Abstract
Objectives
This study investigated whether serum fibroblast growth factor (FGF)-23 concentrations are associated with serum total calcium (tCa) and blood ionised calcium (iCa) concentrations in cats with chronic kidney disease (CKD) and upper urolithiasis.
Methods
Serum samples and the medical records of cats with CKD with nephroliths, ureteroliths or both were investigated retrospectively. Cats with a serum creatinine concentration >250 μmol/l and/or a serum phosphorus concentration ⩾1.50 mmol/l were excluded. Based on cut-offs for serum tCa (2.70 mmol/l) or blood iCa (1.40 mmol/l), cats were divided into the following groups: total hypercalcaemia (H-tCa) (>2.70 mmol/l) and total normocalcaemia (N-tCa) (⩽2.70 mmol/l) groups, or ionised hypercalcaemia (H-iCa) (>1.40 mmol/l) and ionised normocalcaemia (N-iCa) (⩽1.40 mmol/l) groups, respectively. Serum FGF-23 concentrations were compared between groups and correlation analysis was performed.
Results
Thirty-two cats with CKD and upper urolithiasis were included. Serum FGF-23 concentrations in the H-tCa group (median 573 pg/ml [range 125–3888]; n = 12) were significantly higher compared with the N-tCa group (median 245 pg/ml [range 94–627]; n = 20) (P = 0.001). Serum FGF-23 concentrations in the H-iCa group (median 1479 pg/ml [range 509–3888]; n = 6) increased significantly compared with the N-iCa group (median 245 pg/ml [range 94–637]; n = 26) (P <0.001). Serum FGF-23 concentrations significantly correlated with serum tCa (r = 0.511, P = 0.003) and blood iCa concentrations (r = 0.425, P = 0.015) but not serum creatinine (r = 0.279, P = 0.122) or phosphorus concentrations (r = 0.208, P = 0.253).Conclusions and relevance Increased serum FGF-23 concentrations were associated with hypercalcaemia independently of creatinine and phosphate status in cats with CKD and upper urolithiasis.
Introduction
Chronic kidney disease (CKD) is a common cause of death in cats. 1 It is defined as a decreased glomerular filtration rate (GFR) and/or renal damage that persists for more than 3 months and is characterised as an irreversible and progressive condition. 2 Upper urolithiasis is one of the causes of CKD, and its prevalence in cats has been increasing recently.3–5 Calcium stones, such as oxalate calcium and phosphate calcium, have been reported to account for a large portion of feline ureterolithiasis. 3
Fibroblast growth factor (FGF)-23 is a phosphaturic hormone that has been identified as an early marker of mineral metabolic disturbances of CKD.6–8 In cats, plasma concentrations of FGF-23 increase as CKD progresses.9–12 The mechanism for elevated blood FGF-23 concentrations in patients with CKD is explained by a compensation for the accumulation of phosphate in the body, declining renal clearance of FGF-23 caused by decreased GFR and a compensatory response to decreased klotho protein in the kidney.6–8,13–15 A number of studies have investigated the association between FGF-23 and phosphate status,16–18 and in patients with CKD, phosphate accumulation in the body is thought to be an important contributor to increased blood FGF-23 concentrations. However, previous studies demonstrated that plasma total calcium (tCa) or blood ionised calcium (iCa) concentrations were significant variables that can be used to independently predict blood FGF-23 concentrations in cats.9,12 Furthermore, a recent study indicated that plasma FGF-23 concentrations increased significantly in cats in which plasma tCa concentration increased after the initiation of a renal diet. 19 However, the regulation system between blood FGF-23 and calcium concentrations has not been described. 16 Based on the findings of previous studies in cats,9,12,19 increased blood calcium concentrations can be associated with increased blood FGF-23 concentrations independently of phosphate status. In particular, in cats with upper urolithiasis, an assessment of calcium metabolism may be important, because the majority of feline ureteroliths have been reported to be composed of calcium stone. 3
The aim of our study was to investigate the association between blood calcium and serum FGF-23 concentrations in cats with CKD and upper urolithiasis. We hypothesised that increased serum FGF-23 concentrations are associated with hypercalcaemia in cats with CKD and upper urolithiasis.
