Abstract
Objectives
The aim of our study was to describe a biomechanical testing protocol to reproduce ex vivo craniodorsal hip luxation specific to the feline model, and evaluate the biomechanical properties of an intact hip joint compared with the fixation strength of two different techniques of extra-articular hip stabilisation.
Methods
Eighteen hip joints (femur and hemipelvis) were harvested from nine mature feline cadavers. CT was performed for each hip joint so that a biomechanical base specific to each joint morphotype could be created using computer-aided design. The biomechanical bases were then produced using a three-dimensional printer to secure the hip joints during testing. A total of 34 biomechanical compression tests were performed. Eighteen compression tests were performed in the control group, of which two fractured. The remaining 16 hip joints were then randomly assigned either to group A (hip joints stabilised with an extra-articular ultra-high molecular weight polyethylene (UHMWPE) implant secured by an interference screw [n = 8]) or to group B (hip joints stabilised with a UHMWPE iliofemoral suture [n = 8]).
Results
Mean ± SD yield, failure load and linear stiffness in the control group were 616 ± 168 N, 666 ± 158 N and 231 ± 50 N/mm, respectively. The relative fixation strength (% of intact joint) before hip luxation in groups A and B was 43.8% and 34.7%, respectively. No statistical difference was found between groups A and B for yield and failure load. However, the reoccurrence of craniodorsal hip luxation was higher in group B than in group A, in 5/8 and 0/8 tests, respectively. Moreover, in group A, the extra-articular UHMWPE implant induced caudodorsal hip luxation, reported as failure mode in 7/8 cases.
Conclusions and relevance
This modified biomechanical protocol for testing craniodorsal hip luxation in a feline model was validated as repeatable and with acceptable variance. The extra-articular UHMWPE implant stabilisation technique proved to be more efficient in avoiding reoccurrence of craniodorsal hip luxation than UHMWPE iliofemoral suture.
Introduction
Coxofemoral luxation is frequent in cats and is the most common type of joint luxation observed in this species; 1 it is most often caused by road traffic trauma.1–3 The choice of surgical treatment depends on the type of hip luxation, which usually occurs in a craniodorsal direction (78.1% of dogs; 72.7–83.7% of cats),2,3 and more rarely in a caudodorsal (0–4% of cats)1–5 or caudoventral (0–4% of cats) direction.1–5 Hip luxation may be managed using various stabilisation techniques, including capsulorrhaphy, 6 prosthetic capsule repair, 7 rectus femoris suture, 8 deep gluteal muscle fixation, 9 the De Vita technique, 10 trochanteric transposition, 11 extra-articular iliofemoral suture 12 secured with anchors, 13 intra-articular replacement of the femoral head ligament with a synthetic ligament secured by toggles3,5,14–19 or transarticular pinning. 20
Intra-articular surgical techniques secured by toggles have provided variable clinical results across studies, with a recurrence rate of hip joint luxation of up to 24.4%,2,3,5,15–19,21 while biomechanical studies have reported promising results in dogs.14,22 However, there is still little information about capsulorrhaphy or extra-articular techniques such as iliofemoral suture and prosthetic capsule repair in long-term clinical and biomechanical studies,7,8,12,23 apart from the work by Singer et al on suture anchor in the canine cadaver pelvis. 24 Moreover, no biomechanical evaluation has investigated hip joint stabilisation techniques applied to the anatomical and biomechanical specificities of cats. The canine studies were performed around 20 years ago with three different biomechanical testing protocols and set-ups on cadaveric models.13,14,22
The aims of this study were: (1) to validate a biomechanical testing protocol to reproduce ex vivo craniodorsal hip luxation specific to a feline cadaver model (validation criteria: test repeatability [low dispersion of linear stiffness, yield load and failure load] and failure mode [desired type of hip luxation obtained in 100% of cases, ie, craniodorsal hip luxation]); and (2) to establish the biomechanical strength of intact hip joints (control group) to induce craniodorsal hip luxation and compare it with two types of extra-articular stabilisation using ultra-high molecular weight polyethylene (UHMWPE) materials: either a UHMWPE implant secured by an interference screw (group A) or an UHMWPE iliofemoral suture (group B).
