimental studies by squeezing or slicing epididymal tissue after routine orchiectomy,67–69 these techniques are not used in clinical practice for AI procedures.
Artificial vagina
This technique is most frequently used in catteries and research colonies, where semen is collected frequently for AI, sperm preservation or other purposes from animals trained for the procedure. By contrast, for a single collection in an untrained cat in the clinical practice setting, this is usually a fruitless method. The principal advantages of using an AV are the low cost of the equipment, and no need for restraint of the cat; in addition, a single technician can perform the collection. Training to use an AV is possible in many cats, and can generally be completed after 2 weeks of gentle handling during exposure to a queen in oestrus. 52
In preparation for the collection, the male is permitted to approach to the female as he would naturally, to encourage maximum sexual arousal. At the point when he mounts the queen, the operator’s gloved hand slips the AV (Figure 3) over the penis and, after few coital thrusts, the ejaculate is collected. If the cat is well trained, it may be possible to collect the semen in the absence of a teaser queen. It is generally possible to repeat the procedure after an interval of about 10 mins. A rate of three collections per week does not impact sperm volume and concentration, but daily collection has been shown to cause a rapid drop in these parameters from day 4. 52
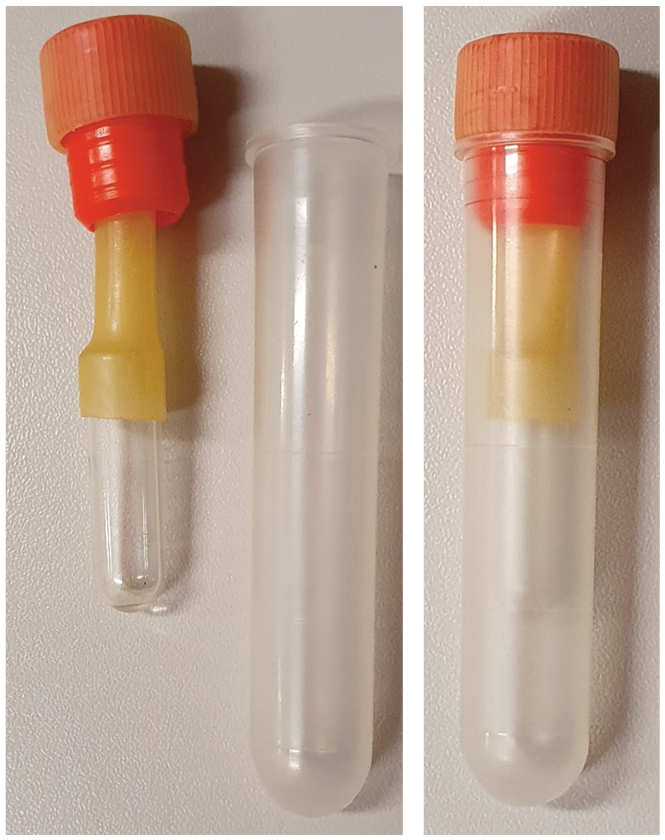
An artificial vaginal created using a 5 ml plastic tube (the external portion), Penrose drain and a small glass tube. The 5 ml tube is filled with water (40°C) to maintain the sperm temperature and reduce the thermal shock. 56 Reprinted from Johnson AK and Kutzier MA. Feline reproduction. Oxfordshire, UK: CAB international, 2022
Electroejaculation
This technique may potentially be used in any male that can be safely anaesthetised 70 and offers advantages over use of an AV in the context of a previously untrained or aggressive male or in the absence of a teaser queen. As such, EE was previously considered the method of choice for elective sperm collection in cats. Moreover, EE safety has been reported in a study showing no significant histological and endoscopic lesions in the rectum following the procedure. 71 The main disadvantages are the necessity to anaesthetise the tom, and the cost of the instrumentation.
Various anaesthetic protocols have been proposed for cats undergoing EE, with ketamine being the most commonly used agent, either administered alone (20 mg/kg IM) or in combination with medetomidine (5 mg/kg IM and 80 u.g/kg IM, respectively). The effects of both agents on the quality of electroejaculated sperm have been reported in a comparative study. 72 Medetomidine, which provides both deep sedation that allows the procedure to be performed and dose-dependent analgesia, permits collection of an ejaculate that is characterised by a higher number of sperm than after ketamine administration. 72 Moreover, while the passage of some sperm into the urinary bladder (retroejaculation) is normal in the cat, medetomidine does not increase the percentage of sperm flowing retrogradely into the bladder. 55 An anaesthetic protocol that includes 30-40 u.g/kg IM of dexmedetomidine combined with 3-5 mg/kg of ketamine IM has been reported for successful semen collection by EE, 3 but further data about the use of this drug for EE in cats (eg, success rate, quality of semen collected) have not been published.
