Abstract
Practical relevance:
Inappetence may have many origins and, as a presenting sign or observation in the hospitalised patient, is common in feline practice. Nutritional assessment of every patient is encouraged, to identify the need for, and appropriate type of, intervention indicated. The impact of malnutrition may be significant on the feline patient, perpetuating illness, delaying recovery, slowing wound healing and negatively impacting gut health and immunity. Delayed intervention may result in the cat’s deterioration; hence prompt control of contributing factors such as the underlying illness, pain, nausea, ileus and stress is vital to optimise voluntary food intake. Management is multimodal, comprising reduction of stress, medications and assisted nutrition in the form of tube feeding or parenteral nutrition. Use of antiemetic, analgesic, prokinetic and appetite stimulant medications may restore appetite, but placement of feeding tubes should not be delayed. Feeding tubes are generally well tolerated and allow provision of food, water and medication with minimal stress, although clinicians must be aware of complications such as stoma site infections and refeeding syndrome.
Clinical challenges:
Cats are vulnerable to malnutrition owing to their unique metabolism and specific nutritional requirements. Moreover, their nature as a species means they are susceptible to stress in the hospital environment, which may result in reduced food intake; previous negative experiences may compound the problem. In particular, an inappropriate clinic environment and/or handling may cause or exacerbate inappetence in hospitalised patients, with negative impacts on recovery. Postponing interventions such as feeding tube placement to await improvement, owing to clinician or caregiver apprehension, may hinder recovery and worsen nutritional deficits.
Evidence base:
The 2022 ISFM Consensus Guidelines on Management of the Inappetent Hospitalised Cat have been created by a panel of experts brought together by the International Society of Feline Medicine (ISFM). Information is based on the available literature, expert opinion and the panel members’ experience.
Keywords
Introduction
Inappetence is a common presenting sign, 1 as well as a frequently observed complication of hospitalisation, 2 in feline patients. Suboptimal nutrition may have many negative effects on cats, in particular, given their higher protein requirements compared with other species, as well as other metabolic adaptations. 3 Additionally, hospitalised cats are vulnerable to stress, which may impact appetite with consequences on recovery from illness, wound healing and immunity. 4 Aside from stress and the underlying pathology, there may be many other factors contributing to a cat’s reluctance to eat. Pain, nausea and ileus must all be managed in order to improve the cat’s food intake, and many strategies are also applicable to outpatients. 5 Nutritional assessment is a vital part of case management, with implications for treatment and prognosis;6–8 and early intervention to encourage voluntary food intake or provision of support with feeding tubes can improve outcome.3,5

These guidelines aim to provide practitioners with tools to identify cats in need of nutritional support, and guidance on how and when to intervene, so that their hospitalised feline patients can avoid the negative consequences of poor nutrition. Accompanying these guidelines are two guides and a video to support clients caring for cats with a feeding tube; and two videos on feeding tube placement and management, which have been created to provide support for veterinary staff (see Appendices 1 and 2, page 640; also available at bit.ly/inappetentcattoolkit).
Unique nutritional and energy requirements of cats
Feline nutritional requirements
Cats require nutrients and energy from their food to survive and thrive, 9 and while some nutrients can be synthesised from precursors (non-essential), others must be provided by the diet (essential). A complete diet is one that provides all essential nutrients in adequate amounts to prevent deficiencies or excesses. For commercial diets, there are industry recommendations and sometimes regulatory guidelines that define what a complete diet is; relevant bodies include the Association of American Feed Control officials (AAFCo) 9 and FEdIAF, 10 the trade body representing the European pet food industry. Nutrients can be classified as macronutrients, or as micronutrients (vitamins and minerals). Macronutrients include protein, fat, digestible carbohydrates and fibre. Protein, fat and digestible carbohydrates also provide energy, 11 with fat providing more than twice the amount of metabolisable energy per gram (8.5 kcal or 35.5 kJ) compared with protein and carbohydrate (3.5 kcal or 14.6 kJ).
Manufacturers have to declare on the cat food label if the diet is nutritionally complete, but it is not easy to determine this from the nutritional information alone, as what is listed is very limited. Cats require about 40 essential nutrients (Table 1), and typical pet food labels are only mandated to list a few (eg, protein, fat) and provide no data on digestibility or bioavail-ability. For therapeutic veterinary diets, product guides provide more information and nutrients are usually presented on an ‘as fed’ and calorie basis, the latter being more helpful when comparing diets, as it allows direct comparison between diets differing in moisture content and energy density. Some therapeutic veterinary diets might not meet all nutrient requirements established by AAFCO/FEDIAF owing to their nutritional strategies to manage disease.
Essential nutrients required in the diet of cats 11

Cats’ nutrient requirements differ from those of dogs, both quantitatively and qualitatively. Requirements also vary depending on the life stage of the cat. Cats are strict carnivores and some of their nutritional peculiarities might be related to their evolution as hunters (Figure 1). 12 For example, cats require higher amounts of protein than dogs (and people). A lack of enzymatic adaptation to dietary protein levels, 13 meaning that the rate of protein breakdown does not adjust according to reduced protein intake, has been proposed to explain these high requirements. The typical prey of cats is low in starch and high in protein,14,15 and it has been hypothesised that the high protein needs are in part to cover glucose requirements via gluconeogenesis. 16 Protein requirements for critically ill cats have not been established, but a common recommendation is to provide 60-80 g/1000 kcal.17,18 This is higher than minimum maintenance protein requirements, likely due to factors such as increased protein losses or altered metabolic and inflammatory pathways. 19 It is postulated, therefore, that the high protein and amino acid requirements are intended to minimise lean mass catabolism during illness.

