Abstract
Objectives
The aims of this study were to evaluate and compare the effects that dexmedetomidine and methadone, either alone or in combination, have on the ocular variables of healthy adult cats when administered intramuscularly, as well as their reversal with atipamezole.
Methods
A randomized crossover blinded study of 10 healthy cats was used to assess the effect of 0.2 mg/kg methadone (MET), 7.5 μg/kg dexmedetomidine (D7), 10 μg/kg dexmedetomidine (D10), 7.5 μg/kg dexmedetomidine and 0.2 mg/kg methadone (DM7) and 10 μg/kg dexmedetomidine and 0.2 mg/kg methadone (DM10) on intraocular pressure (IOP), tear production and pupil diameter (PD). The animals were evaluated for 30 mins. Afterwards, atipamezole was administered and ocular variables were evaluated for 30 mins.
Results
D10, DM7 and DM10 significantly decreased mean IOP but MET or D7 did not. Tear production decreased significantly in all treatments, corresponding to 18%, 59%, 63%, 86% and 98% in MET, D7, D10, DM7 and DM10, respectively. PD increased in all treatments, but MET showed the highest PD. Thirty minutes after atipamezole (RT30), IOP returned to baseline with no difference between groups, and there was a significant increase in tear production, but the means were still different from baseline.
Conclusions and relevance
Dexmedetomidine decreases IOP and tear production but increases PD in healthy cats. Atipamezole can partly reverse those alterations. Low-dose dexmedetomidine (7.5 µg/kg) promotes sedation without changing the IOP. All protocols significantly decrease tear production, and Schirmer tear test after sedation is not representative of non-sedated values. Methadone induces quick onset mydriasis without changing the IOP.
Introduction
Ophthalmic abnormalities in cats are associated with numerous conditions, including infectious diseases,1–3 endocrine disorders4,5 and drug administration.6,7 Thus, patients with ophthalmic alterations, systemic disease and advanced age require accurate eye examinations.8,9
Most cats only require manual restraint for ophthalmic examination, but in fractious cats, sedatives and tranquilizers may be necessary, to improve examination performance. An appropriate protocol decreases stress associated with the eye examination and provides safety to both patient and examiner, causing minimal changes in ophthalmic variables. 10
Alpha(α)2-agonists provide adequate sedation for most purposes in cats, with the advantage of offering reversal with atipamezole. These agents can be used alone or in combination with an opioid.11–13 The association of an α2-agonist and an opioid seems to increase the sedative and analgesic effects with minimal further cardiovascular consequences.14,15
Dexmedetomidine is a synthetic α2-adrenergic receptor agonist with sedative and analgesic properties. 16 This drug also shows more selectivity for α2 receptors in comparison to α1, and greater potency than other drugs that make up the same group, such as xylazine and medetomidine.17,18
Although the effect of several drugs over the ophthalmic variables in dogs is already well known, there are few studies in cats, and these mainly evaluate the effects of multimodal protocols. Thus, this study aimed to determine the effects of intramuscular dexmedetomidine and methadone, either alone or combined, on the ocular variables of healthy adult cats, as well as their reversal with atipamezole.
Materials and methods
This was a prospective, randomized, crossover, blinded study. Ethical approval was granted by the local Committee on Ethics in the Use of Animals of the Universidade Federal do Paraná – Palotina (protocol 46/2019). The owners agreed to participate in the study and signed an informed consent form.
The study population comprised 10 neutered adult domestic shorthair cats. Inclusion criteria comprised ophthalmic variables within the reference interval (RI): Schirmer tear test (STT1; 9–34 mm/min), 19 rebound tonometry (12–32 mmHg) 20 and no alterations on direct ophthalmoscopy, systolic blood pressure (SBP), complete blood count and biochemistry profile (plasma protein, albumin, urea, creatinine, alkaline phosphatase and alanine aminotransferase). All cats were negative for feline immunodeficiency virus and feline leukemia virus.
During the study, the cats were housed individually under a standard controlled environment and received commercial dry food and water ad libitum. Before the study began, the cats were allowed to adapt for 7 days, being handled daily for 3 days to become familiarized with the procedures.
The study took place from 7.30 am to 12.00 am, under controlled humidity, temperature (24°C) and luminosity. Cat-friendly handling techniques were used to minimize stress. Food was withheld for 6 h before each study; water was provided ad libitum.
The study followed a double Latin square design (5 × 5) with repeated measures over time. Thus, all animals were subjected to five treatments, with a washout period of at least 7 days. One examiner performed all the examinations and measurements, and was unaware of the treatments.