Materials and methods
Feline cadaver collection period
Nine mature feline cadavers weighing 3.1 ± 0.9 kg were collected after the animals had been euthanased for reasons unrelated to the present study. No sign of musculoskeletal disease, trauma or a defect in conformation or an area that might suggest a previous scar from trauma was reported on palpation and mobilisation of the hindlimbs and hip joints. These findings were immediately confirmed by individual CT (Canon Aquilion Lightning 32, 120 KVp, 120 mAs, 0.430 × 0.430 × 0.5 mm3 voxel size) to exclude hips affected by osteoarthritis. The same acquisition method was used for each cadaver included in this study, to limit potential biases. Nine data sets of the pelvic region were then recorded with images in DICOM format. Once the cadaveric specimens were checked, their pelvic limbs, including the pelvis and caudal spine, were mass harvested, wrapped in saline-soaked laparotomy towels and stored at −18°C until the day of the biomechanical study.
Segmentation of CT medical imaging
Segmentation of the nine pelvises from the nine CT scan acquisitions was performed with three-dimensional Slicer open source software. It consisted of automated global thresholding and image stacks of 250–1900 Hounsfield units,25,26 followed by manual refinement to improve the fidelity of the three-dimensional reconstruction of each pelvis (Figure 1). Finally, a 0.5 smoothing factor was applied to erase surface defects in each segmentation. Once segmented, each pelvis was split in half longitudinally to obtain two hemipelvises.

Choice of thresholding according to the Hounsfield scale to highlight bone part, applied to the first feline cadaver using three-dimensional Slicer open source software. (a) Transverse CT view of the pelvis; (b) three-dimensional reconstruction of the pelvis; (c) sagittal CT view of the pelvis; (d) dorsal CT of the pelvis
Computer-aided design of specific biomechanical bases
Eighteen specific biomechanical bases, one for each hip joint, were created using computer-aided design (CAD). Starting from a rectangular parallelepiped volume, an adaptation of the grooved rail fixed to the lower support of the testing machine was initially performed. Our objective was to design a biomechanical set-up that could induce craniodorsal hip luxation in a feline cadaver model according to its clinical description.27,28 We chose a vertical alignment of the femur following the axis of the testing machine and corresponding to a hip joint flexor alpha angle of 125° (Figure 2a), associated with an adduction angle of 30° once the samples were placed in the mechanical testing machine with mild hip anteversion. Hemipelvises were correctly placed onto each biomechanical base by Boolean subtraction (Figure 2b). Once designed, polylactic acid (PLA) biomechanical bases were printed in three dimensions (STREAM 30 Dual MK2; Volumic). The three-dimensional printer was configured to maximise the strength and precision (200 µm) of each biomechanical base.

Diagrams of the fifth specific biomechanical base. (a) Profile view showing 125° extension alpha angle of the left hip joint following the vertical mechanical axis of the traction machine; and (b) global view without a cadaveric hip joint
Sample preparation protocol
The anatomical parts of the nine feline cadavers were left to thaw in a fridge at 5°C for 48 h prior to their dissection (Figure 3). Eighteen hip joints (femur and hemipelvis) were harvested from the feline anatomical parts (Figure 3). Soft tissues were removed, except the articular capsule of the hip and the femoral head ligament. Femoral parts were transected at the level of the distal metaphysis. A 1 mm-wide transversal hole was drilled 20 mm distal to the third trochanter to allow the placement of a needle through the bone tunnel (Figure 4). This was carried out to normalise the inclusion level of femoral parts into a metallic mould (15 × 100 mm) with resin (Polyol and Isocyanate; Axson Technologies), already assembled with a universal joint (20 mm) in its distal part to allow both axial rotation and pivoting of the femur in any direction in response to a compressive load. 14

Three-step summary of the scientific methodology: (I) collection; (II) dissection; and (III) biomechanical testing

Normalisation of femoral inclusions using needles placed in a 1 mm bone drill at 20 mm distal to the third trochanter
Biomechanical testing
All tests (n = 34) were performed with a uniaxial testing machine (AGS-X Serie; Shimadzu) at a room temperature of 23°C. The 18 intact control hip joint samples were fixed on their specific biomechanical base by four steel plates with washers through the obturator hole (Figure 5). They were then tested with the same mechanical protocol to obtain biomechanical values of physiological strength required to induce craniodorsal hip luxation. A 30° ± 2° adduction of femurs was carried out with two mechanical processes: (1) an inclination of the mechanical rail of the testing machine corresponding to the gamma angle (15° ± 1°; Figure 5); and (2) a displacement of the biomechanical base on its rail to obtain another 15° ± 1° angle (beta angle; Figure 5). This 30° inclination was switched between tests performed on the right and left hip joint. A 10 mm/min preload was applied to reach 10 N before starting the compression test to failure at 100 mm/min in order to reproduce craniodorsal hip luxation. 14 The end of each compression test was set at 80% of the maximum load recorded by the testing machine software (TrapeziumX; Shimadzu). The 16 luxated joints were then randomly divided into two equally sized stabilisation technique groups (eight luxated hips per group) by a computerised number generator (Research Randomizer; https://www.randomizer.org): group A (stabilisation with an UHMWPE implant fixed with an interference screw) and group B (stabilisation with a UHMWPE iliofemoral suture) (Figure 3).

