Abstract
Objectives
The aims of this study were to describe diseases, complications and outcomes associated with the use of feeding tubes in a population of sick cats with appetite disturbance managed at an internal medicine referral service.
Methods
Clinicopathological data from cats receiving nasoenteral (NE) or oesophagostomy (O) tubes were studied. Cats were categorised according to their underlying disease and divided into subgroups (NE or O tube). The following factors associated with survival were analysed: disease category, type of tube and return to appetite.
Results
Included in the study were 112 cats, representing 118 cases. Of the 118 cases, 98 (83%) and 20 (17%) received NE or O tubes, respectively. The most common underlying conditions were digestive (13.5%), hepatobiliary (11%) and upper urinary tract (11%) disorders. Hepatobiliary (50%) and upper respiratory tract (30%) conditions were predominant in the O tube group. Digestive (15%) and upper urinary tract (12%) diseases were more common in the NE tube group. Complications following tube placement occurred in 22/118 cases (18.6%). The global survival rate was 73% and did not differ between NE (71.4%) and O tube (80%) groups (P = 1.00) or disease categories (P = 0.61). Return of appetite before feeding tube removal occurred in 56 cases (47%), within a median of 3 days (range 1–30) and was not associated with mirtazapine administration (P = 1.00). Appetite returned earlier for cats with NE tubes (3 days, range 1–17) than for those with O tubes (33 days, range 5–60; P <0.05). Voluntary food intake was positively associated with survival (odds ratio 15.17). After discharge and feeding tube retrieval, return of appetite was observed in 67% of cases.
Conclusions and relevance
Feeding tubes were predominantly placed for the management of appetite disturbance in cats with digestive, hepatobiliary and upper urinary tract diseases. While complications were frequent, they were mostly mild, easily managed and did not preclude feeding tube use. Return to appetite occurred in a large proportion of cats during or after assisted enteral feeding and was associated with survival.
Introduction
Anorexia, defined as a lack or loss of appetite, is a frequent presenting complaint in cats and occurs in a large variety of diseases. Owing to their distinctive metabolism, which requires high protein and essential amino acid concentrations, cats are highly susceptible to the effects of starvation.1,2 Prolonged loss of appetite can lead to severe systemic complications, including hepatic lipidosis, intestinal impairment and immunosuppression.2,3
Early refeeding is strongly recommended for any anorectic cat. It improves outcome and shortens hospitalisation in humans, dogs and cats.4–9 In cats with hepatic lipidosis, early enteral feeding has been associated with high recovery rate (up to 88%) in the absence of an identified underlying primary condition. 7
After correction of fluid and electrolyte imbalances, nausea and pain, several strategies can be implemented to enhance food intake. Trying a different food or increasing palatability of the meal is often attempted first.10–12 Some drugs can be tried to stimulate appetite. Mirtazapine and cyproheptadine are the two main medications recommended for cats owing to their proven efficacy and safety. 13 The efficacy and safety of mirtazapine has been demonstrated in healthy cats and in cats with chronic kidney disease, and is associated with a significant increase in the amount of food ingested in 24–91% of cats, with few side effects.14–17 Other drugs, such as benzodiazepines, glucocorticoids, anabolic steroids, megestrol acetate and, more recently, gabapentin have also been suggested.13,14 Capromorelin is another appetite stimulant currently approved for use in dogs and was recently demonstrated to be well tolerated in healthy cats in two randomised, placebo-controlled studies. 18
When appetite stimulants are not an option or fail to resolve anorexia, feeding tubes are indicated to provide enteral nutrition. Even if there is no consensus guidelines, three or more days of inappetence, meaning consumption of less than the resting energy requirement (RER = 70 × body weight [BW]0.75), should prompt assisted refeeding.11,19 In a cohort of hospitalised anorectic dogs, a significant and positive relationship was documented between caloric intake and outcome. 20 In another study, outcome in hospitalised dogs and cats was better for the animals that received food, regardless of the way it was administered (forced feeding, enteral or parenteral support). 7 However, there was no comparison between refeeding modalities, including nasoenteral (NE) and oesophagostomy (O) tubes. 7
