Abstract
Case series summary
Cats with non-erosive immune-mediated polyarthritis (IMPA) were identified from seven referral hospitals between 2009 and 2020 for a multicentre retrospective case series. Data were obtained from hospital records and referring veterinarians were contacted for follow-up. Twenty cases were identified: 12 castrated males (60%), one entire male (5%) and seven spayed females (35%). Common clinical signs included lameness (n = 20/20) and pyrexia (n = 10/18). Three cats presented with and two cats developed ligament laxity during treatment. Thirteen cats (65%) were diagnosed with non-associative IMPA and seven (35%) with associative IMPA. Comorbidities identified included chronic enteropathy (n = 3/7), feline immunodeficiency virus (n = 1/7), feline herpesvirus (n = 1/7), bronchopneumonia (n = 1/7) and discospondylitis (n = 1/7). Sampling of the tarsal joints most frequently identified an increased proportion of neutrophils, consistent with IMPA. Eighteen cats (90%) received immunosuppressants. Eleven cats were started on prednisolone; eight had a poor response resulting in the addition of a second agent, euthanasia or acceptance of the persisting signs. One cat received ciclosporin and required an alternative second agent owing to adverse effects. Five cats were started on prednisolone and ciclosporin; three had a poor response and required an alternative second agent. One cat received prednisolone and chlorambucil and had a good response. Two cats (10%) received meloxicam and had a good response, although the clinical signs recurred when medication was tapered. A good outcome was achieved in 14/20 cats (70%) with IMPA. In the cats with a poor outcome 4/6 were euthanased and 2/6 had chronic lameness.
Relevance and novel information
Prognosis for feline IMPA can be good. Multimodal immunosuppression was often required. IMPA should be considered in lame cats, with or without pyrexia, when there is no evidence of trauma or infection. The tarsal joints should be included in the multiple joints chosen for sampling. Ligament laxity can occur in non-erosive feline IMPA.
Keywords
Introduction
Compared with dogs, immune-mediated polyarthritis (IMPA) is a rarely reported condition in cats. 1 It is diagnosed based on the identification of sterile, neutrophilic inflammation in multiple joints, and is defined as erosive or non-erosive based on radiographic findings. 2 IMPA is caused by an aberrant immune response, either targeted at the joints directly or occurring as a result of the deposition of circulating immune complexes within the joints. 1 Non-erosive IMPA can be split into two main subcategories: associative and non-associative. The terms associative and non-associative address the limitations of previous categorisation models as they reflect the spectrum of disease and our incomplete understanding of the pathogenesis of immune-mediated disorders. 3 Cases in which significant comorbidities are identified during investigations are categorised as associative. In associative IMPA, it is considered possible that the comorbidity identified is the underlying cause. However, the comorbidity could be incidental and not the primary cause of IMPA. Cases in which no comorbidities are identified are categorised as non-associative IMPA. In non-associative IMPA, although no comorbidity has been identified in the investigations performed, it does not exclude the possibility of an underlying cause being present that was either not understood or not detected in the investigations performed. IMPA has previously been associated with pneumonia, pyelonephritis, toxoplasmosis, feline leukaemia virus (FeLV), feline infectious peritonitis and myelodysplastic disorders.4,5
There is little published information about the clinical findings in cats with non-erosive IMPA. To our knowledge, at the time of writing, there has only been one previous case series, which included 13 cats with non-erosive IMPA. The cats in that study were identified at two referral centres in the UK between 1977 and 1986. 4
The aims of this case series were to describe the clinical features of associative and non-associative IMPA in cats, as well as the investigations performed, treatment and outcome.
Case series description
Ethical approval was granted by the University of Bristol Animal Welfare and Ethics Review Body (veterinary investigation number 18/068), the Royal Veterinary College Social Sciences Research Ethical Review Board (reference URN SR2019-0482) and the University of Liverpool Veterinary Research Ethics Committee (reference VREC736).
Cases were recruited from seven referral hospitals in the UK from January 2009 to April 2020. Electronic medical records were searched to identify cats diagnosed with IMPA. Cats were included for analysis if they met the following criteria: complete clinical records; no evidence of erosive lesions on CT and/or radiographs of at least one affected joint; an increased proportion of non-degenerate neutrophils (>10% of nucleated cells present) in the synovial fluid of two or more joints with no evidence of intracellular or extracellular bacteria; and a minimum of 2 months of follow-up. 1 Twenty cats met the inclusion criteria.