All cats were randomly allocated to receive the following treatments, intramuscularly (IM): 0.2 mg/kg methadone (MET [Mytedom; Cristália Produtos Químicos Farmacêuticos]); 7.5 μg/kg dexmedetomidine (D7 [Dexdomitor; Zoetis]); 10 μg/kg dexmedetomidine (D10); 7.5 μg/kg dexmedetomidine + 0.2 mg/kg methadone (DM7); and 10 μg/kg dexmedetomidine + 0.2 mg/kg methadone (DM10). After 30 mins, sedation was antagonized with IM atipamezole, with two different doses, depending on the dexmedetomidine dose: D7 and DM7 – 37.5 μg/kg atipamezole (Antisedan; Zoetis); D10 and DM10 – 50 μg/kg atipamezole; MET – 0.1 ml/kg saline. Drug volume was standardized at 0.1 ml with saline.
The animals were evaluated at baseline (T0) and 5, 10, 20 and 30 mins (T5, T10, T20, T30) after drug administration, and 5, 10, 20 and 30 mins (RT5, RT10, RT20, RT30) after reversal. Intraocular pressure (IOP), pupil diameter (PD), non-invasive systolic blood pressure (SBP) and heart rate (HR) were evaluated at all time points. An STT1 was carried out at T0, T30 and RT30.
For the ophthalmic evaluations, cats were manually restrained in sternal recumbency, and for SBP measurement, in right lateral recumbency. All assessments were performed by the same trained researcher, following the same chronological order. Episodes of emesis and sialorrhea were recorded as adverse effects.
IOP was measured with a rebound tonometer (TonoVET), which was calibrated before data collection. Each IOP reading was obtained as the mean of five consecutive measurements from each eye, and the difference between readings was never higher than 5%. The average of each eye from an individual cat was considered separately in the statistical analyses.
Tear production was measured using Schirmer strips. A sterile tear strip was placed in the lateral half of the lower conjunctival sac of both eyes. The wetted length (mm) was measured after 60 s.
PD was obtained using a digital caliper under a fixed light. The caliper was manually adjusted according to the PD in the horizontal meridian.
Non-invasive SBP was measured by Doppler ultrasound (Parks Medicals Electronics). Cuff size was approximately 30% of limb circumference and was placed on the distal third in the antebrachium, in the left arm, at heart height. Earphones were used to minimize stress. Three consecutive measurements were performed, and the mean was recorded.
All statistical analyses were performed with GraphPad Prism 9. The right and the left eyes were considered separately. A D’Agostino–Pearson test was applied to check for normality. Parametric data were subjected to two-way ANOVA with repeated measures was used to analyze treatment and time effects, and interaction. When appropriate, Tukey’s multiple comparison test was applied to compare time within treatments and treatments within time. A χ 2 test was used for pairwise comparisons and Fisher’s exact test for categorical variables to correlate variance proportions between groups. Spearman’s correlation coefficient was used to assess the correlation between the results. A 0.05 level of significance (α) was established for analyses.
Results
A total of 10 cats (five castrated males and five spayed females) were included in the study. All animals were subjected to all treatments. Mean ± SD body weight was 3.5 ± 0.8 kg, and age ranged from 12 to 24 months (15 ± 3 months).
There were no difference in baseline values for IOP, PD, HR, SBP (Table 1) and STT1 among groups (Figure 1), and values were within the RI for the species and age.
Intraocular pressure (IOP), pupil diameter (PD) and cardiorespiratory variables, heart rate (HR) and systolic blood pressure (SBP), determined before drug administration (baseline) and 5 (T5), 10 (T10), 20 (T20) and 30 (T30) mins in 10 cats sedated with methadone and dexmedetomidine, and a combination of dexmedetomidine–methadone

Data are mean ± SD. Different uppercase letters indicate significance in the same row and different lowercase letters indicate significance in the same column (P ⩽0.05)
MET = 0.2 mg/kg methadone; D7 = 7.5 μg/kg dexmedetomidine; D10 = 10 μg/kg dexmedetomidine; DM7 = 7.5 μg/kg dexmedetomidine + 0.2 mg/kg methadone; DM10 = 10 μg/kg dexmedetomidine + 0.2 mg/kg methadone; bpm = beats per min

Boxplot (mean, SD, minimum and maximum) for tear production using Schirmer tear test (STT1) before (T0), after 30 mins (T30) of intramuscular methadone, dexmedetomidine and dexmedetomidine–methadone, and after 30 mins of reversal with atipamezole (RT30). MET = 0.2 mg/kg methadone; D7 = 7.5 μg/kg dexmedetomidine; D10 = 10 μg/kg dexmedetomidine; DM7 = 7.5 μg/kg dexmedetomidine + 0.2 mg/kg methadone; DM10 = 10 μg/kg dexmedetomidine + 0.2 mg/kg methadone
There was no significant differences in IOP between the left and right eye at baseline. A significant decrease in IOP was observed in D10, DM7 and DM10 but not in MET and D7 over time (Table 1). There was no difference between treatments. After reversal with atipamezole, IOP increased significantly in DM7 and DM10 (Table 2). Before reversal (T30), MET had the highest IOP when compared with the other treatments, but at RT5 all treatments returned to baseline with no significant difference between groups.