NE and O tube placement is technically easy and inexpensive, justifying their common use in feline practice.3,21,22 The choice of using either an NE or an O tube is generally based on the clinician’s estimation of anticipated duration of feeding, type of food needed and anaesthetic risk (for O tube placement). While NE tubes are easy to place and do not necessitate general anaesthesia, their small diameter (5–8 F) requires the exclusive use of liquid diet. Medication through smaller tubes could compromise their patency. Moreover, NE feeding tubes only allow short-term nutritional support (<10 days), while O tubes can be used for up to 8 weeks, with appropriate care.2,23,24
Complications associated with enteral feeding are not rare and range from 13% to 71%.25–29 The two most common complications associated with the use of O tubes are infections of the stoma site and tube dislodgement.29,30 Premature removal of the NE tube despite the use of an Elizabethan collar, as well as diarrhoea, are also commonly reported with the use of NE tubes. 27 Moreover, one study demonstrated that the location of the tip of the NE tube (either in the oesophagus or in the stomach) did not influence complication rate. 31
Indications for enteral nutrition are highly variable but have never been reported for cats with NE or O tubes.8,9,23–27,32 Therefore, the first aim of this study was to describe the use of feeding tubes in a population of sick cats with appetite disturbances. We recorded the underlying disease process, the type of tube used (NE or O tube) and the associated complications. The second aim was to report clinical outcomes (survival and return to voluntary food intake) in this population.
Materials and methods
The medical records of cats hospitalised at the Internal Medicine Service of the Veterinary Teaching Hospital of VetAgro Sup Lyon, and managed with either NE or O tubes, between January 2015 and January 2020, for at least 24 h, were retrospectively reviewed. Each new tube placement was recorded as one case. The cases that received NE tube followed by O tube during the same hospitalisation were included in the O group.
The following details were recorded for each case: signalment; duration of anorexia; BW; body condition score (BCS) using a 9-point scale based on a previous publication; 33 physical examination findings; blood work; diagnosis (if achieved); type of feeding tube (NE or O tube); concurrent oral administration of mirtazapine; occurrence and nature of any complication; time from the onset of anorexia/hyporexia to presentation at the university (days); time from presentation at the university to feeding tube placement (days); time from beginning of anorexia/hyporexia to feeding tube placement (days); time from feeding tube placement to spontaneous return of appetite (days); and total duration of feeding tube utilisation (days). Appetite disturbance was qualified as anorexia or hyporexia. In this manuscript, ‘anorexia’ refers to complete loss of appetite and ‘hyporexia’ to partial loss of appetite. A cat could have presented first with hyporexia and then anorexia. Return to voluntary food intake was considered as being soon as the cat started to eat spontaneously even if the RER (RER = 70 × BW0.75) was not achieved. Cats with voluntary food intake were subdivided into two categories: spontaneous feeding either resulting in <50% of RER or >50% of RER.
Clinical signs reported by owners or physical examination abnormalities were used to allocate each case to one of the following disease groups: gastrointestinal; hepatic; pancreatic; triaditis; neoplastic; respiratory; cardiac; urogenital; haematological; endocrine; and infectious disorders. Cats entered the neoplastic category if they had a tumour that could not be included in any aforementioned category. Where there were multiple disorders, the condition that was most likely responsible for the clinical signs prompting hospitalisation was retained. Diseases that did not fit in any of the categories described above were classified as ‘other’. If the diagnosis was not reached, cats were classified in the category ‘unknown’. In this medicine unit, traumatic conditions were not represented.