Cats have higher protein requirements than dogs and humans, likely related to their evolution as hunters. Image courtesy of Sam Taylor
Despite this high protein requirement, cats are metabolically flexible 20 and can adequately metabolise different dietary macronutrient profiles, provided that minimum requirements are met. Also, while carbohydrates are not essential, cats are capable of digesting and utilising dietary starch, as long as it is adequately processed and provided within an adequate range. 21
Other nutrients that are exclusively found or more abundant in animal tissue are essential for cats. For example, cats cannot adequately use beta-carotene (plant based) as a source of vitamin A and require dietary retinol. Moreover, cats have a very low delta-6-desaturase enzyme activity, which limits their fatty acid metabolism; therefore, arachidonic acid is an essential nutrient. Taurine is also essential in cats and most abundant in animal tissue (especially organs), whereas dogs can synthesise it from sulfur-containing amino acids. While vitamin D is an essential nutrient in both dogs and cats, there are additional challenges for cats; namely cats extract dietary vitamin D more efficiently from animal sources (cholecalciferol) than from plant-based ergocalciferol. 22 It is important to note that the fact that cats are strict carnivores does not mean they can survive on meat alone (especially skeletal meat). Nutritional imbalances will develop in cats fed in this manner.23,24
Feline energy requirements
Energy requirements of cats vary depending on several factors, such as age, life stage, breed, activity level and neuter status.11,25 Requirements can be estimated with formulas, and there are different equations depending on the information source. As all estimates are associated with varying degrees of error, 26 they should all only be considered a starting point. Energy requirements can be expressed as maintenance energy requirements in adults or daily energy requirements, and the units are kcal or kJ per day. Resting energy requirements (RER) approximate the basal metabolic needs, important for basic functions, and are commonly used to estimate energy needs of hospitalised cats on cage rest. 6
If energy needs are not met, cats are at risk of undernutrition, which has negative consequences on outcomes. Excess calorie provision can result in overweight and, in critically ill patients (even if underweight), can increase the risk of complications such as hyperglycaemia, vomiting and diarrhoea. 27 Therefore, the veterinary team must be very careful when feeding hospitalised critically ill cats to safely promote calorie and nutrient intake while minimising risks.
Physiology of starvation
When cats are underfed, there is a metabolic response to ensure provision of nutrients for basic functions using body reserves. This response is different when it happens in healthy animals without access to food (‘simple starvation’) compared with hospitalised animals, where undernutrition is secondary to decreased food intake and altered metabolism associated with disease (‘stressed starvation’).1,28
Simple starvation
In healthy animals, dietary nutrients are used to meet immediate needs, sparing endogenous fuels stored as glycogen and adipose tissue. Excess energy can be stored as fat. When there is food deprivation, the use of endogenous fuels is required to maintain body functions. Over time, there is a shift from using mixed sources (glycogen, protein, fatty acids) to using fatty acids as a primary fuel source, and a simultaneous decrease in the basal metabolic rate to conserve resources (such as lean body mass). Glycaemia will be maintained via hepatic glycogen (glycogenolysis) and, when these stores are depleted, gluconeogenesis, using substrates resulting from the catabolism of adipose (glycerol) and lean tissue (glycogenic amino acids, lactic acid and pyruvate). Glucose can also be recycled – with energy cost – to ensure provision for glucose-dependent tissues. Some of these tissues, like the brain, can adapt to using ketone bodies as an energy source.
Stressed starvation
Hospitalised cats usually have access to food but can have a reduced or absent appetite. Such patients may be in a hypermetabolic state, induced by inflammatory mediators and stimulation of the sympathetic nervous system.29,30 This can increase energy expenditure and proteolysis, leading to a negative energy and nitrogen balance, with no adaptive reduction in the metabolic rate. Despite this effect on energy needs, overall energy expenditure of hospitalised cats is usually lower than maintenance, likely due to a reduction in physical activity during cage rest. However, the accelerated loss of lean body mass can negatively affect immune function, wound healing and, potentially, overall survival.31,32 The specific disease(s) of the patient will also influence the type and degree of metabolic and hormonal alterations. Nutritional support can help to provide substrate and energy to the patient, but disease management is required to reverse the hypermetabolic state.


Causes of inappetence in cats
There are numerous causes of inappetence in cats (see box). Certain cases of reduced food intake are functional, and have a more obvious cause, such as jaw fractures or other facial injuries; other conditions may lead to dysphagia, where the cat is interested in food yet unable to eat (eg, periodontal, oral or neurological disease). This may be termed ‘pseudoanorexia’.
In general, the most common causes of inappetence in hospitalised cats are conditions leading to nausea (with or without vomiting), pain or ileus, or the stress of hospitalisation itself (discussed in more detail later).3,35 Certain conditions can affect appetite directly via inflammatory cytokines (eg, neoplasia). 36
Non-specific causes of inappetence
There are important non-specific factors commonly associated with inappetence in hospitalised cats that must not be overlooked, as summarised in Figure 2 and described below.

Factors commonly associated with inappetence in hospitalised cats. All must be managed to encourage voluntary food intake. (See also Figure 6 for causes of distress in hospitalised cats.) Image courtesy of Sam Taylor
Nausea and vomiting
Many underlying conditions causing inappetence also lead to vomiting. When vomiting is not present, inappetence may sometimes be the only sign of nausea. Other signs include ptyalism (Figure 3), lip licking, turning away from food and retching at the sight/smell of food. 37 Management of the underlying disease is optimal, but an antiemetic treatment trial is also indicated; medication is selected based on the individual patient’s condition, and may include maropitant, metoclopramide, mirtazapine and ondansetron (Table 2). 38

Signs of nausea may include ptyalism or more subtle signs (inappetence, lip licking and turning away from food). Image courtesy of Serge Chalhoub
Commonly used antiemetics, prokinetics and appetite stimulants for the management of inappetence in cats*

Information contained in this table represents a consensus of opinion and experience of the guidelines panel members. Treatment is at the discretion of the attending veterinary surgeon. See text for further discussion of these agents
CRI = constant rate infusion; IV = intravenous; IM = intramuscular; SC = subcutaneous; PO = oral; GI = gastrointestinal
Pain
Pain assessment and provision of adequate multimodal analgesia, selected and dosed to avoid adverse effects, will optimise voluntary food intake. Pain scoring systems, such as the Feline Grimace Scale, 60 UNESP-Botucatu multidimensional feline pain assessment scale short form 61 and Glasgow composite pain scale-feline, 62 should be used regularly to score pain in hospitalised cats and assess the response to analgesia.63,64 An analgesic trial may be considered in inappetent patients, selecting agents based on the cat’s clinical condition and any contraindications.
Gastrointestinal dysmotility
Gastrointestinal (GI) dysmotility may be a common sequela of critical illness and surgery in several species, including cats;42,53 it is reported, in particular, in association with pancreatitis and hepatic lipidosis, 37 but is also associated with electrolyte abnormalities, medications such as opioids, primary GI disease and systemic disorders. 42 Ileus (decreased/lack of motility), with fluid and gaseous distension of the bowel, causes pain and inappetence, as well as fluid sequestration. Affected cats may show signs of nausea, abdominal distension, regurgitation, abdominal discomfort and diarrhoea, and are at risk of aspiration pneumonia. Additionally, ileus has a significant effect on the cat’s nutritional status, reducing voluntary food intake and limiting the amount of food that can be given via a feeding tube, for example.5,42
Diagnosis of ileus is generally subjective, as evaluation of GI motility beyond ultrasound examination and radiography is unlikely to be available outside specialist practice. Contrast radiography is generally no longer used due to the risk of aspiration pneumonia, as well as stress and food aversion associated with administration of oral contrast media. Ultrasonography may reveal an inappropriate degree of gastric filling, reduced gastric contractions and reduced intestinal peristalsis. Normal rates of peristalsis are 4-5 contractions/min for the proximal duodenum and 1-3 contractions/min for the rest of the small intestine. A normal stomach should contract 4-5 times/min if it contains some food, and less frequently if empty. 42
Management with pharmaceuticals to promote gastric emptying, such as metoclo-pramide, erythromycin or cisapride (Table 2), together with early enteral nutrition, is indicated, along with suction of fluid from the GI tract, if appropriate, and use of antiemetics and analgesics to promote comfort. Other types of GI dysmotility may result in inappetence in hospitalised cats, including gastro-oesophageal reflux, which is reported in critical patients 53 and during anaesthesia, 65 and may result in oesophagitis.
General considerations for management of inappetence in hospitalised cats
The risk of developing inappetence may be reduced by evaluating prescribed medications and how they are administered, and optimising treatments early on to promote appetite. Many commonly used medications may cause inappetence (see box on page 617); hence, reviewing prescriptions, discontinuing redundant and unnecessary drugs, switching from oral to IV or SC administration, if appropriate, and avoiding drug interactions may result in increased voluntary food intake. Bitter-tasting tablets can be put into gelatin capsules and bitter liquids should be avoided. Food offered to hospitalised cats should be palatable and good quality. However, feline food preferences are set at an early age, and may result in neophobia (dislike of anything new or novel; ie, preference for certain tastes and textures of food); 66 hence, presenting hospitalised cats with unfamiliar tastes and textures may result in inappetence and food aversion. This can commonly occur when a dietary history is not obtained, diets prescribed for the underlying condition are novel and unpalatable to the patient, or when hospitalised cats are force- or syringe-fed. Thus, such diets should not be introduced while the cat is hospitalised, and force-feeding is never indicated.
In addition to the management of nausea, pain and ileus, fluid deficits should be corrected along with electrolyte abnormalities. Dehydration may result in various negative clinical consequences, including inappetence. Hypokalaemia has been associated with reduced appetite 67 and ileus, 42 and is common in critically ill cats; 68 hence, serum potassium should be assessed and deficiencies corrected. Similarly, supplementation with cobalamin in vitamin B12-deficient cats may result in clinical benefits, including improved appetite.69,70
Constipation is common in hospitalised human patients, particularly older adults, 71 for reasons including dehydration, pain, electrolyte abnormalities, inactivity and immobility, and constipating drugs such as opioids. The same is likely true for feline patients, where risk factors including chronic kidney disease (CKD), obesity, older age, osteoarthritis and other comorbidities may contribute. 72 Additionally, cats may be reluctant to use litter boxes if they are used to defecating outdoors, or if the litter type/box is unfamiliar. If possible, caregivers should be asked to provide familiar litter, or soft sand litter should be used, 2 and other risk factors for constipation managed. Frequency of defecation should be documented in the hospital notes and laxatives administered if required.
Stress reduction should be considered with any cat in hospital, especially the inappetent cat. Strategies to reduce stress include noise reduction, gentle handling and provision of comfortable bedding, clean litter and somewhere to hide (see later).

