Abstract
Case series summary
The objective of this case series was to describe the presentation, surgical treatment and outcome of a congenital intrahepatic portosystemic shunt (IHPSS) in 12 cats. A retrospective case series of cats undergoing surgical treatment for an IHPSS was undertaken. Signalment, clinical signs, imaging, surgical treatment, complications and short-term outcome (<30 days) were obtained using medical records. Long-term outcome (>1 year after first surgery) was obtained, where possible, using a health-related quality of life owner questionnaire. Seven cats were diagnosed with a left divisional shunt, three with a central divisional shunt and two with a right divisional shunt using intraoperative mesenteric portovenography. Three cats tolerated complete acute suture attenuation, eight cats underwent partial suture attenuation, four of which received complete suture ligation at a second surgery, and one cat underwent partial attenuation with a thin film band. Six cats (50%) developed post-attenuation neurological signs (PANS) after first surgery and two cats (17%) died or were euthanased due to severe PANS. Long-term outcome was available for eight cats (67%), with a median follow-up time of 1743 days (range 364–2228), and was described as excellent in five cats (63%), fair in two cats (25%) and poor in one cat (12%).
Relevance and novel information
Few papers exist that describe the presentation, intraoperative imaging, treatment and outcome of IHPSSs in cats. This is the first to describe surgical attenuation with a thin film band in a cat with an IHPSS. This case series reports excellent long-term outcomes in a majority of surgically treated cats with IHPSS.
Introduction
Congenital intrahepatic portosystemic shunts (IHPSSs) are rarely diagnosed in cats, and 73–100% of feline congenital portosystemic shunts (CPSSs) in the literature are extrahepatic;1–7 therefore, minimal information specific to feline IHPSSs is currently available. The aim of this case series was to describe the clinical presentation, intraoperative imaging, surgical treatment, and short- and long-term outcomes of a series of cats with an IHPSS.
Case series description
The medical records of all cats with a diagnosis of CPSS that underwent surgical attenuation at the Queen Mother Hospital for Animals, UK between 2004 and 2020 were reviewed. The inclusion criteria were cats diagnosed with an IHPSS via intraoperative mesenteric portovenography. Ethical approval was provided by the Royal Veterinary College Ethics and Welfare Committee (URNSR2021-0016).
Information collected from the medical records included signalment, body weight, pre- and postoperative clinical signs, pre- and postoperative medical management, ultrasound and/or CT findings, intraoperative mesenteric portovenography findings, method of shunt attenuation and perioperative complications.
Presenting clinical signs were categorised into five categories according to the body system involved: neurological; gastrointestinal; urinary tract; musculoskeletal (growth); and other. 8 A four-point grading system for hepatic encephalopathy9,10 was used to categorise cats before initiation of medical management. Clinical response to medical management instigated prior to surgery was also graded. 11
Surgical attenuation of the IHPSS was performed similarly to previously published reports. 12 Intraoperative mesenteric portovenography using a mobile image intensifier (Siremobil; Siemens) was used prior to and during temporary complete shunt occlusion to confirm the presence of an IHPSS, assess shunt morphology, confirm vessel occlusion at the correct site and assess intrahepatic portal vasculature. Complete shunt closure was achieved by suture ligation using two metric polypropylene (Prolene, Ethicon). Partial shunt attenuation was achieved using two metric polypropylene (Prolene; Ethicon) suture or an autoclaved, double-folded, 4 mm wide, clear, non-proprietary, synthetic polymer thin cellophane band with a uniform thickness (mean ± SD 55.3 ± 18.33 μm) thin film band partially tightened around the shunt using at least two metal clips, ensuring there was no acute portal hypertension, as described by Tivers and Lipscomb (determined by assessing a combination of parameters, including arterial blood pressure, central venous pressure, portal pressure, intrahepatic portal branching, colour of the pancreas and small intestines). 13 An additional two metric polypropylene suture was left loosely in situ around the shunt in case a second surgery was required.
Neurological signs after surgery were categorised according to severity and the degree of medical intervention required to control clinical signs, as described in a previous study. 9 Antiepileptic drugs were administered to effect according to clinician preference, if neurological signs developed postoperatively. Short-term clinical outcome (<30 days after first surgery) for surviving cats was graded as good (resolution of clinical signs), moderate (improvement in clinical signs) or poor (no improvement in clinical signs or relapse). 11 Long-term outcome was a minimum of 1 year after the first surgery and assessed where possible using a standardised owner questionnaire, modified from a published health-related quality of life questionnaire used in dogs with an extrahepatic portosystemic shunt (EHPSS). 14 Long-term outcome was then categorised based on a previous study: 15 excellent (resolution of clinical signs with no medical management); fair (resolution of clinical signs with continued medical management); or poor (recurrent or persistent clinical signs with poor response to surgery).