Cats were categorised according to the disease category leading to appetite disturbance and the type of feeding tube used (NE or O tube). The feeding plan was the same for all cats in this population. At admission, the RER was estimated based on the following formula: RER = 70 × BW(kg)0.75. When the appetite disturbance lasted <5 days, the amount of food provided per day was progressively increased to reach 100% of RER after ‘X’ days, with ‘X’ corresponding to the number of days of anorexia. When the appetite disturbance lasted >5 days, the amount of food provided per day was progressively increased to reach 100% of RER over 5 days. Daily calorie intake was divided into 4–5 meals per day. The amount of food per day and the number of meals per day varied depending on the cat’s tolerance (induction of vomiting, abdominal discomfort, gastroesophageal reflux).
Statistical analyses
The statistical analyses were performed using R statistical software (Development Core Team 2008). 34 The data were generated for the general population, for cats with an NE tube and for cats with an O tube. For continuous variables (age, weight, BCS, temperature, time of anorexia, time before feeding tube placement, time before feeding, time before feeding tube retrieval), normality was assessed using the Shapiro–Wilk test. The median and interquartile ranges (IQRs; first and third quartiles) were reported for all data, because some variables were not normally distributed. For categorical variables (sex, vaccination, deworming status, lifestyle, clinical signs, haematological and biochemical abnormalities, use of mirtazapine, survival and complications), the respective frequencies were estimated using an exact binomial test. The cats in each subgroup (NE vs O) were compared using the non-parametric Wilcoxon Mann–Whitney test for continuous variables, and the Fisher’s exact test (with subsequent estimation of odds ratios [ORs]) for categorical variables. For all data evaluated, a P value <0.05 was considered to be statistically significant.
Results
Population
One hundred and twelve cats were included in the study, representing 118 cases of feeding tube placements, with six cats included twice at two different visits. Of the 112 cats, there were two intact females, one intact male, 60 neutered females and 49 neutered males. Eight breeds were represented, including 91 domestic shorthairs, eight Birmans, six Siamese, two Chartreux, two Maine Coons and one each of British Shorthair, Bengal and Norwegian. Median age was 9 years (range 1–18). Median BW was 3.8 kg (range 1.7–10) and median BCS was 4/9 (range 1–9).
