Abstract
Practical relevance:
Increases in cat ownership worldwide mean more cats are requiring veterinary care. Illness, trauma and surgery can result in acute pain, and effective management of pain is required for optimal feline welfare (ie, physical health and mental wellbeing). Validated pain assessment tools are available and pain management plans for the individual patient should incorporate pharmacological and non-pharmacological therapy. Preventive and multimodal analgesia, including local anaesthesia, are important principles of pain management, and the choice of analgesic drugs should take into account the type, severity and duration of pain, presence of comorbidities and avoidance of adverse effects. Nursing care, environmental modifications and cat friendly handling are likewise pivotal to the pain management plan, as is a team approach, involving the cat carer.
Clinical challenges:
Pain has traditionally been under-recognised in cats. Pain assessment tools are not widely implemented, and signs of pain in this species may be subtle. The unique challenges of feline metabolism and comorbidities may lead to undertreatment of pain and the development of peripheral and central sensitisation. Lack of availability or experience with various analgesic drugs may compromise effective pain management.
Evidence base:
These Guidelines have been created by a panel of experts and the International Society of Feline Medicine (ISFM) based on the available literature and the authors’ experience. They are aimed at general practitioners to assist in the assessment, prevention and management of acute pain in feline patients, and to provide a practical guide to selection and dosing of effective analgesic agents.
Keywords
Introduction
The feline population has grown significantly in recent years, with a current estimate of around 70 million pet cats in the USA and Canada.1,2 These individuals will require veterinary care and the majority undergo at least one surgery (ie, elective neutering) during their lifetime. Others will have medical conditions or trauma requiring hospitalisation, diagnostic examinations, intravenous treatments and palliative care. Effective acute pain management is required in most, if not all, of the above scenarios and must be an integral part of feline practice. Pain has a negative impact on feline welfare. It is a basic requirement that each feline practitioner performs pain assessment during every physical examination, in addition to recording the temperature, pulse, respiration (TPR) and conducting a nutritional assessment. This requires proficient recognition and management of pain in cats, aspects of which are species-specific.

Our knowledge of feline pain management has progressed over recent years. Review articles have been published on different aspects of pain management in cats.3–6 Moreover, there are now three scales with reported validity for acute feline pain assessment that take into consideration facial expressions and the cat’s unique behaviour.7–10 All three provide clinical guidance on the need for analgesia when a cut-off value is reached. However, rescue interventions may be administered even when the cut-off is not reached if the veterinarian feels that the cat could be in pain. The advent of valid pain assessment systems has improved our ability to recognise and assess pain in the clinical setting. It has also led to more robust research and clinical studies, and provided more objective outcome measures for approval of feline-specific drugs.
We no longer rely on extrapolation of scientific information from dogs for analgesic techniques and dosage regimens in cats. The published literature contains several feline-specific pharmacokinetic-pharmacodynamic studies involving analgesics. Opioids and nonsteroidal anti-inflammatory drugs (NSAIDs) with market authorisation specifically for cats are now available to the feline practitioner, and cat owners have come to expect safe and effective pain relief for their pets.11,12 Historically, however, feline pain management was neglected. 13 Thankfully this attitude has changed and advancements continue to be made, 14 but there are still areas for improvement. For example, pain assessment is not commonly performed in the clinical setting and validated pain assessment tools are not widely implemented in small animal practice. 15 Many veterinarians do not administer or prescribe analgesics to cats after discharge and following routine procedures (eg, neutering). 16
Here, the International Society of Feline Medicine (ISFM) provides its first guidelines on acute pain management using current evidence-based information and expert consensus. The Guidelines identify areas where critical research is needed and emphasise pain management as a key component of feline welfare. Throughout, the utility of a multi-modal and preventive analgesic approach is highlighted, involving non-pharmacological and pharmacological therapy, cat friendly handling techniques, and respect and empathy for feline patients.
Assessment and recognition of pain in cats
Principles of pain assessment
Cats are unique and so is their behaviour, especially when fear, anxiety and stress are present in the hospital setting. The assessment and treatment of pain is only one part of the pain management ‘puzzle’; every manipulation and physical examination should be performed using cat friendly handling techniques (Figure 1). 17 Pain management is not only about giving an analgesic drug: the emotional needs of the hospitalised cat must be considered (Figure 2), and the individual itself should always be treated with respect and empathy.

