Abstract
Objectives
The aim of the study was to evaluate cardiac size and early growth through echocardiographic, body weight (BW), body condition score (BCS), morphometric and biomarker changes in cats followed from 6 to 24 months of age.
Methods
Twenty-four female European shorthair colony cats were evaluated at birth for BW and at 6, 12, 18 and 24 months of age for BW, BCS, head length (HL) and head width (HW), N-terminal pro B-type natriuretic peptide (NT-proBNP), insulin-like growth factor-1 (IGF-1) and echocardiographic measurements.
Results
BCS, HW, left ventricular free wall in diastole, left atrium diameter and aortic diameter increased significantly between 6 and 12 months, while BW, HL and interventricular septum in diastole increased significantly between 6, 12 and 18 months, and BW decreased significantly between 18 and 24 months. NT-proBNP decreased significantly between 6 and 12 months. IGF-1 increased significantly between 6 and 12 months but decreased significantly between 12 and 18 months.
Conclusions and relevance
This study prospectively evaluated changes in echocardiographic measurements, BW, BCS, HL, HW, IGF-1 and NT-proBNP in cats during the first 2 years of life. Results show a comparable change over time for different variables. These findings contribute to the understanding of a possible relationship between cardiac measures and body size from young age through to adulthood.
Introduction
There is a possible interaction between body size and cardiac health in cats. Cats with hypertrophic cardiomyopathy (HCM) are skeletally larger (ie, larger heads, vertebrae and longer humeri) and heavier at diagnosis.1–4 They are also heavier at an early age than cats without HCM. 5 A potential mechanism for this interaction involves insulin resistance and/or the growth hormone–insulin-like growth factor (IGF)-1 axis, as binding of insulin and IGF-1 to their receptors on the cardiomyocyte stimulates myocardial protein synthesis and can cause ventricular hypertrophy.6–8 Some, but not all, studies have identified insulin resistance and elevated growth hormone or IGF-1 concentrations in cats with HCM,2,4,5,9 and cats with asymptomatic HCM can have higher body condition score (BCS), serum insulin and circulating cardiac biomarkers. 10
Multiple studies have identified associations between body weight (BW) and left ventricular measurements in healthy cats and cats with HCM.11–19 These studies had differences in study design, with primarily intact cats that were all or mostly adults, including single or different breeds, and different sex ratios. All these studies examined the cats at one single time point, and did not report BCS, making it impossible to evaluate how many of the cats in these previous studies were ideal BW or overweight/obese, which would identify possible confounded associations between BW and echocardiographic measurements. A previous study in cats with asymptomatic HCM showed significant associations between circulating cardiac biomarkers, echocardiographic variables, BW and BCS. 10
Programming is the process of long-term effects from a positive or negative event during a sensitive or critical period of development. More specifically, programming can result from early life experiences and impact the development of subsequent cardiac disease. Fetal programming has been shown in several animal and human studies, showing, among others, the effect of alterations in maternal nutrition on fetal growth and heart disease. 20 In cats, a possible relationship between growth and cardiac measures can be extrapolated from the associations between BW and left ventricular measurements at adult age, and the possible interaction between body size and cardiac health. Although previous studies have provided information on growth in cats with HCM 5 or left ventricular (LV) hypertrophy, 2 growth was evaluated retrospectively. One study of Maine Coon cats retrospectively collected information on BW at 6 and 12 months of age and showed that cats with HCM were larger at 6 and 12 months than cats without HCM. 5 Another study looked at the effect of growth on cardiac health at adult age. 2 Cats aged 3–7 years in a colony were retrospectively reviewed for BW at 6, 12 and 18 months of age, and underwent echocardiography, blood analysis and morphologic evaluation. In that study, 50% of cats had echocardiographic evidence of LV hypertrophy, which was significantly associated with head width (HW), BW, N-terminal pro B-type natriuretic peptide (NT-proBNP) and IGF-1 concentrations. However, echocardiography was only performed at a single time point with cats at different ages. Other limitations of the study were that BW was not available for all cats until 6 months of age, BCSs were not available during growth, and cats ate a variety of diets during growth and throughout adulthood. A prospective study evaluating BW, skeletal size, BCS and echocardiographic measurements might be a next step in better understanding the relationship between cardiac measures and body size from a young age through to adulthood. Therefore, the objective of this study was to prospectively evaluate changes in echocardiographic measurements, BW, BCS, morphological variables and circulating blood marker during the first 2 years of life in cats.