Twelve cats were treated surgically for an IHPSS. Details of each cat’s signalment, clinical signs, medical management and imaging findings are outlined in Table 1. Median age at time of first surgery was 0.9 years (range 0.3–5.9). Median body weight at time of first surgery was 3.2 kg (range 1.5–3.9 kg). Median duration of clinical signs before diagnosis was 73.5 days (range 1–1030). All cats had abnormal neurological clinical signs on presentation, which varied in severity (Table 1). Ten cats (83%) had ptyalism. Five cats presented with ‘other’ clinical signs, which included copper-coloured irises (n = 3/12; 25%) and ascites (n = 1/12; 8.3%). Three cats presented as an emergency, two with hepatic encephalopathy signs that improved with lactulose and antibiotic treatment. One cat presented with acute collapse, pyrexia and severe non-regenerative anaemia (packed cell volume 10%), which required a blood transfusion for stabilisation.
Signalment, presenting clinical signs, medical management, response to medical management and imaging findings in cats with an intrahepatic portosystemic shunt

Categorised into five categories according to the body system involved 8
Categorised according to a four-point grading system for hepatic encephalopathy before initiation of medical management9,10
Categorised according to a previously published study 11
MN = male neutered; NA = not applicable; BSH = British Shorthair; FN = female neutered; DSH = domestic shorthair; ME = male entire
All cats received medical management prior to surgery for a median duration of 43 days (range 8–150). Antibiotics were given in all cats except one: amoxicillin–clavulanic acid (12.5–25 mg/kg PO [Clavaseptin; Vetoquinol UK]) was given in seven cats (58%) and ampicillin (10–20 mg/kg PO [Amfipen; MSD Animal Health]) was given to four cats (33%). Ten cats (83.3%) were fed a low-to-medium protein commercial diet and two cats (16.6%) received no special diet as part of their medical management. All cats received lactulose (0.5–2 ml PO q8h) prior to surgery. Four cats received antiepileptic drugs preoperatively: three received levetiracetam (20–30 mg/kg PO [Keppra; UCB Pharma]) and one received levetiracetam with phenobarbitone (1.5–3 mg/kg PO [Epiphen; Vetoquinol UK]). By the time of first surgery, four cats (33%) had a good response to medical management, five cats (42%) had a moderate response to medical management and three cats (25%) had a poor response to medical management.
Intraoperative mesenteric portovenography identified seven cats (58%) with a left divisional IHPSS, three cats (25%) with a central IHPSS and two cats (17%) with a right divisional IHPSS. Cats diagnosed with a left divisional IHPSS (Figures 1 and 2) were found to have a dilated inverted C-shaped anomalous vessel from the left portal vein that inserted on the left side of the caudal vena cava. Left divisional shunts were dissected extravascularly immediately cranial to the liver on the left side at their insertion on to a focal dilation (ampulla) representing a confluence of the shunt, caudal vena cava, left hepatic vein and left phrenic vein. Two cats with a right divisional shunt (Figure 3) were found to have a long, dilated, tortuous continuation of the right intrahepatic portal vein through the right lateral lobe of the liver, which was dissected extravascularly immediately cranial to the liver on the right side at the insertion on the caudal vena cava. Three cats with a central divisional shunt (Figure 4) were found to have a dilated central portal vein branch becoming confluent with the caudal vena cava in one continuous straight line without deviation to either side. The central intrahepatic shunts were dissected extravascularly after parting the quadrate and right medial liver lobes to visualise the shunt at its insertion on the caudal vena cava.