In cats undergoing EE, the stimulus voltage and number of electrical stimuli appear to influence the number of sperm collected, but not the volume of the ejaculates.51,73
For sperm collection by EE, an electroejacu-lator connected to a rectal probe (1 cm diameter, 12 cm long) is required, equipped with two or three longitudinal stainless steel electrodes. After inducing anaesthesia, the rectum is emptied of faeces (which would decrease conductivity of the probe) and the glans penis is cleaned using saline-moistened gauze. The lubricated probe, with electrodes oriented ventrally, is gently introduced into the rectum to a depth of 7-8 cm and pushed ventrally to ensure good contact with the rectal mucosa and the pelvic plexus; this network of nerves is located dorsally to the membranous urethra, between the prostate and bulbourethral gland. The prepuce is retracted to expose the glans and a sterile Eppendorf tube is gently positioned onto the penis before starting the procedure (Figure 4).

Semen collection by electroejaculation. A lubricated probe has been inserted 7-8 cm into the rectum and a sterile Eppendorf vial has been gently positioned onto the penis
Different electrical protocols have been reported in the literature and the one proposed by Howard et al in 1990 52 is the most commonly used in practice (see box). It is recommended that Eppendorf tubes are changed after each set of stimuli, because the higher voltages can induce urine emission, leading to contamination of samples. The male responds to electrical stimulation with a rigid and symmetrical extension of both hindlimbs. Assessment of this extension is very important. An absent, weak or asymmetrical response suggests poor contact between the electrodes and rectal mucosa due to incorrect positioning of the rectal probe or the presence of faecal material.

Semen collection by urethral catheterisation after pharmacological induction
The sperm collection technique of UrCaPI, described in the past couple of decades, 56 overcomes some of the most important disadvantages of the the AV and EE techniques. UrCaPI does not require any specific equipment (AV or electroejaculator) or a trained cat (necessary for AV collection) and can be performed in any country without restrictions. Although semen collected by UrCaPI shows macroscopic and microscopic differences compared with samples collected using other techniques (see later), it can be cryopreserved 62 and used for in vitro fertilisation 62 or AI, 51 similar to sperm collected with an AV or EE. This technique represents an important tool in clinical practice, offering a cost-effective and simple means of collecting an ejaculate. A peculiarity of this technique is that it produces small-volume ejaculate samples with a very high concentration of spermatozoa. 45 This can be considered an advantage for artificial fertilisation and in vitro procedures, but it requires very careful sample handling to avoid damage to the spermatozoa. Depending on the volume and concentration of sperm collected, the sample may be diluted with a small amount of tris-glucose-citrate (TGC) or Tyrode’s albumin lactate pyruvate (TALP) medium, 62 ’ 74 which will also enable sample evaluation without utilising an excessive proportion of the ejaculate.
The reported dose of medetomidine for this technique is 130 u.g/kg IM. This stimulates contraction of the vas deferens, with consequent release of sperm cells into the urethra, without inducing ejaculation. 56 Despite many anaesthetists being reluctant to use this dose, it has been proven to be safe and well tolerated. 57 As always, a cardiological examination before anaesthesia is recommended to exclude individuals with cardiovascular disease. Results of sperm collection after administration of high doses of medetomidine (120 u.g/kg) or dexmedeto-midine (60 u.g/kg) and low doses of the two drugs (50 [ig/kg and 25 [ig/kg, respectively) have been compared. 61 It was observed that high doses of both drugs allow the collection of good quality sperm, while low doses were in general unsatisfactory in terms of both sperm collection and sedation level. In addition to these protocols, other doses of medetomidine (such as 80 or 100 u.g/kg) and dexmedetomidine (such as 5 or 25 u.g/kg in combination with another anaesthetic agent to reach an adequate level of sedation) are reported in the literature for successful semen collection by UrCaPI. 60 ’ 64 ’ 65
Once adequate sedation has been achieved, an open-ended tom cat urinary catheter is introduced gently and slowly into the urethra to a total depth of 9 cm, while the prepuce is pulled caudally to distend the urethra; note that by not exceeding this depth, the tip of the catheter is prevented from reaching the bladder. Sperm collect inside the catheter by capillary action (Figure 5) and are transferred to an Eppendorf tube using an insulin syringe.