Materials and methods
Twenty-four female European shorthair cats from 11 different litters (1–5 cats per litter) were included in the study. The study was approved by the Royal Canin Ethics Committee and the Animal Use and Care Advisory Committee of Pays de la Loire (France; reference 01934.01). Cats were habituated to human contact and manipulation between birth and 6 months of age, and all cats were group housed in a colony in compliance with European Union regulations and were fed ad libitum. Cats were fed a growth/reproduction diet (Royal Canin Mother and Babycat; Royal Canin SAS) from birth until weaning, a growth diet (Royal Canin Kitten; Royal Canin SAS) from weaning until 10.5 months of age and a commercial adult diet (Royal Canin Neutered Young Male; Royal Canin SAS) from 10.5 to 24 months of age. All cats were neutered at 8 months of age. BW was recorded at birth. BW, BCS (9-point scale), morphometric measurements, echocardiography, and blood analysis for NT-proBNP and IGF-1 were obtained at 6, 12, 18 and 24 months. Measurements were performed in conscious cats with no sedation. Morphometric measures included head length (HL) and HW. HL and HW were measured according to previously described techniques. 4 Head measurements and BCS were performed by the same person at each time point. All cats underwent physical examination and echocardiography (two-dimensional [2D]-mode, M-mode and colour flow Doppler echocardiography [GE Vivid 7 Dimension; General Electric Systems], performed by a single board-certified veterinary cardiologist [DJC]) using a 7.5 mHz probe on Harmonic mode-octave at the highest frame rate available. Cats were scanned from beneath while in right lateral recumbency to obtain 2D-mode and M-mode images from right parasternal views. Loops were recorded of the right parasternal long-axis four-chamber view, the right parasternal long-axis LV outflow (‘five-chamber’) view, the right parasternal short-axis view at the level of the papillary muscles and the right parasternal short-axis view at the level of the aortic valve. M-mode images were guided from 2D-mode images of the right parasternal short-axis view at the level of the papillary muscles. 21
Measurements were made from recorded images. LV wall thickness measurements were made from 2D-mode and M-mode images. 2D-mode maximal LV wall thickness was measured on the first frame after mitral valve closure on the long-axis four- and five-chamber view or at the frame with the largest end-diastolic LV internal diameter in diastole (LVIDd) in the short-axis view at the level of the papillary muscles. A leading-edge to leading-edge method of measurement was used, being careful to exclude the pericardium, false tendons or papillary muscles. M-mode measurements were taken in a right parasternal short-axis view at the level of the papillary muscles using the leading-edge to leading-edge method. 21 At least three measurements were made of the thickest region identified for each view of the end-diastolic interventricular septum (IVSd) and LV free wall (LVWd), recording the largest repeatable value. All cats were assessed for focal wall hypertrophy from the right parasternal long-axis inflow and outflow views.
The size of the left atrium (LA) was assessed using two separate methods: using 2D-mode images from a right parasternal short-axis view to calculate the ratio of diastolic LA diameter to aortic root (Ao) diameter (LA:Ao) measured on the first frame after aortic valve closing and using a right parasternal long-axis four-chamber view to measure the diameter of the LA measured parallel with the mitral annulus in the last frame before mitral valve opening. 3 At least three measurements were made of each variable, recording an average value for each. The presence or absence of systolic anterior motion of the mitral valve was assessed on a 2D-mode right parasternal long-axis LV outflow view, using cine loop played back at reduced speed and by visualisation of characteristic colour Doppler flow.21–23 Simultaneous electrocardiographic monitoring was not possible owing to cat compliance. Unsedated blood pressure was measured by a single veterinarian in a quiet environment using Doppler technique, using the mean value of three separate measurements.
Blood was collected after food restriction for approximately 10 h at each time point for NT-proBNP and IGF-1. EDTA plasma was collected at specified time points and stored at −20°C for batch analysis. Analyses for IGF-1 (IGF-1 RIA CT; Mediagnost) and NT-proBNP (Feline CardioPet NT-proBNP; IDEXX Laboratories) were performed by commercial laboratories.