Nutritional assessment of the hospitalised cat
Nutritional status should be measured as the fifth vital assessment (after temperature, pulse, respiration and pain assessment) and should be evaluated in every patient. 73 This is of particular importance in critically ill and hospitalised patients,3,5 which are at high risk of inadequate food intake. 7 Such an assessment facilitates identification of cats with, or at risk of, malnutrition and those in need of nutritional support. Nutritional assessment will enable informed decision-making on choice of diet, the need for, and type of, assisted feeding required and which patients will benefit from early intervention. Evaluating a patient’s nutritional status is challenging, but in practical terms will include assessment of the cat’s weight, and body condition and muscle condition scores, as well as a full physical examination and recording of a dietary history. Additional factors that may be included to assess the risk of malnutrition include the presence of hypoalbuminaemia, severe vomiting and/or diarrhoea, and the predicted duration and severity of illness. Patients may then be designated as low, moderate or high risk (see box on page 620). 5


(a) Body condition score chart and (b) muscle condition score chart. Images from the Global Nutrition Committee Toolkit provided courtesy of the World Small Animal Veterinary Association (WSAVA). Available at the WSAVA Global Nutrition Committee Nutritional Toolkit website: http://www.wsava.org/nutrition-toolkit. Accessed 20 October 2021.© WSAVA (image a) and Tufts University, 2014 (image b)
On the basis of the nutritional assessment, cats with malnutrition can be promptly supported, and those at risk of malnutrition identified to allow early intervention, which can result in more positive outcomes. 5 Nutritional assessments should be repeated during hospitalisation, as the patient’s clinical status, appetite and other factors alter over time and with treatment. 75 The feeding plan can then be adjusted accordingly.
Nutritional support of the hospitalised cat: optimising the clinic environment to encourage voluntary food intake
Caring for cats appropriately means first understanding what a cat is, why it behaves the way it does and how that may impact efforts to meet its welfare needs in the clinic, including successfully providing nutrition.
Pet cats retain many of the characteristics of their wildcat ancestors, which are considered solitary survivalists. Cats are territorial, obligately carnivorous and, while they can be social with their own (and other) species, they are selectively so. Territoriality is a very important consideration in pet cats, as they are ‘place bonded’. 76 Many cats do not cope well with change, having to share their territory with unknown cats, or other stressors. 77 Cats need free and immediate access to their resources, and a sense of autonomy is also integral to feline wellbeing. Communication may be subtle and different from that recognised in more socially obligate species, and is aimed at maintaining personal and resource safety in a non-contact/non-confrontational way. 78
The predatory nature of cats, and how and where they acquire and consume food (see box), are important considerations in cat friendly feeding.
Feline senses
Cats’ special senses are integral to feline behaviour, including their communication. Cats have highly attuned vision, hearing and olfaction, as well as tactile capability (particularly via their pads and vibrissae; Figure 5), all of which have developed to create proficiency in hunting and survival. Being in the clinic setting can overwhelm these special senses and cause distress, as the environment can be intense, unfamiliar and signal danger, with no option to escape. Understanding these species-specific characteristics, as well as providing for individual cats’ needs within the clinic setting, is vital to ensure mental wellbeing, which is inextricably linked to both physical health and appetite.

Cats have highly attuned senses, including vision, hearing, olfaction and touch (pads and vibrissae), all of which should be considered when managing hospitalised patients. Image courtesy of Sam Taylor

