Abstract
Practical relevance:
Understanding the normal reproductive anatomy and physiology of the male cat is important for successful breeding. Veterinarians may be called in to troubleshoot when fertility and pregnancy rates decrease in a cattery. By understanding the normal physical parameters, as well as breeding behavior, the veterinarian is better equipped to assess the infertility issue. Also, clients are increasingly requesting breeding soundness examinations prior to mating or purchase. Semen collection is more difficult in the cat than in the dog but can still be accomplished in a clinical setting and provides important information when confronted with breeding males.
Aim:
The aim of this review is to provide a practical overview of the breeding male. The reader will receive information on time of puberty, normal reproductive anatomy (both external and internal), and breeding management practices to optimize fertility. The most up-to-date semen collection techniques are discussed, as are sedation/anesthesia options. Also, historical literature on the basic anatomy of the male reproductive system is reviewed, such as how the penile spines work and when they appear; this information is specific to the cat.
Evidence base:
The information in this article is based on the author’s experience, as well as drawing on historical and current literature, and provides the most up-to-date review as possible.

Anatomy
For a diagram of the reproductive anatomy of the tom cat, see Figure 1.
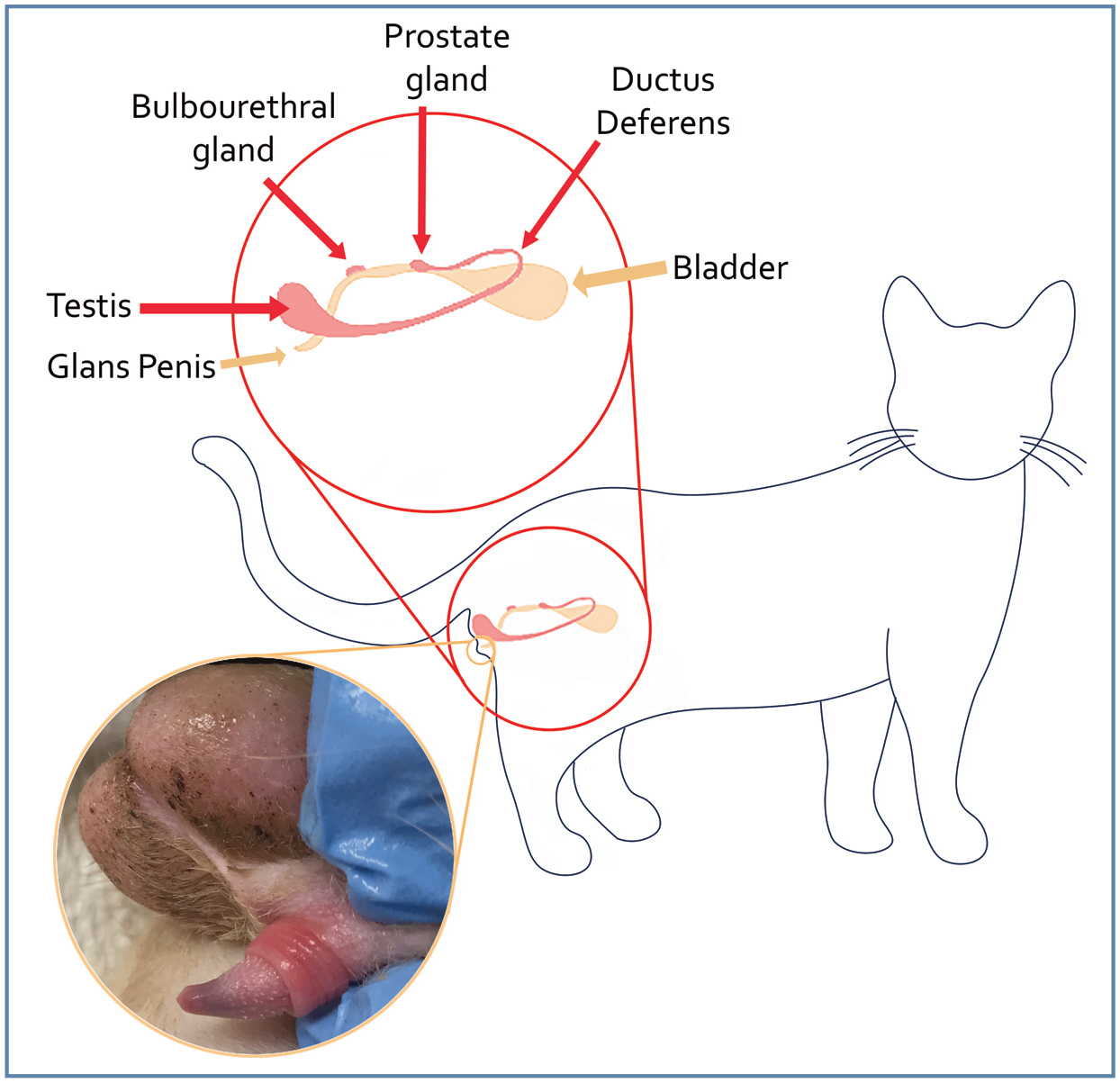
Reproductive anatomy of the tom cat. Figure designed by Jamie M Douglas
Penis
The penis is located ventrally to the testicles and points caudally unless erect (Figure 2). Prior to puberty, the penis is unable to be fully extended due to an adhesion of the balanopreputial fold (Figure 3). Because the breakdown of the fold is dependent on androgen secretion, if the cat is castrated before the breakdown occurs, the adhesion will remain. 1 The free portion of the penis of the tom is approximately 5–10 mm in length and 4–5 mm in diameter at the base. Once andro-gens are present in the postpubertal tom, penile spines become apparent on the glans penis, starting at the base and extending distally to cover approximately two-thirds of the penis (Figure 4). The tip of the penis is free of spines and has a smooth appearance.
