Abstract
Objectives
Early diagnosis of arterial hypertension is essential to prevent target organ damage. In humans, retinal arteriolar narrowing predicts hypertension. This blinded prospective observational study investigated the retinal vessel diameters in senior and geriatric cats of varying systolic blood pressure (SBP) status and evaluated retinal vascular changes in hypertensive cats after treatment.
Methods
Cats with a median age of 14 years (range 9.1–22 years) were categorised into five groups: group 1, healthy normotensive (SBP <140 mmHg; n = 40) cats; group 2, pre-hypertensive (SBP 140–160 mmHg; n = 14) cats; group 3, cats with chronic kidney disease (CKD) and normotensive (n = 26); group 4, cats with CKD and pre-hypertensive (n = 13); and group 5, hypertensive cats (SBP >160 mmHg, n = 15). Colour fundus images (Optibrand ClearView) were assessed for hypertensive lesions. Retinal vascular diameters and bifurcation angles were annotated and calculated using the Vascular Assessment and Measurement Platform for Images of the Retina annotation tool (VAMPIRE-AT). When available, measurements were obtained at 3 and 6 months after amlodipine besylate treatment.
Results
Ten hypertensive cats had retinal lesions, most commonly intraretinal haemorrhages and retinal exudates. Arteriole and venule diameters decreased significantly with increasing age (–0.17 ± 0.05 pixels/year [P = 0.0004]; –0.19 ± 0.05 pixels/year). Adjusted means ± SEM for arteriole and venule diameter (pixels) were 6.3 ± 0.2 and 8.9 ± 0.2 (group 1); 7.6 ± 0.3 and 10.1 ± 0.4 (group 2); 6.9 ± 0.2 and 9.5 ± 0.3 (group 3); 7.4 ± 0.3 and 10.0 ± 0.4 (group 4); and 7.0 ± 0.3 and 9.8 ± 0.4 (group 5). Group 1 arteriole and venule diameters were significantly lower than those of groups 2 and 4. Group 2 arteriole bifurcation angle was significantly narrower than those of groups 1 and 3. Post-treatment, vessel diameters decreased significantly at 3 and 6 months in seven hypertensive cats.
Conclusions and relevance
Increased age was associated with reduced vascular diameters. Longitudinal studies are required to assess if vessel diameters are a risk indicator for hypertension in cats.
Introduction
Systemic arterial hypertension (SAH) in cats can lead to blindness and other target organ (the kidneys, brain, heart and vasculature) damage (TOD); therefore, successful management of this condition depends upon early identification, diagnosis and well-monitored response to treatment.1–4 Diagnosis is typically based on serial blood pressure (BP) measurements and regular monitoring.2,4 The American College of Veterinary Internal Medicine (ACVIM) guidelines recommend annual screening of cats and dogs aged 9 years or older. 2 However, various publications suggest that cats are typically presented late in their disease and already have signs of TOD.5–8
Ocular fundus lesions and pathomechanisms in hypertensive cats have been classified into three categories similar to the disease in humans: hypertensive retinopathy, hypertensive choroidopathy and hypertensive optic neuropathy.9,10 Near-constant retinal blood flow is maintained despite fluctuations in BP due to a phenomenon termed autoregulation, by which the vascular diameter and resistance are adjusted to maintain tissue perfusion.10,11
Ophthalmoscopically, early changes in the retinal vessels may be difficult to recognise and include vascular narrowing or beading and straightening of the arterioles due to immediate vasoconstriction to maintain tissue perfusion.1,10,11 As the retinal vessel walls become more damaged owing to the increased vessel wall pressure and vessel wall necrosis, further changes may include vasodilation, aneurysmal dilations, tortuosity and haemorrhages.1,9–11
Photograph-based methods for evaluation of the retinal vasculature through automated and semi-automated retinal analysis systems are widely used to diagnose SAH in humans. Generalised arteriolar narrowing has been associated with a high risk of SAH.12–15
The Vascular Assessment and Measurement Platform for Images of the Retina annotation tool (VAMPIRE-AT) is computer software designed for semi-automatic morphometric measurements of the retinal vasculature that has been used in various vascular conditions in people, including hypertension, coronary heart disease, stroke and diabetes.16–20 The use of this annotation tool has previously been described in a group of healthy and hypertensive cats in Italy and it was shown that arteriolar diameters were significantly narrower in the hypertensive group. 21
The purpose of the present study was to determine whether differences in retinal vascular diameters of senior and geriatric cats were associated with varying BP. If persistent hypertension is associated with retinal arteriolar narrowing, then measurement of arteriolar diameters might be useful in differentiating truly hypertensive cats from those with white coat hypertension.
Secondary outcome measures were used to evaluate the retinal vascular diameter changes of hypertensive cats after treatment with amlodipine besylate.
Materials and methods
Cats aged >9 years presented to the Royal Veterinary College’s (RVC) Beaumont Sainsbury Animal Hospital and Bow PDSA Pet Hospital (primary UK-based veterinary practices) were included in the study. Average BP readings were obtained with a sphygmomanometer and non-invasive Doppler detector (model 811-B; Parks Medical Electronics). 22 Cats underwent fundus photography, blood and urine testing (biochemistry, urinalysis and total thyroxine if hyperthyroidism was suspected clinically) every 6 months for healthy cats and every 4 months for cats with chronic kidney disease (CKD). Cats were considered azotaemic CKD if the plasma creatinine concentration was ⩾177 µmol/l in conjunction with a urine specific gravity <1.035 or plasma creatinine concentration ⩾177 µmol/l on two consecutive occasions 2–4 weeks apart. Ethical approval was granted by the RVC Ethic Committee (URN: 20131258). Cats suspected of hyperthyroidism were excluded.