Mesenteric portovenogram imaging of a left divisional intrahepatic shunt in a cat (cat 6) that tolerated complete acute shunt ligation. (a) Preocclusion; (b) post-complete acute occlusion
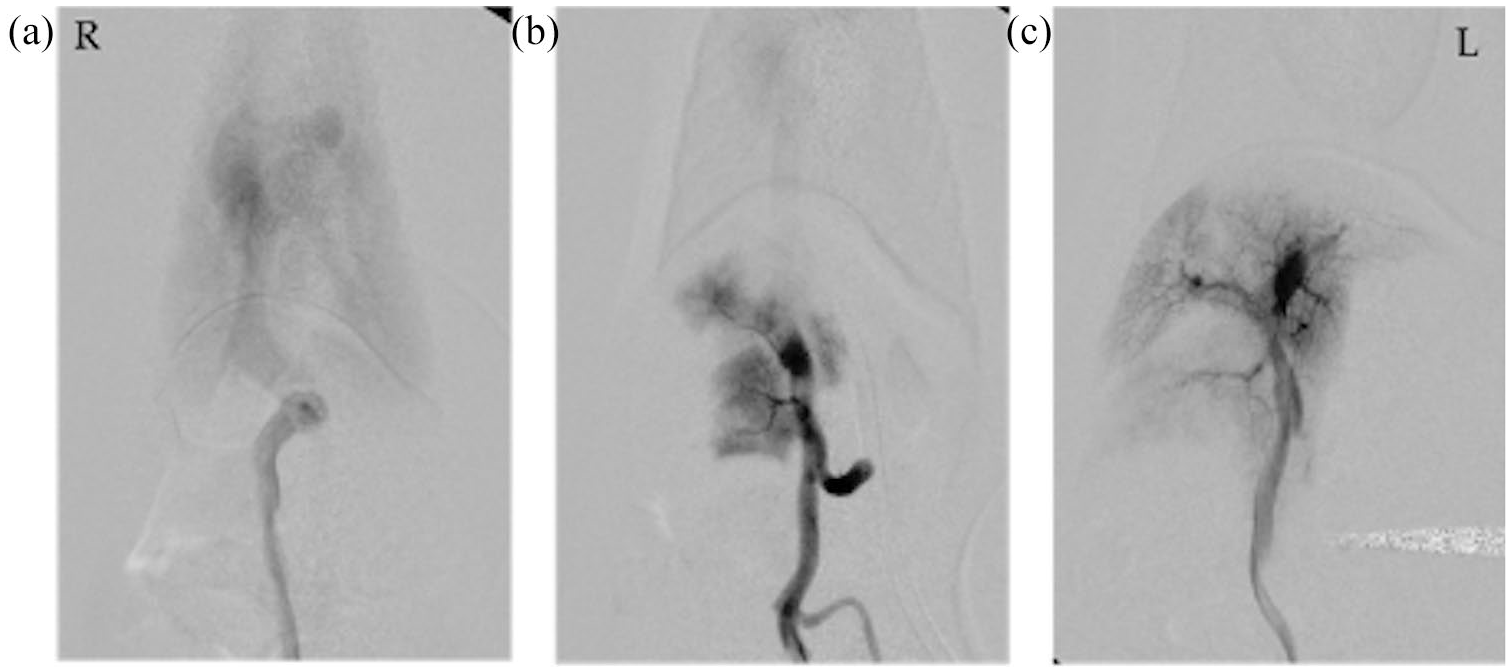
Mesenteric portovenogram imaging of a left divisional intrahepatic shunt in a cat (cat 11) that underwent staged suture ligation consisting of partial suture ligation at the first surgery and complete suture ligation at a second surgery. (a) Preocclusion (first surgery); (b) post-occlusion (first surgery; complete, temporary); (c) fully occluded at second surgery. Note the increased intrahepatic portal branching without portal vein and splenic vein backfilling at second surgery

Right divisional shunt in a cat (cat 8) that received partial suture ligation at the first surgery and only tolerated further partial suture ligation at the second surgery. (a) Preocclusion (first surgery); (b) post-occlusion (first surgery); (c) preocclusion (second surgery); (d) post-occlusion (second surgery)

Central divisional shunt in a cat (cat 7) that received thin film banding at the first surgery. The second surgery demonstrated the shunt was closed and multiple acquired shunts had developed. (a) Preocclusion (first surgery); (b) post-occlusion (first surgery; complete, temporary); (c) shunt closed (second surgery) and multiple acquired shunts
Details of the surgeries performed, complications and outcome for each cat are summarised in Table 2. Three cats (25%) tolerated complete acute shunt closure with polypropylene suture, and nine cats (75%) received partial attenuation with either suture or thin film band (one cat) at first surgery. Six cats underwent a second surgery. Median duration between the first and second surgeries was 120 days (range 98–364). Four cats with a partial suture ligation at first surgery tolerated complete shunt closure at second surgery (Table 2, Figure 2). One cat was only able tolerate further partial suture attenuation as temporary complete shunt occlusion during surgery resulted in signs of acute portal hypertension (Figure 3). The cat receiving a thin film band partial attenuation became anorexic 224 days following surgery; a second surgery revealed 2–3 large acquired extrahepatic shunting vessels draining into the azygous vein cranial to the diaphragm with no flow through the original intrahepatic shunt (Figure 4). One intraoperative complication was reported at second surgery, where haemorrhage occurred during dissection of adhesions around the shunt to access the pre-placed Prolene suture from the first surgery; a new ligature was placed instead. The cat’s packed cell volume reduced to 15% during surgery but recovered without a blood transfusion.