Statistical methods
Linear mixed models were used for modelling the effect of time (6, 12, 18 and 24 months) on heart rate (HR), blood pressure, the echocardiographic measurements (2D-mode LVIDd, 2D-mode IVSd, 2D-mode LVWd, Ao, LA, LA:Ao, M-mode IVSd, M-mode LVIDd and M-mode LVWd), BW, BCS, morphological variables (HL and HW) and blood markers (IGF-1 and NT-proBNP). NT-proBNP data were log-transformed for respecting model assumptions (normally distributed residuals and homoscedasticity). Tukey’s honestly significant difference (HSD) test was applied for multiple comparisons between time points and the level of significance was
Results were obtained in RStudio Version 1.1 (http://www.rstudio.com). Linear mixed models were calculated from the lme4 function of LmerTest package.
24
Tukey’s HSD test was applied from emmeans function from emmeans package (https://cran.r-project.org/web/packages/emmeans/emmeans.pdf, R package version 1.4.1). Results are expressed as median and range (minimum–maximum). Level of significance was set as
Results
Median birth weight was 0.125 kg (range 0.100–0.170 kg). All 24 cats completed the follow-up between 6 and 24 months of age. Changes over time in BW, BCS, HL and HW are described in Table 1 and Figure 1. BW continuously increased significantly between 6 and 18 months and decreased significantly thereafter between 18 and 24 months. BCS and HW increased significantly between 6 and 12 months; however, HL increased significantly between 6, 12 and 18 months. The prevalence of cats that were overweight or obese (ie, >5/9 BCS) was 38% at 6 months, 79% at 12 months, 88% at 18 months and 88% at 24 months.
Body weight (BW), body condition score (BCS), head length (HL), head width (HW) and blood markers at 6, 12, 18 and 24 months of age in 24 healthy cats

Data are presented as median (range). For each parameter, where there are significant differences (
IGF-1 = insulin-like growth factor-1; NT-proBNP = N-terminal pro B-type natriuretic peptide

Head width (mm) and head length (mm) at 6 (M06), 12 (M12), 18 (M18) and 24 (M24) months of age. The boxes represent median and 25th–75th percentile, bars represent 1.5 × 75th–25th percentile and points are values outside of these ranges
None of the cats had evidence of structural heart disease on echocardiography at any time point, and focal wall hypertrophy was not observed. Changes in HR, blood pressure (mmHg) and echocardiographic measurements over time are shown in Table 2. Blood pressure increased significantly between 6 and 12 months. HR decreased significantly between 6 and 24 months. 