Ninety-eight cases (83%) had an NE tube placed and 20 (17%) an O tube. Among the 20 cases that had an O tube placed, 13 (65%) had been previously managed with an NE tube and seven (35%) were only managed with an O tube.
Clinicopathological findings
Clinical signs
Clinical signs are presented in Table 1. For the global population, the median time of appetite disturbance before admission was 7 days (range 1–95).
Clinical signs of the 118 cases prior to presentation

Haematological and biochemical abnormalities
Haematological and biochemical abnormalities are presented in Table 2. The main biological abnormalities included increased alanine aminotransferase (65%), hyperbilirubinaemia (59%), anaemia (49%), hypoalbuminaemia (48%), hyperglobulinaemia (42%), hypokalaemia (41%) and hyperglycaemia (41%).
Haematological and biochemical data of the general population at admission
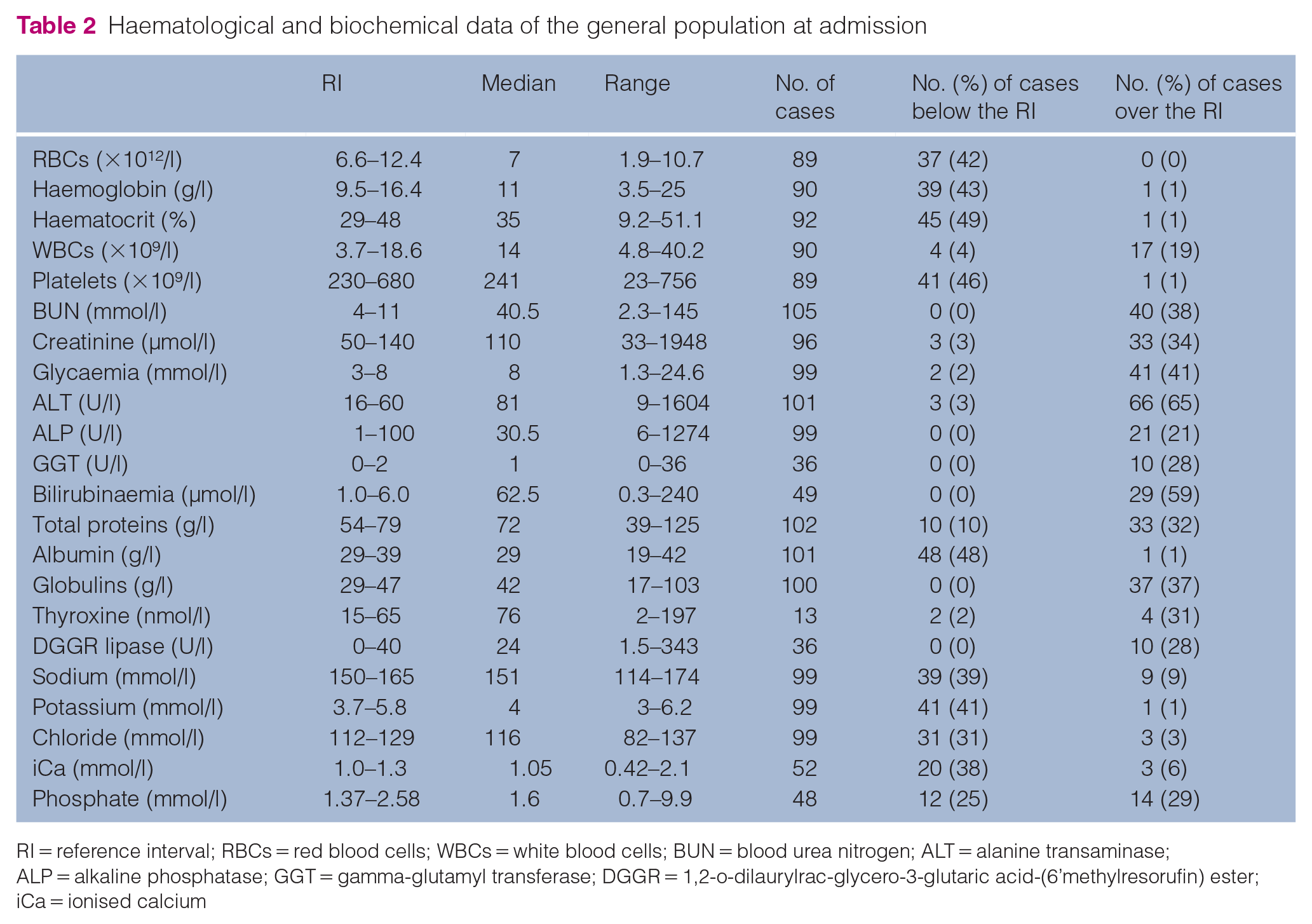
RI = reference interval; RBCs = red blood cells; WBCs = white blood cells; BUN = blood urea nitrogen; ALT = alanine transaminase; ALP = alkaline phosphatase; GGT = gamma-glutamyl transferase; DGGR = 1,2-o-dilaurylrac-glycero-3-glutaric acid-(6’methylresorufin) ester; iCa = ionised calcium
Disease categories
Disease categories are presented in Table 3. The main diseases associated with feeding tube placement were digestive (13.5%), hepatobiliary (11%) and upper urinary tract (11%) diseases.
Underlying disease processes that had required placement of a feeding tube in 118 cases

Comparison between NE and O tube groups
The subgroups (NE and O tube) were similar regarding age (P = 0.31) and sex (P = 0.32). BW and BCS were significantly lower in the NE group than in the O group (P = 0.03 and P = 0.05, respectively). Time between admission and tube placement in cats only managed with an NE or an O tube did not differ (median 0.5 days and 0 days, respectively; P = 0.70). For the 13 cats first managed with an NE tube followed by an O tube, median time between admission and O tube placement was 6 days (range 2–10). Median time of appetite disturbance before tube placement was similar between the NE and the O group (median 7 days and 10 days, respectively; P = 0.85).