Stress and distress
Distress refers to an unpleasant emotional experience that is deleterious to mental well-being. Distress may encompass anxiety (anticipation of an aversive event), fear (including perceived threats), frustration (inability to attain something wanted or needed, or escape from something aversive), as well as the negative emotional component of physical pain. 78 Emotions are not mutually exclusive, and an anxious or fearful cat may also be feeling frustrated if, say, they cannot find a place of safety, or prevent physical contact in a handling situation. Frustration increases the vigour of behaviour (often leading to dangerous situations), as cats try to find ways to gain or avoid specific outcomes.
Engaging behaviours (seeking desired outcomes, such as social or tactile interaction) are often seen in positive emotional states, whereas negative emotions tend to correlate with protective behaviours (eg, aggression, hiding). Emotional arousal level is also important to monitor, as patients in states of high arousal may be less inclined to prioritise food intake. Emotional distress and physio-logical stress may occur in tandem, making it difficult to untangle clinical (physiological and behavioural) signs such as tachycardia or hyperglycaemia, and also significantly affecting mental wellbeing, food intake and therefore recovery from illness.4,77,78,84 Figure 6 illustrates potential causes of distress and, in turn, reduced food intake in hospitalised cats.
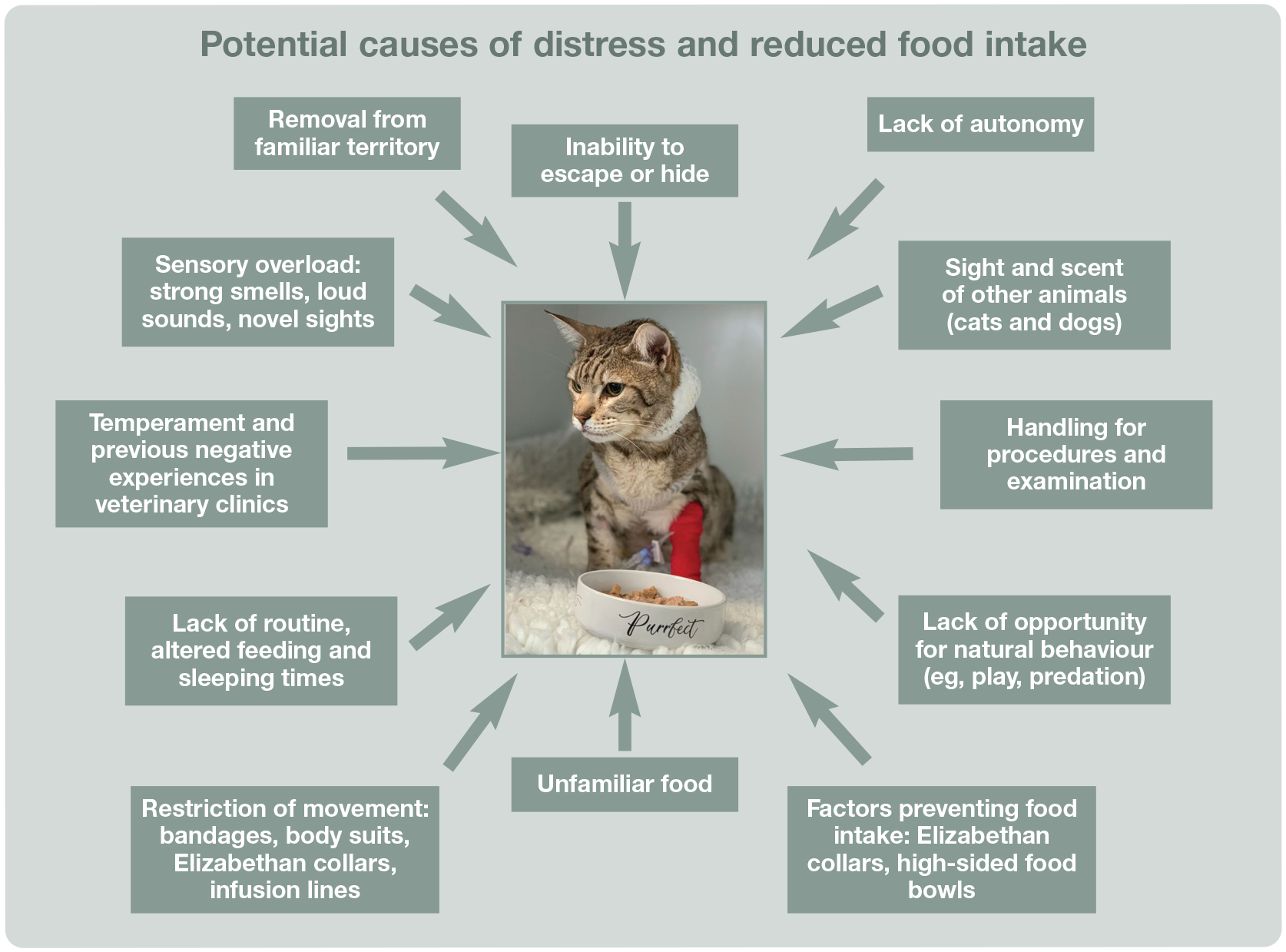
The hospital environment contains multiple factors that can cause distress for cats. Image courtesy of Sam Taylor
Cat friendly strategies for feeding hospitalised patients
A cat friendly ethos should be at the centre of care, aiming to avoid as many environmental and interaction stressors as possible. Solutions are multifaceted – some examples of ways of preventing or alleviating distress in hospital are given in the box on pages 623 and 624.
In general, aim to address the underlying emotional state, where possible, through combined environmental management, human interactions and meeting of cats’ needs.4,78,82,85,88,89 At all times, good nursing and monitoring of a cat’s emotional and physical health, as well as response to ‘realtime interactions’, is needed. Additionally, proactive feeding techniques, such as placement of feeding tubes (see later), may be required to improve the cat’s physical condition and mental wellbeing and, in turn, encourage voluntary food intake. In many cases, the fewer interactions and handling events, the better.


All hospitalised cats should have somewhere to hide, and also perch, within their cage. Even something as simple as a cardboard box will reduce stress and encourage voluntary food intake. (a) A Cat’s Protection ‘Cat Castle’; (b) a cat’s own carrier used as a bed and perch in the hospital cage; (c) a simple cardboard box; (d) the front of the cage can be partially covered with a towel or blanket. Images courtesy of Sam Taylor (a and c), Nicki Reed (b) and Lumbry Park Veterinary Specialists, UK (d)

Cage layout can be optimised to reduce stress and encourage voluntary food intake, ideally by providing somewhere to hide and perch, and allowing food and water to be positioned away from litter trays and each other. Additionally, the front of the cage can be covered to offer more privacy. Image courtesy of Lumbry Park Veterinary Specialists, UK

Wide-brimmed ceramic bowls are preferred to avoid the cat’s whiskers touching the sides of the bowl; this is particularly important for brachycephalic cats. Image courtesy of Linda Ryan

Maximise positive experiences and interactions, if accepted by the cat. This cat particularly enjoyed being groomed and would often start to eat after being brushed in the cage. Image courtesy of Sam Taylor
Nutritional support of the hospitalised cat: appetite stimulants
Appetite stimulants can be helpful in the hospitalised feline patient as a means of maintaining caloric intake. Indications and contraindications for use of appetite stimulants are outlined in the box (for dosing information, see Table 2). It is important to remember that these medications should not replace a diagnostic work-up for the underlying condition, and that conditions that can affect appetite (dehydration, anaemia, nausea, pain, fever, etc) should be addressed.
Currently there are two drugs that are approved for use in cats: mirtazapine (Mirataz; Dechra, USA, Canada and Europe) and capromorelin (Elura; Elanco, USA). Other putative appetite stimulants (steroids [anabolic or corticosteroids], megestrol acetate, propofol, diazepam, B vitamins) have not been assessed for efficacy or have significant side effects that likely outweigh their potential benefits.
Oral mirtazapine
Oral mirtazapine can be an effective appetite stimulant in cats, but higher dosages are more commonly associated with side effects (hyper-excitability, vocalisation, tremors) attributable to serotonin syndrome.49,98 Thus, smaller, more frequent dosages (2 mg/cat q24h in the absence of contraindications) are recommended to maintain efficacy while minimising adverse effects. In young normal cats with no pre-existing liver or kidney disease, the half-life of oral mirtazapine is short enough that it can be administered daily. 98 Although more commonly used as an appetite stimulant, oral mirtazapine also demonstrates antiemetic properties, likely acting at the 5HT3 receptor similar to ondansetron. 99
Mirtazapine is most commonly used in elderly and medically compromised patients, and medical status should be taken into account when prescribing. Due to renal elimination, cats with CKD have significantly longer clearance and higher drug exposure than age-matched controls; 100 thus, a 1.88 mg dose of oral mirtazapine is commonly given q48h in cats with kidney disease. The half-life of oral mirtazapine is also prolonged in cats with liver disease (alanine aminotransferase [ALT] >200 IU/l or total bilirubin >1 mg/ dl). 50 Although there is correlation between mirtazapine half-life and alkaline phosphatase, ALT and total bilirubin concentrations, the alterations in metabolism appear variable and it is challenging to predict which cats would be the most affected. At this time there is no information that indicates that mirtazapine is contraindicated in hospitalised liver or kidney patients, merely that the dose and dosing interval should be carefully considered. When behavioural side effects are noted, a 50% dose decrease is recommended.