Biomechanical set-up with decomposition of 30° ± 2° adduction of the femur angle: gamma angle (15° ± 1°) and beta angle (15° ± 1°) used to measure compressive load applied to intact (left cadaveric hip joint presented here) and stabilised feline cadaver hip joints
Description and implantation of the extra-articular UHMWPE implant
In group A, cadaveric hip joints were stabilised with a UHMWPE implant (Novalig 2000 Platine; Novetech Surgery). This synthetic implant is manufactured by weaving and braiding UHMWPE medical-grade filaments that are already in use for orthopaedic implants in several medical applications, such as anterior cruciate implants. 29 Once braided, the synthetic implant is sterilised with ethylene oxide. It comprises three parts: a cortical button (12 × 4 mm; Figure 6I[1]) preassembled on a flat implanted section (22 cm × 3.4 mm; Figure 6I[2]). The UHMWPE implant ends in an 18 cm-long puller wire, which allows for its insertion into the bone tunnels. The implanted section is secured by a titanium interference screw (3 × 11 mm; Figure 6I[3]).
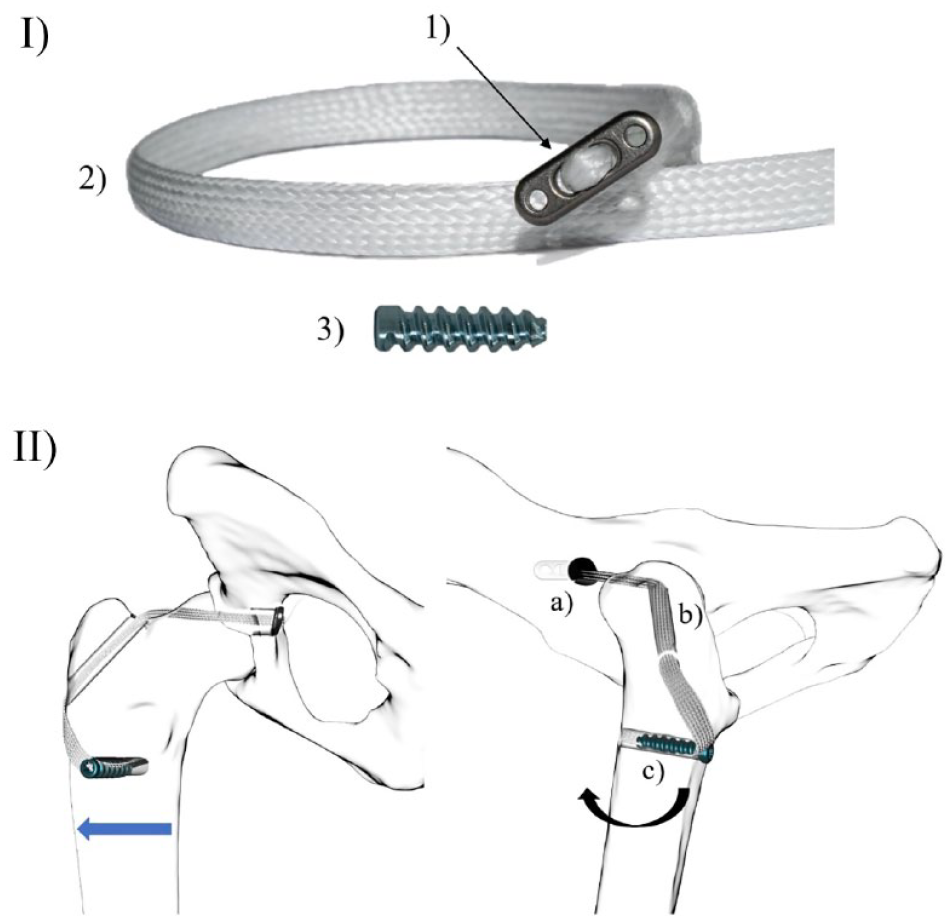
(I) Ultra-high molecular weight polyethylene (UHMWP) implant (Novalig 2000 platine) made of (1) a cortical button preassembled to (2) a flat implanted section; (3) 3 × 11 mm titanium interference screw used as fixation system. (II) Diagram of an extra-articular hip joint stabilisation using a UHMWP implant for craniodorsal hip luxation. (a) A 4 mm-wide ilium tunnel was drilled at 10 o’clock dorsally to the origin of the articular coxal muscle and the rectus femoris muscle, close to the joint; (b) 2.5 mm trochanteric bone tunnel; and (c) 2.5 mm bone tunnel drilled through the femoral metaphysis. Blue arrow points to mild adduction of the coxofemoral joint once stabilised. Black arrow indicates internal rotation of the coxofemoral joint once stabilised
Regarding the implantation of this UHMWPE implant, a 4 mm-wide ilial tunnel was drilled at 10 o’clock dorsal to the origin of the articular coxal muscle and the rectus femoris muscle, as close as possible to the joint, in order to drill through the iliac part of the acetabulum in a cranio-dorsal direction (Figure 6II[a]). The cortical button of the UHMWPE implant was inserted lateromedially and secured to the inner cortical part of the ilium (Figure 6II[a]). A second 2.5 mm bone tunnel was drilled from the level of the greater trochanter in a lateral (centre of the origin of the vastus lateralis) to medial (dorsal and as close as possible to the neck) direction (Figure 6II[b]). A third 2.5 mm bone tunnel was drilled in a laterocaudal-to-cranial direction of the femoroproximal metaphysis (Figure 6II[c]). The entry point was defined as the caudal part of the vastus lateralis origin, next to the gluteal tuberosity (on the linea aspera), and the exit point was centred on the sagittal plane