The distribution of disease categories within the NE and O groups is presented in Table 4. Digestive (15.3%) and upper urinary tract (12.2%) diseases were the most frequent categories for the cats with an NE tube. Hepatic (50%) and upper respiratory tract (30%) categories were the most frequent categories for the cats with O tube.
Underlying disease processes according to the type of feeding tubes
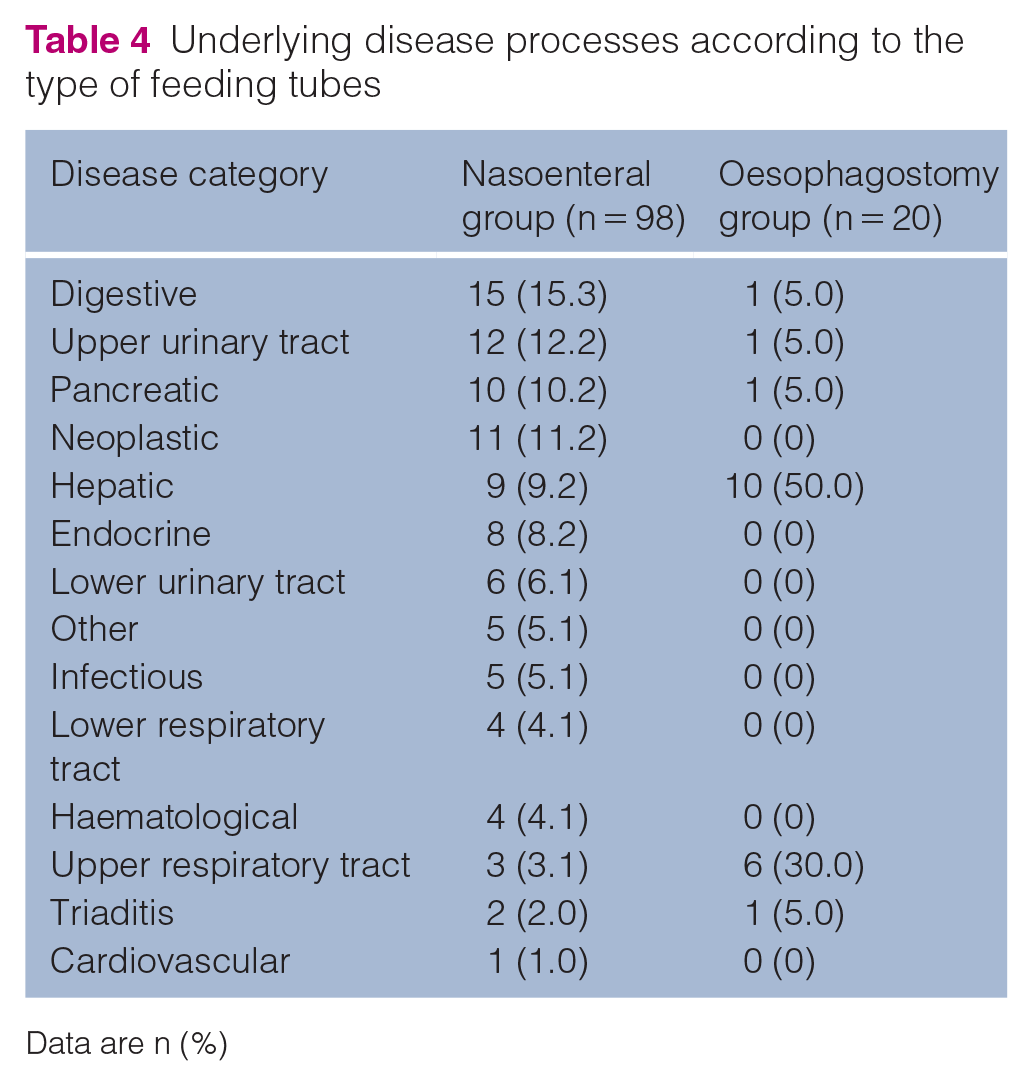
Data are n (%)
Complications
For the global population, complications following tube placement occurred in 22/118 cases (18.6%), and are summarised in Table 5. For the cases only managed with NE tubes, the complication rate was 15% (n = 15/98). Complications in cases only managed with O tubes were reported in 43% of cases (n = 3/7). For cats managed with an NE tube followed by an O tube, the complication rate was 31% (n = 4/13).