Details of the first and second surgeries performed, complications and clinical outcome for cats with intrahepatic portosystemic shunts 1 year after first shunt attenuation surgery was performed
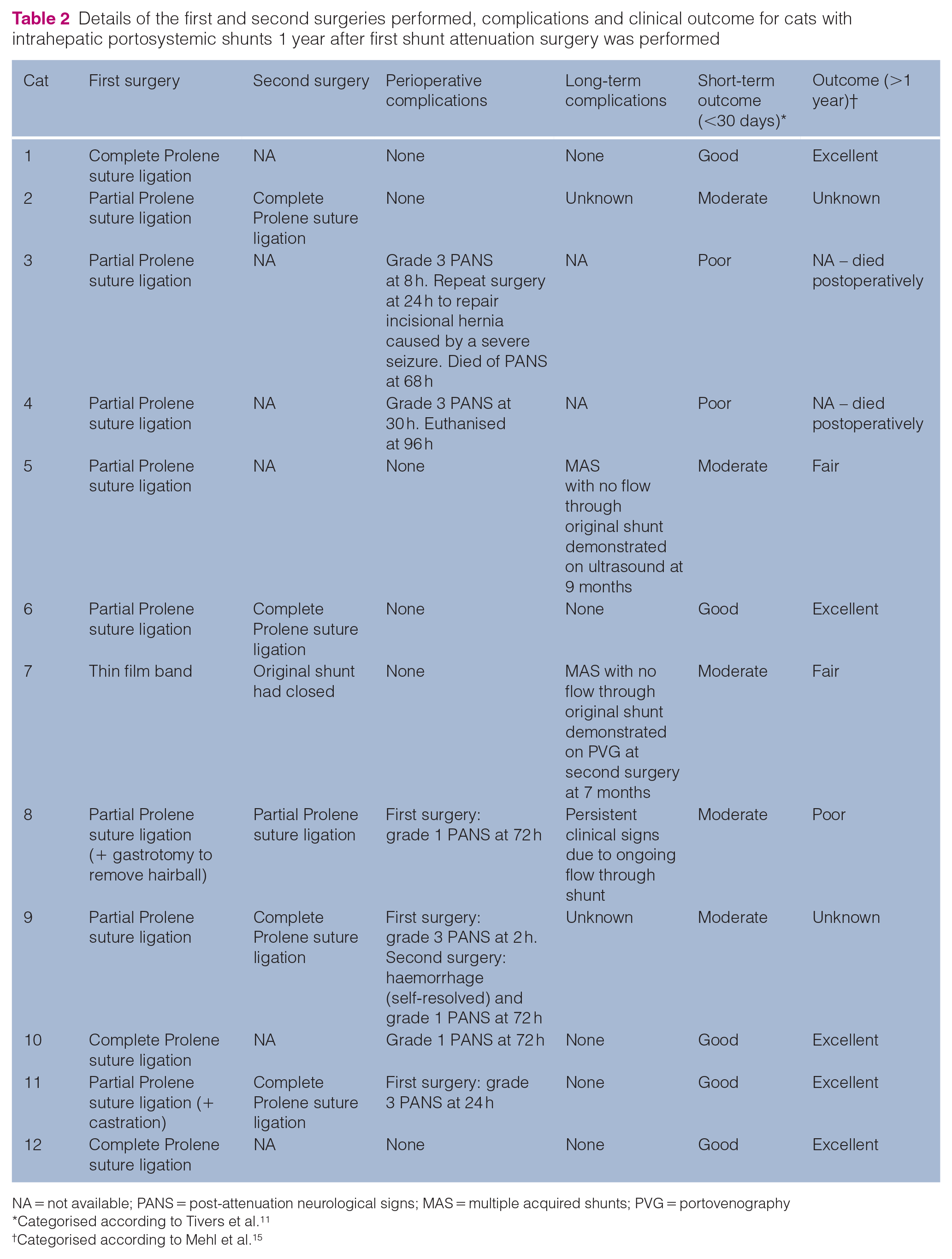
NA = not available; PANS = post-attenuation neurological signs; MAS = multiple acquired shunts; PVG = portovenography *Categorised according to Tivers et al.11†Categorised according to Mehl et al.15
Categorised according to Tivers et al. 11
Categorised according to Mehl et al. 15
Cats were hospitalised for a median duration of 6 days (range 4–7). Six cats (50%) developed post-attenuation neurological signs (PANS) following their first surgery, two of which resulted in death. Severity of PANS at the time of onset was grade 3 in four cats and grade 1 in two cats. The median time to onset of PANS was 24.5 h (range 3–72 h). Cat 3 developed grade 3 PANS 8 h after partial suture attenuation surgery, resulting in an incisional hernia, which required surgical repair 24 h following its shunt surgery, after which it died of cardiorespiratory arrest secondary to PANS. Cat 4 developed grade 3 PANS 30 h after the first surgery and was euthanased. Of the cats that underwent second surgery, only one developed PANS (grade 1, reduced vision), which had partially responded to phenobarbitone at the time of discharge.
Short-term outcome after the first surgery for cats was good in five cats (42%), moderate in five cats (42%) and poor in the two cats that died (17%). Five cats that received partial shunt attenuation continued to have clinical signs related to their IHPSS within the short-term period following their first surgery, which included seizures (1/5), vomiting (1/5), inappetence (1/5), blindness (1/5), haematuria (1/5) and mild neurological signs such as tremors and hypersalivation (2/5). The cat that developed seizures after the first surgery (partial suture attenuation) had an abdominal ultrasound 250 days postoperatively, which revealed multiple acquired extrahepatic shunting vessels medial to the right kidney, in addition to continued flow through the original intrahepatic shunt. A second surgery was not attempted, and the cat was continued on medical management.
Long-term information (>1 year after first surgery) was available for eight cats (67%), two cats were lost to follow-up and two died of PANS. Seven cats had long-term information provided by the health-related quality of life questionnaire and one cat had information in the clinical notes describing an excellent long-term outcome (absence of clinical signs without medical management) 1 year after its first surgery. Median follow-up