Cats were classified into five groups according to their systolic BP status and risk of TOD based on the ACVIM guidelines. Group 1 contained normotensive cats (systolic BP [SBP] <140 mmHg); group 2 included pre-hypertensive cats (SBP 140−160 mmHg); group 3 cats had CKD and were normotensive; group 4 cats had CKD and were pre-hypertensive; and group 5 contained hypertensive cats (SBP >160 mmHg). 2 Cats were considered hypertensive if their SBP was >160 mmHg on at least two occasions 1–2 weeks apart, or they had a single SBP >160 mmHg in association with the presence of TOD. Age, sex, weight, medication and general health were recorded.
Colour fundus images were obtained without prior pharmacologic mydriasis using the Optibrand ClearView (1280 × 1024 pixels) fundus camera. Blood vessel diameters and bifurcation angles were measured by two annotators blinded to the cats’ identification, BP readings and clinical status.
When possible, hypertensive cats were also further evaluated at 3 and 6 months after treatment with amlodipine besylate 0.625–1.25 mg/cat q24h (Amodip; Ceva).
Image analysis
A photograph-based subjective assessment was initially performed recording the presence of vascular tortuosity, haemorrhages, retinal oedema, hyper-reflectivity, chorio-retinal changes and retinal exudates.
The VAMPIRE-AT (https://vampire.computing.dundee.ac.uk/) was provided by its developers (TM, ET). Instructions regarding the use of this annotation tool were available on the online platform, and its use in cats has been described elsewhere. 21
Briefly, an image centred on the optic nerve head was selected, followed by identification of the optic disc and the four standard measurement areas (SMA A, B, C and D), the annuli where summative width measurements are defined in most studies of the human retina (Figure 1).15,17 The vessel diameter of the dorsal venule and arteriole was measured at four locations (SMA A, B, C and D) equidistant from the border of the optic disc. Another assessor (OD) measured the bifurcation angles between the daughter vessels for both vessel types at the first branching point. The vessel diameter readings were averaged across the vessel segment for comparison. Comparison was also performed between the readings obtained at locations closest to and furthest from the optic disc (SMA A and SMA D, respectively).
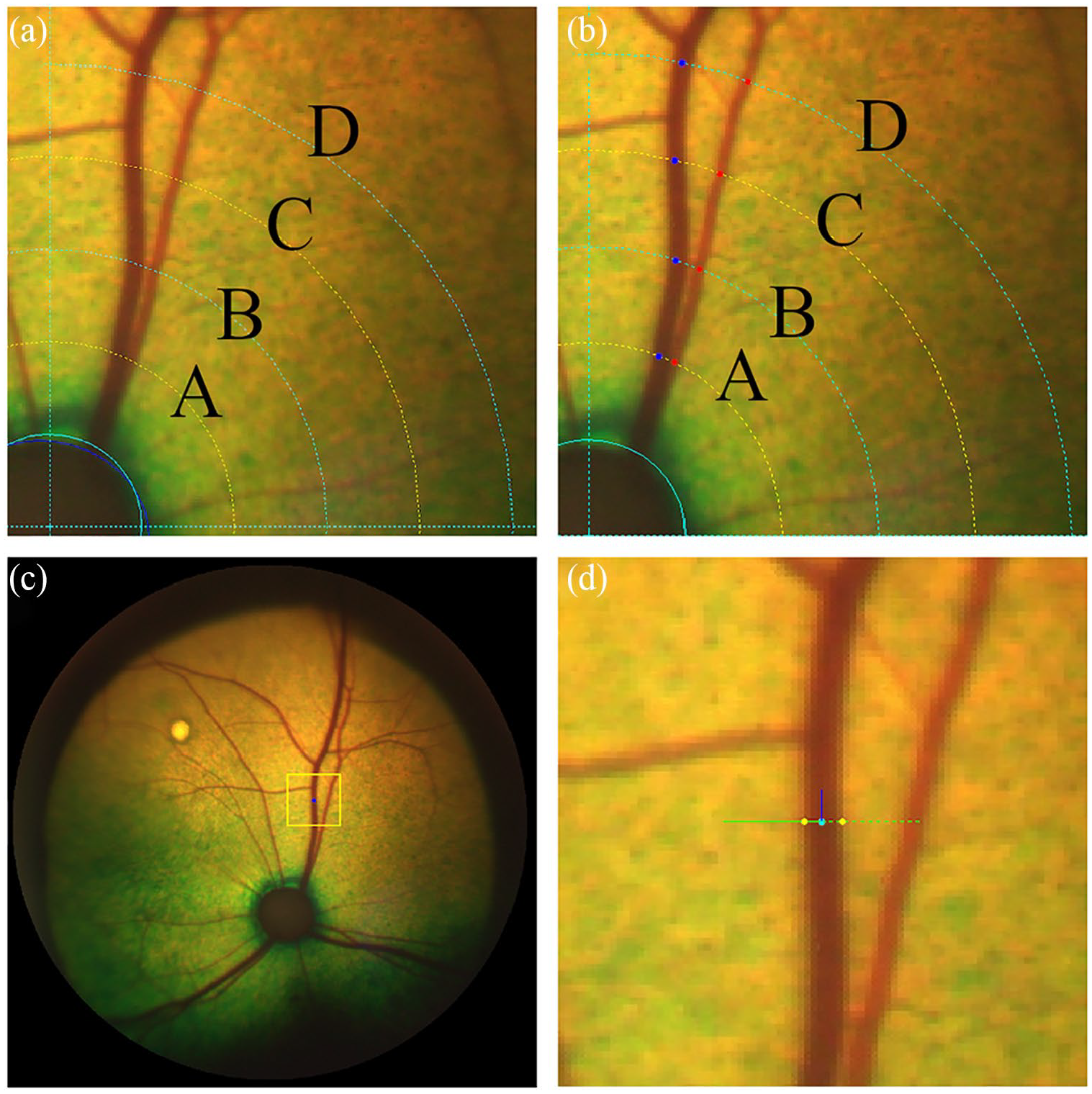
Annotation of vessel diameter using the Vascular Assessment and Measurement Platform for Images of the Retina annotation tool (VAMPIRE-AT). The circles in (a) and (b) show the retinal coordinates identifying standard measurement areas (SMA) A, B, C and D. (a) Manual outlining of the optic disc followed by generation of the five equidistant circles (defining SMA A, B, C, D). (b) Manual selection of the centre of the arteriole (red dots) and venule (blue dots) at four locations (SMA A, B, C and D) equidistant from the optic disc. (c) Zooming in on the area of interest to visualise the vessel segment. (d) Manual selection of the outline of the vessel walls; the blue line represents the direction of the vessel centreline, estimated at the green point (centre of vessel); the green line is perpendicular to the vessel direction; the yellow points indicate the estimated positions of the vessel borders and their distance is the estimated vessel width
Inter- and intra-observer reliability
Images from up to 10 randomly selected cats were used for intra- and inter-observer agreement assessment. In addition to the lead investigator (AE), two annotators (EJ and UD) recorded the vessel diameter of selected images at the same locations in order to evaluate inter-observer reliability. Annotators were all blinded to the cats’ BP readings and clinical status.