The following details were extracted from the medical records of all cats: signalment; presenting clinical signs; onset and duration of clinical signs; treatment prior to referral; initial physical examination findings; results of diagnostic investigations; final diagnosis; treatment; and outcome.
Information regarding outcome was recorded from referral hospital follow-up appointments and/or communication records or via telephone updates with the primary care practice. If a patient had died, the reason for euthanasia or cause of death was recorded. The outcome was considered good if clinical signs of IMPA were completely controlled while on medication without significant side effects, if medications could be tapered without a relapse in clinical signs, if death occurred for reasons unrelated to IMPA or if a relapse occurred but was successfully managed with treatment and clinical signs resolved. Outcome was considered poor in cats in which clinical signs of IMPA were not able to be controlled with treatment, or if death was considered to be related to IMPA or treatment. Relapse was defined as a recurrence in clinical signs relating to IMPA.
Statistical analysis was performed using IBM SPSS version 26. Normality was assessed for continuous variables with the Shapiro–Wilk test. Results were reported as mean ± SD if normally distributed and as median (range) if they were not.
Signalment
The mean age of the included cats was 8.8 ± 4.8 years. Median body weight was 4 kg (range 2.11–8.8). The most common breed was domestic (n = 13 cats [65%]; 11 [55%] domestic shorthairs and two [10%] domestic longhairs). The remaining cats included three Maine Coons (15%) and one each (5%) of the following: Ragdoll, Siamese, Persian cross and Bengal cross. There was a male predominance, with 12 castrated males (60%), one entire male (5%) and seven spayed females (35%). As the breed and sex distribution for each hospital included in this study was unknown, it is not possible to confirm that males or Maine Coon cats were significantly over-represented.
History and treatment before referral
A summary of presenting signs is shown in Table 1. Twelve cats (60%) had acute onset of clinical signs (<24 h) and the remaining eight (40%) had a more insidious onset of signs (range 2 weeks to 1 year). The most common presenting sign was lameness (reported in all cats). In seven cats (35%), the lameness was present in a single limb and three cats (15%) were non-ambulatory. Rectal temperature at the onset of clinical signs was recorded in 18 cases. Ten cats (55%) were pyrexic (temperature >39.2°C), 6 seven (39%) were normothermic and one (6%) was hypothermic.
Summary of presenting clinical signs in 20 cats with immune-mediated polyarthritis

Treatment prior to referral was highly variable. Eighteen cats received treatment prior to referral; treatment data were unavailable for one cat and one cat was referred without any treatment other than fluid resuscitation. The most common treatments were non-steroidal anti-inflammatory drugs (NSAIDs), administered to 13 cats (68%) and antimicrobials administered to 11 cats (58%). Opioid analgesia was administered to nine cats (47%). Corticosteroids were administered to three (16%), gastroprotectants to one (5%) and codeine to one (5%).
Diagnostic tests
Complete blood count (CBC), serum biochemistry, joint fluid cytology, abdominal imaging and joint imaging were performed in every cat. Thoracic imaging was performed in 18/20 cats. Other specific diagnostic tests are documented in Table 2 and the imaging modalities used are summarised in Table 3.
Diagnostic tests performed in 20 cats diagnosed with immune-mediated polyarthritis

Data are n (%)
CBC = complete blood count; FIV= feline immunodeficiency virus, FeLV = feline leukaemia virus; ANA = antinuclear antibody
Diagnostic imaging performed in 20 cats diagnosed with immune-mediated polyarthritis

Data are n (%)
NA = not applicable
The most common abnormal findings on CBC were neutrophilia in 10 cats (six non-associative and four associative) and lymphopenia in six cats (four non-associative and two associative); three cats (two non-associative and one associative) had a mild anaemia (packed cell volume >20% and ⩽24%, respectively; reference interval [RI] 24–45. The most common serum biochemistry abnormalities were hyperglobulinemia in six (five non-associative and one associative) and hypoalbuminemia in five (three non-associative and two associative). Of the six cats with hyperglobulinemia, the elevation was mild (46 to ⩽50 g/l) in four, moderate (50 to <55 g/l) in one and severe (>55 g/l) in one (RI 25–45). Of the five cats with hypoalbuminemia, it was mild (20 to ⩽26 g/l) in four and moderate (15 to <20 g/l) in one (RI 25–45).