of the cranial part of the diaphysis (Figure 6II[c]). The third tunnel was tapped (3 mm diameter). The UHMWPE implant was passed through the second and third tunnels, ensuring that the flat part of the implant remained in contact with the bone and hip joint surface. It was tightened with a zero-version angle to the hip, which induced slight internal rotation and abduction of the femur. The suture was then secured with a 3 × 11 mm interference screw (Novetech Surgery; (Figure 6I[3]).
To identify the precise nature of the failure mode, a single 4-0 polypropylene suture (Prolene; Ethicon) was used as a marker for each hip in group A. It was systematically placed at the exit point of the femoral tunnel at the origin of the vastus lateralis to check the precise nature of the failure mode by reviewing the video footage recorded during each compression test frame by frame.
Description and implantation of the UHMWPE iliofemoral suture
In group B, cadaveric hip joints were stabilised with a UHMWPE iliofemoral suture (Fibertech; Novetech Surgery). 12 Suture material of size 5 metric was used for this purpose (Figure 7I). The UHMWPE fibre used to make the implant (Figure 6I) and the suture (Figure 7I) was the same. UHMWPE monofilaments were woven to obtain a suture thread, which was then mounted on two curved needles and secured by knots (Figure 7I).

(I) Five metric ultra-high molecular weight polyethylene (UHMWPE) suture preassembled to two curved needles (Fibertech). (II) Diagram of extra-articular hip joint stabilisation using UHMWPE iliofemoral suture for craniodorsal hip luxation, following the technique described by Meij et al.12 (a) A 2.7 mm ilial tunnel drilled at the origin of articular and rectus femoris muscles; (b) a 2.7 mm femoral bone tunnel. Blue arrow shows mild adduction of coxofemoral joint once stabilised. Black arrow shows internal rotation of coxofemoral joint once stabilised
First, a 2.7 mm-wide iliac tunnel was drilled through the cranial part of the acetabulum between the rectus femoris and the origin of the coxal muscle (Figure 7II[a]). Then, a second 2.7 mm-wide femoral tunnel was drilled under the great trochanter at the origin of the vastus lateralis. Drilling was performed in a caudocranial direction and perpendicular to the anatomical axis of the femoral neck (Figure 7II[b]). The UHMWPE thread (Fibertech 5 Metric; Novetech Surgery) was passed through both tunnels in a figure-of-eight mode, using the technique described by Meij et al. 12 After tightening with a zero-version angle to the hip (slight internal rotation and abduction of the femur), the UHMWPE suture was tied by five surgeon’s knots (Figure 7II).
An indelible felt-tip mark was made as close as possible to the first knot to check the precise nature of failure mode by reviewing the video footage recorded during each compression test frame by frame.