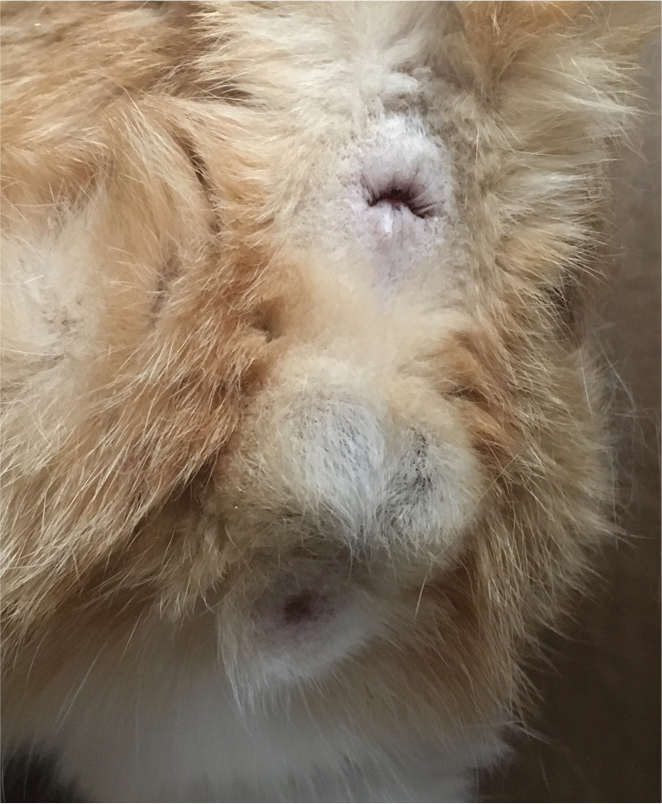
Normal anatomy of a mature intact torn. The two testicles are descended into the scrotum, located just ventral to the anus. The penis is ventral to the testicles and points caudally unless erect

The partially exposed penis of a 4-month-old intact male tom. This male is prepubertal and therefore the adhesion of the balanopreputial fold has not yet been broken down (arrow). Only the distal tip of the penis can be visualized

The penis of a normal intact male. The preputial fold (portion of the prepuce that covers the non-erect penis) can be seen as a smooth area at the base of the penis. The glans penis is exposed showing the androgen-dependent spines pointing caudally along the shaft, and the smooth tip
The penile spines, which measure 0.1–0.7 mm in length, comprise connective tissue covered by cornified epithelium, which imparts rigidity. They point caudally, away from the tip of the penis, and lay flat against the penis unless it is erect. During erection, engorgement of the erectile tissue causes the base of the spines to tighten and thus the spines protrude outward from the engorged penis. 2 The spines are dependent on androgens; therefore, once the tom is castrated, they will regress within 5–6 weeks. If testosterone is administered parenterally, the spines will become visible again. 3
Some tom cats will have an os penis, a small bone (3–5 mm) within the glans portion of the penis. Unlike in dogs, the development of the os penis is an age-related change that is due to the ossification of the distal end of the septum between the penile corpus caver-nosum; an os penis is therefore rarely seen in young cats. 4 If present, it can be observed on radiographs as a sliver of bone pointing cau-dally within the penis. 5 The erectile portion of the penis is made of two corpora cavernosa and a corpus spongiosum surrounding the urethra.
Testicles
The testicles are located within a thin scrotum ventral to the anus. Each testicle is separated within the scrotum by a median septum comprised of fibrous connective tissue that divides the scrotum in half. The testicles are more round than oval in shape. On ultrasound, there is a clear mediastinum visualized as a hyperechoic line through the central portion of the testicle. The mediastinum represents the center area of collecting ducts (rete testes), which funnel sperm into the epididymis.

The testicular parenchyma is homogeneous and delineated by the tunica albuginea, which is observed ultrasonographically as a hyper-echoic structure surrounding the testicle. On cross section, the tunica albuginea appears as a hyperechoic line encircling the testicular tissue (Figure 5).