Case classification
Cats were split into two subcategories of IMPA, based on the final diagnosis made by the clinician: associative or non-associative. Thirteen cats (65%) were presumed to have non-associative IMPA. Comorbidities were identified in the remaining seven cats (35%), categorised as associative IMPA. The comorbidities identified in the seven cats with associative IMPA included gastrointestinal disease (n = 3), feline immunodeficiency virus (FIV) (n = 1), chronic feline herpesvirus (n = 1), bacterial bronchopneumonia (n = 1) and discospondylitis (n = 1).
Joints affected
All cats had an increased proportion of non-degenerate neutrophils identified on cytology of ⩾2 joints. Neutrophils made up >10% of the total nucleated cell count on cytology in all affected joints. 1 No infectious organisms were identified on cytology or culture of any joint aspirates; however, only nine cats had joint fluid culture performed. Total nucleated cell counts were not available for all samples owing to the small volume of synovial fluid obtained in most cases.
The median number of joints sampled was four (range 2–10). The joints sampled most commonly were the carpus (15 cats; 75%), stifle (15 cats; 75%) and tarsus (14 cats; 70%). The elbow was sampled in eight cats (40%) and the shoulder was sampled in one cat (5%). All tarsal joints that were aspirated had an increased proportion of non-degenerate neutrophils identified. The proportion of joint aspirates with findings suggestive of IMPA (neutrophils >10% of the nucleated cell count and no intra- or extracellular bacteria) obtained in this cohort is outlined in Figure 1.
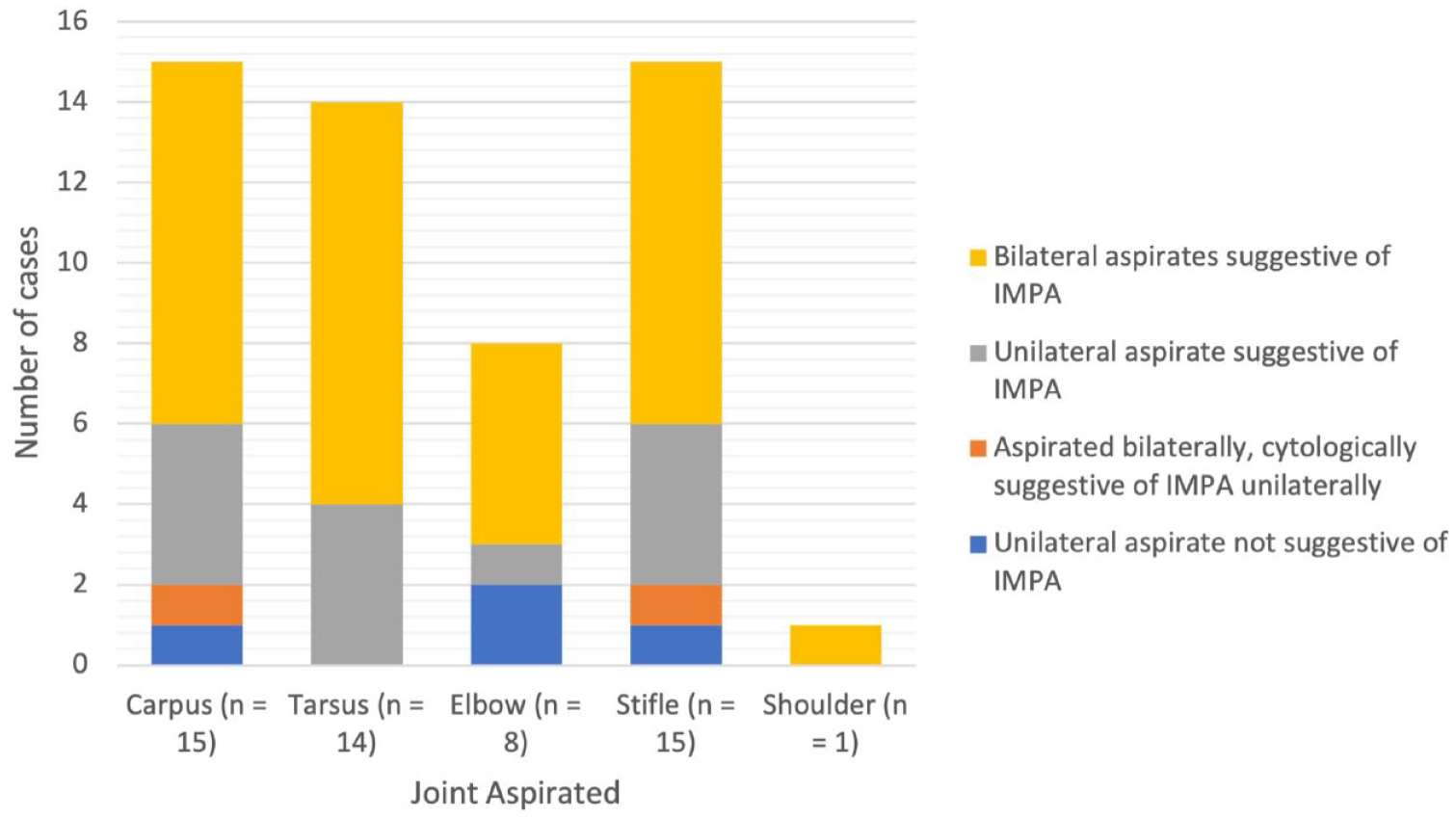
Proportion of joint aspirates yielding synovial fluid samples cytologically suggestive (neutrophils >10% of the nucleated cell count and no intra- or extracellular bacteria) of immune-mediated polyarthritis (IMPA) in 20 cats subsequently diagnosed with IMPA based on the involvement of ⩾2 joints