Feline friendly handling techniques should be used for every interaction with cats. This will ensure optimal healthcare by reducing the cat’s stress, improving personnel safety and providing a positive outcome for the patient, client and veterinary team. After observation of the cat’s normal behaviour, the patient is calmly approached and stroked, and the painful area is gently palpated. Image courtesy of Paulo Steagall

The hospital environment has an influence on the patient’s pain experience. A cat ward should be a quiet and comfortable area, considering the specific needs of this species. A hospital cage equipped with a cardboard box provides an elevated area where the cat can perch (‘a safe place’). Simple enrichment measures, such as this, can improve the hospital experience and reduce stress. Image courtesy of Paulo Steagall
Pain assessment is the first step towards appropriate treatment. Failure to recognise pain is likely the primary cause of inadequate treatment (oligoanalgesia; ie, undertreatment of acute pain). 16 Recognition of pain requires understanding of this species’ reactions within a hospital setting. Signs of pain may be subtle and misinterpreted; for example, when a cat simply reduces movement and ‘freezes’ due to fear. However, the veterinary care team and owners, too, can be trained to improve their pain assessment skills and several websites are available for this purpose.18–20 Used in isolation, physiological variables are not reliable in acute pain assessment, since they can be affected by disease, fear and stress.21–23 However, appetite may be a ‘pain indicator’, as painful individuals generally do not eat. 24
Overall, acute pain assessment is subjective, although subjectivity and bias can be reduced with specific guidelines and assessment tools (see later). In cats, and other animals, all assessments depend on the observations of a veterinary professional or carer and hence are termed ‘observer-related outcomes’. Assessment includes, but is not limited to, observation of posture, general behaviour, comfort with interactions, activity, attitude, body position and facial expressions. In conjunction with this, a dynamic and interactive approach is recommended, usually involving greeting the cat while performing gentle palpation of the wound and/or suspected painful area. This is an important step, otherwise evoked pain may be overlooked; it should be possible to palpate around surgical wounds without a strong reaction from the cat if pain is well controlled (Figure 3).

Clinical pain assessment incorporates 1) observations of posture, general behaviour, comfort, activity, attitude, body position and facial expressions, and 2) a dynamic and interactive approach involving greeting the animal and performing gentle palpation of the wound/painful area. The response elicited during touch and palpation may help with clinical decisions and with determining the sensory-discriminatory domain of pain (intensity, location and duration). (a) A healthy cat is examined prior to surgery. After observing the cat’s normal behaviour, the cage is opened, and the patient is gently approached and stroked. (b) The cat responds to the interaction with the observer and shows a friendly demeanour. This should be maintained after surgery if the cat is not painful. Images reproduced from Steagall and Monteiro (2019) 5
Pain-related behaviours have been described in detail in cats (Table 1), particularly in those undergoing abdominal surgery and dental extractions (Figure 4).25,26 The development of new behaviours or loss of previous behaviours beyond the immediate postoperative period may be indicative of pain. For example, a friendly, pain-free cat normally interacts with members of the veterinary care team, tolerates gentle abdominal palpation and displays normal behaviours such as stretching, playing and yawning. The cat may sleep during hospitalisation in a ‘bagel’, ‘croissant’ or ‘pretzel’ curled-up position (Figure 5). These individuals usually have an interest in their surroundings and respond to stroking. Eating and grooming are normal behaviours that should be present in the postoperative period in comfortable cats. When a cat is painful, these behaviours may quickly disappear. It is important to note that the entire veterinary care team should be trained, and actively involved, in feline pain assessment.
Behaviours suggestive of acute pain in cats, depending on the source of pain