Data analysis
All analyses were carried out using R software version 3.6.1. Data obtained included BP status, age, breed, sex, weight, annotator, vessel type, eye, vessel diameter and bifurcation angle (Phi2). Linear mixed-effects models were used to evaluate differences in vessel diameters and bifurcation angles between the groups, vessel type and their interaction; cat ID was included as a random effect to account for the repeated measures. Fisher’s least significant difference was used for post-hoc comparison. Results were reported as means ± SEM. The significance level was set at 5%. The inter-class correlation coefficient and its 95% confidence interval (CI) in vessel diameter between the annotators were reported.
Results
Signalments of the 107 cats (82 domestic shorthairs [DSHs], 11 domestic longhairs, two Birman, two Siamese, two Maine Coon, two Persian and one each of Birmilla, British Shorthair, Burmese, Norwegian Forest Cat, Oriental Cat and Tiffany Cat) in the five BP groups are summarised in Table 1. One DSH cat was included in both groups 3 and 4 as its BP increased from normotensive to pre-hypertensive status during the study period. Normotensive cats were significantly younger than those in the other groups (P value ranged from <0.001 to 0.03), with a median age of 12.2 years (range 9.2−17.8 years). Cats with CKD and pre-hypertension (group 4) were the eldest with a median age of 16.3 years (range 10.1−22 years).
Descriptive statistics of age, body weight and sex among the five groups of cats
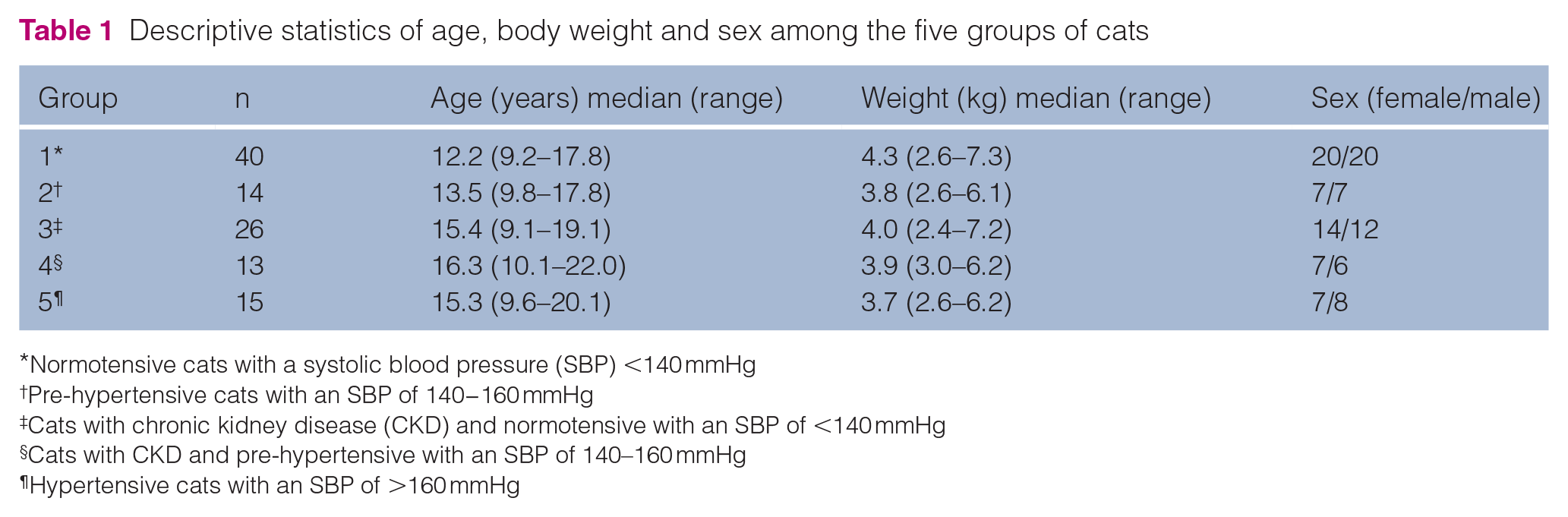
Normotensive cats with a systolic blood pressure (SBP) <140 mmHg
Pre-hypertensive cats with an SBP of 140−160 mmHg
Cats with chronic kidney disease (CKD) and normotensive with an SBP of <140 mmHg
Cats with CKD and pre-hypertensive with an SBP of 140–160 mmHg
Hypertensive cats with an SBP of >160 mmHg
Group 5 hypertensive cats had idiopathic hypertension (n = 9) and International Renal Interest Society stage 2 CKD (n = 6). There was no association between body weight and BP in any of the groups.
Intra- and inter-observer reliability
Intra-observer (AE) correlation was 0.80 (95% CI 0.62–0.89) between two repeated vessel diameter assessments that were performed 3 days apart. Inter-observer correlation coefficients were 0.73 (95% CI 0.62–0.81) between the annotators UD and AE and 0.77 (95% CI 0.44–0.89) between AE and EJ (Figure 2). EJ consistently underestimated the vessel diameters for both arterioles and venules vs AE.