2D-mode LVWd, LA, Ao diameter, 2D-mode LVIDd, M-mode LVIDd and M-mode LVWd increased significantly between 6 and 12 months; however, 2D-mode IVSd increased significantly between 6, 12 and 18 months (Table 2 and Figure 2). Blood pressure was ⩾180 mmHg in one cat at 18 months of age and two cats at 24 months of age. No cat had a cardiac murmur at the ages of 6 and 12 months, one cat had a murmur (I/VI) at 18 months but not at 24 months and two cats had a murmur (I/VI) at 24 months of age. No cat had a gallop sound at 6 months, one cat had a gallop sound at 12 months but not at 18 months, and one cat had a gallop sound at 24 months of age.
Heart rate (HR), blood pressure (BP) and echocardiographic measurements (in mm) at 6, 12, 18 and 24 months of age in 24 healthy cats
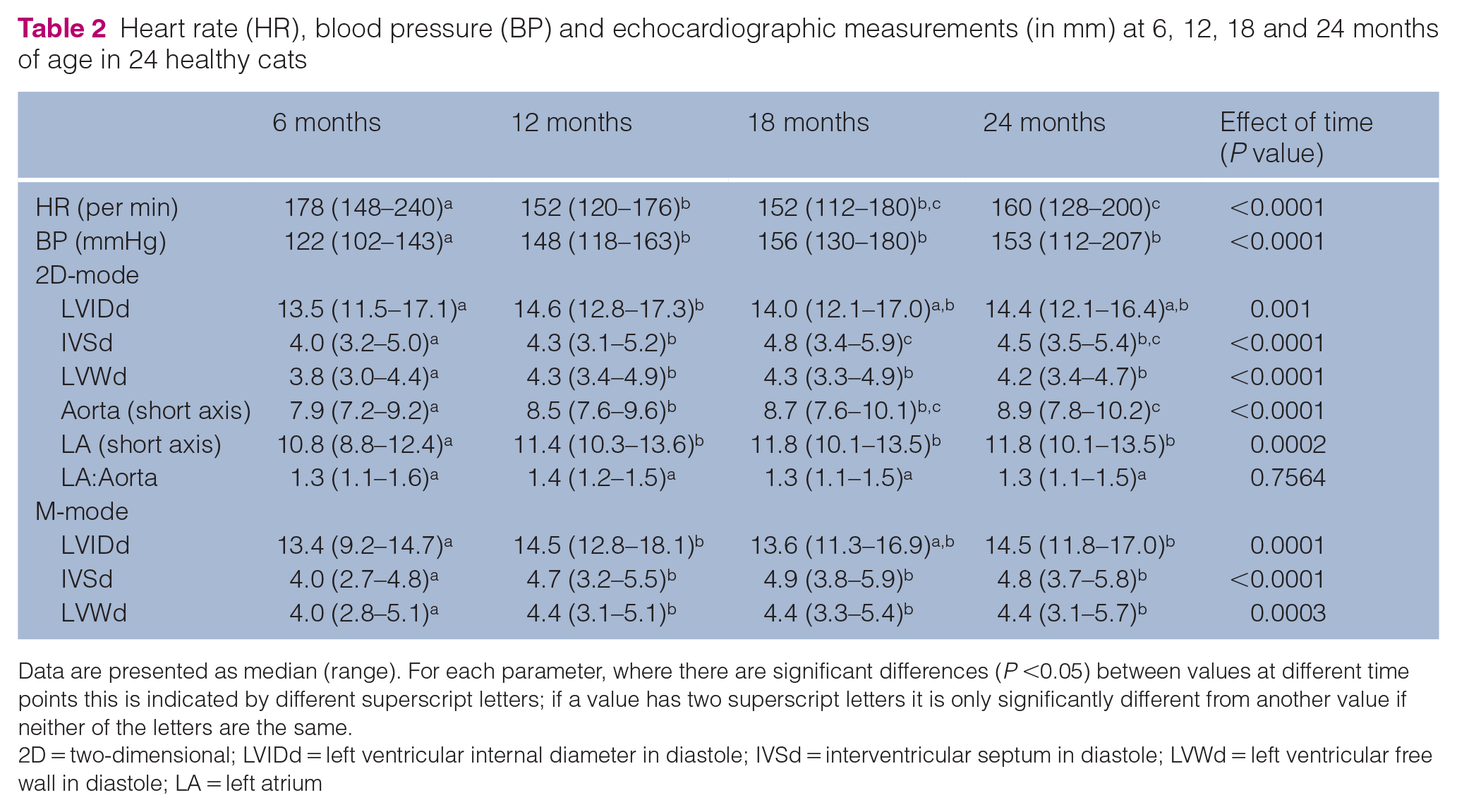
Data are presented as median (range). For each parameter, where there are significant differences (
2D = two-dimensional; LVIDd = left ventricular internal diameter in diastole; IVSd = interventricular septum in diastole; LVWd = left ventricular free wall in diastole; LA = left atrium

Two-dimensional (2D)-mode interventricular septal thickness in diastole (IVSd; mm) and 2D-mode left ventricular free wall thickness in diastole (LVWd; mm) in 24 cats at 6 (M06), 12 (M12), 18 (M18) and 24 (M24) months of age. The boxes represent median and 25th–75th percentile, bars represent 1.5 × 75th–25th percentile and points are values outside of these ranges
NT-proBNP concentrations decreased significantly between 6 and 12 months (

N-terminal pro B-type natriuretic peptide (NT-proBNP; pmol/l) and insulin-like growth factor 1 (IGF-1; ng/ml) in 24 cats at 6 (M06), 12 (M12), 18 (M18) and 24 (M24) months of age. The boxes represent median and 25th–75th percentile, bars represent 1.5 × 75th–25th percentile and points are values outside of these ranges
Discussion
The results of this study showed significant changes over time for echocardiographic measurements, BW, BCS, morphological measures and blood markers, although these changes did not follow the same process over time. LVWd, Ao, HW, BCS and IGF-1 increased until 12 months, with IGF-1 decreasing thereafter until 18 months. IVSd, HL and BW increased until 18 months, with BW decreasing thereafter until 24 months. NT-proBNP and HR were the only variables that decreased after 6 months, until 12 months of age and 24 months of age, respectively. Despite these different changes over time, some variables of both echocardiographic and morphometric measures, such as BW and BCS, did follow the same course of changes; these included LVWd and HW, and IVSd and HL. LVIDd, both by 2D-mode and M-mode, significantly increased between 6 and 12 months, although there was no significant change at 18 or 24 months compared with 6 or 12 months. The reason for this is unclear and might be due to measurement variability and/or power of the statistical analysis.