Complications associated with feeding tubes

Data are n (%)
NE = nasoenteral; O = oesophagostomy; NA = not applicable
Refeeding syndrome was suspected in one cat after 10 days of enteral feeding, including 7 days with an NE tube followed by 3 days with an O tube. The cat had appetite disturbance for 75 days before admission and a hepatic lipidosis was diagnosed. On day 10, the cat became severely lethargic and presented vomiting. Blood work revealed hypokalaemia (2.7 mmol/l), hypophosphataemia (1.06 mmol/l), hypomagnesaemia (0.4 mmol/l), hyperglycaemia (11.7 mmol/l) and anaemia (haematocrit 10.6%). The cat was managed by intravenous (IV) and oral potassium, IV magnesium supplementation, oral phosphorus supplementation, IV B1 and B6 vitamin supplementation, constant rate infusion of metoclopramide, IV maropitant citrate and fluid therapy. Enteral feeding via the O tube was continued. Anaemia required a whole-blood transfusion. The cat survived to discharge, and spontaneous food intake was observed on day 29.
Outcomes
For the global population, spontaneous return to appetite before feeding tube removal occurred in 56/118 cases (47%), including 49/98 (50%) cases in the NE group and 7/20 (35%) in the O group (P = 0.33). Median time before return to voluntary food intake was reported in 52/118 cases (44%) and was 3 days (range 1–30 days) for the whole population. Return to voluntary food intake occurred in a median time of 3 days (range 1–16) for the NE group and 14 days (range 7–30) for the O group (P <0.05).
The amount of food ingested when the tube was retrieved was reported in 48 cases for which return to voluntary intake occurred. The percentage of RER covered was estimated as <50% in 22 cases (46%) and >50% in 26 cases (54%). A comparison in the RER coverage between cats with NE and O tubes is presented in Table 6. These results were not statistically different between groups (P = 0.1).
Amount of food ingested according to the type of feeding tube

RER = resting energy requirement; NE = nasoenteral; O = oesophagostomy
Thirty-nine cases (33%), including 31 in the NE tube group and eight in the O tube group, received mirtazapine during hospitalisation. Mirtazapine was not associated with return to voluntary food intake (P = 1.00) or amount of food consumed when reported (P = 0.53) (Table 7).
Amount of food consumed with or without oral administration of mirtazapine

RER = resting energy requirement
The decision to remove the tube occurred in 88/118 cases (75%), including the 56/88 cases previously reported with spontaneous return of appetite and 32/88 that did not recover appetite. The 30 cases for which the tube was not removed died or were euthanased. For the global population, median time before removal was 5 days (range 1–61). It was 4 days (range 1–17) for the NE group and 33 days (range 5–61) for the O group (P <0.05). Of those 88 cases, 86 survived to discharge (98%). The survival rate was 72.9% for the 112 cats included in this study and did not differ between the NE (71.4%) and O groups (80%; P = 1.00). Survival was also similar among disease categories (P = 0.61). Resolution of anorexia was positively correlated with survival (OR 15.17, 95% confidence interval 4.21–83.94).
Of the 32 cases that survived to discharge and did not experience voluntary food intake before tube removal, follow-up was available for 12 (37.5%). Return of appetite was observed in 8/12 cases (66.7%) in a median time of 2 days (range 1–12). The last 4/12 cases remained anorexic (33.3%). Two were euthanased and two underwent another tube placement within 72 h after initial discharge.
Discussion
Few studies have documented the use of feeding tubes in cats. Therefore, the first aim of this study was to describe the use of NE and O feeding tubes, including underlying disease processes and complications of feeding tube placement in a large population of cats. Complications were common and occurred in 18.6% of cases.