Data processing
During the biomechanical compression tests (n = 34), data acquisition was performed using TrapeziumX software (Shimadzu). Linear stiffness, yield and failure load were recorded in each test. Linear stiffness was assessed by calculating the slope of the load displacement curve in its linear interval in each compression test. Yield load was defined as the load at which the first deviation from linearity occurred on the load displacement curve. Failure load was defined as the maximum load measured during each test, corresponding to hip luxation. Failure mode was defined as luxation of the samples. Parameters were systematically verified after each test by a European veterinary surgeon specialist (EV) and confirmed by video footage (iPhone XS; Apple) synchronised with the testing machine software (TrapeziumX; Shimadzu). Excel (Microsoft) and Matlab (R2017b; MathWorks) were used for data processing. The validation of the biomechanical testing protocol was evaluated in terms of test repeatability (low dispersion of linear stiffness, yield load and failure load) and failure mode (desired type of hip luxation obtained in 100% of cases; ie, craniodorsal hip luxation).
Statistical analysis
The normal distribution of the data could not be confirmed statistically by posteriori verification with the Lilliefors test (Matlab R2017b; MathWorks). Differences in linear stiffness, yield and failure load between groups were compared using the non-parametric Mann–Whitney U-test. A P value of <0.05 was considered to be statistically significant. A Cohen’s effect size and an a posteriori statistical power analysis were performed with GPower 3.1.9.7, to verify the size of our groups and the statistical values obtained.
Results
All results are summarised in Table 1. The altered biomechanical testing protocol described in this study to induce craniodorsal hip luxation in a feline cadaver model reports 16/18 cases of the desired failure mode (ie, craniodorsal hip luxation). In the other two cases, the pelvis was fractured during the biomechanical compression test, making the data from these trials unusable and resulting in their withdrawal from the study (Figure 3). Two other pelvic fractures were reported in groups A and B, also excluding the use of these two data sets.
Results of biomechanical testing of intact hip joints and extra-articular stabilisation surgical techniques
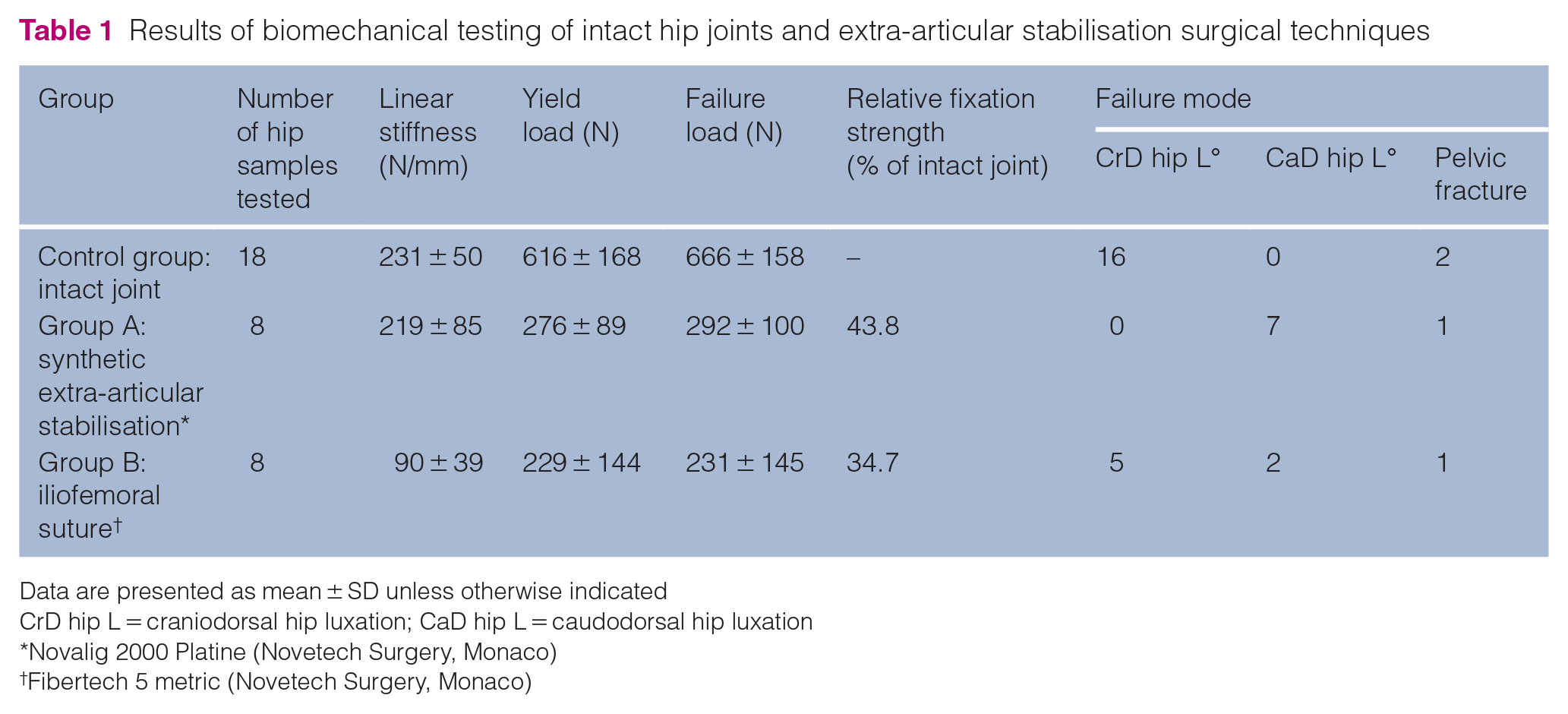
Data are presented as mean ± SD unless otherwise indicated
CrD hip L = craniodorsal hip luxation; CaD hip L = caudodorsal hip luxation
Novalig 2000 Platine (Novetech Surgery, Monaco)
Fibertech 5 metric (Novetech Surgery, Monaco)