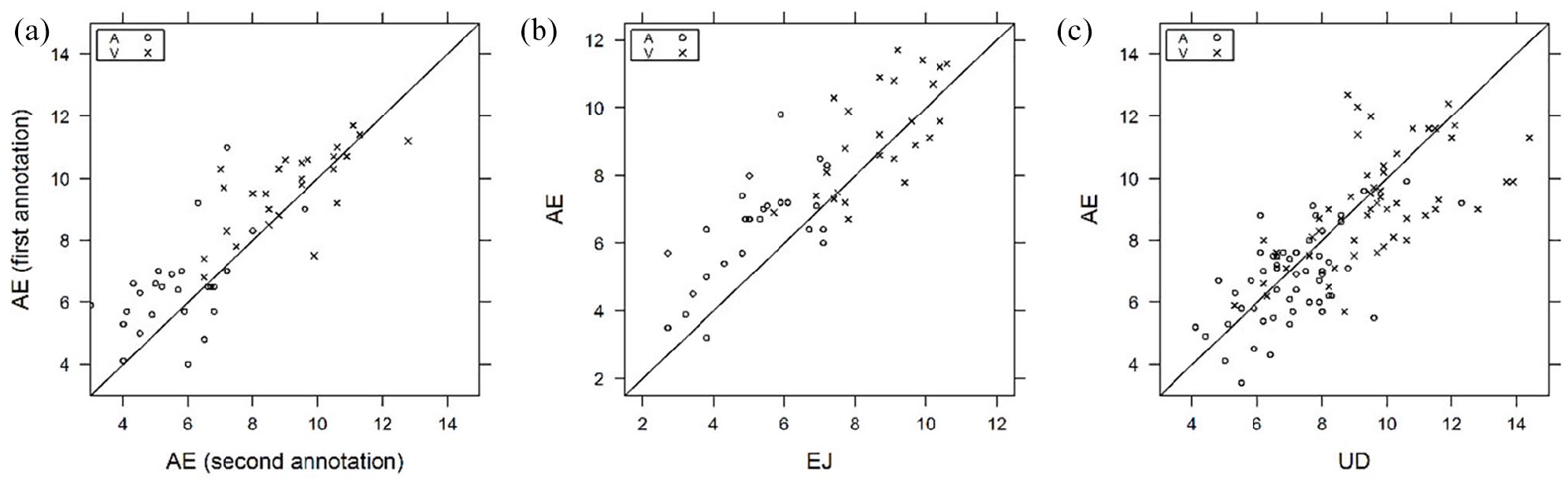
Graphs depicting the intra-observer agreement (AE, graph [a]) for 10 cats and inter-observer agreement for 9 and 10 cats, respectively, for retinal arteriole (A, open circles) and venule (V, crosses) diameters between annotators AE and EJ (graph [b]) and UD and AE (graph [c])
Vessel diameter
Fundus images (n = 3921) from 107 cats were assessed. The venule diameters were larger than the arterioles in all the groups (P <0.0001) (Figure 3). After correcting for age, group 1 arterioles were significantly smaller than those of groups 2 (P = 0.0007), 3 (P = 0.049) and 4 (P = 0.008) (Table 2). Group 1 cats had smaller venule diameters than those of group 2 (P = 0.009) and 4 (P = 0.015). There was no overall effect of weight on the arteriole or venule diameters (P = 0.2 and P = 0.45, respectively).
Age and weight corrected mean and SEM of the arteriole and venule diameters

Normotensive cats with a systolic blood pressure (SBP) <140 mmHg
Pre-hypertensive cats with an SBP of 140−160 mmHg
Cats with chronic kidney disease (CKD) and normotensive with an SBP of <140 mmHg
Cats with CKD and pre-hypertensive with an SBP of 140–160 mmHg
Hypertensive cats with an SBP of >160 mmHg
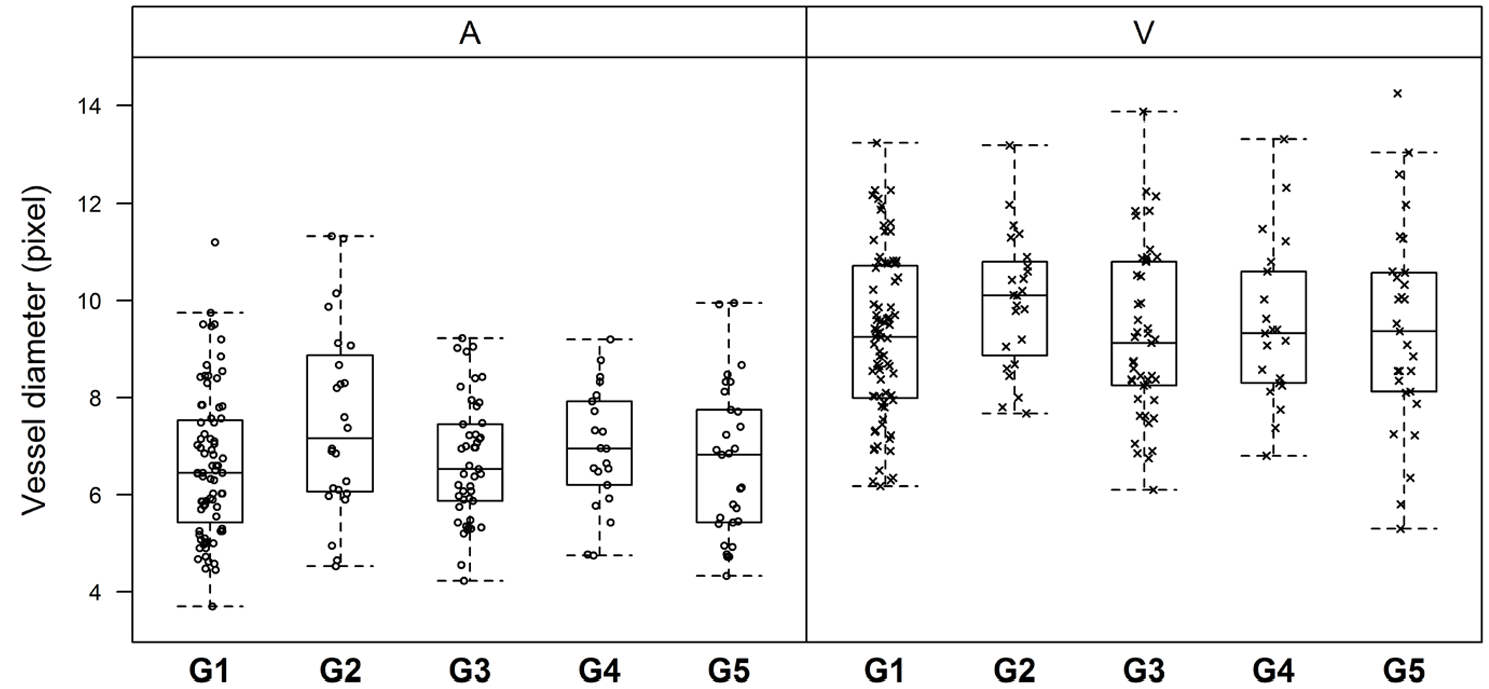
Box plots representing the distribution of the retinal arteriole (A, open circles) and venule (V, crosses) diameters (in pixels) in each group (group [G] 1 = normotensive cats with a systolic blood pressure [SBP] <140 mmHg; G2 = pre-hypertensive cats [SBP 140−160 mmHg]; G3 = cats with chronic kidney disease [CKD] and normotensive; G4 = cats with CKD and pre-hypertensive; G5 = hypertensive cats [SBP >160 mmHg])
Increased age significantly reduced both the arteriole and venule diameters by −0.17 ± 0.05 pixels/year (P = 0.0004) and −0.19 ± 0.05 pixels/year (P = 0.001), respectively (Figure 4).

Representation of cats’ averaged retinal arteriole (A, solid line, open circles) and venule (V, dashed line, crosses) diameters showing that vascular diameters were reduced with increased age
Group 1 venule diameters were significantly smaller compared with groups 2 (P = 0.0026), 3 (P = 0.047), 4 (P = 0.024) and 5 (P = 0.0091) at the SMA D location (Table 3). The group 1 diameters were significantly smaller compared with those of group 2 (P = 0.0036), and slightly smaller than those of groups 3 (P = 0.066), 4 (P = 0.032) and 5 (P = 0.044) at the SMA A. Furthest from the optic disc (SMA D), the group 1 arteriole diameters were significantly smaller compared with those of group 2 (P = 0.007).
Age and weight adjusted mean and SEM of the retinal arteriole and venule diameters closest and furthest from the optic disc corresponding to the standard measurement areas A (SMA A) and D (SMA D), respectively
Normotensive cats with a systolic blood pressure (SBP) <140 mmHg
Pre-hypertensive cats with an SBP of 140−160 mmHg
Cats with chronic kidney disease (CKD) and normotensive with an SBP of <140 mmHg
Cats with CKD and pre-hypertensive with an SBP of 140–160 mmHg
Hypertensive cats with an SBP of >160 mmHg
