Abstract
Objectives
The aim of this study was to ascertain the effect of a drop of timolol 0.5% ophthalmic solution on the systolic function of the left ventricle (LV) and left atrium (LA), and to confirm if timolol helped appraisal of diastolic function by reducing heart rate (HR) and separating the transmitral outflow waves from tissue Doppler imaging (TDI).
Methods
A total of 41 client-owned healthy cats underwent two echocardiograms 20 mins apart. The timolol group (33 cats) received a drop of timolol solution after the first examination. Standard and speckle-tracking echocardiography evaluated the LV and LA function of both groups at the two time points evaluated.
Results
Timolol reduced HR (19%), and fractional shortening from LV (20.3%) and LA (16.6%). Septal S' decreased by 51% (from 7.7 to 5.2 cm/s) and lateral S’ dropped by 43.1% (7.3 to 5.1 cm/s). Most longitudinal techniques did not change after timolol, including the mitral annular plane systolic excursion from the interventricular annulus, tricuspid annular plane systolic excursion, LV longitudinal strain and LV tissue motion annular displacement. The isovolumic relaxation time increased by 15.2% (from 54 to 64.6 ms), with most cats presenting this variable above the reference (>60 ms). Timolol did not support diastolic assessment, enabling evaluation in only 2/11 cats when using lateral TDI and 1/9 cats using septal TDI. Regarding side effects, miosis occurred in 18 cats (54.5%).
Conclusions and relevance
Timolol reduced systolic function, decreasing standard echocardiographic variables. Regarding diastolic evaluation, although timolol decreased HR, it did not separate the mitral diastolic waves, as expected.
Introduction
An echocardiogram in cats usually activates the fear response, causing an increase in heart rate (HR),1,2 systemic blood pressure (SBP) 3 and respiratory rate. Tachycardia is an undesirable consequence of stress, as it can impede or complicate diastolic evaluation. 4 At a lower HR, there are two transmitral outflow waves: the E wave, which corresponds to the filling of the left ventricle (LV) in early diastole, followed by the A wave, with the peak resulting from atrial systole.5,6 In most cats with normal diastolic function, E is greater than A (E/A>1). An HR above 170–180 beats per min (bpm) during echocardiography in cats leads to fusion of the mitral outflow waves (EAfus). The same occurs with tissue Doppler imaging (TDI): the summation of waves with tachycardia (E’A’fus). This technique reflects the myocardial velocity rather than blood movement by placing the Doppler gate at the mitral annulus (interventricular septum [IVS] or free wall [FW]). The E’ wave, called early diastolic mitral annular velocity, is produced during early LV diastole, while A’ reflects annulus movement during contraction of the left atrium (LA). 5 Normally, E’ is expected to be greater than A’, although a slightly inverted ratio (E’/A’ between 0.8 and 1.0) can be considered normal in cats with a high HR. 4
Fused waves do not allow diastolic assessment, which is essential for diagnosis, as the most prevalent heart diseases in cats (ie, cardiomyopathies with hypertrophic or restrictive phenotypes) start with impairment of diastolic function.4,7,8 It is highly recommended that diastolic function is evaluated and its pattern classified using a combination of spectral Doppler and TDI. Precocious detection of dysfunction can help diagnose early-stage disease. In cats that have already developed signs of congestive heart failure or arterial thromboembolism (ie, those classified as stage C), diastolic information helps establish a prognosis. 9
An alternative to avoiding summation and enabling diastolic assessment is to apply a vagal maneuver at the time of mitral outflow Doppler or TDI acquisition. 10 It requires the presence of an assistant to perform the parasympathetic stimulation correctly at the appropriate moment. Another option is to administer timolol ophthalmic solution to block the sympathetic tone and decrease the HR, separating the transmitral and TDI waves. 11 The topical application of timolol has a systemic absorption,12,13 reducing the HR when administered alone in cats 11 or associated with topically applied carbonic anhydrase inhibitors, such as dorzolamide or brinzolamide. 14
Timolol maleate is the most frequently used drug in managing open-angle glaucoma, 15 although long-term treatment may have adverse effects in humans, such as low systolic or diastolic blood pressure. 16 A non-selective beta-adrenergic antagonist such as timolol may reduce the contraction force of the heart,17,18 compromising the commonly used systolic variables. In consequence, it may also modify the LA activity, directly affected by the LV. 19
This study assumed that timolol would alter cardiac function in cats undergoing an echocardiogram. In this case, the objective was to determine how much a single drop of timolol interferes with the systolic variables obtained by standard and speckle-tracking echocardiography. Furthermore, it was expected that the echocardiographic indices of the control group would not change after 20 mins of waiting and second handling, even though it could be stressful for cats.