Statistical differences were found between the control group and extra-articular stabilisation techniques (groups A and B) for yield and failure load (Table 2). Regarding linear stiffness, there were statistical differences between the control group and group B, and between groups A and B. The aforementioned statistical results were supported by a strong Cohen’s effect size and a satisfactory a posteriori statistical power analysis (Table 2). In addition, no other statistical differences were found between groups A and B for linear stiffness, yield and failure load, owing to low values for Cohen’s effect size and a posteriori statistical power analysis (Table 2).
Results of Cohen’s effect size and a posteriori statistical power analysis performed for each Mann–Whitney test

P <0.05 was considered significant
Fixation strength (% of intact joint) based on failure load in the control group was 43.8% and 34.7% in groups A and B, respectively.
We observed a difference in failure mode between group A (caudodorsal hip luxation in 7/8 cases) and group B (craniodorsal hip luxation in 5/8 cases). In group A, the fixation remained intact with no rupture or slippage of the UHMWPE implant through the tunnel at the bone/interference screw/implant interface (Figure 6I). In group B, there was no breakage of the thread or knots used for iliofemoral suture. However, we observed minor slippage of the knots, thus compromising stabilisation.
There was a greater reoccurrence of craniodorsal hip luxation in group B than in group A (in 5/8 and 0/8 tests, respectively). These failure load results correspond to a reoccurrence of hip joint luxation of 292 ± 100 N (group A) and 231 ± 145 N (group B), thus representing 9.6 and 7.5 times, respectively, the weight of the ex vivo cats tested during this biomechanical study.
Discussion
This study has described and validated a technique for performing biomechanical testing of feline cadaveric coxofemoral joints. We also used this technique to compare the functional differences between two separate extra-articular stabilisation techniques for coxofemoral luxation. Our key findings were that there were 16/18 cases of desired failure mode (ie, craniodorsal hip luxation); statistical differences were found between the control group and the extra-articular stabilisation techniques (groups A and B) for yield and failure load (Table 2); and a difference in failure mode between group A (caudodorsal hip luxation in 7/8 cases) and group B (craniodorsal hip luxation in 5/8 cases) associated with greater reoccurrence of craniodorsal hip luxation in group B than in group A (in 5/8 and 0/8 tests, respectively).