One previous study that excluded Maine Coon cats 4 identified an association between HCM and HW and HL, and a study in 28 cats of varying breeds (including four Maine Coon cats) only showed an association between LV hypertrophy and HW. 2 The results of the current study contribute to the hypothesis of a relationship between cardiac and body size, not only in cats with cardiac pathology but also in healthy cats. Because cats in the current study were only followed until 24 months of age and none of the cats had developed cardiac pathology at that age, it is unclear whether the observed changes over time for the different variables have a predictive value for the development of cardiac pathology later in life.
IGF-1 increased significantly until 12 months and decreased significantly thereafter until 18 months. Two previous studies in cats showed a significant association between IGF-1 and LV hypertrophy 2 or HCM, 5 and the results described here could contribute to the general understanding of the relationship between cardiac measures and body size and the mechanism behind this relationship. It can be hypothesised that an increasing IGF-1 concentration and/or variation in IGF-1 concentration have an influence on cardiac measures. To our knowledge, no studies have reported IGF-1 concentrations during growth in cats. However, in humans, serum IGF-1 concentrations increase during growth, with peak values at puberty. 28 In the current study, the highest mean IGF-1 concentration occurred at 12 months of age. None of the cats showed signs of hypersomatotropism, 29 and therefore acromegaly was not suspected in these cats.
NT-proBNP was the only variable that decreased over time, with a significant decrease until 12 months of age. It is important to note, however, that only two cats had NT-proBNP concentrations >100 pmol/l at any time point, and they were without cardiac abnormalities or an identified cause for elevated NT-proBNP. Previously, NT-proBNP was shown to be significantly associated with measures of cardiac size in cats with LV hypertrophy 2 or HCM.30,31 NT-proBNP is secreted from cardiac myocytes during cardiac myocyte stretch, pressure overload and in response to neurohormonal stimuli, 30 which are all processes that may intermittently occur during cardiac growth. While other studies of NT-proBNP have included at least some cats younger than 2 years of age,26,31,32 none has reported NT-proBNP concentrations for healthy cats during the first 2 years of life. The cut-off value of 100 pmol/l for detection of preclinical cardiomyopathy was determined in a study with cats classified as clinically healthy or with a different severity of HCM. The cats in the control group were assigned to different age groups (<3 years, 3–<7 years and >7 years). 26 The cats from the control group were significantly younger than the HCM groups, although age was not significantly associated with NT-proBNP concentrations. It is unclear whether the cats in the group aged <3 years had a comparable age to the cats described here, and whether the cut-off value of 100 pmol/l can be used to classify growing cats.
BW increased significantly until 18 months and decreased significantly until 24 months, while BCS only increased significantly until 12 months. BCS showed a non-significant decrease between 18 and 24 months, which might explain the reported decrease in BW. It should be noted that 88% of cats in the current study were overweight by the time they were 18 months of age, likely owing to the cats being fed ad libitum since birth. Therefore, the BW of a cat at 18 or 24 months of age does not necessarily reflect this cat’s body size and cardiac measures. Previous studies have shown associations between BW and LV measures in healthy cats,11,13,16–18 although the study described here is the first to examine cats at different time points in life and including BCS. BW, head size, NT-proBNP and IGF-1 have been associated with LV hypertrophy or HCM in previous studies.2,4,5 The association between obesity and LV hypertrophy has been described in humans, 33 dogs 34 and cats, 5 although it is still unclear whether this also exists for healthy cats with normal cardiac function.
One notable finding from the current study was wide variation in the cats’ BW, growth rates and BCS (Table 1), even though the cats had identical housing, handling and were all fed the same diet ad libitum. This may be due to genetic factors as cats were from 11 different litters, or owing to individual variability, although the sample size was too small to evaluate these factors in more detail. Male cats are predisposed to HCM.1,19,35 All cats in the current study were female so their risk of developing thickened cardiac walls may have been lower than in the general population. Studying the role of early growth and nutrition on the heart in a controlled situation is advantageous, although results would need to be confirmed in a home environment and in cats of different breeds and sex.