Transdermal mirtazapine
Transdermal mirtazapine achieves therapeutic serum concentrations, resulting in appetite stimulation, weight gain and improvement in body condition score in cats.51,52,101,102 A clinical trial performed in 177 cats (83 mirtazapine, 94 placebo) with >5% unintended weight loss demonstrated that 2 mg applied daily to the inner ear pinnae for 14 days resulted in significant weight gain. 102 The most common adverse event was mild erythema at the application site (17.4% placebo, 10.4% mirtazapine). Cats with kidney disease experienced similar therapeutic efficacy without an increase in behavioural side effects (vocalisation, hyperexcitability). Therefore, for most CKD patients, daily dosing appears appropriate with this formulation of the drug.
For clinicians used to using transdermal mirtazapine, it should be noted that the appetite effect after administration may be more subtle in some patients than seen with oral mirtazapine. This is likely due to a flatter drug con centration curve and lower peak serum concentrations, a phenomenon that also results in fewer adverse effects with transdermal administration.52,101 In hospitalised patients, perfusion status and body temperature should be taken into account when determining whether transdermal mirtazapine is appropriate, as poor peripheral perfusion may have an unknown effect on absorption.
Commonly, the hospitalised patient is sent home with appetite stimulant therapy to ease the transition to the home environment and promote caloric intake during the recovery period. Mirtazapine (oral or transdermal) is an effective appetite stimulant in cats with
CKD and results in significantly increased appetite and weight when administered at a dosage of 1.88 mg PO q48h for 3 weeks.51,103 Long-term use is common in patients with chronic disease and anecdotally is not associated with negative repercussions.
Capromorelin
Capromorelin is a ghrelin receptor agonist, and thus acts directly as an orexigenic compound, stimulating appetite in a similar fashion as ghrelin, while also acting to stimulate growth hormone and insulin-like growth factor-1. 57 A clinical trial performed in 112 cats with CKD (71 capromorelin, 41 placebo) with >5% unintended weight loss demonstrated that 2 mg/kg PO q24h for 56 days resulted in significant weight gain. 104 The most common adverse events were vomiting, hypersalivation and lethargy.55,104 Use in cats with acromegaly is contraindicated, and caution should be exercised in patients with diabetes as hyperglycaemia has been documented.56,104 Transient bradycardia and hypotension have also been documented in feline patients. Therefore, it is recommend that this medication not be used in patients with significant systemic compromise, which likely precludes its use in hospitalised patients. 104
Cyproheptadine
Cyproheptadine is not approved for use in cats, but historically has been commonly used as an appetite stimulant, with anecdotal efficacy (1−4 mg/cat q12–24h). 58 No studies have determined what dose alterations should be made in patients with kidney disease (cyproheptadine undergoes some renal elimination), and it is not recommended for use in cats with hepatic lipidosis due to anecdotal concerns for liver failure. The medication may take a few days to reach effective therapeutic levels and the primary adverse effect is sedation, which may preclude its use in hospitalised patients. 58 Cyproheptadine is the recommended antidote for mirtazapine toxicity; therefore, the two drugs should not be used concurrently.

Nutritional support of the hospitalised cat: feeding tubes
Nutritional support can be provided enterally or parenterally – selection of route depends on patient nutritional assessment. Table 3 provides details, including advantages and disadvantages, of different types of feeding tubes.
Advantages and disadvantages of different types of feeding tubes in feline patients

GI = gastrointestinal
Types of feeding tube
Feeding tubes are available commercially for the veterinary market, or human paediatric tubes may be used. Feeding tubes may be made of red rubber, polyvinyl chloride (PVC), polyurethrane or silicone elastomer. Poly-urethrane is generally the preferred material as it is stronger than silicone, allowing the tube to have thinner walls and a wider internal diameter, with less chance of kinking. Silicone percutaneous endoscopic gastrostomy (PEG) tubes may deteriorate before polyurethane, 105 and PVC tubes can stiffen over time and cause irritation. Red rubber feeding tubes are flexible but can become brittle and split, requiring replacement; however, they can be more cost-effective.
Naso-oesophageal and nasogastric feeding tubes
Naso-oesophageal (NO) or nasogastric (NG) feeding tubes are suitable for short-term feeding (generally fewer than 5 days)5,106 in the hospital setting (Figure 11) and when a cat is not a candidate for anaesthesia. They may also be used short term in sick cats to improve clinical condition and allow anaesthesia for longer term tube placement. However, their narrow diameter reduces diet choices to those liquid enough to pass down the tube without obstruction. In terms of selecting NO vs NG tubes, Yu et al (2013) 107 compared the two types of tube feeding in dogs and found no difference in complication rate despite concerns regarding reflux of gastric secretions with NG tubes, and similar findings were reported in a recent study including cats. 108 Gastric reflux into the oesophagus is more likely a concern with larger bore feeding tubes. 109 Nasogastric feeding has been studied in 55 cats with pancreatitis, with minimal complications. 110 NG tubes have the advantage of the option to aspirate residual gastric content, which can be particularly useful in cats with ileus. 5

Naso-oesophageal (NO) feeding tubes. (a) Cat with an NO feeding tube in place. Suitable for short-term feeding and easy to place, this tube is secured with tape and tissue glue. (b) This NO feeding tube is secured with sutures. Note the use of a softer fabric Elizabethan collar to prevent interference, while avoiding the stress associated with hard plastic collars and still allowing the cat to eat. Another method of securing NO tubes is the use of staples. With all methods, the aim should be to avoid patient distress/discomfort and interference with the whiskers or line of vision. Note that nasogastric tubes can be secured similarly. Images courtesy of Sam Taylor (a) and Lindsey Dodd (b)
With careful placement and use, complications of NO or NG tubes are likely to be minor. Obstruction and dislodgement are the most common complications, occurring in 8-13% of cats (see box on page 628).110,111 Epiphora may be noted on the side where the tube has been placed. Vomiting and diarrhoea have been reported in cats fed via such tubes, 111 but may have many contributory factors. Irritation of the nose, self-limiting haemorrhage and sneezing may occur during placement. Gentle, cat friendly handling and light sedation (eg, gabapentin, butorphanol), if required, can reduce stress while placing the tube.


Inadvertent tracheal intubation with resultant pneumonia is a major complication of use of NO/NG tubes, but can be avoided by carefully checking tube position at placement and also before every use, as vomiting between feeds could dislodge the tube into the trachea. 109 Appropriate use of antiemetics (see Table 2) can help to reduce this complication. Placement of an NO or NG tube can be confirmed with thoracic radiography or fluoroscopy at the time of insertion, but given that these tubes tend to be placed in conscious cats, and the requirement for checking correct placement before every feed, the protocols outlined in the box above are recommended.

Cats should be offered food, under supervision, while the NO/NG feeding tube is in place. Removing Elizabethan collars, or using soft fabric versions, may help to encourage voluntary intake. If cats are still not eating adequately voluntarily when the NO or NG tube has been in place for 5 days, consideration should be given to placing a more medium-term tube such as an oesophagostomy (O) tube. Some cats may be deterred from eating by the presence of the tube; hence, tube removal and ‘testing’ of appetite may be needed, with the tube replaced if intake remains inadequate.
Oesophagostomy feeding tubes
O tubes are a useful and well tolerated way to provide enteral nutrition to cats (Figure 12). Placement requires general anaesthesia but, with training, can be rapid. Moreover, these larger bore tubes allow a greater variety of diets and slurries to be fed, and also facilitate water and medication administration. Consideration should be given to placement when the opportunity arises; for example, when cats with current or predicted reduced food intake are anaesthetised for imaging or surgery. O tubes may be used for an extended period of time and can be better tolerated than NO tubes in some patients owing to the lack of fixings on the cat’s face; also they can be used by caregivers at home, with minimal training and care requirements. Elizabethan collars may not be needed, as patient interference can be prevented using dressings or purpose-made fabric collars. In the panel members’ experience, if dressings are kept to a minimum, tension on anchoring sutures is avoided, and the cat is comfortable, interference tends to be minimal.