Ultrasound image of a cross section of both testicles in a tom. The mediastinum can be visualized in the center of each testicle (black arrows). The tunica albuginea can be seen as a bright white line encircling the testicles (white arrows)
Testicular measurement (see ‘Measuring the testicles’ box) allows estimation of the volume of testicular tissue present and is performed as part of a breeding soundness examination. Average daily sperm output for a tom has been estimated at 32 million. 7

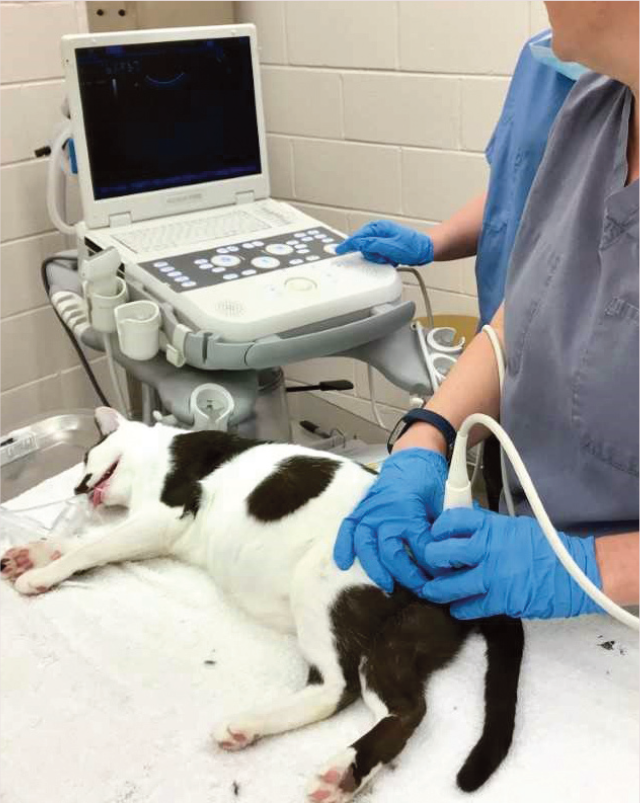
Set-up for testicular ultrasound in an anesthetized tom cat. The testicle to be measured is isolated, dampened with alcohol, and the ultrasound probe is placed perpendicular to the measurement being obtained. In this image, the width of the testicle is being measured. The cat is under general anesthesia because an electroejaculation procedure followed this examination. For measurement only, heavy sedation can be administered as an alternative
Epididymis and sperm maturation
Following formation in the testicle, the sperm travel to the epididymis, which is the place of sperm maturation and is comprised of the caput (head), corpus (body) and cauda (tail). The epididymis is closely connected to the testicle, with the caput located on the dorso-medial aspect of the cranial edge of the testicle. The corpus courses dorsolaterally to the testicle, and the cauda is located on the dorsocaudal portion of the testicle. The cauda passes medially to the testicle and then dorsally, becoming the ductus deferens.
Sperm maturation in the tom requires 47 days from formation in the testicle to release into the epididymis. Epididymal transit and maturation requires an additional 10–12 days, making the entire spermatogenic cycle approximately 60 days. As a sperm matures through the epididymis, a cytoplasmic droplet migrates. This droplet is formed as the sperm changes from a round cell to an elongated cell. The extra cytoplasm is not needed in the mature streamlined spermatozoon and is present as a ‘droplet’ of cytoplasm on the exterior of the cell. The droplet, which is located close to and below the spermatozoal head (proximal droplet) when released from the efferent ducts into the epididymis, then migrates from a proximal to a distal location at the junction between the midpiece and tail as the spermatozoa transition into the body of the epididymis. The distal droplets are shed during ejaculation. 8
Seminal fluid originating from the epi-didymis and testicle is high in alkaline phosphatase (ALP). In cases of azoospermia (no sperm in the ejaculate), the fluid obtained during semen collection can be submitted for routine ALP analysis. If the fluid originated from the epididymis, the ALP will be extremely high (>1000 U/l, often >10,000 U/l) and therefore ALP analysis can be used as a diagnostic test to confirm complete ejaculation.