Bifurcation angles
Bifurcation measurements at the first branching point were available for 67 cats where the image quality allowed it (Figure 5 and Table 4). The angle between the daughter vessels varied widely, with no observed effects of age and weight. The venule bifurcation angles were not different between the groups. Overall, there were some differences in the arteriole bifurcation angles between the groups (P = 0.047), with group 2 angles significantly smaller compared with those of groups 1 (P = 0.0074) and 3 (P = 0.0052).
Age and weight corrected mean and SEM of the bifurcation angle of retinal arterioles and venules by blood pressure groups

Normotensive cats with a systolic blood pressure (SBP) <140 mmHg
Pre-hypertensive cats with an SBP of 140−160 mmHg
Cats with chronic kidney disease (CKD) and normotensive with an SBP of <140 mmHg
Cats with CKD and pre-hypertensive with an SBP of 140–160 mmHg
Hypertensive cats with an SBP of >160 mmHg

Distribution of the bifurcation angles of retinal arterioles (A, open circles) and venules (V, crosses) in each group (group [G] 1 = normotensive cats with a systolic blood pressure [SBP] <140 mmHg; G2 = pre-hypertensive cats [SBP 140−160 mmHg]; G3 = cats with chronic kidney disease [CKD] and normotensive; G4 = cats with CKD and pre-hypertensive; G5 = hypertensive cats [SBP >160 mmHg])
Amlodipine treatment
Seven of 15 hypertensive cats were re-evaluated at 3 and 6 months after treatment with amlodipine besylate. Of these, six cats had an SBP <160 mmHg after 3 months. Comparing pre- and post-treatment observations (Table 5), the arteriole and venule diameters were decreased at 3 and 6 months post-treatment (P = 0.03 for arterioles and P = 0.0009 for venules at 3 months, and P <0.0001 for both vessels at 6 months).
Mean and SEM of retinal arteriole and venule diameters among the seven hypertensive cats with 3- and 6-month post-treatment observations

Overall, vessel diameters of cats after amlodipine treatment approached the mean values of the normotensive cats. The venule diameters at SMA A and the arteriole diameters at SMA D were not different between groups (Table 6). When comparing the measurements closest to the optic disc (SMA A), the arteriole diameters were significantly reduced at 6 months after treatment (P = 0.026). Furthest from the optic disc (SMA D), the venule diameters were decreased at 3 (P <0.0001) and 6 months (P <0.0001).
Mean and SEM of the retinal arteriole and venule diameters closest to (standard measurement area [SMA] A) and furthest from (SMA D) the optic disc in the seven hypertensive cats with 3- and 6-month post-treatment observations

Owing to image quality, only six hypertensive cats had bifurcation angle data post-treatment (Table 7). Amlodipine treatment had no effect on the bifurcation angles both at 3 and 6 months post-treatment (P = 0.59 and P = 0.43 for arterioles and venules, respectively). Neither vessel type showed significant angular changes and large SEMs were observed for the bifurcation angles.