Oesophagostomy (O) feeding tubes. (a) A polyurethane O tube coiled and kept in place with a purpose-made fabric collar (Kitty Collar). (b) An O tube with additional length removed (note this tube is secured with a finger trap suture to the periosteum of the wing of the atlas, as preferred by some clinicians). (c) A red rubber O tube covered with a dressing. Images courtesy of Sam Taylor (a), Danielle Gunn-Moore (b) and Serge Chalhoub (c)
The most common complications are stoma site infection and tube dislodgement. Two studies of O tubes have shown stoma site infections in 17.8% and 12.1% of cats, respectively,113,114 with Breheny et al (2019) 114 reporting that cats receiving glucocorticoids or oncolytic agents were more likely to develop an infection. Guidance for management of stoma site infections is given in the box on page 631.

Tube migration or dislodgement in cats that are vomiting may occur and resuturing may be required. More serious complications of O tubes include damage to the vascular structures in the neck causing haemorrhage (thought to be a rare occurrence), and tracheal intubation. Other, minor complications include head swelling (often with excessively tight dressings), kinking of the tube and obstruction. A rare complication documented in the literature was Horner’s syndrome and Pourfour du Petit syndrome following assumed injury to the vagosympathetic trunk associated with O tube placement in a cat. 115 Transient Horner’s syndrome alone has been observed by one of the panel members (ST) and also recorded once in the literature. 116 Such complications are likely due to neuropraxia; generally there is spontaneous resolution without specific treatment.
After placement, O tube position is confirmed with thoracic radiography (Figure 13), fluoroscopy or endoscopy. A further check of tube placement is also made prior to every feed to confirm no inadvertent endotracheal intubation (see box on page 629).
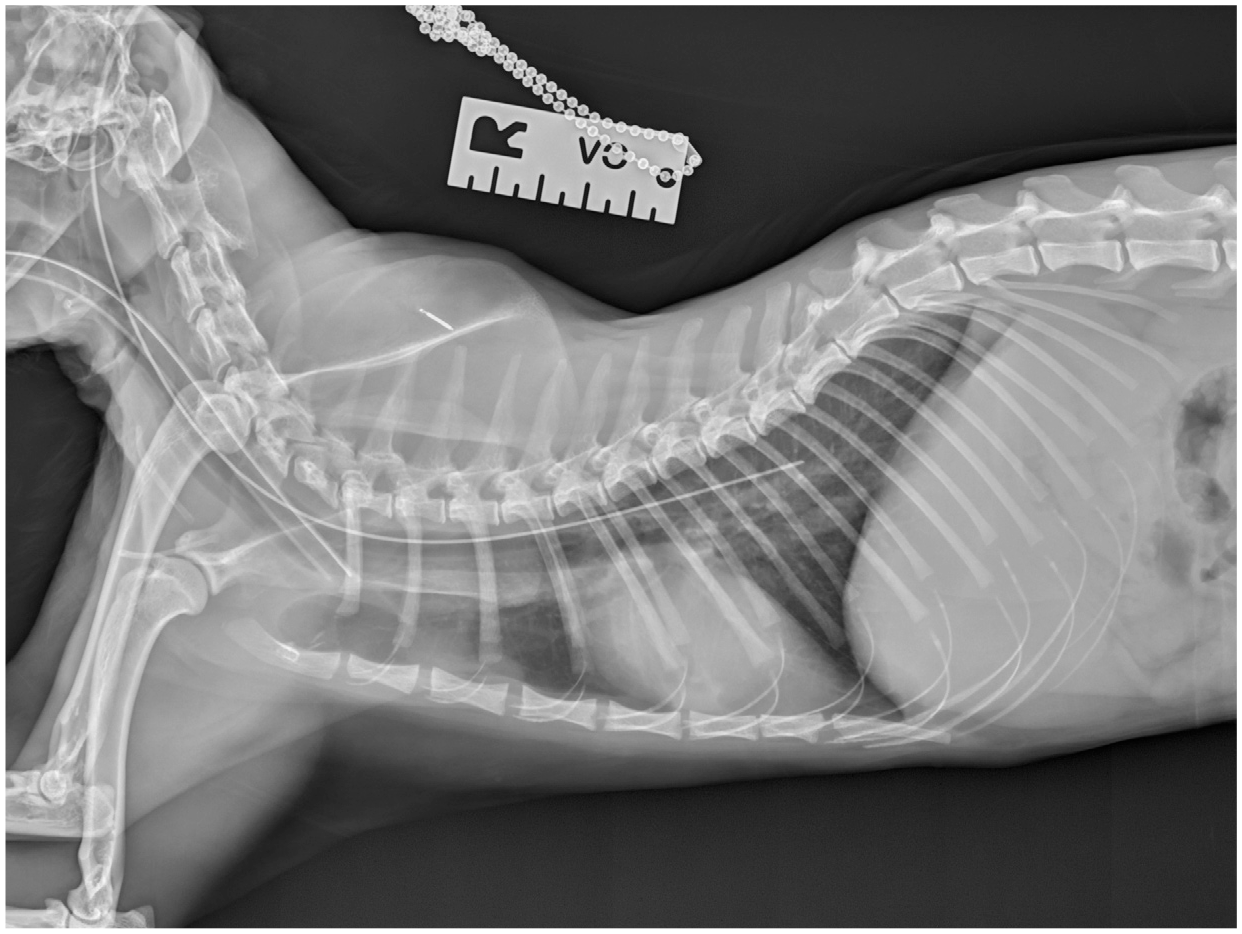
Correct placement of an O tube confirmed with radiography. The tube should terminate in the distal third of the oesophagus. Image courtesy of Sam Taylor


Oesophagostomy tube site cellulitis (a) and abscess (b). Topical antibiotic therapy may be adequate to manage infection, along with increased cleaning of the site. However, infection may necessitate tube removal in some cases, as well as sedation or anaesthesia for surgical debridement/flush. Culture and sensitivity testing should be performed where possible to enable appropriate antibiotic selection. Antibiotic-impregnated discs (c) can be placed around the stoma site. Images courtesy of Sam Taylor (a and c) and Carolyn O’Brien (b)
Cats with O tubes in situ should be encouraged to eat voluntarily by offering food prior to a feed. Intake should be recorded, and the volume fed via the tube adjusted accordingly. Once voluntary intake has been satisfactory (75–100% of RER and adequate for maintenance or gain of weight) for at least 3–5 days, and/or the tube is no longer required, it can be removed by cutting the anchoring sutures, and gently pulling out the tube; sedation or anaesthesia is not required. O tubes can be removed at any time, as there is no need for a delay to allow a seal to form (unlike with gastrostomy tubes). 106 The stoma site should be cleaned and covered with a dressing for 24 h. It will heal by second intention (wound closure is not recommended).
Gastrostomy tubes
Gastrostomy tubes (Figure 15a) can be placed surgically or with endoscopic guidance (PEG tube). They can be used long term (in which case they can be replaced with a low-profile tube [Figure 15b]); their large bore makes them suitable for almost any diet, and they can be used by caregivers at home with minimal training. 117 Salinardi et al (2006) 118 examined complications of PEG and surgically placed gastrostomy tubes and found no difference in complication rates and severity scores between the two. Reported complications include injury to abdominal viscera during tube placement, peritonitis, cellulitis, stoma site infection, obstruction, vomiting and metabolic derangements.118,119 Additionally, pressure necrosis of the stomach wall and pyloric outflow obstruction can occur with overly tight or improperly located tubes, respectively. 119 It appears that treatment with corticosteroids might also increase the rate of complications for cats with a PEG tube. In a study by Aguiar et al (2016), 120 the rate of severe complications associated with PEG tubes (in a population of both dogs and cats) was higher in the corticosteroid-treated group.