Materials and methods
Animals
This prospective longitudinal observational study recruited client-owned healthy cats between May and July 2019. Before enrollment, all cats underwent a thorough physical examination, SBP measurement with Doppler, electrocardiography (ECG) and standard transthoracic echocardiography.
The Institutional Animal Care and Use Committee (protocol 014/2019) previously approved all the procedures and they complied with the National Institutes of Health Guide for the Care and Use of Laboratory Animals. Owners were required to formally agree to participate in this research by signing an informed consent form.
An age limitation was adopted to avoid possible changes in drug metabolism in younger or older animals; cats <1 year or >14 years of age were excluded. Cats with an elevated SBP prior to the first echocardiogram (>160 mmHg) did not participate, nor did any cat with a history or signs of heart disease or other chronic illness, or that had received any treatment in the past 6 months.
Echocardiography
All cats underwent two echocardiographic examinations with only gentle restraint and no sedation. Positioning followed the recommendations of the Echocardiography Committee of the Specialty of Cardiology of the American College of Veterinary Internal Medicine. 20 The same operator (GLRT) performed all examinations and measurements.
A drop of timolol 0.5% ophthalmic solution containing approximately 0.17 mg timolol (1 mm = 30 drops) was instilled at the left eye immediately after the first echocardiogram (control timolol). A second echocardiogram (timolol) was performed 20 mins later, following the same protocols mentioned. The control group consisted of eight cats not receiving the timolol eyedrop, submitted to two echocardiograms 20 mins apart (control 1 and control 2, respectively).
LV systolic function
LV M-mode measurements at the papillary level were determined, as described previously, 21 allowing the calculation of the most common systolic variables, such as fractional shortening (FS) and ejection fraction (EF). The end-diastolic and end-systolic LV diameters were measured using a leading-edge-to-leading-edge technique, with a simultaneous ECG allowing us to distinguish the two cardiac phases. The cut-off point for hypertrophic cardiomyopathy (HCM) was considered to be 5.5 mm for any LV image, 22 including IVS thickness in the LV outflow tract view from the right parasternal window.
Evaluation of the longitudinal systolic function of the LV required acquisition of apical four-chamber (AP4) and two-chamber (AP2) images from the left parasternal window. At least five cardiac cycles were recorded to allow off-line calculation of tissue motion annular displacement (TMAD) measures and the longitudinal strain (LSt) values, considering the mean of AP4 and AP2 values as global indices. In addition, AP4 was applied to measure the mitral annular plane systolic excursion (MAPSE) from the IVS annulus and FW annulus, the tricuspid annular plane systolic excursion (TAPSE), and the mitral annular velocities with the gate of investigation on the IVS and FW annulus, as described elsewhere.5,23
Both LV TMAD and LV LSt required the aortic valve closure time, measured from the apical five-chamber image. It corresponds to the time (ms) from the beginning of the QRS complex to the end of the aortic valve spectra obtained with the pulsed Doppler gate positioned distally to the valve. 24
The hinge points of mitral valve leaflets (septal and lateral) and the LV apex epicardial region were the three regions of interest (ROIs) used to calculate the LV LSt 25 (Figure 1a), which was measured automatically by the equipment software (QLAB ACMQ; Auto Cardiac Motion Quantification). Manual corrections were made whenever the automatic myocardial tracking was obviously incorrect. For LV TMAD, the same three ROIs were defined (Figure 1b). The displacement of both annuli towards the apex (in mm) was automatically calculated. Also, the software defined a virtual midpoint between the two annular ROIs (LV MP TMAD) and calculated its displacement towards the LV apex (mm), as well as the proportional displacement of that midpoint concerning the total length of the LV (LV MP% TMAD).