Retinal arteriole and venule bifurcation angle pre- and post-treatment observations for six hypertensive cats

Data are mean ± SEM
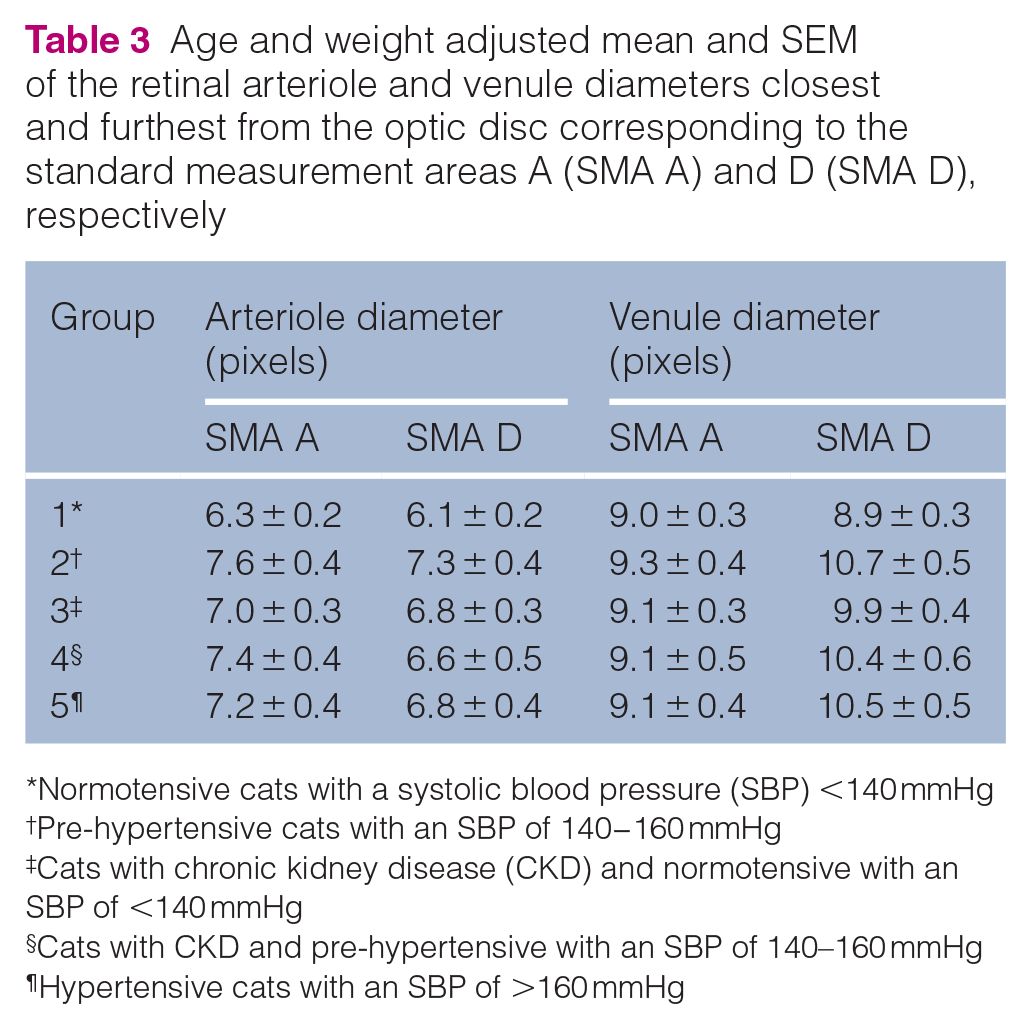
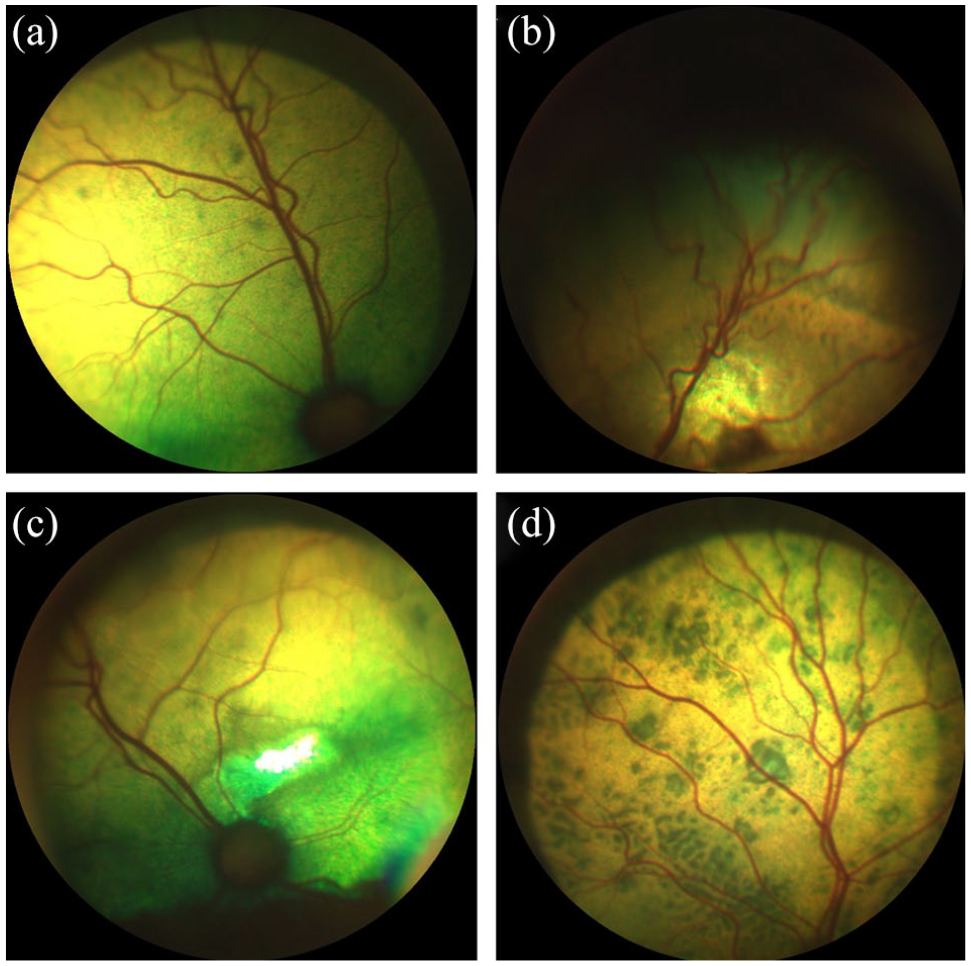
Retinal changes were present in 10/15 hypertensive cats (Table 8, Figure 6): arteriolar tortuosity; intra-retinal haemorrhages; retinal oedema; hyper-reflective areas; and multifocal subretinal exudates. Multifocal subretinal exudates (causing bullous retinal detachments) were also present in one group 2 and one group 3 cat (Figure 7). Intraretinal haemorrhages were also recorded in one group 1 cat (Figure 8), two group 2 cats and one group 4 cat. Arteriolar beading was observed in one pre-hypertensive group 2 healthy cat (Figure 9). Arteriolar tortuosity was also recorded in five group 1 normotensive cats (Figure 10), one group 2 cat, four group 3 cats and one group 4 cat. Two normotensive cats and one normotensive CKD cat had areas of retinal oedema.