period for seven cats was 1743 days (range 364–2228). Long-term outcome was excellent in 5/8 cats (63%), fair in 2/8 cats (25%) and poor in 1/8 cats (12%). The five cats with excellent long-term outcomes displayed no clinical signs related to their IHPSS, were not on any medical management and all received complete attenuation in either one or two surgeries. Both cats with a fair long-term outcome developed multiple acquired shunts (MAS) after their first partial attenuation surgery. One cat was on medical management (lactulose, levetiracetam and a commercial low-protein diet) with good control of their clinical signs unless medical management was withdrawn. The other cat was on lactulose and a commercial low-protein diet with overall good control of clinical signs except for monthly occurrences of diarrhoea and inappetence. The cat with a poor long-term outcome had only tolerated a second partial suture ligation and was on long-term medical management (lactulose, commercial low-protein diet and amoxicillin–clavulanic acid) with persistent seizures, head pressing, disorientation, aggression, ataxia, hypersalivation, collapsing episodes, lethargy and vomiting.
All owners reported that they were satisfied with their decision for surgical treatment of their cat’s IHPSS and felt that the surgery had improved their cat’s quality of life overall compared with before surgery.
Discussion
This study describes 12 cats treated surgically for an IHPSS, of which the majority of cats survived the perioperative period and had excellent long-term outcomes, which is consistent with the relatively small number of cats with IHPSS reported in the literature to date.12,16–18
The postoperative period following the first surgery was associated with the greatest risk in the present study, with 50% of cats developing PANS, two of which died. This was the only serious complication that developed, despite surgical treatment of IHPSS considered to be more challenging compared with EHPSS. 19 Numerous studies report PANS as the most common short-term complication in cats with a CPSS.1,5,9,20–22 It has been suggested that later published studies report a higher incidence of PANS in cats with CPSS due to a better understanding of this complication and the ability to detect milder abnormal neurological presentations such as tremors or behavioural changes.1,9 Similar to previous studies, a majority of cats in our study were able to recover from PANS, with excellent long-term outcomes.1,5,9,20 The occurrence of PANS after a second surgery was much lower (one cat), potentially due to the improvement in liver function following partial shunt attenuation.