Apical four-chamber (AP4) images demonstrating: (a) longitudinal strain of the left ventricle (LV); (b) tissue motion annular displacement (TMAD) of the LV; (c) TMAD of the left atrium (LA) in a patient with a low heart rate (150 beats per minute [bpm]) and a visible plateau (dashed line); and (d) TMAD of the LA in a cat with a faster heart rate (186 bpm), with diastasis determined by pausing the electrocardiogram (ECG) cursor just before the P wave on the ECG (white circle). TMAD AP4 MV1 (in blue) describes the displacement of mitral septal annulus towards the LV apex; TMAD AP4 MV2 (in orange) is the displacement of mitral lateral annulus towards the LV apex; TMAD AP4 MV midpoint (in white) signifies the displacement of a virtual point determined in between the septal and lateral hinge points of mitral annulus towards the LV apex. In (c) and (d), the larger arrows designate the total displacement of the mitral annulus towards the farthest point of the LA (LA D TMAD); the shorter ones represent systolic displacement to the same point (LA S TMAD)
LA function
LA FS was estimated based on the minimum and maximum LA diameter from the right parasternal short-axis view image at the aortic valve level, using anatomic M-mode. 26 The ratio between the LA and aorta was measured at the maximum LA diameter (LAD), right after the T wave from the ECG, as described previoulsy. 27 The LAD measure was acquired from the right parasternal long-axis four-chamber view, being performed as explained before, with a maximum reference value of 1.6 cm. 28 The AP4 image was used to measure LA dimensions at three different moments based on the LA cycle (before P wave, before QRS complex and after T wave), enabling later calculation of the passive (pEF), active (aEF) and complete (cEF) LA EF, as detailed elsewhere. 29
To calculate LA LSt and LA TMAD, the first two ROIs had the same position as the LV at the mitral leaflet hinge points, while the third ROI was defined at the cranial inner edge of the LA. The total displacement (LA D TMAD) between the maximal negative and positive positions was the average of the displacements documented individually for each of the two annular points. The systolic displacement (LA S TMAD) was the average movement of the two ROIs between diastasis and the maximal positive position. In humans, diastasis is easily recognized by a plateau wave (Figure 1c). 30 However, as the mean HR in cats is approximately three times higher, a plateau phase might not happen. In these cases, it is possible to determine the diastasis based exclusively on the simultaneous ECG, pausing the marker immediately before the P wave (Figure 1d).
Statistical analysis
The study aimed to determine whether ophthalmic timolol would cause differences in the echocardiographic variables of cats after its application. The same group of animals was compared before and after application (paired quantitative variable). The sample size was calculated using GPower 3.1.9.4, 31 with an expected effect of 80%, a sampling power of 95% and an error of 5% for quantitative measurements and using two dependent samples, reaching a total number of 23 cats for the timolol group.
Analysis was conducted in R 4.0.5 (R Core Team 2021). 32 Initially, a descriptive analysis of the data was accomplished with an estimate of the mean, median, SD, range and interquartile range of the quantitative variables, and simple and relative frequencies of the qualitative variables.
The Shapiro–Wilk normality test was performed to determine the parametric and non-parametric approaches to evaluate the quantitative variables. For variables with a normal distribution, the difference between two independent groups was verified with a Student’s t-test or paired t-test for dependent groups (crossover). For variables without a normal distribution, this difference was verified using the Mann–Whitney or Wilcoxon U-test. P values <0.05 were considered to be significantly different. Finally, Spearman’s correlation was adopted to analyze the relationship between HR and body weight (BW) for both timolol and control groups.
Results
Forty-one client-owned healthy cats were enrolled in the study. The timolol group was composed of 33 cats, including 18 females (mean age 6.1 years [range 1–13]; mean weight 5.1 kg [range 2.1–11]). This group included Persian (n = 2), Maine Coon (n = 1), Siamese (n = 1) and mixed breed (n = 29) cats. Eight mixed-breed (including six females) were included as a control group (mean age 8.8 years [range 3–13]; mean weight 4.5 kg [range 2.1–7.6]).
Most data were normally distributed, except BW, HR, LA and aorta measurements on the right transversal image, isovolumic relaxation time (IVRT), TDI velocities, MAPSE IVS, MAPSE FW and TAPSE.
The mean SBP (140 mmHg) of the control groups (control timolol and control 1) did not differ significantly. All animals had normal sinus rhythm on ECG recording.
The echocardiographic indices of the control group presented no significant differences between the two examinations performed after a 20 min interval (control 1 and control 2). On the contrary, the echocardiogram of cats after timolol administration showed differences in many indices, as shown in Table 1 and Figure 2. Figure 3 displays the variations in some of the variables affected by timolol (control timolol and timolol).