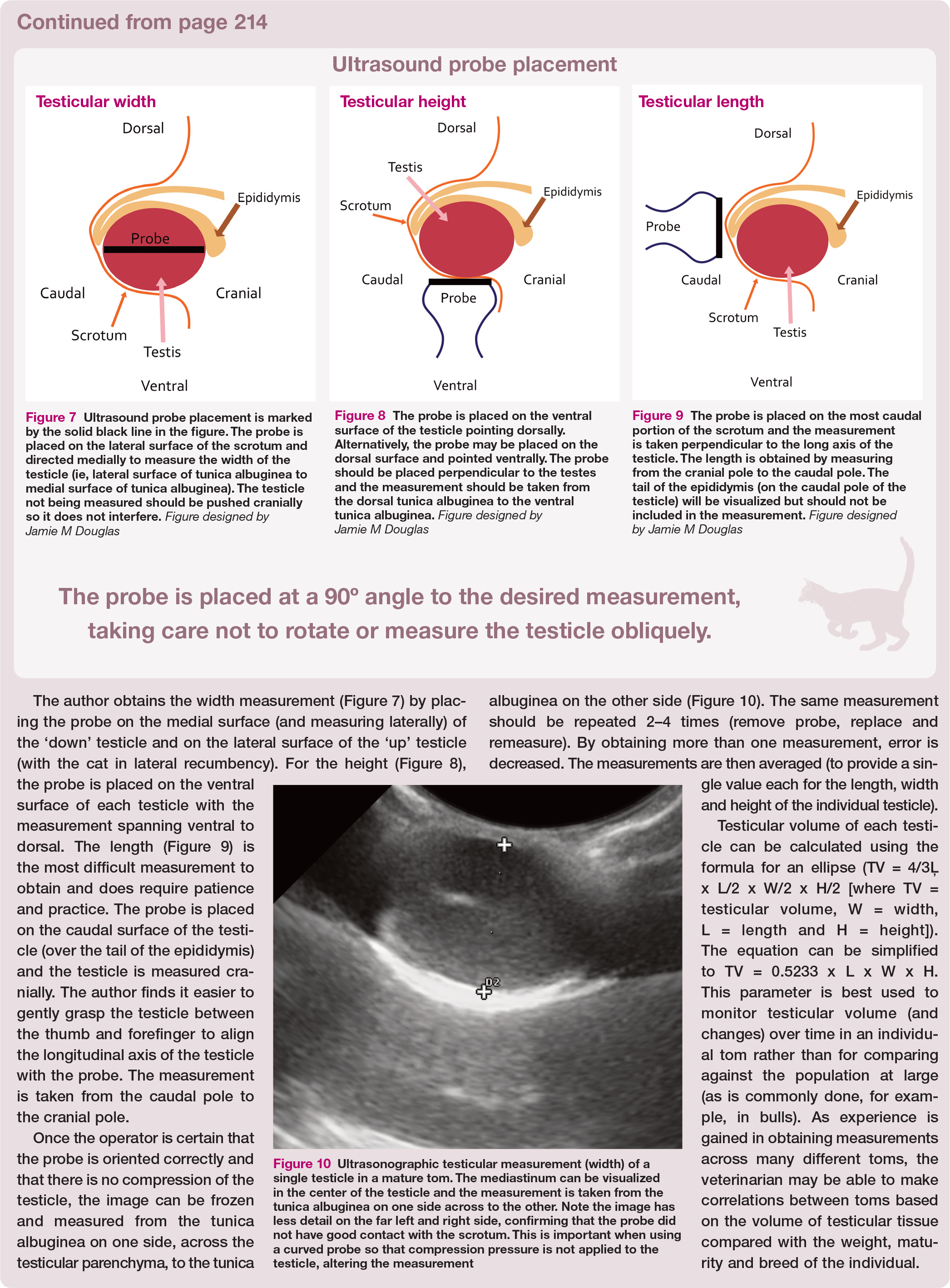

Ultrasound probe placement is marked by the solid black line in the figure. The probe is placed on the lateral surface of the scrotum and directed medially to measure the width of the testicle (ie, lateral surface of tunica albuginea to medial surface of tunica albuginea). The testicle not being measured should be pushed cranially so it does not interfere. Figure designed by Jamie M Douglas

The probe is placed on the ventral surface of the testicle pointing dorsally. Alternatively, the probe may be placed on the dorsal surface and pointed ventrally. The probe should be placed perpendicular to the testes and the measurement should be taken from the dorsal tunica albuginea to the ventral tunica albuginea. Figure designed by Jamie M Douglas

The probe is placed on the most caudal portion of the scrotum and the measurement is taken perpendicular to the long axis of the testicle. The length is obtained by measuring from the cranial pole to the caudal pole. The tail of the epididymis (on the caudal pole of the testicle) will be visualized but should not be included in the measurement. Figure designed by Jamie M Douglas
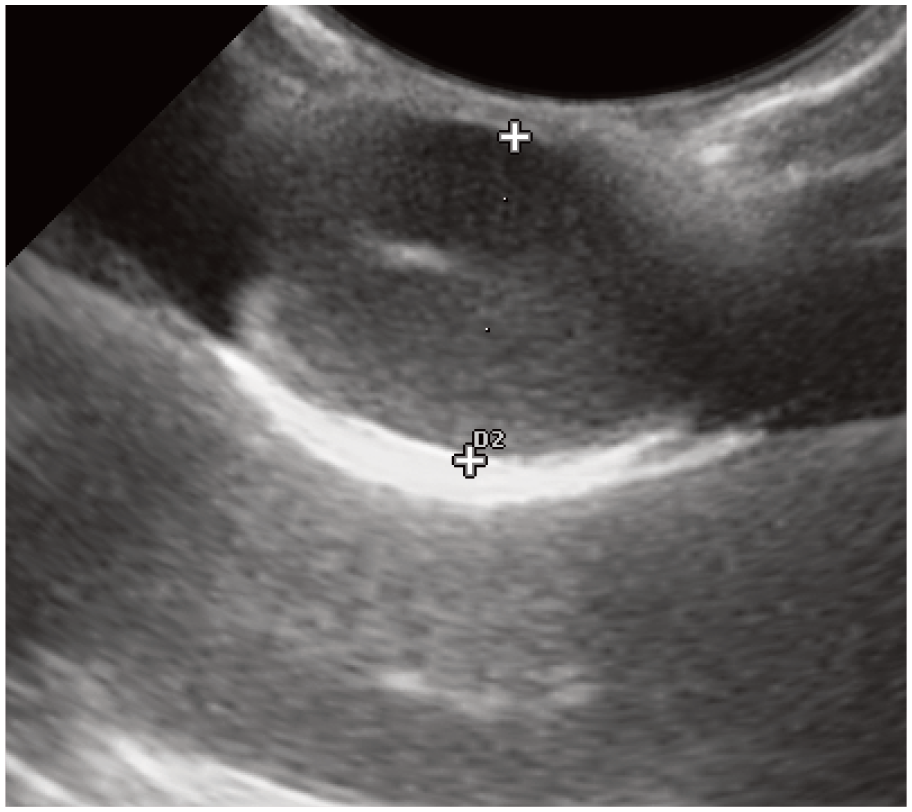
Ultrasonographic testicular measurement (width) of a single testicle in a mature tom. The mediastinum can be visualized in the center of the testicle and the measurement is taken from the tunica albuginea on one side across to the other. Note the image has less detail on the far left and right side, confirming that the probe did not have good contact with the scrotum. This is important when using a curved probe so that compression pressure is not applied to the testicle, altering the measurement
Accessory sex glands
Tom cats have a prostate and bulbourethral glands as the only accessory sex glands.
The prostate, which is located approximately 2–3 cm caudal to the bladder, is made up of two cranial and two caudal lobes (each 1 cm long) and surrounds the urethra dorsally and laterally. Prostatic fluid makes up the majority of the seminal fluid obtained with an ejaculate. The prostate is androgen-dependent and therefore atrophies following castration. This is a little described organ in the cat and problems associated with the prostate are much rarer in the cat than observed in the dog, with only a few case reports describing pathology (adenocarcinoma, prostatitis).
The bulbourethral glands are paired organs located just lateral to the urethra at the base of the penis near the ischial arch and distal to the anus. The glands are approximately 4–5 mm in diameter and circular. The pudendal nerve passes over the ventral portion of the glands. 2 There are no reports to date of abnormalities associated with the bulbo-urethral glands in the tom.
Collection of an ejaculate
Semen collection is an important diagnostic tool to assess the reproductive health and potential fertility of a tom. Semen collection and analysis should be performed prior to the tom being used for breeding for the first time and when the fertility of the tom is questioned (eg, no pregnancies or low litter size).