Intraocular pressure (IOP), pupil diameter (PD) and cardiorespiratory variables, heart rate (HR) and systolic blood pressure (SBP), determined before drug administration (baseline), 30 mins after protocols (T30) and 5 (RT5), 10 (RT10), 20 (RT20) and 30 (RT30) mins after reversal with atipamezole in 10 cats sedated with methadone and dexmedetomidine, and a combination of dexmedetomidine–methadone
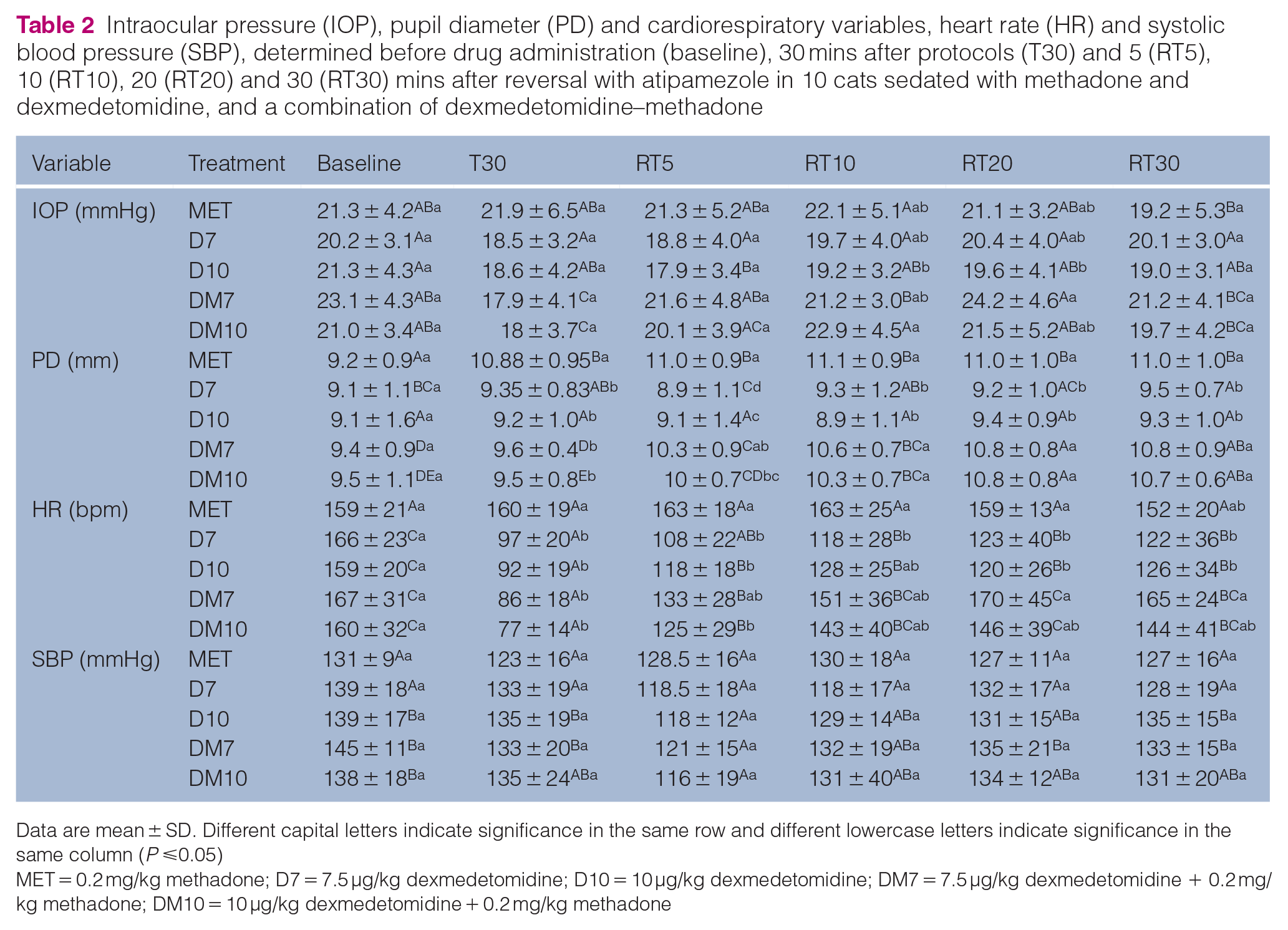
Data are mean ± SD. Different capital letters indicate significance in the same row and different lowercase letters indicate significance in the same column (P ⩽0.05)
MET = 0.2 mg/kg methadone; D7 = 7.5 μg/kg dexmedetomidine; D10 = 10 μg/kg dexmedetomidine; DM7 = 7.5 μg/kg dexmedetomidine + 0.2 mg/kg methadone; DM10 = 10 μg/kg dexmedetomidine + 0.2 mg/kg methadone
There was no significant difference in the STT1 between left and right eyes at any time point. Tear production progressively decreased in MET from T30 (14 ± 5 mm/min) to RT30 (11 ± 4 mm/min) vs baseline (19 ± 7 mm/min). In D7, D10, DM7 and DM10, tear production significantly decreased at T30 (7 ± 5, 6 ± 4, 2 ± 4 and 0 ± 1 mm/min, respectively). A drug and dose-dependent decrease in tear production was observed at T30: DM10 showed the lowest tear production (18/20 eyes had tear production of 0 mm/min); the combination of dexmedetomidine and methadone (DM10) showed significantly lower tear production than single drug dexmedetomidine (D10) or methadone (MET); and MET showed the highest tear production (Figure 1). After reversal with atipamezole, there was a significant increase in tear production at RT30, but the means were still different from baseline, with the lowest rate in DM10 (8 ± 7 mm/min) and the highest in D7 (12 ± 6 mm/min).
There was no significant difference in PD between eyes at any time point. PD significantly increased at T5 for all treatments. PD progressively increased in MET and showed significantly bigger PD compared with all other treatments. D7, D10, DM7 and DM10 caused an increase in PD, which peaked at T5, followed by a gradual decrease (Table 1). After reversal with atipamezole, PD progressively increased in DM7 and DM10 (Table 2). Comparisons between groups showed significantly increased PD in MET compared with D7, D10 and DM10 at RT5, but at RT10, RT20 and RT30, PD increased in DM7 and DM10, and was similar to MET.