Standard and speckle-tracking echocardiographic variables in the timolol group, before the drug administration (control timolol) and after (timolol), and in the control group, before (control 1) and after (control 2) the 20 min interval

P value <0.05
HR = heart rate; LAD = left atrium diameter; LA FS = left atrium fractional shortening; IVSd = interventricular septum thickness end diastole; LVIDd = left ventricular internal dimension at end-diastole; LVPWd = left ventricular posterior wall thickness at end-diastole; IVSs = interventricular septum thickness at end-systole; LVIDs = left ventricular internal dimension at end-systole; LVPWs = left ventricular posterior wall thickness at end-systole; FS = fractional shortening; EF = ejection fraction; IVRT = isovolumic relaxation time; MAPSE = mitral annular plane systolic excursion; FW = free wall; IVS = intraventricular septum; TAPSE = tricuspid annular plane systolic excursion; AP4 = apical four-chamber; LV = left ventricle; LSt = longitudinal strain; MP = midpoint (virtual midpoint determined between the septal and lateral points of the mitral annulus); TMAD = tissue motion annular displacement; LA D TMAD = total displacement of the mitral annulus towards the farthest point of the left atrium; LA S TMAD = systolic displacement of the mitral annulus towards the farthest point of the left atrium; LA = left atrium; cEF = complete ejection fraction; aEF = active ejection fraction; pEF = passive ejection fraction; AP2 = apical two-chamber
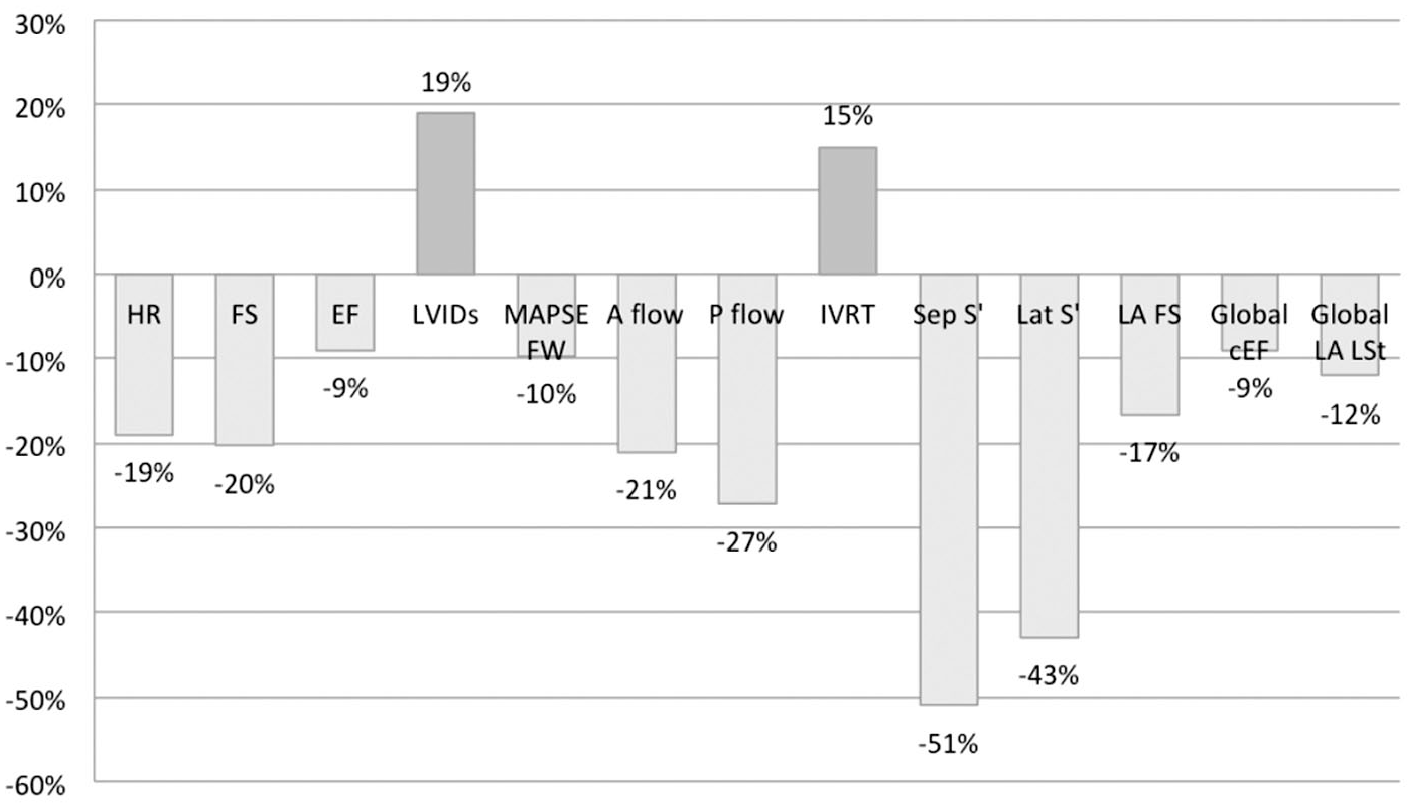
Percentage of change in echocardiographic indices significantly affected by timolol administration.