Artificial vagina
The best sample is obtained by allowing the tom to ejaculate naturally into an artificial vagina (AV). The AV is most commonly constructed using an Eppendorf tube and a rubber pipette bulb. The male is allowed to mount a queen, and the AV is held in place to facilitate and collect the ejaculate. If the male has been adequately trained to the AV, and there is no queen in estrus available, the male may be allowed to mount a gloved arm or ‘dummy’ such as a stuffed animal that has been housed with queens in estrus. The advantages of using an AV are that semen collection can be performed readily in the unanesthetized tom and it allows a complete ejaculate to be obtained. Dis -advantages are the requirement for training and the frequent necessity for a teaser queen.
In one study, training sessions were scheduled three times weekly for 20 mins. 9 The authors reported a mean of 3.9 months (1.5–5.5 months) before the first ejaculate was produced. Another more recent study used 17 toms for training. 10 Training sessions lasted under 30 mins each and involved exposing the tom to a female in estrus. If collection was not successful, another attempt was performed 2–7 days later. The authors reported a success rate of 85% following an average of 3.8 ± 3.1 training sessions. Training was more successful when conducted by an experienced collector.
Collection using an AV is an excellent method for situations where semen is collected from a single male or group of toms on a regular basis, such as in a cattery or research colony, but is impractical for a single evaluation in a clinical setting using an untrained tom. Factors that may affect the ability of a tom to be trained to an AV are experience of the collector, the overall libido of the cat and aggression to other cats. 10
Electroejaculation
Electroejaculation (EEJ) is the most consistent method of obtaining an ejaculate from a tom that is not trained to an AV. The procedure requires general anesthesia. This author’s preferred anesthesia protocol includes dex-medetomidine (30-40
Catheterization
In a clinical setting, the most practical method to obtain a semen sample is urethral catheter-ization. This does not require extra equipment or a trained tom and can be performed in general practice. The procedure was first described by Zambelli and colleagues in 2008. 13 Cats are heavily sedated with either medetomidine (130–140 µg/kg IM) or dexmedetomidine (65–70 µg/kg IM). Once the sedation has taken full effect (10–20 mins), an open-ended tom cat catheter is inserted 8–9 cm into the urethra, taking care not to enter the bladder. Once inserted, the catheter is then immediately removed. The sperm located within the urethra will enter the catheter by capillary action and can be collected. After removal, the catheter should be flushed with extender into a small tube and the sample analyzed for the presence of sperm.