Gastrostomy feeding tubes. (a) This cat was diagnosed with oesophagitis and was in poor body condition and benefited from placement of a gastrostomy feeding tube at the time of surgery for intestinal biopsy. Dressing of gastrostomy tubes can be challenging as dressings tend to ‘bunch’ cranially to the hindlimbs. Body suits may be tolerated by some cats, but care must be taken to avoid chronic stress associated with dressings/ clothing while the tube is in place. (b) Cat with idiopathic megaoesophagus and a low-profile PEG tube. This cat was fed via the tube for 7 years. Images courtesy of Sam Taylor (a) and Elise Robertson (b)

When patients have both feeding tubes and IV lines, particularly central venous catheters, ensure they are appropriately labelled. Image courtesy of Dan Chan
Gastrostomy tubes cannot be removed for at least 10–14 days post-placement to allow a seal to form at the gastrostomy site. 119 Depending on the tube type, the mushroom tip may need to be removed endoscopically when the tube is cut, to prevent intestinal obstruction, although some types can be removed using gentle traction. As described above for O tubes, voluntary food intake should be adequate for 3–5 days prior to removal to ensure the tube is no longer required.
Jejunostomy and nasojejunal tubes
These types of tube are rarely placed in feline patients. Despite a suggestion that bypassing the stomach and pancreas may be beneficial in the management of pancreatitis, gastric outflow obstruction or gastroparesis, 121 research in humans with pancreatitis suggests that jejunal (or parenteral) feeding is not associated with a better outcome than NG tube feeding. 122 There has been little research in cats, but the same is likely to be true. Reported complications associated with jejunostomy tube use include cellulitis, tube dislodgement and migration, vomiting, dehiscence, peritonitis and tube obstruction. 123
Parenteral nutrition
Parenteral nutrition (infusing nutrients intravenously) can be used as an alternative in patients unable to tolerate enteral feeding; for example, those with intractable vomiting and diarrhoea, lack of gag reflex, presenting a high anaesthetic risk or recovering from severe gastric or intestinal disease and coagulopathy. Parenteral nutrition may be associated with a higher rate of complications than tube feeding, 74 is more expensive and limited in availability. For these reasons – as well as on a physiological level (see page 633) – enteral feeding remains preferable in the majority of cases. However, safe administration of parenteral nutrition is possible and readers are referred to other sources for further details. 18
Nutritional support of the hospitalised cat: creating a practical feeding plan
When to intervene
Given the metabolic and pathological changes that occur in cats following prolonged fasting, 31 nutritional interventions (eg, placement of feeding tubes or initiating parenteral nutrition if the enteral route is contraindicated) should be implemented no later than 3 days after the cessation of eating. When cats are presented for veterinary assessment of inappetence, it is important to take into consideration the number of days without normal food intake at home that have already elapsed, in addition to the number of days they have consumed less than the RER while hospitalised.



Before the implementation of a feeding plan, cats should have been rehydrated, have had any major electrolyte and acid-base disturbances addressed and be cardiovascularly stable. Maintenance of normotension is desirable prior to enteral feeding in humans, 124 as enteric vasodilation could further reduce systemic blood pressure, and similar is likely the case in feline patients. See the box above for considerations influencing choice of diet.
Which route to use
Feeding into the GI tract will have the most benefit to the patient and so enteral nutrition should be pursued in most cases, unless there are apparent contraindications. 5 Enteral nutrition stimulates Gl motility, enteric blood flow and local immunity, activates neuro-endocrine pathways, promotes a favourable micro bio ta, reduces oxidative stress and imparts trophic effects on mucosal epithelium, thereby supporting enteric barrier function. 125 Even when patients can only tolerate a portion of the energy needs enterally, this route should be used as many of the benefits of enteral nutrition can still be realised. 126 Other factors to consider include the cat’s medical condition, anticipated duration of support, and advantages and disadvantages of each tube (Table 3). In the event that cats show no tolerance to enteral feeding (eg, persistent vomiting despite antiemetic therapy), parenteral nutrition should be considered.
How much to feed
The primary purpose of nutritional support is to stabilise the patient’s nutritional status rather than replenish lost body condition due to illness. Therefore, initial daily nutritional targets should be conservative. The aim should be to meet the RER within 3 days of initiating nutritional support, if tolerated by the patient (see box on page 634); for example, feed one-third RER on day 1 and, in the absence of adverse effects, increase to two-thirds on day 2. 5 In more debilitated patients or in the presence of significant morbidity (eg, severe GI dysfunction, major electrolyte and nutrient deficienrcies, severe starvation), a more conservative approach may be necessary and meeting RER may take several additional days. 127 Following stabilisation of nutritional status (ie, stopping further weight loss) and when the cat is in recovery from the primary disease, energy targets can be increased to restore normal body condition.
In growing animals, once stabilised, energy targets will need to exceed RER – kittens may require 200% RER to support growth. Cats recovering from particularly catabolic conditions (eg, thermal burns, large exudative wounds) may also have energy targets that far exceed RER (ie, >200%). 128 However, as mentioned, overfeeding is associated with complications, so increases should be made slowly and only once the cat is stable.
How to determine a feeding schedule
Generally, patients that are beginning to eat should have small but frequent meals, and this holds true for those being tube fed. Most animals being fed via feeding tubes can tolerate meals as boluses over 10–15 mins several times a day (eg, 3–6 meals per day). The decision regarding the frequency of feeding depends on patient factors (eg, tolerance to feeding volume, calories required) as well as practical considerations (eg, nursing care availability overnight, ability to use feeding pumps). Some patients may tolerate enteral feeding delivered as a constant rate infusion (CRI; Figure 17) better than bolus feeding (eg, less vomiting or regurgitation). There is currently no evidence supporting one modality over the other, so this needs to be determined on an individual basis. 129 Patients receiving a CRI should be positioned upright or with their head/neck supported, to avoid regurgitation.

Some patients with severe vomiting or regurgitation, for example, may benefit from being fed via a constant rate infusion, which can be facilitated with standard syringe drivers. Image courtesy of Dan Chan

Once feeding the full RER (as above, enteral nutrition should be introduced over several days), and provided there are no complications, the total maximum volume per bolus feed for a cat ranges between 5 and 15 ml/kg; if feeding via CRI, rates between 3 and 8 ml/h are generally well tolerated. Figure 18 illustrates a recording sheet for calculation of RER and feeding requirements, as well as for recording feeds, and is designed to complement general hospital records for cats receiving assisted nutrition.

Example feeding record for a hospitalised patient with a feeding tube. This record sheet is available to download from the supplementary material (see page 636)
What to monitor in patients with feeding tubes
Tips for cat friendly tube feeding are given in the box on page 635. Patients should not experience any distress (gagging, coughing, retching, vocalising, struggling) during tube feeding; if any distress is noted, feeding should stop immediately, and the feeding tube checked for dislodgement. For O and gastrostomy tubes, the stoma site should be assessed for signs of infection. Body weight, hydration status and vital signs should be checked daily; body condition should be evaluated weekly. Patients should be monitored for signs of overhydration (eg, chemosis, gelatinous skin turgor, clear nasal discharge, increased respiratory rate, crackles on thoracic auscultation), since tube feeding and flushing of feeding tubes after each meal will likely mean daily fluid requirements are exceeded and can therefore lead to fluid overload in cats predisposed to this complication (eg, cardiac disease). This is a particular concern in patients concurrently receiving intravenous fluid therapy, which should be adjusted accordingly.
Hospitalised cats with feeding tubes that are able to eat orally should be offered food prior to feeding to encourage voluntary intake. Using soft fabric Elizabethan collars (as pictured in Figure lib) or removing them and monitoring the cat, in combination with other techniques to reduce stress (see box on pages 623 and 624), may help encourage eating.