Boxplots representing echocardiographic indices altered significantly after timolol administration. Comparison between the first and second echocardiograms in the timolol group (control timolol and timolol) and the control group (control 1 and control 2).
Table 2 presents the assessment results of the LV filling pattern before and after timolol administration. The lower HR resulted in an increase in IVRT by 15.2% (from 54.8 to 64.6 ms), with most cats having this variable above the reference (>60 ms) 4 after drug administration. Of 33 cats, only 11 (33.3%) presented fused transmitral waves before timolol administration. Similarly, few diastolic TDI waves were fused on the control echocardiogram (control timolol): only five when measured at the IVS annulus (15.1%) and eight at the FW annulus (24.2%). The ‘impossibility of diastolic assessment’ described in Table 2 refers to the fusion of mitral waves or TDI waves preventing diastolic interpretation, as the presence of unfused waves is required in both techniques.
Echocardiographic diastolic alterations before and after timolol administration

Data are n (%)
IVRT = isovolumetric relaxation time; EAfus = peak velocity of summated E and A waves; E’A’fus = peak velocity of summated E’ and A’ waves; FW = free wall; IVS = interventricular septum
Spearman’s test revealed a moderate negative correlation between HR and BW (r = −0.290 and −0.393, respectively) for both the control and timolol groups, with evidence of statistical significance for the timolol group (P = 0.025).
Concerning the LA TMAD results, most cats did not show a plateau phase (Figure 1d). A horizontal line during LA diastasis similar to that seen in a human graph (Figure 1c) 30 occurred in a few cats: three in the first echocardiogram of the control group (control 1) and two in the second (control 2). In the timolol group, four cats showed a plateau phase on the first echocardiogram (control timolol), increasing to eight on the second (timolol), without statistical significance.
Regarding side effects, miosis occurred in 18 cats (54%) and lasted an average of 18 h, reaching up to 80 h in one cat. A contralateral effect on pupil diameter was not seen in the non-treated eye, ending in anisocoria (Figure 4). Short-lived ptyalism (<3 mins) was observed immediately after timolol application in 4/33 cats (12%).

Left eye of the cat with miosis owing to timolol resulting in anisocoria
Discussion
This investigation sought to assess whether timolol administered in eye drops interfered with cardiac variables assessed by echocardiography. In general, cats are more sensitive to stressful conditions, although they may hide signs of stress owing to their survival strategy. 33 Continued restraint during echocardiography exacerbates sympathetic activation, resulting in tachycardia and changes in several variables to assess cardiac function. The administration of a beta blocker could lead to a condition closer to the normal physiologic pattern, reflecting the condition the cat was in before sympathetic activation, or it could go beyond this point and lead to parasympathetic activation. It is likely that the latter occurred, given the high number of significantly altered variables (17/46) in the second echocardiogram of the timolol group (Table 1). In contrast, in the control group no significant differences were observed between the variables in the two echocardiographs performed. Hence, handling twice for echocardiography and the waiting period in a hospital environment did not stress the cats sufficiently to raise the HR and affect the indices.
Although timolol has its maximum effect in cats between 6 and 12 h after administration, 12 we chose to repeat the echocardiogram 20 mins later to follow the same protocol for the diastolic assessment detailed previously, 11 which coincides with the onset of its action after topical administration. As previously mentioned, timolol applied topically to the eyes has systemic effects, with a reduction in HR being the easiest to observe. Timolol effectively decreased HR by 19%, from ~190 to ~160 bpm. The mean decrease of 30 bpm was close to that previously reported (25 bpm). 11 Interestingly, another research group reported that a drop of timolol gel-forming solution did not affect HR 4 and 8 h later, 34 suggesting the bradycardic effect may not last long.