Three cats (15%) presented with ligament laxity and two (10%) developed ligament laxity 5 months and 12 months into treatment with corticosteroids. Three of the five cats had tarsal joint laxity resulting in a plantigrade stance, and two cats had bilateral carpal laxity and a palmigrade stance (Figure 2). Radiographs were obtained in 4/5 cats after the development of joint laxity and there were no erosive lesions in any of the affected joints. The timing of joint imaging in relation to the onset of joint laxity was variable (2 weeks, 5 weeks, 1 month, and 3 months, respectively). As it can take several weeks for erosive lesions to become visible on radiographs, it is not possible to definitively prove erosive lesions did not develop in the cats imaged. Two cats developed joint laxity while on prednisolone after 5 months and 12 months of treatment, respectively. It is not possible to identify whether the IMPA or prednisolone therapy was the cause of the joint laxity in these cases. However, the cat which developed laxity 5 months into treatment had a chronic 1-year history of lameness without ligament laxity prior to diagnosis, suggesting that prednisolone is more likely to have been the cause of the ligament laxity in this cat. The other cat had a 3-week history of lameness prior to diagnosis and developed ligament laxity after 12 months of prednisolone. The three cats which presented with joint laxity had an acute onset of signs and none had received corticosteroids prior to presentation. No cat underwent surgical arthrodesis. A differential diagnosis for a plantigrade stance in a cat is tibial nerve neuropathy and diabetic neuropathy. None of the three cats were diabetic. Tibial nerve neuropathy was excluded in one cat with evidence of subluxation of the calcaneoquartal joint on CT; the second cat had an unremarkable examination performed by a neurologist and the third cat had excessive range of motion detected on orthopaedic examination.

Carpal hyperextension due to joint laxity in a cat with non-erosive immune-mediated polyarthritis
Infectious disease testing
Infectious disease screening was variable between cases and was dependent on the clinician’s decision. Fourteen of the 20 cats (70%) either tested negative for Mycoplasma species on PCR of joint fluid (n = 10) or received a 2-week course of oral doxycycline without being tested (n = 4). FIV and FeLV testing was performed in 14 (70%) cats; one cat in this case series was positive for FIV infection. The diagnosis of FIV was confirmed with proviral DNA PCR testing in addition to a positive SNAP FIV antibody test.