HR decreased significantly with time after all dexmedetomidine treatments (D7, D10, DM7 and DM10) but not with MET (Table 1). At 5 mins of reversal with atipamezole (RT5), HR increased significantly in D10, DM7 and DM10 (Table 2). In D7 and D10, after 30 mins of reversal (RT30) the values still showed a significant difference from baseline. A combination of methadone and dexmedetomidine (DM7 and DM10) was associated with a steeper decrease in HR. However, after 30 mins of reversal (Table 2), HR was significantly higher in DM7 compared with D7 and D10. A moderate correlation was observed between HR and STT1 (r = 0.52).
No proposed treatment caused significant variation in SBP over time from baseline (Table 1). Reversal caused a significant decrease in SBP in D10 and DM7, although the means remained within the RI (110–132 mmHg) (Table 2). 21
Emesis occurred in 34% (n = 17/50) of the assessments performed in the D7, D10 and DM10 groups, seven in D7 and eight in D10. No emesis was observed in the MET and DM7 groups. No correlation was identified between vomiting and IOP alteration (r = 0.04).
Discussion
Low-dose dexmedetomidine and methadone provides adequate sedation with minimal adverse effects in cats.14,22,23 The doses used in our study were based on previous studies,14,23 with the aim of achieving light-to-moderate sedation without significant cardiovascular effects.
The study was conducted during a fixed period of the day, to avoid any possible interference associated with the circadian rhythm in IOP. 24 The cats were placed in sternal recumbency during data recording, to minimize alteration in IOP by body position. 25
In clinically healthy cats, the mean IOP ranges from 12 to 32 mmHg. 20 In the present study, baseline IOP was 21.4 ± 3.9 mmHg. IOP decreased significantly from baseline in 3/4 of dexmedetomidine treatments (D10, DM7 and DM10), but not with low-dose dexmedetomidine (D7) or methadone (MET), suggesting a dose-dependent and synergistic effect with methadone. 10 Despite the significant decrease in IOP in these treatments, means were within the RI. 26
In general, α2-agonists can decrease IOP due to the depressing sympathetic function and decreased aqueous production, 27 but a direct effect over α2-adrenoceptors in the eye has previously been proposed. 28 In small animals, the previous results vary due to species and drugs, showing a significant decrease in dogs29,30 and no effect in cats. 31 However, this is the first study to report a decrease in IOP secondarily to dexmedetomidine and dexmedetomidine–methadone in cats. Such divergence may be due to difference in measurement times, tonometer type (applanation and rebound) and experimental design.