Subjective photograph-based qualitative analysis of cats and the retinal changes observed in the five groups

Normotensive cats with a systolic blood pressure (SBP) <140 mmHg
Pre-hypertensive cats with an SBP of 140−160 mmHg
Cats with chronic kidney disease (CKD) and normotensive with an SBP of <140 mmHg
Cats with CKD and pre-hypertensive with an SBP of 140–160 mmHg
Hypertensive cats with an SBP of >160 mmHg
Qualitative analysis of the fundus images of hypertensive cats from group 5 showing a range of retinal changes. (a) Mild arteriolar tortuosity and focal chorioretinal pigmentary changes. (b) Intraretinal haemorrhage, arteriolar tortuosity and adjacent focal area of hyper-reflectivity. (c) Focal area of hyper-reflectivity dorsal to the optic disc and mild peripheral arteriolar tortuosity. (d) Multifocal chorioretinal lesions (hypertensive chorioretinopathy)
Multifocal subretinal exudates in (a) one pre-hypertensive healthy cat from group 2 and (b) one normotensive cat with chronic kidney disease from group 3

Intraretinal haemorrhage adjacent to the terminal end of an arteriole in a normotensive cat from group 1

Retinal arteriolar beading (boxcarring) in a pre-hypertensive healthy cat from group 2
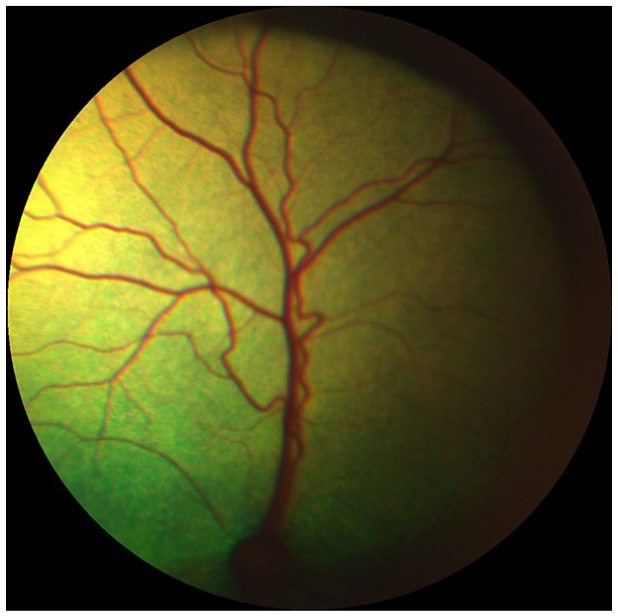
Marked retinal arteriolar tortuosity in a normotensive cat from group 1
Discussion
First, we showed that both retinal arteriole and venule diameters decreased significantly with age by −0.17 ± 0.05 pixels/year and −0.19 ± 0.05 pixels/year, respectively. Increased age and hypertension have been associated with arteriolar narrowing in people.23,24 However, an association between hypertension and reduced arteriolar diameters was not observed in this study population. In elderly people, the effect of hypertension on the retinal arteriole diameters is less marked than in the general population. This finding appears to reflect age-related structural changes in the wall of the arterioles (intimal thickening and medial hyperplasia, hyalinisation and sclerosis), which lead to increased rigidity, loss of elasticity, reduced arterial compliance and arteriolosclerosis.24–27 This concept may explain the lack of association between hypertension and retinal arteriolar narrowing in the present study due to a median age of 14 years in this feline population.
When adjusted for age and weight, the retinal vessel diameters in the normotensive group were narrower than the pre-hypertensive groups. The retinal arteriolar diameters of hypertensive cats were not significantly different from the normotensive cats, which contradicts the current literature. 21 The hypertensive group (n = 15) had a mean arteriole diameter and SEM of 7.0 ± 0.3 pixels, which was larger than the value (3.3 pixels) reported in the literature. 21 The hypertensive group from the previously reported study 21 had a larger and younger sample size (n = 45, median age 138 months [range 120−185 months]) than the hypertensive cats in this study. To add strength to the current study, all the annotators were blinded to the cat’s signalment, BP status and their affiliation to the treatment groups. The inter-observer correlation coefficient was consistent with a good repeatability between the three annotators, despite the reported difficulties in obtaining a clear visualisation of the blood vessel walls. Measurements of the same vessel segment performed by the same annotator at different time points were also consistent with a good correlation coefficient.