Cats should be allowed to adopt a comfortable position when being tube fed, avoiding stress and heavy restraint. This cat is relaxed, accepting being stroked and shows no sign of nausea or pain while receiving an O tube feed. A bed has been placed under the cat’s head to raise it slightly to avoid reflux of food proximally. Image courtesy of Sam Taylor

Refeeding syndrome
Occasionally, severely debilitated cats can develop a life-threatening metabolic condition termed ‘refeeding syndrome’ if fed in excess of what they can process.127,130 Refeeding syndrome has been reported to occur even after short periods of starvation in human patients; 131 however, in a recent feline case series, affected cats had been missing (presumed starved) for more than 3 weeks. 127
This syndrome is characterised by neurological signs (eg, depression, coma), systemic weakness, haemolytic anaemia, glycaemic dysregulation and severe electrolyte abnormalities (eg, hypokalaemia, hypophosphataemia, hypomagnesaemia) due to insulin-driven glucose uptake. Thiamine deficiency may also contribute to neurological signs. 127
If refeeding syndrome is suspected, feeding should cease until the patient is stabilised and the feeding plan adjusted to avoid worsening of this syndrome. Identification of at-risk patients is important and includes cats that have been missing, had a complete lack of food intake, are severely debilitated with significant body condition loss (Figure 20) or those with electrolyte abnormalities prior to commencing feeding. Prevention of refeeding syndrome in such patients should include feeding no more than 20% RER on day 1 and slowly increasing the proportion of RER over 4–10 days depending on clinical response. Empirical supplementation of phosphate at 0.01-0.03 mmol/kg/h, potassium at 0.05 mEq/kg/h and magnesium at 0.01–0.02 mEq/kg/h for the first 24 h of therapy, provided the patient does not have electrolyte values above the reference interval, is recommended. 130 Thiamine can be administered prior to feeding (25 mg total dose, SC or IM) and repeated daily until signs resolve. 130

Severely debilitated cats and those that have been starved (typically after going missing) are at risk of refeeding syndrome. This cat had a body condition score of 1/5 after going missing for several weeks. Image courtesy of Sam Taylor
Summary Points
✜ The 2022 ISFM Consensus Guidelines on Management of the Inappetent Hospitalised Cat provide practical information regarding the frequently observed complication of suboptimal nutrition in hospitalised feline patients, with reach to those managed at home.
✜ Treatment of inappetence should consider the unique metabolism of the feline species and its susceptibility to stress and malnutrition.
✜ Prompt intervention, with feeding tubes if required, can optimise recovery of inappetent patients and expedite discharge from the hospital.
Supplemental Material
sj-docx-1-jfm-10.1177_1098612X221106353 – Supplemental material for 2022 ISFM Consensus Guidelines on Management of the Inappetent Hospitalised Cat
Supplemental material, sj-docx-1-jfm-10.1177_1098612X221106353 for 2022 ISFM Consensus Guidelines on Management of the Inappetent Hospitalised Cat by Samantha Taylor, Daniel L Chan, Cecilia Villaverde, Linda Ryan, Franck Peron, Jessica Quimby, Carolyn O’Brien and Serge Chalhoub in Journal of Feline Medicine and Surgery
Supplemental Material
sj-docx-2-jfm-10.1177_1098612X221106353 – Supplemental material for 2022 ISFM Consensus Guidelines on Management of the Inappetent Hospitalised Cat
Supplemental material, sj-docx-2-jfm-10.1177_1098612X221106353 for 2022 ISFM Consensus Guidelines on Management of the Inappetent Hospitalised Cat by Samantha Taylor, Daniel L Chan, Cecilia Villaverde, Linda Ryan, Franck Peron, Jessica Quimby, Carolyn O’Brien and Serge Chalhoub in Journal of Feline Medicine and Surgery
Supplemental Material
sj-docx-3-jfm-10.1177_1098612X221106353 – Supplemental material for 2022 ISFM Consensus Guidelines on Management of the Inappetent Hospitalised Cat
Supplemental material, sj-docx-3-jfm-10.1177_1098612X221106353 for 2022 ISFM Consensus Guidelines on Management of the Inappetent Hospitalised Cat by Samantha Taylor, Daniel L Chan, Cecilia Villaverde, Linda Ryan, Franck Peron, Jessica Quimby, Carolyn O’Brien and Serge Chalhoub in Journal of Feline Medicine and Surgery
Supplemental Material
sj-pdf-4-jfm-10.1177_1098612X221106353 – Supplemental material for 2022 ISFM Consensus Guidelines on Management of the Inappetent Hospitalised Cat
Supplemental material, sj-pdf-4-jfm-10.1177_1098612X221106353 for 2022 ISFM Consensus Guidelines on Management of the Inappetent Hospitalised Cat by Samantha Taylor, Daniel L Chan, Cecilia Villaverde, Linda Ryan, Franck Peron, Jessica Quimby, Carolyn O’Brien and Serge Chalhoub in Journal of Feline Medicine and Surgery
Supplemental Material
sj-pdf-5-jfm-10.1177_1098612X221106353 – Supplemental material for 2022 ISFM Consensus Guidelines on Management of the Inappetent Hospitalised Cat
Supplemental material, sj-pdf-5-jfm-10.1177_1098612X221106353 for 2022 ISFM Consensus Guidelines on Management of the Inappetent Hospitalised Cat by Samantha Taylor, Daniel L Chan, Cecilia Villaverde, Linda Ryan, Franck Peron, Jessica Quimby, Carolyn O’Brien and Serge Chalhoub in Journal of Feline Medicine and Surgery
Footnotes
Appendix
A guide to placement of a nasooesophageal or nasogastric tube in a cat https://youtu.be/-WfuE8djYos
A guide to placement of an oesophagostomy tube in a cat https://youtu.be/MiNvX2pF6to
A guide to caring for a cat with an oesophagostomy tube https://youtu.be/UsLcTZ8u8Gk
Two videos on feeding tube placement and management, and a video to support clients caring for cats with a feeding tube, can be downloaded from the above links, and are also available as supplementary material at bit.ly/inappetentcattoolkit
Acknowledgements
ISFM is grateful to Royal Canin and Linneaus for contributing to the creation of the feeding tube videos, and to the veterinary clinics involved in the filming. ISFM also thanks Marge Chandler for review of the manuscript.
Supplementary material
✜ Example dietary history questionnaire for caregivers.
✜ Example feeding record for a hospitalised patient with a feeding tube.
Conflict of interest
Franck Peron is an employee of Royal Canin. Jessica Quimby is a consultant for a variety of companies including Dechra, Elanco, Boehringer Ingelheim, Vetoquinol, Zoetis, Purina, Hill’s and Royal Canin. Sam Taylor has worked for various pet food and pharmaceutical companies on a consultancy basis. Cecilia Villaverde has done consulting work for a variety of pet food companies. She develops educational materials for Mark Morris Institute, sits on the scientific advisory board of FEDIAF and is a member of the Global Nutrition Committee of the WSAVA. She participates as a speaker in continuing education events sponsored or organised by pet food companies. The other members of the panel have no conflicts of interest to declare.
Funding
The authors received no financial support for the research, authorship, and/or publication of these guidelines.
Ethical approval
This work did not involve the use of animals and, therefore, ethical approval was not specifically required for publication in JFMS.
Informed consent
This work did not involve the use of animals (including cadavers) and, therefore, informed consent was not required. For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