Adapted from Steagall (2020) 27

(a) A cat with abdominal pain following ovariohysterectomy. The cat is depressed, immobile and silent. The patient has squinted eyes and is in a hunched-up position with head down. (b) A cat with abdominal pain secondary to constipation. The eyes are partially closed (‘feigned sleep’), the ears are pulled apart (ie, distance between the tips of the ears is increased when compared with a non-painful cat), and the muzzle is retracted backwards (whiskers are straight, not loose and curved). (c) Periodontal disease, such as gingivitis and periodontitis, causes pain, inflammation, dysphagia, chronic haemorrhage and weight loss. Painful cats with severe oral disease (eg, requiring multiple dental extractions) have decreased food intake, increased pain scores and present distinct behaviours such as reduced attention to surroundings and spending more time lying down, compared with pain-free cats with minimal periodontal disease. Following dental extractions, this cat scores 9/10 using the Feline Grimace Scale and requires the administration of opioids for pain relief. (d) A cat with abdominal pain can contract or extend its pelvic limbs and/or contracts its abdominal muscles (flank) spontaneously. These cats may be in dorsolateral recumbency with facial expressions of pain. (e) A tense and depressed cat in severe pain after sternotomy. This patient is sitting up due to intense discomfort, reluctant to move and not attentive to the surroundings. The cat received a score of 8/10 using the Feline Grimace Scale, indicating the need for analgesia. The cat remained uncomfortable and bothered by all bandages, catheters and nursing needs, despite infusions of opioids, ketamine and lidocaine, and the administration of NSAIDs and intercostal nerve blocks. This demonstrates that pain can be challenging to treat in some cases. The cat finally improved and analgesic infusions were tapered down before patient discharge. Images (a), (c), (d) and (e) courtesy of Paulo Steagall. Image (b) reproduced from Steagall and Monteiro (2019) 5

The curled-up position of this pain-free cat is a normal posture to conserve body heat and is not usually observed in painful cats. Image reproduced from Steagall and Monteiro (2019) 5
Pain-related behaviours in cats
Pain assessment is best performed on a regular basis. However, the cat should not be disturbed while it is eating, grooming, playing and/or sleeping. The frequency of assessment should be determined based on the pain severity noted on the first assessment after surgery or trauma, the cat’s response to analgesic administration or non-pharmacological intervention and clinical judgement. Cats may be evaluated as soon as 30–45 mins after the end of surgery and then every hour in the initial postoperative period, depending on the analgesic drugs and techniques used. Assessment should be continued to ensure comfort and until the patient is discharged. 26 After discharge, assessment can continue at home with cat carers trained to look for signs of pain.
A detailed review of feline acute pain assessment is available elsewhere. 5

Pain assessment tools
The use of pain assessment tools allows a consistent, practical and more objective approach to pain evaluation. In a busy clinical setting when several team members participate in pain assessment, use of a structured tool reduces inter-observer variability.
There are now three user-friendly pain assessment tools with reported validity in cats (Table 2): the Glasgow composite measure pain scale-feline, 7 the UNESP-Botucatu multidimensional feline pain assessment scale short form (Table 3) and the Feline Grimace Scale.7–9,24,28–32 The challenges facing us are how to disseminate this information, how best to incorporate these tools/systems into practice and how to train the veterinary clinic team. Facial expressions are incorporated into all three of these acute pain assessment tools; the Feline Grimace Scale (felinegrimacescale.com) is based entirely on facial ‘action units’ (Figure 6).8,29,30,33 These scales provide a cut-off/intervention score that suggests administration of analgesia is required (Table 2). Guidance to assist the veterinary nurse or technician in advocating for their patient, and the clinician with decision-making, is a clear benefit of using pain assessment tools. If the cat receives rescue intervention(s), pain assessment is repeated after 30–45 mins to ensure that treatment has been effective.
Overview of feline acute pain assessment tools

Adapted from Steagall (2020) 27
‘Cut-off’ value at which point administration of analgesia is required; note a veterinarian may administer rescue intervention when the cut-off is not reached if it is considered that the cat may be in pain
UNESP = Universidade Estadual Paulista; UFEPS = UNESP Feline Pain Scale
UNESP-Botucatu multidimensional feline pain assessment scale short form (UFEPS-SF)