The second aim of this study was to evaluate survival and return to voluntary food intake in this population. Spontaneous food intake occurred in half of the cases before removal of the tube, and a large proportion of the global population survived to discharge (72.9%). When return to voluntary eating was observed, 54% of cats ate >50% of their RER. Time before spontaneous food intake was significantly shorter when an NE tube was used (4 days) compared with when an O tube was used (14 days). Survival did not depend on the type of feeding tube chosen or disease category, and was positively correlated with resolution of anorexia.
In this study, 118 cases with feeding tube placement were documented, with 83% being NE tubes and 17% being O tubes. Sixty-five percent of cats with an O tube were previously managed with an NE tube. Interestingly, median BW and median BCS were significantly higher for cats with an O tube compared with those with an NE tube. One explanation could be that overweight cats seem to be more susceptible to hepatic lipidosis and so a longer, more aggressive nutrition plan could be preferred.9,35–37 Moreover, the duration of appetite disturbance before tube placement did not vary between cats only managed by NE and O tubes. This might suggest that the type of tube chosen is not only influenced by the duration of anorexia, but rather by other parameters, including the severity of the condition encountered or the BCS of the patient. Interestingly, time of appetite disturbance (7 days for NE tube and 10 days for O tube) was longer than the period of anorexia (3 days) generally accepted as an indication for tube placement. This result seems to be primarily due to the fact that, as a referral service, cats were seen in the first instance by their primary care veterinarian.
In our population, the most common underlying disease processes were digestive, hepatobiliary and upper urinary tract diseases (13.5%, 11% and 11% of cases, respectively). Interestingly, hepatobiliary and respiratory conditions were predominant (50% and 30%, respectively) in the O tube group, and digestive and upper urinary tract diseases were the most common categories (15.3% and 12.2%, respectively) in the NE tube group. To our knowledge, this is the first study to compare disease conditions associated both with O tube and NE tube placement. In a recent study on O tube placement in cats, the repartition of disease categories was slightly different. 26 Indeed, traumatic, neoplastic, pancreatic and gastrointestinal diseases were most common, representing 16.1%, 16.1%, 14.1% and 11.3% of cats, respectively. The differences with our findings could be explained by different populations and definitions of disease categories. Indeed, cats with trauma were not represented in our study, as it was conducted in an internal medicine service. Moreover, only cats with a neoplastic condition that could not fit a previously defined category were classified in the neoplastic subgroup in the present study, which was not the case in the previous study.
We observed that voluntary eating occurred in 48% of cats while the feeding tube was still in place. This result agrees with previous studies that reported spontaneous feeding ranges from 44% to 50%.27,32 The quantity of food ingested at the time of removal of the tubes represented <50% of RER in 46% of cats and >50% of RER in 54%. To our knowledge, this study is the first to document a quantitative evaluation of voluntary food intake during utilisation of feeding tubes in cats. Almost half of the cats ate <50% of RER when their tube was retrieved. For cats remaining anorexic despite assisted nutrition with an NE tube, appetite at home is often tested to rule out appetite disorders related to stress within the hospital areas before drawing any conclusions and considering other means of feeding. Considering the small number of cats managed with an O tube alone, it remains difficult to draw clear conclusions regarding food intake. Median time before tube removal was significantly shorter in the NE group compared with the O group. Obviously, NE tubes are retrieved earlier than O tubes, as they are a more short-term option due to their use and discomfort caused to the patient. An earlier return to voluntary eating for cats within the NE group is also logically expected. Moreover, in our hospital, cats are never discharged with their NE tube.Mirtazapine is a serotonin and presynaptic alpha2-receptor antagonist that acts as an appetite stimulant.
In our study, the use of mirtazapine did not appear to act on qualitative (return to voluntary food intake or not) or quantitative (<50% of RER or >50% of RER) food intake. This finding is unexpected as an appetite-stimulating effect of oral or transdermal mirtazapine has been demonstrated in several studies comparing sick and healthy cats.13,14–17,38 However, to our knowledge, the effect of mirtazapine in cats managed with enteral feeding has never been previously evaluated. It cannot be excluded that the tube in place might alter the response to mirtazapine.