As there was a difference in BW between the animals, the dose of assimilated timolol was not the same, which may have influenced the variations observed. One drop of timolol contains approximately 0.17 mg, with most cats receiving around 0.033 mg/kg, considering an average BW of 5.1 kg. The dose that the lightest animal (2 kg) received was 5.6 times that of the heaviest cat (11 kg): 0.085 and 0.015 mg/kg, respectively. The difference in dose may explain the non-alteration of HR between the two echocardiograms of the heaviest animal; the HR remained at 150 bpm, which is a low HR for the species. However, excluding this outlier cat, the others weighed a maximum of 7.5 kg, and all showed a significant drop in HR after timolol administration. Moreover, Spearman’s test showed a significant negative correlation between HR and BW after timolol, amplifying a tendency seen by the control group, although without statistical significance. The correlation with the control group would also presumably be significant in a larger sample, as previously observed in healthy cats, 22 with HR decreasing as BW increases.
Comparing the results of timolol administration and the vagal maneuver previously investigated is challenging. Their advantages and limitations are pretty balanced, and it is difficult to determine which is more suitable. Timolol required a 20 min waiting interval and the HR reduction lasted throughout the echocardiographic examination. The vagal maneuver produced a more pronounced reduction in HR (42 bpm) within a few seconds, 10 lasting a maximum of 15 s, allowing systolic assessment with usual HR. However, about one-quarter of animals submitted to vagal stimulation had an inadequate response (HR reduction <20 bpm). Although the cited investigation did not observe it, a paradoxical tachycardia might result from the oculocardiac reflex. 35 Besides, to separate both the spectral and TDI waves, it might be necessary to repeat the stimuli, with the risk of reducing the intensity of the decrease in HR caused by fatigue, as reported in children. 36 Finally, the administration of timolol seems to be less invasive and more suitable for stressed cats. They may be disturbed by the mechanical procedure for parasympathetic stimulation in addition to the restraint required for the echocardiography. In dogs, fear and pain may be associated with the procedure, and potential reasons for a paradoxical elevation in HR or the absence of response after the maneuver. 10
Most systolic surrogates decreased with the use of timolol, such as FS and EF (Table 1). The FS, a classical echocardiographic variable used to evaluate radial systolic function, reduced by 20.3% (from 51% to 42.4%), coinciding with what has been previously reported. 11 However, post-timolol FS was still within the reference interval for cats (ie, between 39% and 51%). 22 Timolol is not likely, at least in normal cats, to impair the interpretation of the echocardiogram. Nonetheless, in cats with HCM, the reduction in FS has already been demonstrated to be a prognostic surrogate.9,37 A mean FS of 42.4% is much closer to what has been seen in cats with heart failure owing to HCM than in healthy cats. 38
Interestingly, the variables known to assess longitudinal ventricular systolic function, including LV LSt, MAPSE IVS and TMAD, were less impaired by timolol. A possible explanation would be a more potent action of timolol on the transversal myocardial fibers. In humans, transverse cardiomyocytes have been shown to be significantly more sensitive to beta blockers, with studies suggesting a physiological balance of antagonism between myocytes.39,40 Another explanation lies with HR variation itself, which affects the radial fibers more than the longitudinal ones, as previously described. 41
A surprising result was the significant reduction (9.8%) in MAPSE FW after timolol administration, while there was no corresponding reduction in MAPSE IVS. This finding reinforces the perception that MAPSE does not behave similarly when measured on the FW or the IVS annulus, with the IVS annulus somehow affected by the right ventricle, as previously reported. 42 A previous study with HCM cats concluded that the MAPSE IVS was the only factor predicting pleural effusion on a multivariable regression model, 43 pointing to the influence of the right ventricle on this index. The researchers assumed that the IVS mitral annulus acts differently from the FW, and changes in its longitudinal function could lead to pleural effusion rather than pulmonary edema. In any case, the decrease in MAPSE FW indicated that timolol may also have influenced longitudinal fibers, although not sufficiently to change other longitudinal indices, as previously discussed.
Many LA variables changed significantly after timolol administration. Considerable LA area measures obtained from AP2 and AP4 images increased on the second echocardiogram in the timolol group, affecting LA cEF on both AP4 and AP2. The reductions in LA FS (17%), global LA LSt (12%) and global LA cEF (9%) suggest a compromise in atrial function once timolol was administered. On the contrary, LA TMAD values did not change between examinations, requiring more understanding of this LA assessment technique to justify its non-alteration. Despite presenting potential advantages, such as not demanding high-quality images, the use of TMAD to evaluate the LA function has not been validated in cats, and has previously only been applied to one research group in humans. 30 Interestingly, the reduction in HR promoted by timolol was insufficient to significantly increase the number of graphics, with a plateau phase signaling the diastasis of the LA, as occurs in human LA TMAD, suggesting a need for an even lower HR.