Materials and methods
Case selection
Our study retrospectively investigated medical records and stored serum samples collected from client-owned cats that were diagnosed with CKD and nephroliths and/or ureteroliths between August 2015 and December 2020 at the nephrology service of an animal medical centre at Nippon Veterinary and Life Science University. CKD was diagnosed based on ultrasonographic renal abnormalities with or without renal azotaemia. Renal azotaemia was defined as a serum creatinine concentration >159 μmol/l (the upper limit of the reference interval [RI] at our institution) and urine specific gravity <1.035. Ultrasonographic renal abnormalities included a small-sized kidney, decreased corticomedullary differentiation and/or irregular renal contours. Abdominal ultrasonography was used to determine the presence of renal and ureteral stones, and we collected information from medical records.
The present study included only cats with stable CKD defined as serum creatinine concentration variations <25% within 3 months. Nephroliths and ureteroliths were defined as a hyperechogenic structure with an acoustic shadow in the pelvis or diverticulum of the kidney and ureter, respectively. 20 We classified cats using the 2019 International Renal Interest Society (IRIS) staging of the CKD guidelines based on the following serum creatinine concentrations: stage 1, <140 μmol/l; stage 2, 140–250 μmol/l; stage 3, 251–440 μmol/l; and stage 4, >440 μmol/l. 21 To minimise the effect of GFR and phosphate status on serum FGF-23 concentrations, we excluded cats with stage 3 and 4 CKD and/or a serum phosphorus concentration ⩾1.50 mmol/l from the current study. The serum phosphorus concentration cut-off value was based on the treatment aim recommended by the IRIS CKD guidelines. 22 Cats diagnosed with or suspected to have acute kidney injury, neoplasia or hyperthyroidism were excluded. In addition, we excluded cats that did not undergo blood gas analysis for measuring blood iCa concentration.
Review of medical records and data collection
We collected information on age, sex and neuter status, breed, body weight, serum biochemical analysis, blood gas analysis, abdominal ultrasonography, urinalysis, systolic blood pressure, diet and treatment regimen from the medical records. Serum biochemical analyses were performed using an automated analyser (7180 Biochemistry Automatic Analyzer; Hitachi High-Technologies). Blood iCa and bicarbonate concentrations were measured using a blood gas analyser (GEM PREMIER 3500; Instruments Laboratory). Blood samples for blood gas analysis were collected into heparinised tubes (Fuji Heparin Tube; Fujifilm) and measured within 10 mins of sampling. Abdominal ultrasonography was performed through a standardised protocol, by one veterinarian (YM), using an ultrasonic device (LOGIC S7 Expert; GE Health Japan) with a 3.4–10.8 MHz linear transducer (GE 11L Linear Probe; GE Health Japan). Urine samples were obtained via urethral catheterisation or cystocentesis. Urine chemical analyses were performed using an automated analyser (7180 Biochemistry Automatic Analyzer; Hitachi High-Technologies). Data for the urine protein:creatinine ratio of cats with bacteriuria or pyuria based on microscopic examination of urine sediment were excluded from the statistical analysis. We assessed blood pressure measurements using an oscillometric method (BP100D haemomanometer; Fukuda M-E Kogyo), following the recommendations of the American College of Veterinary Internal Medicine. 23 Information on serum intact parathyroid hormone (PTH) concentrations was collected from the medical records if the measurements were performed in cats with ionised hypercalcaemia to identify the causes of hypercalcaemia. Serum intact PTH concentrations were measured by chemiluminescent enzyme immunoassay (Siemens Immulyze intact PTH III; Siemens Healthcare Diagnostics) at an external laboratory (Fujifilm Vet Systems). Renal diet was defined as a food product for kidney disease fed to a cat that comprised >50% of its caloric intake.
Serum FGF-23 concentration assay
Blood samples for serum FGF-23 concentration analysis were collected with the informed consent of the cats’ owners. Blood samples in tubes containing serum separators were centrifuged at 1181 g for 5 mins, and the obtained serum samples were stored at −30°C for submission to an external laboratory (Fujifilm Vet Systems). Serum FGF-23 concentrations were measured using a sandwich ELISA (MedFrontier FGF23; Hitachi Chemical Diagnostics Systems) reported in a previous study. 12
Statistical analysis
We performed statistical analysis using a commercial software package (SPSS 24 for Windows, IBM Japan). Data normality was evaluated using the Shapiro–Wilk test. Student’s t-test or Mann–Whitney U-test were used for between-group comparisons. Total and ionised hypercalcaemia were defined as >2.7 mmol/l and >1.40 mmol/l, respectively, based on a previous report.24,25 Based on the cut-offs, cats were divided into H-tCa (serum tCa concentration >2.7 mmol/l) and N-tCa (⩽2.7 mmol/l) or H-iCa (blood iCa concentration >1.40 mmol/l) and N-iCa (⩽1.40 mmol/l) groups. We performed correlation analyses using Spearman rank-correlation coefficients. P values <0.05 were considered statistically significant.