Although blood pressure is positively correlated with IOP, 32 in this study, no correlation was observed between IOP and SBP (r = 0.16), suggesting that these effects are minimized by autoregulation of choroidal blood flow as SBP was maintained above 90 mmHg.32,33
Emesis is a possible side effect of dexmedetomidine and opioid administration, 34 having occurred in 17/50 assessments, mainly with dexmedetomidine as a single drug: 70% and 80% of cats in D7 and D10, respectively. Vomiting and nausea can be associated with obstruction of the central venous return or increased central venous pressure, which can increase choroidal blood volume and IOP.32,35 Although some evaluations occurred immediately after the emetic episodes, we did not identify a positive correlation between vomiting and IOP, suggesting that emesis may not increase IOP in healthy cats, or, more likely, that IOP increases during emesis but that the increase is of short duration.
Atipamezole reversed the decrease in IOP in all treatments. Thus, we believe that the decrease in IOP caused by dexmedetomidine is associated with the sedative or hemodynamic effects caused by the drug. The use of the α2-antagonist increased IOP to baseline values, probably by reversing the impact of the α2-agonist.
Methadone did not change IOP during the 60-min evaluation, similar to that reported in dogs. 36 Methadone was not reversed in this study. A transitory increase in IOP was reported previously in dogs and cats treated with methadone, but the presence of cats with corneal sequestration may have influenced their results. 37
Tear production decreased significantly in all treatments, corresponding to 18%, 59%, 63%, 86% and 98% in MET, D7, D10, DM7 and DM10, respectively, at T30 compared with baseline. This reduction in tear production was dependent on drug combination and dose, and may be associated with the decrease of the aqueous component of the tear film. 10 Despite the reported decrease in STT1 by dexmedetomidine in cats,38,39 the opioid combination has potentiated this effect in the present study, as observed in dogs. 10 These results might be attributable to the effects on autonomic regulation of tear production, 40 antinociception, 10 vasoconstriction at the tear gland10,38 or altered metabolism of gland cells, 10 but the exact mechanism remains unclear.
The cardiovascular effects of these sedatives may have affected tear production, as treatments related to more pronounced bradycardia showed a more intense reduction in tear production.
The decrease in tear production was partly reversed by atipamezole, but the results suggest that methadone association, if not reversed, can prolong this effect. This is the first study to report the effects of single-drug dexmedetomidine, methadone and atipamezole in tear production in cats.
Mydriasis occurred in all treatments and was related to either the α2-agonist or the opioid.41,42 The α2-agonist effect is mediated both by central inhibition of parasympathetic tone to the iris and direct sympathetic action of the α2-adrenoreceptors located in the iris, central nervous system or both.28,42,43 The methadone effect was even more pronounced, and possibly occurred through activation of mu opioid receptors. 44 The mechanism of mydriases induced by opioids in cats involves two main pathways: a peripheral component, which includes the release of catecholamines from the adrenal glands; 41 and a central component directly affecting the pupilloconstrictor neuron. 44
It is noteworthy that, despite the significant mydriasis, IOP did not vary, compared with mydriatic drugs, which can induce an increase in IOP in cats. 45 These findings suggest that methadone can be used to induce quick onset mydriasis in cats without changing IOP.
This study is not free of limitations. Invasive blood pressure monitoring would be more reliable, but catheter placement could cause stress and affect other variables. Furthermore, the weekly use of animals in the crossover design would make catheter placement more difficult. Additionally, a more prolonged period of evaluation would reveal the duration of methadone mydriasis in cats.
Conclusions
Dexmedetomidine or dexmedetomidine–methadone can potentially decrease IOP and tear production, and increase PD in healthy cats. Low-dose dexmedetomidine (7.5 µg/kg) promotes sedation without changing the IOP. All protocols significantly decrease tear production, and STT1 after sedation is not representative of non-sedated values. Low-dose methadone can be used to induce quick onset mydriasis in cats without significantly changing IOP.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals and procedures that differed from established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient. The study therefore had prior ethical approval from an established (or ad hoc) committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
This paper was handled and processed by the American Editorial Office (AAFP) for publication in JFMS