Sperm collection is not always successful via catheterization so, if no sperm are collected using this method, it cannot be assumed that the cat is not producing sperm. it should be noted also that these high doses of α-2 agonists produce the best success for obtaining sperm using this technique, but if this protocol is con-traindicated in a patient for medical reasons, other sedation protocols have been tested. in one study, premedication with methadone (0.2 mg/kg) followed by a lower dose of intramuscular dexmedetomidine (5 µg/kg) and intravenous induction with propofol (to effect) consistently produced sperm samples (of a higher volume). 14 This protocol could be used as an alternative for cats that are affected by cardiovascular disease and cannot tolerate high doses of α-2 agonists.
Other techniques
Retroejaculation is reported to occur in the cat. 15 Therefore, after the tom breeds a female, a cystocentesis can be performed to identify the presence of sperm. Motility will be compromised, but this technique allows the confirmation of the production of sperm. Alternatively, the male may be allowed to naturally mate a female and sperm cells may be obtained from her by performing a saline wash of the vagina immediately after mating.
Evaluation of an ejaculate
Following collection, the exact volume of the ejaculate is recorded. This is best done using a pipette and measuring the volume in micro-liters The sample is then extended at least 1:1-1:4 immediately (slow, drop-wise addition, with mixing) with suitable media. This reduces the risk of the small volume evaporating in the collection tube and gives the veterinarian a larger volume to work with. For extender media, the author prefers making Ham’s F-10 with 25 mM of Hepes buffer, 1 mM of pyruvate and glutamine, penicillin/strepto-mycin/neomycin and 5% fetal bovine serum. An alternative that is commercially available is TEST yolk buffer (refrigeration media, Irving Scientific). Analysis of the motion characteristics in this extender may be more difficult due to the interface egg yolk droplets. The extender can be filtered or centrifuged prior to use to reduce the yolk droplets.
Motility (total and progressive) should be recorded by estimation under low power microscopy on a warmed microscope slide or using computer-assisted sperm analysis (CASA) calibrated for feline spermatozoa. Concentration is obtained using a hemo-cytometer at a 1:100 dilution.
Evaluation of sperm morphology is an integral part of semen evaluation. Normal feline spermatozoa have an ovoid head, distinct mid-piece and a long tail (Figure 11). Teratospermia in the domestic cat has been defined as fewer than 40% morphologically normal sperm. 16 The most common abnormalities observed are head and midpiece malformations and tightly coiled tails, indicating aberrant formation of the sperm within the testicle (Figure 12).

Normal feline spermatozoon (eosin nigrosin stain, x 100)
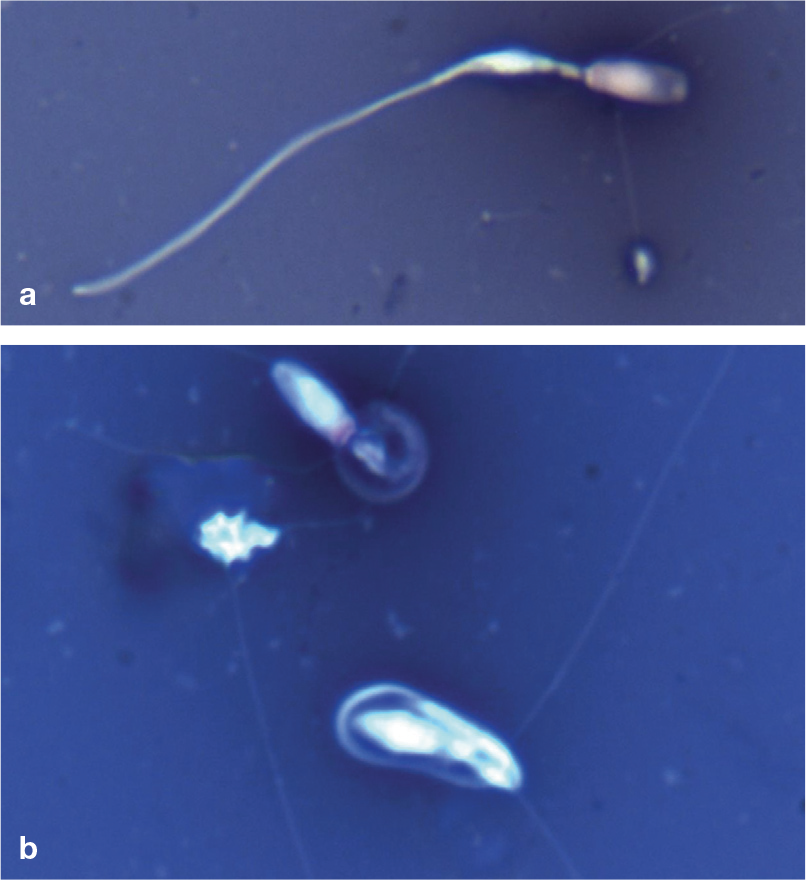
Feline spermatozoal abnormalities. (a) Abnormal head and midpiece (eosin nigrosin stain, x 100). (b) Two tightly coiled tails (eosin nigrosin stain, x 100)
Teratospermia is observed in small populations of cats where inbreeding or excessive line breeding has occurred, as reported by Pukazhenthi and colleagues. 17 More specifically, this research showed that a single generation of inbreeding (offspring bred to parent) produced male offspring with fewer than 15% morphologically normal sperm compared with 55% morphologically normal sperm in control animals, indicating that loss of genetic diversity leads to increased teratospermia in as little as one generation. 17 In other species, the number of morphologically normal sperm and progressive motility of the sperm are closely correlated. In the cat, many teratospermic ejaculates demonstrate adequate motility (>70%), in spite of low numbers of morphologically normal sperm. Although not always routine, other measured parameters of the ejaculate may include pH, osmol ality, membrane integrity, sperm chro-matin structure, bacterial culture or seminal plasma chemistry.