Of the 17 variables that changed after timolol administration (Table 1), peak systolic mitral annular velocity presented the most remarkable change, as determined by tissue Doppler (S’). The S’ obtained from the interventricular annulus reduced by 51% (from 7.7 to 5.2 cm/s) and 43.1% from the FW (from 7.3 to 5.1 cm/s). Consequently, timolol lowered the S’ velocity to <5.2 cm/s, below the reference interval. 5 Similar results on S’ velocity have been reported previously.5,8 There is a positive relationship between the S’ wave and HR; however, it is impossible to delimit how much the reduction in HR may have played a role in such a marked decline. It seems that a combination of factors contributed, such as a systolic impairment evidenced by the lower FS. Of note, cats with HCM are known to have lower S’ wave velocity.5,23 In such patients, timolol might complicate the interpretation of the echocardiogram as the cardiologist will not be able to confirm if a reduced myocardial velocity is a result of the illness or the drug.
The pulmonary and aortic outflow velocities also decreased (by 27.2% and 21.3%, respectively) with timolol administration. This reduction appeared to be a consequence of the reduction in HR. A previous study with dogs concluded that an increase in HR leads to a faster aortic and pulmonary flow velocity, 44 insinuating a directly proportional relationship between them.
Regrettably, a low frequency of fused mitral outflow waves was documented in the first echocardiogram (Table 2). We speculate that having cats undergo a series of procedures (auscultation, hair clip, ECG and SBP measurement) before the echocardiogram might have contributed to their adaptation to the environment. Also, timolol administration did not separate most EAfus and E’A’fus, which impaired the LV filling pressure assessment. In those cats in which fused mitral or TDI waves were observed (11/33, using TDI IVS [33.3%]), timolol produced the desired separation in 3/11 (10.1%). In a previous study with timolol, 13/20 cats had a transmitral fusion, and successful separation was achieved in eight (62%). 11 Investigation with vagal maneuvers had better results, effectively separating 71% of the fused transmitral flows and 72% of the TDI velocity. 10
The fusion of transmitral waves may be caused by increased HR or indicate diastolic impairment. A previous study found that cats with HCM tend to have more EAfus at a lower HR than healthy cats, suggesting that summation may indicate diastolic impairment. 5 However, as the present study aimed to evaluate healthy cats, a more likely explanation for the low success of timolol might be due to a combination of factors: an insufficient reduction in HR; few animals with fused waves on the control echocardiogram (control timolol); and the separation of only one variable (mitral outflow or TDI), not both simultaneously.
A noteworthy result was the increase in IVRT post-timolol (Table 2). IVRT is a diastolic variable influenced by HR, lasting longer as HR decreases. 4 Combined with other variables, an enlarged IVRT reveals a diastolic dysfunction. 45 Eight cats in the timolol group had increased TRIV (>60 ms) before instillation (24%), which were maintained in the experiment because they did not manifest alterations in any other variable and had an HR low enough to justify an increase in this index. However, enlarged IVRT more than doubled after using the drug, rising to 20 (60.6%). Consequently, timolol administration might lead to a misinterpretation of the cat’s echocardiogram as enlarged IVRT in these cases reflects the action of the beta blocker and is not associated with a diastolic impairment.
The administration of timolol ophthalmic solution was initially proposed to assist in the early diagnosis of HCM 11 as diastolic dysfunction secondary to hypertrophy is characteristic of this disease. 46 However, timolol might not be indicated in cats with HCM as it increases IVRT and interferes negatively with the systolic function of LV and LA. The administration would likely make the diagnosis of subclinical HCM more challenging. In patients already diagnosed with HCM, a low LA FS – a classical indicator of poor prognostic9,37 – could be due to timolol action and not to the disease itself. There are no data on the effect of timolol in cats with advanced cardiomyopathy.