Treatment and outcome
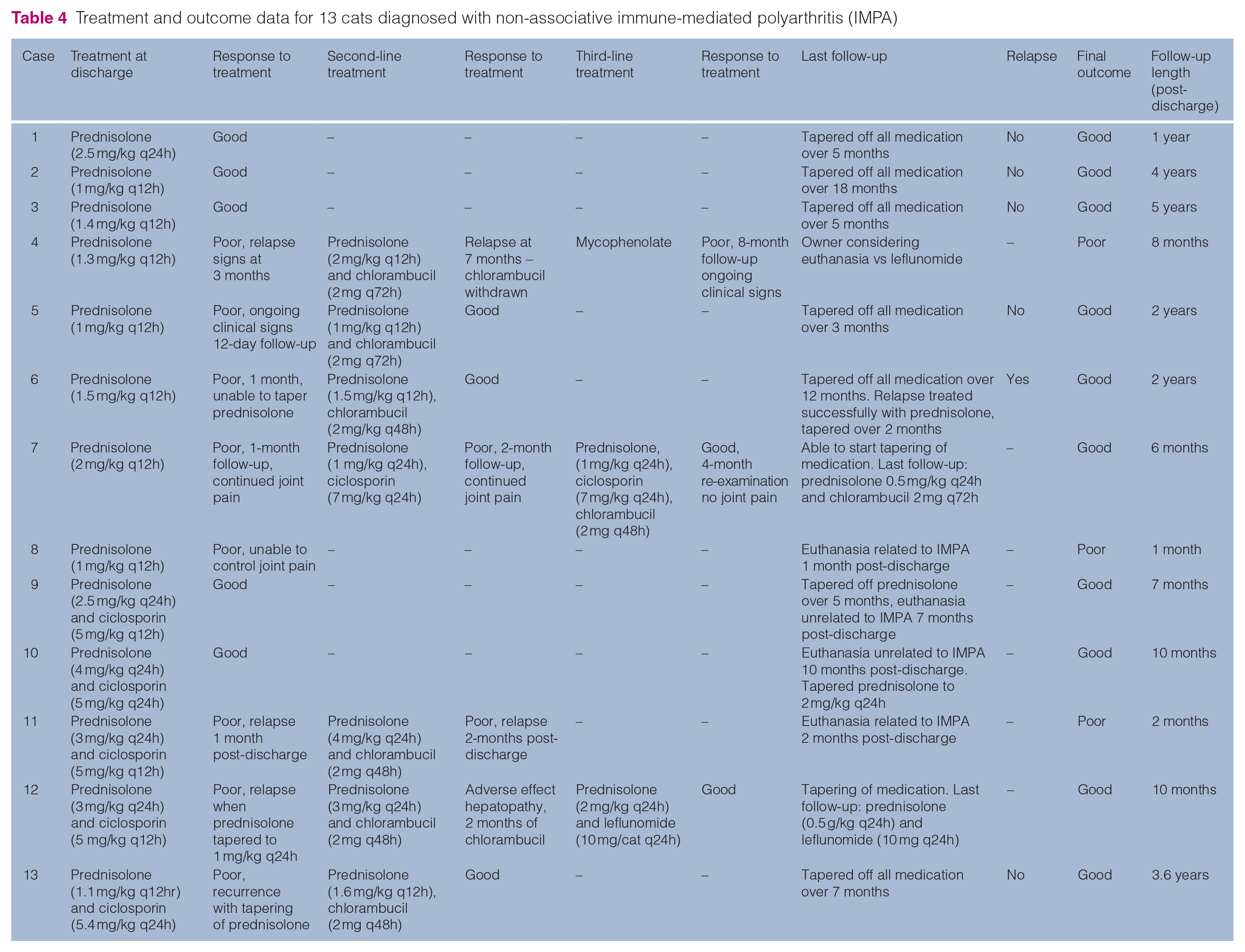
A summary of the treatment and outcomes in the non-associative IMPA cases is found in Table 4 and associative IMPA cases in Table 5. Specific treatments administered for the comorbidities identified in cases of associative IMPA are included in Table 5.