Breeding behavior
In a cattery, the male will establish his territory. The female (with few exceptions - eg, shy) should be brought to the male’s location for breeding. Once exposed, the male will approach and evaluate the behavior of the female. If the female is receptive, the male will bite the back of the neck and the female will assume an estrus position (crouched front end, elevated hindquarters with tail to the side) (Figure 13). The male will mount the female and spend several minutes getting into position and placing his penis near the vulva. Once intromission occurs, the male will thrust several times and ejaculate rapidly. The female emits a low growl that is followed by a louder yowl. Once ejaculation is complete, the male will release the female and move quickly away. As discussed in the accompanying review on the queen in this series, the female will show a typical ‘after reaction’ (rolling, licking the vulvar area, rubbing on objects), while the male remains distant. Once recovered, the male will remount and ejaculate again, typically many times per day.
Ovulation occurs within 36 h of the luteiniz-ing hormone (LH) release, which is triggered by sufficient stimulation from mating. Once she has ovulated, the queen will no longer accept the male. The female may be placed with the male for a period of time (days or weeks) until no further mating is observed or may be placed with the male for several hours on subsequent days until she no longer accepts the male. The latter allows for determination of a more accurate due date for the queen.
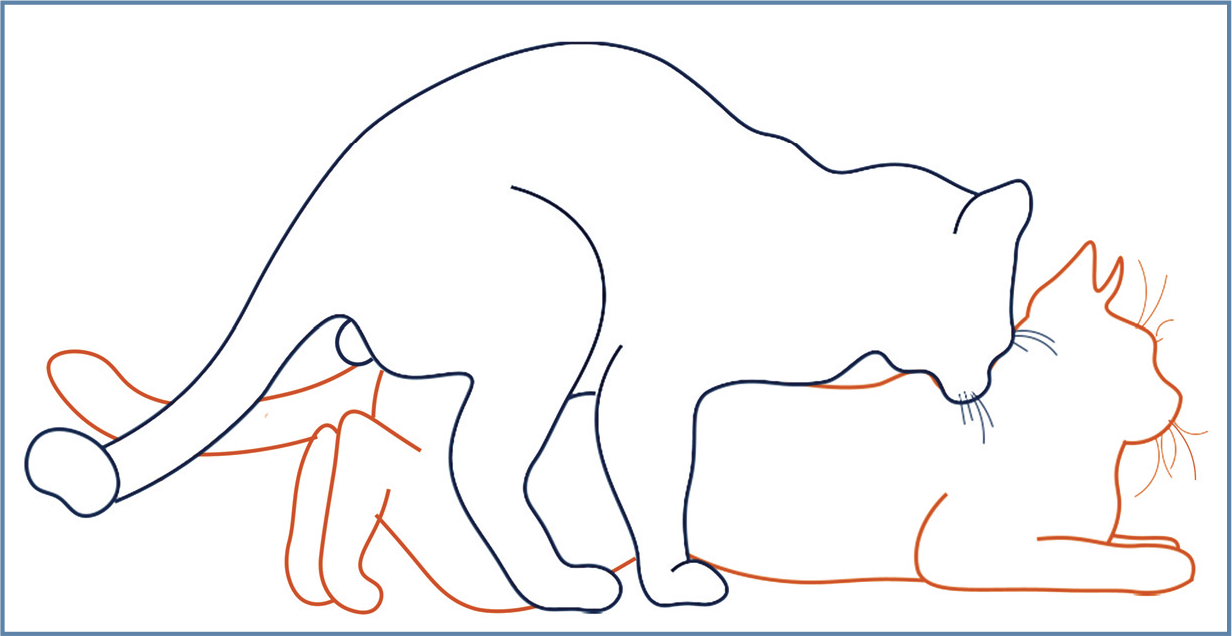
Diagram of the breeding position of the male and female. The female adopts a position of lordosis with elevated hindlimbs while the male bites the back of the neck to hold the female in place. Figure designed by Jamie M Douglas
Breeding management
Breeding age
It is recommended that a tom not be used for breeding until 1 year of age to allow time for him to mature physically and be able to produce adequate numbers of fully mature spermatozoa. However, many catteries will begin breeding males around the age of 9–10 months when the ejaculate sperm numbers may still be low. Because of multiple mating opportunities, these young males are often capable of producing a pregnancy. The presence of penile spines will confirm that the tom has reached puberty but does not guarantee that the tom is mature enough to produce adequate spermatozoa to achieve pregnancies.