Results
The serum samples of 86 cats with CKD were available for serum FGF-23 concentration measurements. Of the 86 cats, 46 were excluded because they were grouped in IRIS stage 3 and 4 CKD or did not have nephroliths or ureteroliths. In addition, three cats that did not undergo blood gas measurement analysis and five cats with a serum phosphorus concentration ⩾1.50 mmol/l were excluded. Therefore, 32 cats with IRIS CKD stage 1 or 2 and upper urolithiasis were included in this study. All cats with CKD were diagnosed based on renal ultrasonographic abnormality with or without renal azotaemia. None of the cats with CKD were diagnosed on the basis of the presence of proteinuria alone. Of these 32 cats, six had nephroliths, eight had ureteroliths and 18 had both nephroliths and ureteroliths. Six cats were grouped in IRIS stage 1 CKD and 26 cats were stage 2.The median age of the 32 cats was 6.7 years (range 1.3–11.8). Median weight was 4.3 kg (range 1.9–7.9). Breeds included domestic shorthair (n = 15), American Shorthair (n = 7), Scottish Fold (n = 4), Munchkin (n = 3), Russian Blue (n = 1), Maine Coon (n = 1) and Egyptian Mau (n = 1). There was one intact male, 19 castrated males, two intact females and 10 spayed females.Thirteen cats were treated with a renal diet. Six cats were fed a dry-format renal diet composed of 77.5 mg/100 kcal phosphorus and a calcium:phosphorus ratio of 1.9:1.0 (RENAL Feline Dry; Royal Canin Japan). The phosphorus content and calcium:phosphorus ratio of the renal diets of three cats were 100 mg/100 kcal and 1.8:1.0 (RENAL Selection Feline Dry; Royal Canin Japan), 116 mg/100 kcal and 1.4:1.0 (Hill’s Prescription Diet k/d Feline chicken dry; Hill’s-Colgate Japan) and 129 mg/100 kcal and 1.3:1.0 (Hill’s Prescription Diet k/d early support Feline chicken dry, Hill’s-Colgate Japan). Detailed information about the renal diets in the other four cats was unknown. Serum FGF-23 concentrations in the cats fed a renal diet (median 463 pg/ml [range 125–3888]) were significantly increased compared with those in cats not fed a renal diet (median 244 pg/ml [range 94–1783]) (P = 0.03). None of the included cats received phosphate binders.
According to serum tCa concentrations, 12 cats were placed in the H-tCa group and 20 in the N-tCa group (Table 1). Eight cats (67%) in the H-tCa group were fed a renal diet vs five (25%) in the N-tCa group. In the H-tCa group, three cats were fed RENAL Feline Dry, one was fed RENAL Selection Feline Dry and detailed information about the renal diets in the other cats was unknown. Serum FGF-23 concentrations in the H-tCa group (median 573 pg/ml [range 125–3888]) were significantly higher compared with those of the N-tCa group (median 245 pg/ml [range 94–627]; P = 0.02) (Table 1, Figure 1a).