Semen collection by urethral catheterisation after pharmacological induction. Semen enters the catheter (arrow) by capillary action. Here, the catheter has been pulled caudally to demonstrate the sample collected
At the end of the procedure, medetomidine or dexmedetomidine are antagonised with ati-pamezole. The intramuscular dose of atipamezole (in ug) is 2.5 times that of the previous dose of medetomidine, or 5 times that of the dose of dexmedetomidine hydrochloride.
Sperm evaluation and insemination dose
A basic semen analysis (spermiogram) should include evaluation of both macroscopic and microscopic parameters. Despite the fact that even a visually normal sample with good quality sperm does not confirm that an individual is fer-tile, 75 macroscopic evaluation, including appearance and volume, is fundamental to assess its quality parameters and suitability for use for AI. Consideration should be given to the collection method used, however, as this can affect the characteristics of normal semen (see box). Parameters that should be checked by microscopy include sperm motility, morphology and viability. In specific circumstances, it may also be necessary to evaluate pH, osmolarity, sperm bacteriology, membrane integrity and semen chemistry. In practice, however, particularly when feline semen is collected for AI using UrCaPI, the clinician often has insufficient sample volume for a complete examination. This represents an important limitation, as does the difficulty of maintaining constant environmental conditions (temperature, presence of oxygen, etc).
Conception rates reported in the literature vary between studies and depend on the dose of fresh or frozen/thawed semen, and whether it was deposited using intravaginal or intrauterine techniques.49,76–78 Recommended minimum sperm dose, based on semen type and also on the site of sperm deposition during AI, is presented in Table 1. Using fresh sperm, intravaginal insemination doses with 20 x 106, 40 x 106 and 80 x 106 motile spermatozoa in a volume of 50-100 u l have been reported to produce conception rates of 6.6%, 33.3% and 77.8%, respectively, in cats. 79 The higher insemination dose was comparable with the number of spermatozoa collected in two consecutive ejaculations, 80 and it seems reasonable to assume that cats copulate several times during oestrus probably as a means of maximising sperm dose. 79 A conception rate of 80% has been reported following surgical insemination of 8 x 106 fresh sperm in a volume of 30 u l into one uterine horn. 76 The requirement for 10 times the number of sperm to achieve high fertilisation rates with intravaginal (80 x 106) vs intrauterine (8 x 106) insemination in the cat is also reported for the dog.81,82
Minimum sperm dose in relation to semen type and site of insemination

Conception after artificial intrauterine insem ination (AIUI; see later) using 2.4-19.2 x 106 fresh sperm has also been reported. 77 In one study of transcervical uterine insemination, a dose of 200 u l of fresh or frozen/thawed semen containing 10 x 106 or 30 x 106 motile sperm, respectively, produced a conception rate of 100% in queens where transcervical catheterisation was possible. 83 In a separate study, sequential uterine endoscop-ic transcervical insemination was reported in three queens using 8.1-53.3 x 106 fresh motile spermatozoa in 18-32 u l of semen collected by UrCaPI; each queen was inseminated twice and the conception rate was 100%. 36


Semen collected (from left) by UrCaPI, and EE after the first, second and third sets of electrical stimuli
Artificial insemination techniques
Fertilisation in the cat is possible using both artificial intra-vaginal insemination (AIVI) and AIUI techniques. As noted earlier, and based on investigations performed under similar experimental conditions, approximately 10 times more fresh sperm are needed for AIVI than for AIUI to obtain an 80% conception rate in the cat.76,79 Additionally, Tsutsui et al 78 demonstrated that approximately five times more frozen sperm were needed compared with fresh sperm to achieve a 57.1% conception rate with AIUI. Early reports detailed surgical procedures for intrauterine insemination, but techniques for transcervical insemination have subsequently been described, avoiding surgical risks and postsurgical complications. Laparoscopic oviductal AI has also been reported using low sperm numbers (2 x 106 per insemination dose) and resulting in high fertilisation and pregnancy rates; 84 to date, however, this remains an experimental procedure.