In humans, the retinal vascular diameter is computed on the basis of measurements carried out in a single, arbitrary selected point of the vessel for analysis. 15 However, symmetry of the measurements, and hence interchangeability of the right and left eye, still requires further studies in humans. 28 No significant differences in the retinal arteriole diameters were found between the left and right eyes in the present study, which is in line with the current literature. 21 The arteriole diameters were slightly smaller and venule diameters were slightly larger furthest from the optic disc. However, the normotensive healthy cats had smaller vessel diameters than the other groups, independent of the location of the measurements. The BP in one normotensive CKD cat progressed to the pre-hypertensive status within 3 months, and therefore this cat appeared in both groups 3 and 4. A longitudinal study would be required to evaluate if the cats with the smallest arteriolar diameters go on to develop hypertension to be able to demonstrate that arteriolar narrowing is a predictor of hypertension.
Computed-based methods are used to automate the analysis of retinal images in humans. The main VAMPIRE software tools (VAMPIRE 3.1 and VAMPIRE-WEB) showed excellent inter-observer reliability in several studies in humans.29–31 The VAMPIRE-AT provided for this study has been previously validated for use in cats and proved useful for objective evaluation of retinal vascular changes associated with hypertension. 21
One limitation of the study is that pixel measurements can vary with the image resolution, the refraction index and the clarity of the ocular media, which in humans play an important role related to magnification effects from the camera. 32 In images acquired by fundus cameras used currently in human ophthalmology (~3000 × 3000 pixels, 45° field of view), the camera resolution can influence the accuracy of the morphometric vascular measurements. 33
Variations in the vessel calibre may also be influenced by diurnal fluctuations of BP and physiological blood flow parameters such as oxygenation and shear stress. 34 A telemetric study of normotensive cats showed that BP generally does not vary much throughout the day and appears to be higher when the cats are active. 35
Ocular lesions have been reported in hypertensive cats with prevalence rates as high as 100%.6–8,36,37 Ten of 15 hypertensive cats had retinal changes of which intra-retinal haemorrhages and bullous retinal detachments were most common. Interestingly, arteriolar beading and retinal haemorrhages were also observed in pre-hypertensive and normotensive cats. Multifocal subretinal exudates were reported in a healthy pre-hypertensive cat and a normotensive cat with CKD. This suggests that hypertension-associated retinal changes may occur at lower BP levels or that cats may have experienced damaging BP spikes prior to their presentation. Vascular tortuosity was present in five normotensive healthy cats and four pre-hypertensive cats with CKD, with only two hypertensive cats having tortuous arterioles, suggesting that this phenomenon may not be associated with hypertension in the cat.
Although CKD is a common concurrent disease associated with increased BP, its direct effect on the retinal vessel diameters and angles in cats is currently unknown. It is also unknown whether CKD and SBP have independent effects on blood vessel measurements. Because the effect of anxiety on BP in cats is unpredictable, this potential measurement error makes it challenging to use measured SBP values directly in the analysis. Our classification of cats according to their SBP and CKD status reflected the disease diagnosis in clinics, and allowed the evaluation of diameters and angles variation as risk indicators for hypertension in cats.
A post-hoc comparison revealed a tendency of re-duced arteriolar bifurcation angles in the pre-hypertensive group vs the two groups with cats with an SBP <140 mmHg. Furthermore, the hypertensive cats had narrower bifurcation angles than healthy normotensive cats, which is consistent with human ophthalmology reports. 38 However, the prognostic value of identifying hypertensive cats based on the bifurcation angles may be small owing to the presence of a large variation even in the normotensive group and the small number of hypertensive cats.
As a calcium channel blocker, the amlodipine relaxes the tone of arterioles and causes a reduction in the arterial BP with a dilating effect on cardiac and vascular smooth muscle cells, thereby improving vascular resistance. 39 Hyperthyroidism causes profound cardiovascular changes and while hypertension does sometimes occur in this condition the major feature of thyrotoxicosis is a profound reduction in systemic vascular resistance. 40 Therefore, the reason for exclusion of the hyperthyroid cats was that in most hypertensive conditions one would expect vascular resistance to be increased, whereas in hyperthyroidism this is decreased. A vasodilation effect on the retinal arterioles has been reported in several studies in hypertensive humans.41–43 Such an effect was not observed in the hypertensive cats after treatment; on the contrary, the arteriole diameters were decreased at 3 and 6 months after treatment. Six of seven hypertensive cats had controlled BP and therefore responded to amlodipine treatment. With antihypertensive treatment, the retinal vascular diameters of hypertensive cats approached the values of the normotensive cats and therefore an effect on the vascular diameter was seen. The lack of vasodilation effect may be due to altered calcium ions transport in the retinal vascular smooth muscle cells or a counter-regulatory response that had masked such dilation.44,45
Conclusions
Using a computer-assisted imaging method to measure the retinal vessel diameters from photographs, we observed that the diameters of retinal arterioles and venules narrowed with increasing age. Although elderly cats had decreased vascular diameters, an association of vessel diameter with hypertension was not observed.
The VAMPIRE-AT may be useful to monitor the treatment effect on the retinal vasculature over time. Further studies are warranted to determine the prognostic value of assessment of retinal vascular architecture in the senior and geriatric cats for diagnosis of early hypertension.
Footnotes
Acknowledgements
The authors would like to acknowledge the Royal Veterinary College Geriatric Cat Clinic, Mrs Nicola Lotter and Dr Emily Jeanes for their contributions.
Author note
Preliminary results were presented, in part, at the 2019 European College of Veterinary Ophthalmologists conference in Belgium.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Ceva Santé Animale (2110.RES.4685).
Ethical approval
This work involved the use of non-experimental animals only (including owned or unowned animals and data from prospective or retrospective studies). Established internationally recognised high standards (‘best practice’) of individual veterinary clinical patient care were followed. Ethical approval from a committee, while not specifically required for publication in JFMS, was nonetheless obtained, as stated in the manuscript.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedure(s) undertaken. No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