Our research was based on three biomechanical publications on canine cadaver models.13,14,22 Two of the three publications were selected because they described a biomechanical method allowing complete hip joint testing.13,14 To our knowledge, there has not been any biomechanical testing of extra-articular fixation of hip luxation in dogs or cats until now. These stabilisation techniques have the advantage of preserving the joint in case of infection, which is classically reported as occurring more frequently with braided materials.16,30–32 Although Smith et al demonstrated good tolerance of intra-articular UHMWPE implants in the knees of dogs, 33 the issue of the tolerance and wear of such material inside the joint remains unresolved. 34 Our research team attempted to set up these two biomechanical testing methods, but major information was lacking and our preliminary results were unsatisfactory. Indeed, we had difficulty inducing hip luxation (without bone conflict between the upper part of the acetabulum and the femoral head) and, when we succeeded, luxation was not homogeneous as caudodorsal, pure dorsal or craniodorsal hip luxation occurred. We therefore decided to modify the biomechanical study based on these two studies.13,14 The major differences were: (1) reversing the position of the hip joint along the z-axis; (2) increasing the hip extension angle from 105° to 125° and inducing a 30° adduction of the femur in order to mimic the pathophysiological conditions of craniodorsal hip luxation (ie, forced abduction with external femoral rotation during weightbearing, occurring during a telescoping movement, or violent or abnormal weightbearing while being out of balance);13,14,28 and (3) designing specific biomechanical bases by CAD thanks to three-dimensional reconstruction carried out for each feline cadaver, in order to reduce the biases linked to the inclusion of the hemipelvises in a resin matrix. 13 The crosshead speed chosen to produce hip luxation was the same as in previous studies (ie, 100 mm/min).13,14,22 The results obtained in the control group demonstrate the efficiency of the biomechanical protocol both in terms of test repeatability (low dispersion of linear stiffness, yield load and failure load) and failure mode (desired type of hip luxation obtained in 100% of cases; ie, craniodorsal hip luxation [Table 1]).
Flynn et al reported mean ± SD failure loads between 915 ± 66 N and 1008 ± 83 N on craniodorsal hip luxation canine cadaver models (Beagles weighing between 8.3 kg and 13.8 kg). 14 We found failure loads of 666 ± 158 N for our feline ex vivo models (mixed-breed cats weighing 3.1 ± 0.9 kg), which is 30% lower. Despite the differences in biomechanical testing protocols and the failure load results of our tests simulating craniodorsal luxation on intact cat hip joints compared with those obtained on a cadaveric canine model in similar conditions, 14 the failure load:body weight ratio seems to indicate a higher resistance to craniodorsal hip joint luxation in cats than in dogs.
Regarding relative fixation strength (% of intact joint) based on failure load in the control group, the results from groups A (43.8 %) and B (34.7 %) were in the same range of surgical intra-articular stabilisation technique strength as reported in previous publications on canine cadaver models.13,14,22 Flynn et al reported results ranging from 33.6% for intra-articular hip stabilisation using a toggle pin and a 50 lb nylon prosthesis to 46.7% using a toggle pin and a braided polyester prosthesis. 14 Our findings show that these two extra-articular stabilisation techniques tested in feline cadavers seem to allow the maintenance of coxofemoral co-optation based on a biomechanical model without active stabilisers, thus reproducing the in situ surgical reality in the immediate postoperative period. Indeed, our failure load results correspond to a reoccurrence of hip joint luxation (292 ± 100 N in group A and 231 ± 145 N in group B). The force applied to reach failure mode in groups A and B was 9.6 and 7.5 times, respectively, the force experienced through weightbearing in the limbs of the cats tested in this experiment.
The video footage showed the failure mode of each compression test in detail. In group A, no slippage or cortical button damage of the UHMWPE implant was detected with frame-by-frame analysis. This shows the benefit of an UHMWPE implant fixed by an interference screw into a tunnel perpendicular to the axis of the mechanical load 35 vs a tunnel following its axis.36–38 The second part of the fixation was a cortical button, which was already tested biomechanically with the same type of implant in a synthetic CCL reconstruction technique. 39 It resisted a 800 N load before damage and progressive slippage was observed.
In group B, minor progressive slippage of the UHMWPE suture knots was noted during each compression test, without causing any breakage of the iliofemoral suture, but leading to the reoccurrence of hip luxation. We locked the UHMWPE with five surgical knots according to the published recommendations.40,41 However, even if UHMWPE sutures are stronger than braided polyester sutures, they have a greater tendency to slip, 42 which could explain the difference in failure mode.
Interestingly, in canine biomechanical model studies, failure of intra-articular toggles most commonly occurs with breakage of the suture.13,14 In feline clinical studies, the same failure mode was described for toggles.3,5,16 Regarding extra-articular stabilisation, a clinical series published by Meij et al, 12 including cats, reported breakage of the iliofemoral suture as the most common cause of fixation failure. Cyclic loading in clinical conditions and the use of UHMWPE fibre in our study could explain this difference in failure mode.
Regarding linear stiffness, no statistical difference was found between the control group and group A, unlike between the control group and group B and between groups A and B. This shows that stabilisation of the hip joint using a UHMWPE implant tends to approximate the physiological ligament stiffness and capsular stabilisation of intact hip joints in feline cadavers.