Treatment and outcome data for 13 cats diagnosed with non-associative immune-mediated polyarthritis (IMPA)
Treatment and outcome data for seven cats diagnosed with associative immune-mediated polyarthritis (IMPA)

FHV = feline herpesvirus; FIV = feline immunodeficiency virus; CIE = chronic inflammatory enteropathy
All cats with presumed non-associative IMPA were treated with immunosuppressants. Eight of the 13 cats were started on prednisolone monotherapy. Only three cats (38%) had a good response to treatment; of the other five cats, four were administered second-line immunosuppressants (chlorambucil [n = 3] and ciclosporin [n = 1]) in addition to the prednisolone and one was euthanased. Two cats responded well to the addition of chlorambucil and one failed to respond to both chlorambucil and also to an alternative second agent (mycophenolate). The cat started on ciclosporin as a second agent failed to improve but responded well to an alternative second agent (chlorambucil) alongside prednisolone. Five cats were started on treatment at diagnosis with prednisolone and ciclosporin as a second agent. Two of these cats (40%) responded well; the remaining three were started on an alternative second agent (chlorambucil) alongside prednisolone. Of the three cats started on chlorambucil, one responded well, one was euthanased when failing to respond and one developed adverse effects with chlorambucil but responded well to an alternative immunosuppressive agent (leflunomide) together with the prednisolone.
Five of the seven cats with associative IMPA were treated with immunosuppressants. Two cats received prednisolone monotherapy and showed a poor response to treatment, one cat failed to respond to the addition of chlorambucil despite successful treatment of the concurrent pneumonia with antibiotic therapy, and was euthanased. The second cat suffered adverse effects and was not started on alternative treatment due to poor compliance; this cat had evidence of ongoing IMPA despite resolution of the clinical signs of the concurrent chronic enteropathy. One cat was started on prednisolone monotherapy at an anti-inflammatory dose. This patient did not respond to treatment and was euthanased within 2 days; this cat tested positive for FIV. One cat received ciclosporin monotherapy, developed adverse effects within 5 days of treatment and responded well to chlorambucil monotherapy alongside resolution of the clinical signs of the concurrent chronic enteropathy. One cat was treated with prednisolone and chlorambucil alongside resolution of the clinical signs of the concurrent enteropathy and responded well to treatment.
Overall, a good outcome was achieved in 14/20 (70%) cats with IMPA: 10/13 cats (77%) with non-associative IMPA and 4/7 cats (57%) with associative IMPA. Two of the 14 cats with a good outcome were not treated with immunosuppressive agents and received meloxicam alone, although both cats displayed recurrence of clinical signs if the meloxicam was withdrawn. In one cat immunosuppression was avoided due to chronic feline herpesvirus and concerns of recrudescence. The second cat was not given immunosuppressants due to concerns that it would exacerbate the suspected concurrent discospondylitis. The remaining 12 cats with a good outcome received immunosuppressants. Seven cats (35%) were tapered off all medications and had no reported relapses during a follow-up period between 4 months and 5 years (median follow-up 2 years). One cat was tapered off all medications and had a single relapse 5 months after, which was treated successfully with prednisolone monotherapy. Two cats were responding well to treatment and were being successfully tapered off medication at the time of writing (6 months and 10 months post-diagnosis, respectively). Three cats were euthanased for reasons unrelated to IMPA and at the time of euthanasia all cats had no clinical signs of IMPA.
Discussion
This is the largest case series to date describing the clinical features and outcome of cats with non-erosive IMPA. In this study, non-associative IMPA was found to be more common than associative IMPA. In a previous case series, 7/13 cats (54%) had associative IMPA with comorbidities identified at the time of IMPA diagnosis. 4 Overall, the prognosis for cats with non-erosive IMPA appeared to be favourable, with most responding well to treatment and 70% having a good outcome.
Only 5/13 (38%) cats with non-associative IMPA and 1/5 (20%) cats with associative IMPA treated with immunosuppressants responded well to first-line treatment. The remaining 12 cats received additional immunosuppressive agents or were euthanased. The need for multimodal immunosuppression did not always result in a poor outcome and overall, of the nine cats who received additional agents, six (67%) had a good outcome.
In the previous case series by Bennett and Nash, 4 all 13 cats with IMPA were treated with prednisolone monotherapy (1 mg/kg q12h, tapered over 6 weeks when possible). In this case series, 6/13 cats (46%) showed a good response to treatment and were successfully tapered off prednisolone. The remaining seven cats were euthanased as a result of the IMPA owing to either a poor response to treatment (n = 3) or an inability to taper the dose of prednisolone (n = 4). The results of our study could suggest that cats with IMPA may not respond well to first-line treatment, but this does not necessarily predict the final outcome and second-line treatment should be considered.