Variables in the N-tCa and H-tCa groups
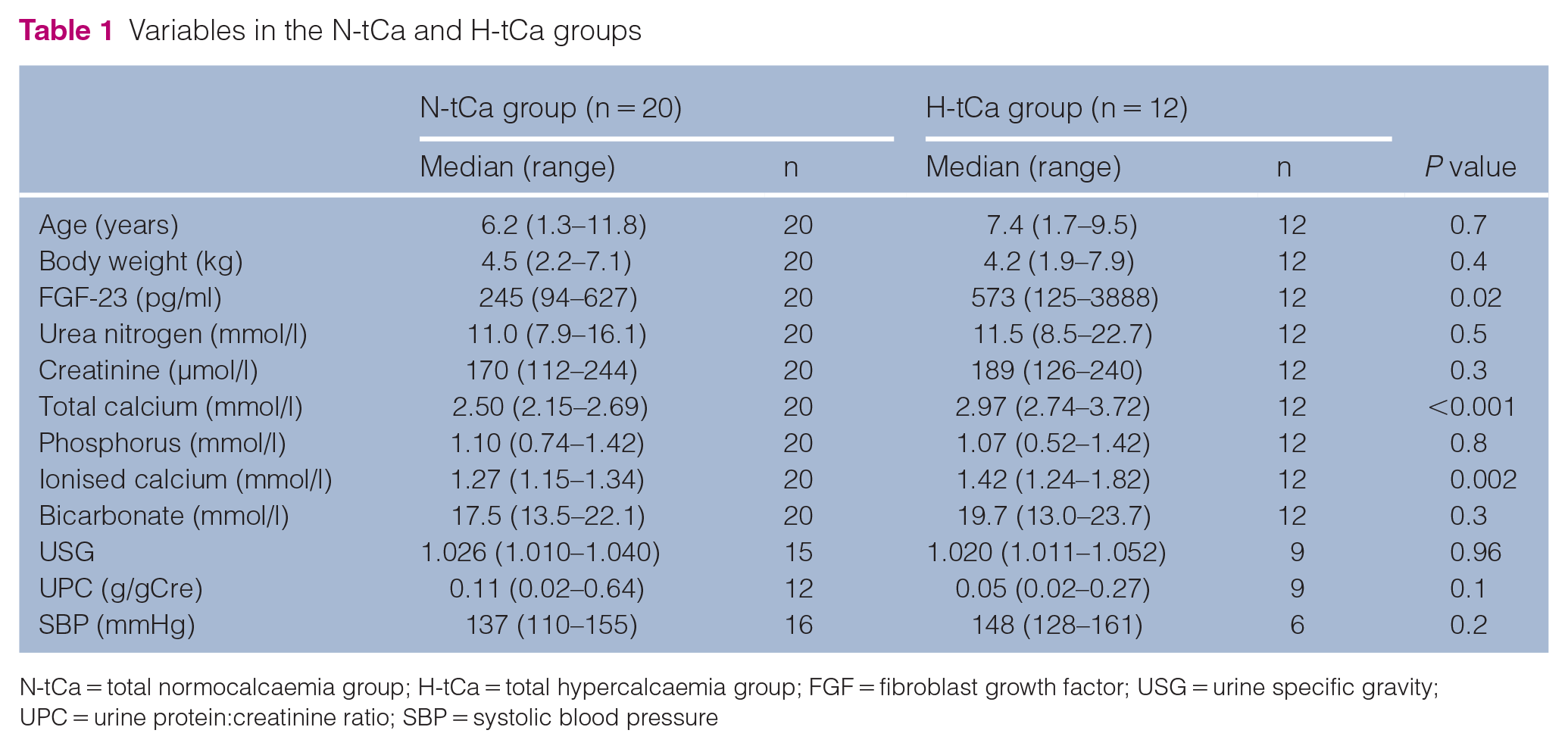
N-tCa = total normocalcaemia group; H-tCa = total hypercalcaemia group; FGF = fibroblast growth factor; USG = urine specific gravity; UPC = urine protein:creatinine ratio; SBP = systolic blood pressure

Serum fibroblast growth factor (FGF)-23 concentrations in cats with chronic kidney disease and upper urolithiasis. (a) Serum FGF-23 concentrations in cats with a total calcium (tCa) concentration ⩽2.70 mmol/l (N-tCa) and >2.70 mmol/l (H-tCa). (b) Serum FGF-23 concentrations in cats with an ionised calcium concentration (iCa) ⩽1.40 mmol/l (N-iCa) and >1.40 mmol/l (H-iCa). The boxes represent the 25th and 75th percentiles, and the central lines in the boxes represent the median values. The whiskers extend from the minimal to the maximal values. The dots represent each measurement. Significant values (P <0.05) are indicated with asterisks
Based on blood iCa concentrations, six cats were divided into the H-iCa group and 26 into the N-iCa group (Table 2). Five cats (83%) in the H-iCa group were fed a renal diet and eight (31%) in the N-iCa group received renal dietary treatment. In the H-iCa group, three cats were fed RENAL Feline Dry, and detailed information about the renal diets in the other cats was unknown. Five cats in the H-iCa group underwent measurement of serum intact PTH concentration. All serum intact PTH concentrations (median 5.8 ng/l [range 5.6–6.1]) were lower than the RI (8.0–25.0 ng/l) at the laboratory. Serum FGF-23 concentrations in the H-iCa group (median 1479 pg/ml [range 509–3888]) were significantly higher compared with those of the N-iCa group (median 245 pg/ml [range 94–637 pg/ml]; P <0.001) (Table 2, Figure 1b).