Many males produce adequate sperm and remain fertile up to 10–12 years of age or older, although older toms do tend to have an increased incidence of age-related testicular degeneration. A decrease in sperm quality may occur at any point in the lifetime of a tom, and semen quality should be examined if infertility is suspected.
Breeding soundness examination
When a male will be used for breeding, a breeding soundness examination (see box) should be performed to ensure the tom has adequate semen quality to produce offspring. A breeding soundness examination is recommended each year to assess any changes in the tom over time. This examination is essential any time fertility is questioned; for example, when there have been missed pregnancies or a small litter size. A subfertile or sterile tom is disastrous in a cattery because mating the queen will cause ovulation but will not lead to a pregnancy. The queen will then enter a pseudopregnancy where cyclicity will not return until 40–50 days later. If this happens repeatedly, the queen is exposed to prolonged progesterone episodes without a pregnancy, risking endometrial hyperplasia and further reducing the queen’s fertility.

A thorough general physical examination should be performed to assess for any non-reproductive abnormality. This may include a minimum database including blood work (complete blood count and serum chemistry). Toms should be vaccinated according to current feline guidelines. A meticulous reproductive examination should consist of manual palpation of the testicles, assessing size, texture and symmetry, followed by ultrasound examination to check for any irregularities within the testicular parenchyma. As described earlier, measurements of the testicles should be taken, and length, width and height of each testicle compared. The penis should be evaluated for any discoloration, discharge and the presence of spines. Exteriorization of the penis may be difficult in the unsedated animal, so this portion of the examination is often best accomplished just prior to sperm collection when the tom is sedated or anesthetized.
Housing
Ideally, toms should be maintained in a separate location away from breeding queens. The presence of a tom near breeding females has been shown to increase the incidence of spontaneous ovulation in the queen. 18 In the author’s experience, a tom that has had exposure to a female in estrus may no longer tolerate group housing with other males and might require single housing. The demeanor and interaction of group-housed toms must be monitored after breeding to reduce the risk of inter-male aggression.
Genetic variability
Many catteries with purebred cats may have narrowed genetic lines in the hopes of producing that ‘perfect’ cat. The issue of reduced genetic variability can lead to severely impaired semen quality, usually terato-zoospermia. As an example, this has been demonstrated in the cheetah and Florida panther, with global population numbers for the latter dropping to fewer than 100 individuals before gradually increasing again. The lack of genetic variation in these two species has resulted in a high proportion of abnormal sperm within the ejaculates of the remaining males (up to 70% for the cheetah 17 and 90% for the Florida panther 19 ).
The coefficient of inbreeding (COI) should be determined for any potential breeding pair. The COI is a number between 0 and 1, where 0 indicates that the animals have no common ancestors and 1 is an exact genetic clone of the original animal. The COI can also be expressed as a percentage (0-100%). This parameter shows how genetically close the parents are to each other and therefore what to expect in the offspring. Many breeders cross related parents to select a certain trait (coat color, hair length, etc). A COI <0.05 (5%) is ideal. A COI >0.1 (10%) has been associated with increased rate of negative genetic traits and decreased offspring viability. When a high COI is already present in a given breed, mating animals with the lowest COI will help diversify the genetics throughout the breed. COI can be calculated using formulas published online. 20
In cats, it appears that despite the high degree of teratozoospermia and limited genetic variability, fertility can be maintained. Cats experiencing decreased numbers of normal spermatozoa may have a compensatory mechanism to overcome this default and maintain fertility. By increasing the number of copulations per female, the male increases the overall number of normal sperm available in the female. 17 It has also been shown that teratozoospermic cats experience an overall decrease in cell apoptosis (cell death) during the first two meiotic divisions of spermato-genesis and produce 30% more spermatids (early sperm cells) per spermatocyte than normospermic cats. 7
Key Points
✜ Management of a well-run cattery involves equal consideration of both the female and the male.
✜ Optimizing the health and genetic variability of cats within a breeding program will yield the best results.
✜ Many underlying causes of infertility can be a direct result of improper management of the breeding animals, including the males.
✜ Annual breeding soundness examinations are highly recommended in each male to assess changes in semen quality or testicular volume.
✜ By knowing what is normal for an individual male, minor changes in fertility or semen quality will be more obvious and can be addressed earlier.
Footnotes
Conflict of interest
The author declared no potential conflicts of interest with respect to the research, authorship and / or publication of this article.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
Ethical approval
This work did not involve the use of animals and therefore ethical approval was not specifically required for publication in JFMS.
Informed consent
This work did not involve the use of animals (including cadavers) and therefore informed consent was not required. No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