The ideal situation would be to adopt a technique to assess diastolic function independent of preload and HR. One method that meets this requirement is assessment of the flow velocity profiles of the left anterior descending coronary artery via transesophageal echocardiography. 47 Another, more promising relaxation index, is the velocity of propagation of the mitral flow (Vp), 48 obtained during transthoracic echocardiography. It measures the rapid filling phase determined by M-mode color Doppler echocardiography, with its application reported in humans with HCM 49 and dogs with dilated cardiomyopathy. 50 A recent study in HCM cats concluded that the decline in Vp is correlated with the degree of increase in LA, which is significantly associated with the progression of diastolic dysfunction in HCM. 45
Interestingly, two cats appeared much calmer on the second echocardiogram. Their HR decreased slightly more than the mean, from 240 to 185 bpm (22.9%) and from 180 to 140 bpm (22.3%). Hence, it is plausible that the observed relaxation resulted from a beta blocker anxiolytic effect 51 and not from the reduction in HR itself. Behavioural assessment was not part of the experimental design, and the tranquillizing effect may have been present in other cats and gone unnoticed. Although this relaxation was not perceived in the control group, it is not possible to exclude the cat’s acclimatization in reducing anxiety.
In this investigation, a drop of timolol did not cause any clinically relevant adverse effects. All the cats likely had miosis on the eye that received the drug (Figure 4), but it was only observed in 18/33 cats (54.5%), resulting in anisocoria as the contralateral non-treated eye was not affected.11,33 A previous study of the administration of timolol in normotensive eyes in healthy cats reported a reduction in pupil diameter of 38.7% at 30 mins after treatment in all cats. 12 Despite going unnoticed by a significant number of owners, anisocoria was a disturbing feature for some of them, who reported non-specific behavioral changes, such as hiding or inactivity, both common alterations in cats exposed to a hospital environment.
Considering that only healthy cats were included, some possible side effects were not observed, such as bronchoconstriction, 52 which is an important reason for contraindication of timolol for cats with feline asthma. 15 This eye solution might also be avoided in elderly cats, as its continued use has been linked to syncope, 53 postprandial head-drops 54 and atrioventricular block 55 in older people. Some studies do not recommend timolol for long-term treatment of glaucoma in cats with cardiac diseases,15,56 but nothing is known about the effect of a single administration in cats with advanced heart failure. However, some researchers hypothesize that the negative inotropic effect of timolol may help to elucidate the patient’s response to beta blockade, particularly in cats with obstructive cardiomyopathy. 11 In the cited study, all six cats with evidence of dynamic obstruction or hypertrophic obstructive cardiomyopathy on baseline echocardiogram had relief of that obstruction after the administration of timolol.
Moreover, some episodes of ptyalism were reported, which is in line with preliminary data. 11 This excess salivation was not an action of timolol, but simply a normal cat response to the bad taste of the eye solution that passed through the tear duct to the back of the throat. However, as this reaction was short-lived, it may have gone unnoticed in most cats, as they waited in their crate for the second echocardiogram.
There were some limitations to this study. First, despite all efforts to select only healthy individuals, occult cardiomyopathies might not have been identified. Another limitation was the variation in the dose administered, which could be resolved by choosing only cats with similar BWs or manipulating the ophthalmic solution to administer a similar dose for all. The small number of cats with EAfus and E’A’fus biased the evaluation of timolol as a facilitator in assessing LV filling pressure. In this case, the owner’s awareness that their cat would undergo two echocardiograms may have discouraged them from bringing stressed cats to hospital environments, with only calmer cats participating in the experiment. A less pronounced fear response would result in a control echocardiogram with a lower HR and separate transmitral flow. An intraocular pressure measurement after the second echocardiogram could have added relevant information connecting the heart effect and reduction in eye pressure.
Conclusions
A drop of timolol 0.5% ophthalmic solution reduced HR and interfered negatively with the LV and LA systolic function in healthy cats. It affected many variables commonly used in standard echocardiography, such as FS and EF. All the cited changes are not desirable in evaluating cats with suspected cardiomyopathy, with a risk of misinterpretation. The influence of timolol was milder on the echocardiographic indexes known to assess the longitudinal systolic function of the LV, such as TAPSE, MAPSE IVS, LSt and TMAD. The MAPSE FW and de S’ wave were two exceptions, with both decreasing significantly after the administration of timolol.
Finally, despite not being the objective of the present study, it was found that diastolic evaluation was not benefited by timolol as the decrease in HR was not enough to separate the transmitral waves. On the contrary, the drug significantly enlarged the IVRT, which was used to classify diastolic function.
Footnotes
Accepted: 7 February 2022
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals and procedures that differed from established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient. The study therefore had prior ethical approval from an established (or ad hoc) committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
This paper was handled and processed by the American Editorial Office (AAFP) for publication in JFMS