Variables in the N-iCa and H-iCa groups

N-iCa = ionised normocalcaemia group; H-iCa = ionised hypercalcaemia group; FGF = fibroblast growth factor; USG = urine specific gravity; UPC = urine protein:creatinine ratio; SBP = systolic blood pressure
Serum FGF-23 concentrations were significantly positively correlated with serum tCa concentrations (r = 0.511, P = 0.003) and blood iCa concentrations (r = 0.425, P = 0.02) (Table 3, Figure 2a,b). In addition, serum FGF-23 concentrations were significantly correlated with body weight (r = −0.349, P = 0.03) and urine specific gravity (r = 0.561, P = 0.005).
Univariable correlation analysis between fibroblast growth factor-23 and other variables


Scatter plots of serum fibroblast growth factor concentration vs (a) serum total calcium concentration and (b) blood ionised calcium concentration in cats with chronic kidney disease and upper urolithiasis
Discussion
Our study showed that in cats with CKD and upper urolithiasis, serum FGF-23 concentrations were significantly increased in the hypercalcaemic cats compared with normocalcaemic cats, and positively correlated with serum concentrations of tCa and blood iCa. To minimise the effect of GFR and phosphate status on FGF-23, we excluded cats with IRIS stages 3 and 4 CKD and/or serum phosphorus concentration ⩾1.50 mmol/l; therefore, serum FGF-23 concentrations were not associated with serum creatinine and phosphate concentrations in the population under study.
In humans, the detailed mechanism of the regulation of FGF-23 and calcium remains unknown. One factor that promotes the secretion of FGF-23 and an increase in blood calcium concentrations is calcitriol.16,26 Vitamin D supplementation can increase blood FGF-23 concentrations; however, our study did not include cats that received vitamin D therapy. In addition, PTH causes both increased blood FGF-23 and calcium concentrations.8,26 In patients with CKD, accumulation of phosphate into the body and decreased synthesis of calcitriol contribute to secondary hyperparathyroidism, resulting in hypercalcaemia. 8 Thus, one reason for the association between increased serum FGF-23 concentrations and hypercalcaemia is an indirect relationship through secondary hyperparathyroidism. However, increased serum intact PTH concentrations were not found in any of the tested cats (5/6). Thus, the association between increased serum FGF-23 concentrations and hypercalcaemia seen in our study cannot be explained by PTH.
In a study in mice, researchers reported that dietary calcium supplementation increased blood FGF-23 concentrations and mRNA expression of FGF-23 in bone, independently of vitamin D receptors. 27 A similar result was shown in another study using rats with calcium loaded by continuous-rate intravenous infusions. 28 These studies in rodents suggest that increased blood calcium concentrations directly elevate blood FGF-23 concentrations.27,28 In addition, a recent study in cats with CKD showed a significant increase in plasma FGF-23 concentrations in cats with increased plasma tCa concentration by renal diet. 19 At this time, increased blood FGF-23 concentrations in patients with CKD has been thought to be predominantly caused by reduced renal clearance via decreased GFR and the accumulation of phosphate in the body.13–15 For this reason, we included only cats with IRIS stage 1 and 2 CKD and serum phosphorus concentrations <1.50 mmol/l, in order to minimise the effect of GFR and phosphate status on serum FGF-23 concentrations. The results of our study suggest that hypercalcaemia is associated with higher serum FGF-23 concentrations independently of GFR and phosphate status in cats with CKD and upper urolithiasis. Therefore, increased blood calcium concentrations can directly increase blood FGF-23 concentrations in cats, as in rodents. Because our study was cross-sectional, we could not determine a causal relationship between increased serum FGF-23 concentrations and hypercalcaemia. Thus, future longitudinal studies are needed to investigate whether hypercalcaemia in cats is a risk factor for increased blood FGF-23 concentrations.