The survival rate in this population of cats managed by NE and O tubes was 72.9%, which is comparable to those registered in previous canine and feline case series with enteral feeding, ranging from 65.3% to 83%.7,26,27 Moreover, the mortality rate did not significantly differ according to the type of tube or among disease categories. In a previous study assessing the effect of nutritional support including enteral and parenteral nutrition and forced feeding on hospital outcome, outcome was mainly related to disease severity. 7 Indeed, survival might rather be influenced by the severity of the disease process itself rather than the type of tube chosen. However, disease severity was not assessed in our population of cats, and it precludes further conclusions regarding factors influencing survival. Resolution of anorexia was significantly and positively associated with survival in our study. This result is in accordance with previous studies, which have demonstrated that persistent anorexia can lead to negative effects on wound healing, immune function, skeletal and respiratory strength and overall prognosis.13,39,40
Complications associated with enteral feeding were common and occurred in 18.6% of cases. In our study, the complication rate associated with the use of NE tubes was 15%, with self-removal of the NE tube being the most common (14.2%). This complication rate appears to be lower than previously reported with NE tubes (42–71%).27,32 However, the prevalence of NE tube removal is similar to that documented in other studies (11–18%), and diarrhoea, which was the main complication previously reported with feeding tubes (25–27%), was not noted in this study. In our study, complications associated with the use of O tubes occurred in 43% of cases, which is in the range of frequencies reported in the literature (13–71%).25,26,28–30 As in two recent studies, infection of the stoma site (14.3%) and partial tube dislodgement or complete removal (28.6%) were the two most common complications associated with the use of O tubes.26,30
Interestingly, refeeding syndrome was diagnosed in one cat in our study, which led to an overall prevalence of 0.8%. In our case, duration of appetite disturbance before tube placement was 11 weeks, which was similar to the results of a recent study (range 3–104). 41 This cat presented electrolyte disturbances (hypokalaemia, hypophosphataemia and hypomagnesaemia), hyperglycaemia and anaemia, which is in accordance with previously reported biological abnormalities.41,42
The main limitation of our study was its retrospective design, which led to missing information. The percentage weight loss since the beginning of appetite disturbance was not documented. It would have been a more interesting parameter to assess than BW or BCS, as this parameter is dynamic and could reflect disease evolution. Moreover, disease severity, usually determined by clinical scores, was not evaluated. It was an important confounding factor, as disease severity could have influenced the choice for a specific feeding option and global prognosis. Furthermore, owing to the retrospective design, the occurrence of diarrhoea as a complication of enteral feeding could not be included, as it was inconsistently reported in the medical database. Another limitation is that many cats (18.6%) were lost to follow-up after tube removal, which affected precise evaluation of outcomes associated with the use of NE tubes and O tubes. Then, even if the effect of mirtazapine was quantitatively investigated by evaluation of RER coverage, a slight improvement of food intake (+10–20%) could have been missed. Finally, our study was conducted in the internal medicine service of a tertiary veterinary teaching hospital, which led to selection bias, with the exclusion of traumatic diseases, for instance. Therefore, the results cannot be generalised to all anorectic cats with enteral feeding.
Conclusions
Enteral feeding was well tolerated in cats, and diseases associated with the use of feeding tubes were highly variable. Minor complications were not infrequent, but were easily managed. Even if return to voluntary food intake occurred in less than half of the cases before tube removal, it was reported in a larger proportion of cases thereafter. This suggests that spontaneous food recovery can be attempted at home in some cases. Mirtazapine appeared safe in cats with feeding tubes but was not associated with resolution of anorexia.
Footnotes
Author note
This original study was presented as a poster at the 2020 ECVIM-CA congress, using preliminary data collected from January 2015 and July 2018.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
This paper was handled and processed by the European Editorial Office (ISFM) for publication in JFMS