There are other important limitations to the current study. Most importantly, cats were only studied until 2 years of age so it is not known if any of these cats will develop HCM or LV hypertrophy later in life. Only 24 cats were included, which limited the possibility of performing multivariable analysis for associations between the complete set of variables in this study. Longer, longitudinal studies in a larger group of cats are needed to determine the relationship between early growth and the development of HCM or other cardiac pathologies over the course of cats’ lifetimes. Another limitation is the variability in obtaining and measuring ultrasound images, although all measurements were performed by a single cardiologist to reduce variability. Intra-observer variability of echocardiographic measurements was investigated by Chetboul et al, 36 showing that increased experience of the observer decreases the coefficient of variation of within- and between-day repeated measurements. The board-certified veterinary cardiologist (DJC) has extensive experience in performing echocardiography in cats, thereby limiting the possible influence of variability on the results. The results of this study may also not be generalisable to pet cats, given that these were of a single breed from 11 litters and were housed in a colony situation, within a controlled environment. However, the feeding situation is not unlike that in many households where cats are fed ad libitum. In one study of Maine Coon cats, the percentage of cats fed ad libitum was 89% during growth and 90% as adults. 5 Evaluations did not begin until 6 months of age, so very early differences in growth may have been missed and should be considered for evaluation in future studies. Nonetheless, although cats are clinically considered to reach maturity by 1 year of age, physeal closure of some long bones in the cat does not occur until as late as 25 months of age, so some growth is possible after 1 year. In fact, skull bones may fuse even later with the sphenoid, frontal, parietal and temporal bone not fusing until 2–4 years of age, 37 which could explain the increased head size between 12 and 18 months of age. In the current study, BW continued to increase until the 18-month time point, which could be partially due to continued growth. BCS increased only until 12 months of age; therefore, the increase in BW due to the development of obesity is less likely. Despite the evaluations starting only at the age of 6 months, this study is still the first to describe echocardiographic measurements in cats repeatedly examined at different time points in early adult life. Only one measure of skeletal size (ie, head size, as assessed by HL and HW) was used. 4 Other studies have looked at humerus length or vertebral size, which may provide useful information as it is likely to be less influenced by breed than head size.4,5 In addition, the cats in the current study were fed the same way with a diet that was changed at three time points during the study. Diet has been described to alter echocardiographic measurements in cats with HCM 38 and rodent models of cardiomyopathy;39,40 therefore, this could also have influenced the results of the study. Different diets or even keeping the cats on the same diet for the duration of the study may have yielded different results. A variety of other factors could have influenced the results, including genetic and epigenetic influences, as well as behavioural factors that could influence food intake and, therefore, growth.
Conclusions
This study prospectively evaluated changes in HR, blood pressure, echocardiographic measurements, BW, BCS, HL, HW, IGF-1 and NT-proBNP in cats during the first 2 years of life. Results show that cardiac measures, BW, BCS and morphometric measures follow a comparable change over time. These findings contribute to the understanding of a possible relationship between cardiac measures and body size from young age through adulthood.
Future studies including a larger number of cats and more time points earlier and later in life are needed to determine any potential relationship between early growth and echocardiographic measurements as indicators of the development of heart disease.
Footnotes
Acknowledgements
The authors wish to thank Professor P Nguyen and Mr S Ninet from Oniris, Nantes, France, for their support with this study.
Author note
This paper was presented, in part, at the 2015 ACVIM Forum, Indianopolis, IN, USA, and at the 2016 WALTHAM International Nutritional Sciences Symposium, Chicago, IL, USA
Conflict of interest
In the past 3 years, Dr Freeman has received research funding or provided sponsored lectures or consulting services for Royal Canin, Hill’s Pet Nutrition, Nestlé Purina PetCare and Aratana Therapeutics. Drs van Hoek and Laxalde are employees of Royal Canin SAS. Dr Rush has received research funding or provided sponsored lectures or consulting services for Royal Canin, Nestlé Purina PetCare, Aratana Therapeutics, IDEXX and Boehringer Ingelheim. Dr Connolly received payment from Royal Canin SAS for performing the echocardiography in this study.
Funding
This study was supported by Royal Canin SAS.
Ethical approval
This work involved the use of experimental animals and the study therefore had ethical approval from an established committee as stated in the manuscript.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedure(s) undertaken (either prospective or retrospective studies). No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