There was higher reoccurrence of craniodorsal hip luxation in group B than in the control group with the failure mode (5/8 and 0/8 tests, respectively), and most cases in group A failed due to caudodorsal luxation (7/8 tests). The stiffness of the UHMWPE implant prevents the reoccurrence of craniodorsal hip luxation thanks to its shape, size and dorsal position above the femoral neck. Once the UHMWPE implant is in place, caudodorsal luxation of the hip is the most reported failure mode (ie, 7/8 cases and 1/8 pelvic fractures). Indeed, the use of UHMWPE and interference screws prevented craniodorsal luxation in all cases. As pure caudodorsal re-luxation is rare in feline clinical cases (from 0% to 4% in the literature),1–5 this biomechanical failure mode needs to be clinically investigated.
These biomechanical findings are interesting, as it appears that the extra-articular UHMWPE implant better stabilises the craniodorsal margin of the joint. In vivo studies are warranted to establish its clinical impact.
Several limitations of this study should be highlighted. First, we were able to verify statistical differences between controls and groups A and B based on values of Cohen’s effect size and a posteriori statistical power analysis (Table 2). No statistical difference was found between groups A and B in terms of linear stiffness, yield and failure load (Table 2). However, it is possible that the number of trials per group (n = 7) was too small to identify any difference. If we take the example of failure load results in group A and group B, an a priori statistical power analysis revealed that 42 biomechanical trials would have been necessary to obtain significant comparison results.
Second, reusing the control specimens (cadaveric feline luxated hips) to perform a second biomechanical test after stabilising the luxated hips may also create a bias. The first biomechanical test may have locally damaged the hip joint surface, the femur and/or the acetabulum, despite our visual macroscopic control of each anatomical part. However, control group samples have already been reused in other biomechanical studies without any major impact on the results.14,22
Third, the size and shape of the UHMWPE implant and the suture used in this study were not the same. Although we did not observe any breakage of either type of UHMWPE material, the capping of the joint, the implantation and the fixation systems differed in group A and group B, which could have caused different failure modes. Although these two techniques are considered to be extra-articular, it may be preferable to distinguish them more clearly.
Fourth, the load applied through the cadaveric hip joints in this set-up does not respect the mechanical axis of a conventional compression test as a vector force is applied to the femoral bone. In this biomechanical condition, the measured force may be overestimated by the testing machine, as the load truly applied to the hip itself is likely to be lower than the force measured.
Finally, the stiffness of our biomechanical bases made by three-dimensional PLA printing is also a limitation. Bases made of titanium would have been stiffer, but this option had to be abandoned owing to cost constraints.
Conclusions
The results obtained with this biomechanical testing protocol showed good repeatability of the desired failure mode for intact joints (ie, craniodorsal hip luxation on an ex vivo feline model associated with low result dispersion), thus validating the protocol. The load necessary to induce craniodorsal hip luxation in an ex vivo feline model devoid of periarticular muscles has been established (ie, 666 ± 158 N), according to our non-axial biomechanical set-up. In this biomechanical condition, the measured force may be overestimated by the testing machine, as the load truly applied to the hip itself is likely to be lower than the force measured.
Statistical differences were found between the control group and extra-articular stabilisation techniques (groups A and B) for yield and failure load. For linear stiffness, there were statistical differences between the control group and group B and between groups A and B.
No statistical difference was found either in yield or failure load between the two extra-articular stabilisation techniques (groups A and B) owing to the small number of biomechanical trials performed (n = 7), thus generating low Cohen’s effect size values and a posteriori statistical power analysis. However, discrepancies in failure mode were observed regarding the type of hip luxation occurring after stabilisation. Although no reoccurrence of craniodorsal hip luxation was observed with extra-articular UHMWPE implant stabilisation, the reoccurrence of craniodorsal hip luxation was observed in approximately 70% of cases with the UHMWPE iliofemoral suture technique.
Additional biomechanical testing is required to highlight differences in biomechanical behaviour between groups A and B before considering in vivo studies to confirm these findings.
Footnotes
Acknowledgements
We thank Mrs Cooke-Martageix for English language editing, Mr Blache for the statistical analysis and Mr Roume for the computer-aided design of the biomechanical bases.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by Novetech Surgery, who provided human resources in the person of Mr Bastien Goin for data processing and writing of this scientific article during his thesis.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (either prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