The most commonly identified comorbidity in our case series was gastrointestinal disease. None of the cats in this case series was diagnosed with neoplasia. This contrasts with the case series in which Bennett and Nash 4 reported that 4/13 cats with IMPA had associated neoplasia. However, one cat in the current study was euthanased within 4 months of diagnosis as a result of suspected lymphoma, based on the development of a marked circulating lymphocytosis. Unfortunately, flow cytometry and histopathology were not performed, and this was not confirmed.
FIV was identified as a comorbidity in one cat in this population. Immune-mediated disease in cats with FIV most commonly occurs secondarily to excessive antibody production in response to chronic infection. This results in hypergammaglobulinemia and immune complex deposition.7–9 This cat did not receive immunosuppressive therapy; the poor outcome may reflect the presence of the FIV infection, or the absence of immunosuppressive therapy. To our knowledge, FIV has not been previously reported in association with non-erosive IMPA. FIV has been associated with feline chronic progressive polyarthritis, which is an erosive form of IMPA. 10 Six cats in this study were not tested for FIV and, considering it is often an asymptomatic infection, it is possible the true prevalence in this population was underestimated. In the previous case series, Bennett and Nash 4 reported three cats with IMPA were FeLV positive; however, the FeLV vaccination only become widely available after the previous case series was reported, which may have affected the prevalence of FeLV in the population at that time.
In the present study, 10/18 (55%) cats in which rectal temperature was recorded were pyrexic on presentation. This is similar to the previous case series where 8/13 cats (61%) were found to be pyrexic. 4 As a result, the absence of pyrexia cannot be used to exclude IMPA in a cat presenting with lameness. This also correlates with the current literature on canine IMPA. Pyrexia is reported in approximately 50% of dogs with IMPA, which is a common cause of ‘pyrexia of unknown origin’ (PUO).1,11,12 In a study of 101 dogs with PUO, IMPA was found to be the diagnosis in 20% and some of these dogs presented without any obvious evidence of joint pain or inflammation on physical examination. 13 In a recent study of 106 cats with PUO, IMPA was the diagnosis in only 3% of these cases. 6 The difference in the prevalence is likely to be reflective of the variability in the incidence of canine and feline IMPA and the higher prevalence of infectious diseases in cats. 14
Septic bacterial arthritis occurs most commonly as a result of cat bites and usually affects one joint in a single limb. 14 Interestingly, seven cats in this population presented to their primary care practice with single-limb lameness despite an increased proportion of non-degenerate neutrophils being identified within synovial fluid samples taken from multiple joints. IMPA in cats should therefore not be excluded based on involvement of a single limb alone and further investigations should be performed if there is no evidence of a wound or bacterial infection. Another interesting observation is the high yield of an increased proportion of non-degenerate neutrophils within the tarsal joints of cats in this study; this has also been found to be the most commonly affected joint in dogs. 12
Two cats in this study were treated with NSAIDs and the clinical signs of IMPA such as lameness and reluctance to walk resolved while on treatment in both cats. Attempts to withdraw NSAIDs were unsuccessful owing to the recurrence of clinical signs. These findings could reflect that in these cases there was an ongoing inflammatory arthropathy and the NSAIDs were treating the joint pain successfully rather than the underlying pathology. Unfortunately, neither cat had repeat joint aspirates performed, so it was not possible to confirm this. One of these cats had concurrent discospondylitis with IMPA; it is therefore possible that the relapse in clinical signs of IMPA seen in this cat was as a result of failure to treat this comorbidity. However, this was considered less likely as the clinical signs of the discospondylitis completely resolved with antibiotic treatment. A positive response to treatment with NSAIDs in cats presenting with lameness of unknown origin should also not be used to exclude IMPA and further investigations should be considered if relapse of clinical signs is seen when NSAIDs are withdrawn.
Mycoplasma species have been implicated in rare cases of feline polyarthritis. 14 Mycoplasma gateae has been identified in the synovial fluid of a small number of cats with naturally occurring erosive arthritis and has also been experimentally reproduced with intravenous inoculation into healthy cats.15,16 Mycoplasma felis has been documented to cause non-erosive monoarthritis in two immunocompetent cats and has been cultured from a cat with polyarthritis and suspected severe immunocompromise.17,18 Infectious disease screening for Mycoplasma species was variable in this population. It was not possible to definitively conclude that the six cats that did not receive testing or treatment for Mycoplasma species were not infected. However, 5/6 cats had a good response to immunosuppressive treatment, making Mycoplasma species infection unlikely in any of the cases.