Artificial intravaginal insemination
The first feline pregnancies after AIVI, using fresh or frozen/thawed semen, were reported in the 1970s by Sojka et al 1 and Platz et al. 49 The procedures described are simple and the equipment needed is generally readily available. Sojka and colleagues 1 used a 20 G, 9 cm-long needle with a bulb, connected to a 0.25 ml syringe. More recently, a 1.5 mm diameter, 9 cm-long nylon probe connected to a 1.0 ml syringe was used for insemination by Tanaka et al. 79 During vaginal insemination, sperm is deposited in the anterior vagina or posterior cervix. It is usually recommended that the queen is under general anaesthesia or heavy sedation for the procedure.1,49 She is then generally maintained in dorsal recumbency with the hindquarters elevated for 15-20 mins post-insemination to minimise sperm backflow.49,79
Artificial intrauterine insemination
In 1992, conception was reported after surgical uterine deposition of sperm using laparoscopy. 77 This AIUI procedure was performed for research purposes and has not been applied in clinical practice. AIUI after laparoto-my has also been described, involving infusion of semen into the uterine horn with the greatest number of ovarian follicles or ovulations.78,81
Various techniques for uterine insemination by cervical catheterisation have been proposed, and different catheters have been developed. Catheterisation has been performed blindly lumen using a 2 mm glass speculum, 85 and also with the aid of a modified polypropylene urinary catheter (2.7-2.8 mm) used as speculum,21,86 to facilitate the insertion of a 3 Fr tom cat catheter through the cervix; these methods are unsuitable for animals with a narrower cranial vagina lumen (<2 mm).87,88 In 2001, another procedure for uterine insemination by transcervical catheterisation was proposed by one of the present authors (Zambelli). 87 This method allowed the uterine lumen to be reached using a 3 Fr catheter, modified with the addition of a rounded tip needle (0.65 mm diameter) at the cut end, inserted into the vagina and passed through the cervix guided by transrectal palpation.87,88 Success rates for cervical catheterisation using this method depend on training and experience of the practitioner. 88
More recently, a study has described endo-scopic transcervical catheterisation for AI in cats. 36 For this procedure, the queens were positioned in sternal recumbency with the pelvis slightly elevated using a cushion (Figure 7). A human semi-rigid sialendoscope (Karl Storz, Germany; 120 mm long, 1.1 mm diameter, 0° direction of view, one operative channel) was inserted through the vestibule and moved forward in the vagina until the dorsal fold and cervix were visualised. Transcervical catheterisation was performed using a modified version of the catheter designed earlier by Zambelli et al: 88 a 3 Fr, 11 cm-long tom cat urinary catheter with a 100 mm stainless steel, rounded tip needle (0.65 mm diameter) inserted at its cut end (Figure 8).