This is the first study to show intraoperative images of mesenteric portovenography of left, right and central IHPSS in cats. The distribution of IHPSS locations in the present study is similar to that previously described, with left divisional IHPSS (patent ductus venosus) being the most common.12,18,23,24
In dogs, open surgical treatment of IHPSS vs EHPSS is generally considered to be more challenging owing to an increased risk of haemorrhage when dissecting around the intrahepatic shunt, especially a central divisional IHPSS.11,25 This has led to less invasive methods of intrahepatic shunt attenuation, such as endovascular coil embolisation being developed for dogs, and, more recently, for cats.18,26–29 Interestingly, in this study the authors considered the extravascular dissection of IHPSS in cats less challenging than in dogs owing to increased separation and mobility of the liver lobes, often resulting in less hepatic parenchyma covering the intrahepatic shunt, especially the central divisional shunts.
Suture ligation with the aim to achieve complete shunt closure at either first or second surgery was the surgical technique used in most cats with IHPSS in the present study. Only a small proportion of dogs and cats can tolerate complete acute closure attenuation of their IHPSS,12,25,30 which is consistent with the results in our study where only 3/12 (25%) cats were able to achieve this. One cat did not tolerate complete shunt closure at a second surgery and this cat had poor long-term outcome due to persistent clinical signs.
To our knowledge, this is the first study to report the use of a thin film band in feline IHPSS. The main advantage of gradual occlusion for CPSS in dogs and cats is the ability to achieve progressive complete or near complete shunt attenuation in one procedure.15,28,29 Some studies report that the use of gradual occlusion techniques, such as cellophane band and ameroid constrictor, may produce faster shunt attenuation than desired.15,31,32 An experimental study of thin film occlusion of intra-abdominal veins in cats demonstrated immediate complete closure of the intra-abdominal vein in two cats postoperatively and only 50% of cats had marked or complete vessel occlusion after 8 weeks. 32 Another study reported complete failure of the cellophane band to produce shunt closure in one cat. 20 Use of a thin film band may be unpredictable in cats when it comes to the degree of shunt attenuation it is able to produce.
In the present study, two cats (17%) developed MAS after their first surgery. Other studies have reported MAS in up to 23% of cats with CPSS following shunt attenuation.18,20,22,33 Both cats with MAS in this study had different surgical techniques performed and were reported to have fair long-term outcomes despite their MAS, achieving adequate control of their clinical signs with long-term medical management. This differs from the cat with persistent flow through its original IHPSS and poor long-term control of its clinical signs. Authors of the present study have noted that cats and dogs with acquired shunts tend to have more favourable outcomes on medical management than those with persistent flow through their original shunt, and this has been described elsewhere.18,20 It is possible that acquired shunts may play a less significant role in diverting portal blood flow away from the liver compared with the original hepatic shunt.
Long-term outcome in the present study used owner questionnaires, where possible, to assess quality of life, which has been used in previous studies for both dogs and cats with CPSS.1,11,14,34 The lack of consistent repeat imaging or liver function tests is a limitation of the present study. Other limitations of this study are the small sample size and retrospective study design, where the type and quality of data in the clinical records cannot be controlled. Furthermore, despite all cats being treated in one institution, the surgeries and postoperative care were not standardised. Grading systems were taken from previously published studies in dogs with portosystemic shunts; however, this could be considered a limitation as dogs and cats have slightly different neurological presentations and postoperative neurological complications. Therefore, future studies should include an established cat-specific grading system that allows better comparison between the different studies. Owing to the rarity of IHPSSs in cats, a multi-institution study, ideally prospective, is required to determine the optimal treatment and factors influencing outcome for IHPSSs in cats.
Conclusions
There is very little clinical research specific to feline IHPSSs and this study reports in detail the presentation, intraoperative imaging, treatment and outcome of 12 cats with an IHPSS. The majority of cats survived the short-term period following surgical attenuation of their IHPSS and had an excellent long-term outcome.
Footnotes
Author note
This paper was presented as an oral abstract presentation at the 2022 British Small Animal Veterinary Association (BSAVA) congress.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
This paper was handled and processed by the European Editorial Office (ISFM) for publication in JFMS