Five cats in this study presented with, or developed, joint ligament laxity while on prednisolone therapy. Ligament laxity has been identified in multiple cases of feline erosive polyarthritis such as feline rheumatoid arthritis and feline periosteal proliferative polyarthritis.2,10,19 To our knowledge, ligament laxity has not been previously documented in cases of feline non-erosive IMPA, but has been reported in five dogs with non-erosive IMPA. 20 The prognosis for dogs with ligament laxity and non-erosive IMPA is poor. 20 The outcome in the five cats in this study with ligament laxity was variable. One cat was euthanased owing to the severity of clinical signs within 2 days of diagnosis. Two cats required multimodal immunosuppression due to frequent relapses and poor response to treatment. Unfortunately, it is not possible to comment on the ability to withdraw medication in either case, as both were still receiving treatment at the time of writing. One of the cats with ligament laxity had associative IMPA and responded well to meloxicam. The final cat responded poorly to prednisolone monotherapy and had severe adverse effects; further immunosuppression and NSAIDs were recommended but not provided. This cat had ongoing chronic lameness on follow-up but received no further treatment.
There are several limitations to the current study. As this study was retrospective and spanned several referral centres the investigations and treatment provided were not standardised. It is therefore difficult to directly compare the efficacy of specific treatments, and treatment recommendations cannot be established. Future prospective studies would be required to investigate treatment protocols. In addition, cases were recruited from a referral population only. It is considered possible that some cases of IMPA identified in primary care practices might have responded more favourably to prednisolone monotherapy and would not have been presented to a referral centre, thus leading to a selection bias in the cases included in this study. However, the number of cases in this cohort that had received prior immunosuppressive therapy was low (three cats; 15%). Most cats presented to the referral centres soon after the onset of clinical signs, making the findings of this study relevant to both referral and primary care practices. Some cats showed significant adverse effects with ciclosporin therapy and were changed to an alternative second agent. Blood ciclosporin levels were not monitored in these cats so it was not possible to confirm if high levels were present in these cases.
IMPA is poorly defined in the veterinary literature, with no consensus on the diagnostic criteria. A cut-off of two affected joints to make a diagnosis of IMPA has commonly been used and accepted as an inclusion criteria in dogs and was extrapolated to this study.11,12,21 In the human literature the involvement of five joints is required to make a diagnosis of polyarthritis. 22 It is therefore possible that the cases in this study with less than five affected joints (nine cats) were misdiagnosed as IMPA. In addition, owing to the small volume inherently available from joint aspirates in cats, total nucleated cell counts were not available for all cases.
Cases were divided into associative and non-associative IMPA based on the diagnosis made by the primary clinician. In associative IMPA the comorbidities identified were not proven to be responsible for the non-erosive IMPA. In addition to this, some cases of associative IMPA did not resolve despite resolution of the comorbidities; this could suggest that the disease identified was incidental and not related to the IMPA. In two cases, thoracic imaging was not performed so it was not possible to exclude a comorbidity present in the thorax for these cases. One case without thoracic imaging was considered to have non-associative IMPA and it is therefore possible this case was categorised incorrectly as a result. Infectious disease screening was highly variable and so the prevalence of diseases such as FIV could not be established.
A further limitation in this study is the lack of follow-up arthrocentesis samples to definitively prove remission and cases of relapse. In this study the presence of relapse was based on the recurrence of clinical signs. Despite these limitations this study provides useful information on the presenting signs, treatment options and outcomes in cases of feline IMPA.
Conclusions
The prognosis for feline IMPA can be good; however, multimodal immunosuppression is often required in order to control clinical signs associated with disease. Sampling of the tarsal joints most frequently identified changes consistent with IMPA in this cohort. Ligament laxity can occur in non-erosive IMPA but does not appear to significantly impact overall prognosis. IMPA should be considered in lame cats, with or without pyrexia, when there is no clear evidence of trauma or infection.
Footnotes
Author note
This paper was presented in part as an oral presentation at the 2019 BSAVA conference.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