Queen positioned for endoscopic transcervical catheterisation. The endoscope is introduced at the level of the dorsal vulvar commissure, and then, under direct endoscopic observation, the catheter is passed through the cervix and reaches the uterine
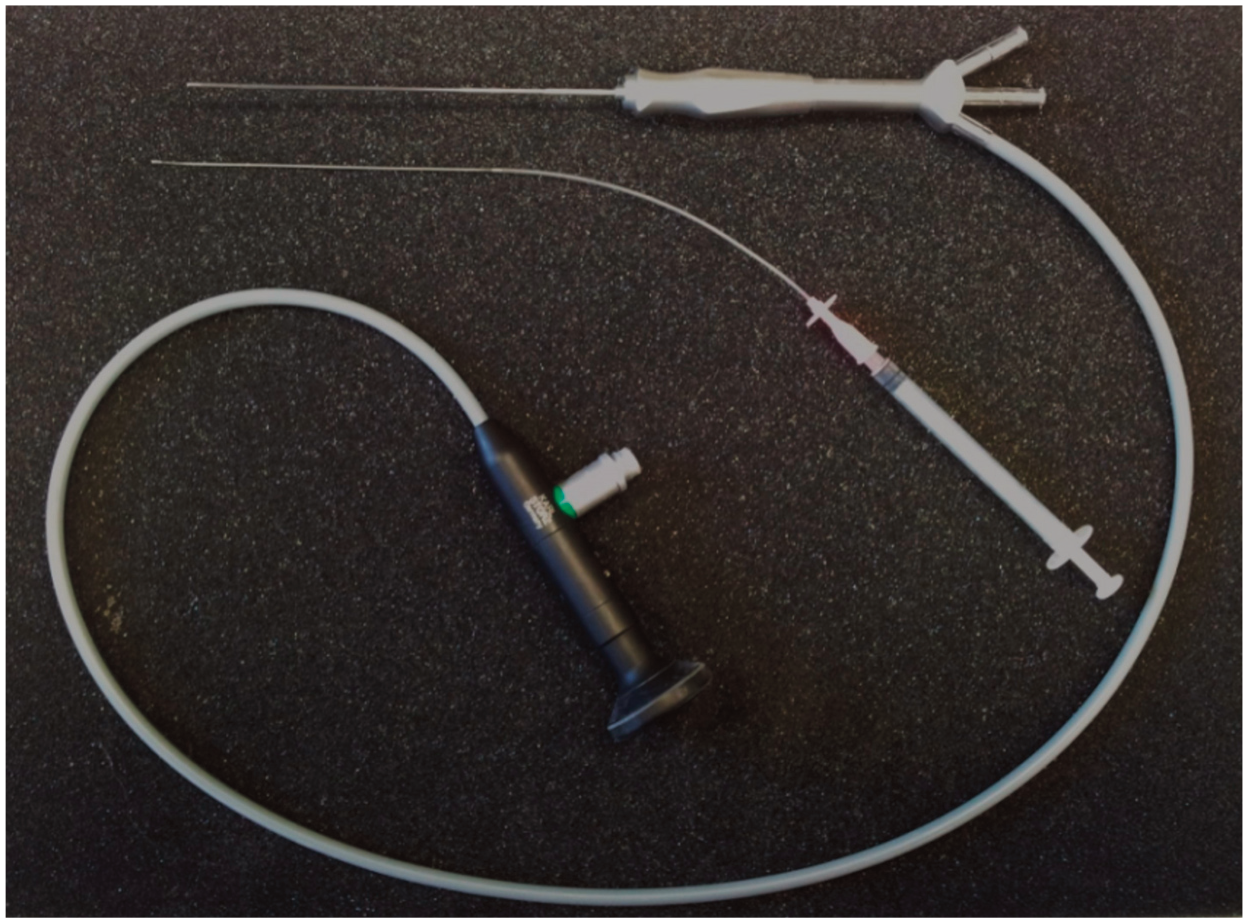
Sialendoscope and specially designed catheter used for endoscopic transcervical insemination
The stainless steel needle connected to the catheter was inserted with the endoscope through the vaginal lumen and under endoscop-ic observation (Figure 9) the catheter was moved through the cervical ostium; the endoscope was then removed. This newer technique of endo-scopic transcervical catheterisation was successful in 12/14 animals (85.71%). 36 In one queen, the cranial vagina was too narrow for insertion of the endoscope together with the catheter, while in another queen, cannulation did not succeed because of excessive inclination of the cervical axis and the narrowness of the genital tract.

Endoscopic view of the (a) cranial vagina and (b) vestibule of a queen. The labels correspond to the anatomical landmarks shown in the gross specimen in (c). 1 = cervix; 2 = vaginal dorsal medial fold; 3 = cranial vagina opening; 4 = urinary meatus; 5 = catheter used for endoscopic transcervical insemination
Key Points
✜ There is increasing demand from domestic cat breeders for AI techniques that are already commonly used in dogs.
✜ Before moving on to examination of the reproductive system, pertinent aspects of the breeding management, husbandry and physical examination findings should be evaluated and recorded.
✜ AI can be performed in queens in natural or induced oestrus, with induction of ovulation on the second or third day of oestrus. Stimulation induced by deslorelin is comparable with a physiological oestrus.
✜ UrCaPI is a simple method of semen collection and one of the most widely used techniques, both in domestic cats and wild felids.
✜ A basic semen analysis should include evaluation of macroscopic parameters, such as appearance and volume, and microscopic parameters, such as motility, morphology and viability.
✜ The recommended insemination dose (volume and total number of spermatozoa) varies depending on the semen type used (fresh or frozen/thawed) and the site of sperm deposition during AI.
✜ Endoscopic transcervical catheterisation is an effective technique as it allows cervical cannulation in most queens, and should be easy for an endoscopist to perform. It is a suitable method for AIUI, reducing the risks related to surgical uterine insemination.
✜ AIVI and endoscopic transcervical catheterisation are, to date, the techniques typically used when good quality and poor quality semen, JHH respectively, has been collected.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work did not involve the use of animals and therefore ethical approval was not specifically required for publication in JFMS.
Informed consent
This work did not involve the use of animals (including cadavers) and therefore informed consent was not required. No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