Upper urolithiasis is a cause and complicating factor of feline CKD, with increasing prevalence.3,4 Thus, its importance as a complication of CKD has grown. Previous studies have reported that most feline ureteroliths consist of calcium stones. 3 Hypercalcaemia can contribute to hypercalciuria and predispose to calcium urolithiasis. 5 Calcium oxalate stone has been reported to be the most common composition in feline ureteroliths, followed by mixed calculus of calcium oxalate and calcium phosphate. 3 FGF-23 is a phosphaturic hormone that enhances urinary phosphate excretion.6–8 Thus, increased blood concentrations of FGF-23 can promote the formation of calcium phosphate stones through excessive urine phosphate saturation. In fact, a previous human study reported that serum FGF-23 concentrations were significantly higher in cases with calcium nephrolithiasis and increased urinary phosphate excretion than in stone formers without urinary phosphate loss, and in humans without nephrolithiasis. 29 This study suggests that increased serum FGF-23 concentrations can be a risk factor for calcium nephrolithiasis. Our study showed that hypercalcaemia could be associated with increased serum FGF-23 concentration in cats. Therefore, increased serum FGF-23 concentrations resulting from hypercalcaemia may promote calcium and phosphate excretion into the urine and increase the risk of calcium phosphate urolithiasis in cats.
We believe the results of our study are important for the interpretation of increased serum FGF-23 concentrations in cats with CKD. Various studies have reported the benefit of a renal diet in cats with CKD, and dietary management is now commonly used for cats with CKD.2,30–33 Because increased plasma FGF-23 concentrations have been associated with a decreased survival time and increased progression of CKD in cats,10,34 FGF-23 can be a biomarker that predicts prognosis in feline CKD. This suggests that reducing the FGF-23 concentration in the blood can improve the prognosis of cats with CKD. In addition, a previous study reported that a renal diet in cats with CKD decreased plasma FGF-23 concentrations. 30 Therefore, FGF-23 may be useful as an indicator of phosphate restriction intervention in cats with CKD. However, our study suggests that hypercalcaemia can also contribute to increased serum FGF-23 concentration, independently of phosphate metabolic disorder in cats. It is known that a renal diet causes hypercalcaemia in some cats with CKD. 31 In addition, Geddes et al reported that attenuating dietary phosphate restriction improved ionised hypercalcaemia in cats with CKD. 35 The same research group reported that cats with lower plasma phosphorus and potassium concentrations had a higher risk of increased plasma tCa concentrations by renal diet. In addition, this research showed that in cats with increased plasma tCa concentrations after the initiation of a renal diet, plasma FGF-23 concentrations significantly increased. 19 Given the present and previous studies,19,31,35 FGF-23 cannot be used as an indicator of phosphate-restricted treatment in some cats with CKD (ie, cats with hypercalcaemia associated with renal diet). Hypercalcaemia caused by a renal diet can worsen calcium uroliths in cats. Thus, when cats with CKD and calcium urolithiasis have hypercalcaemia associated with a renal diet, attenuating phosphate restriction might be needed, even if the blood concentration of FGF-23 increases.
Our study had some limitations. First, the sample size, especially in cats with ionised hypercalcaemia, was small. Secondly, because the present study had a retrospective design, some data were missing. In particular, the serum intact PTH concentration in a cat with ionised hypercalcaemia was not available. Therefore, our study did not determine whether PTH was associated with the cause of hypercalcaemia in this cat. In addition, our study did not evaluate vitamin D metabolites. Vitamin D metabolites are needed to reveal the mechanism of the association between increased serum FGF-23 concentrations and hypercalcaemia. Moreover, because ureteroliths cause ureteral obstruction, which results in decreased GFR, the influence on GFR and serum FGF-23 concentration in cats with ureteroliths could differ from that in cats with nephroliths alone. However, because the present study included cats with stable CKD, ureteroliths were unlikely to influence the results. Thirdly, our study did not determine urolith constituents. Although the present study assumed the composition of the upper urolith in cats as calcium stone as described in the Discussion, cats with struvite or urate stones could have been included in the study.
Conclusions
In cats with CKD and upper urolithiasis, serum FGF-23 concentrations were significantly higher in those with hypercalcaemia compared with normocalcaemia. In addition, serum FGF-23 concentrations were significantly positively correlated with serum tCa and blood iCa concentrations but not with serum creatinine and phosphorus concentrations.
Footnotes
Acknowledgements
Conflict of interest
Yuichi Miyagawa is in receipt of speaker honoraria from Fujifilm Vet Systems.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