From Belli et al (2021) 9 *
Tick where applicable
UNESP = Universidade Estadual Paulista; UFEPS = UNESP FEline Pain Scale
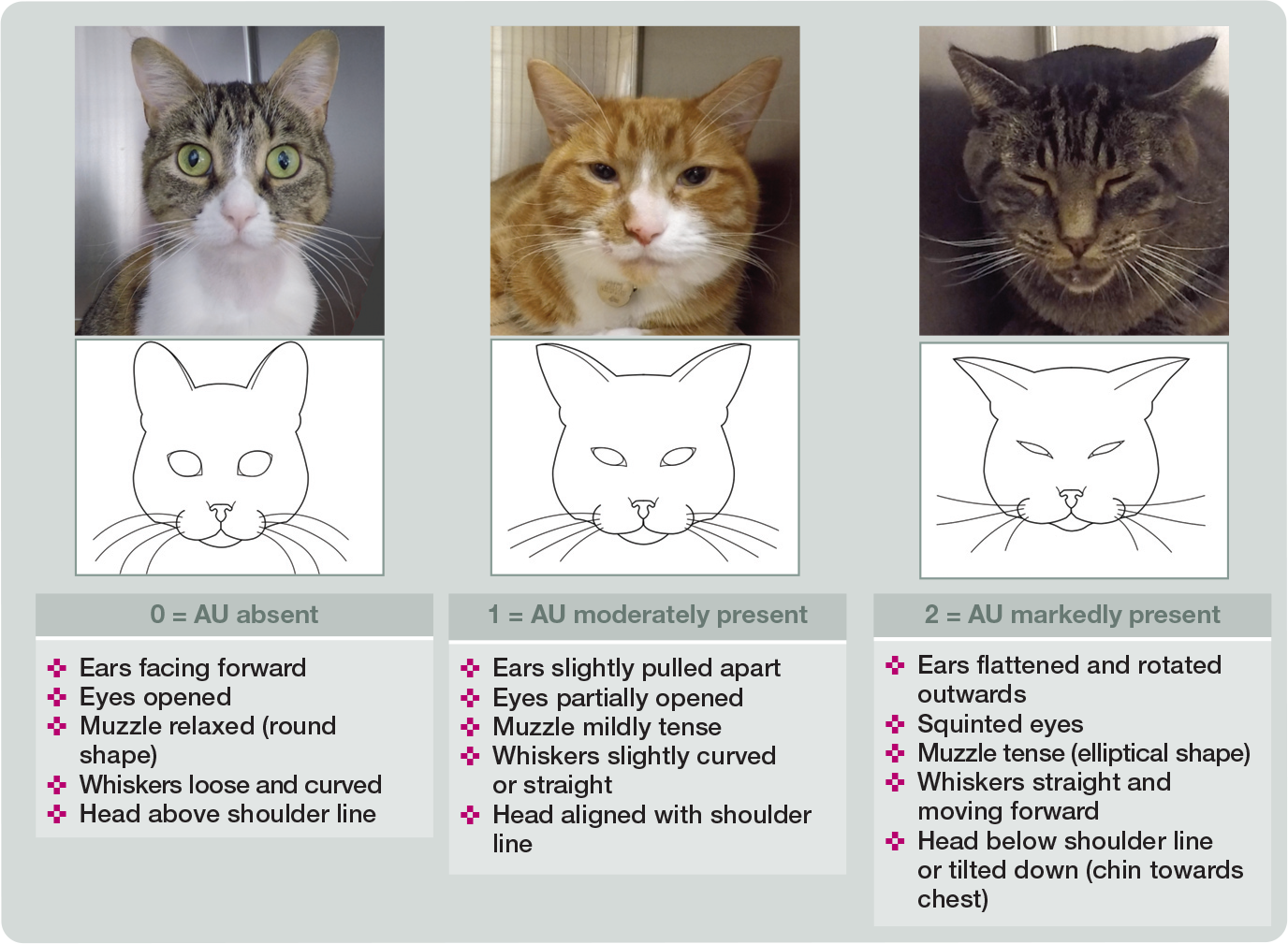
Illustration and description of the Feline Grimace Scale. This pain assessment tool is composed of five action units (AU) (ear position, orbital tightening, muzzle tension, whisker position and head position). Each one is scored from 0 to 2: 0 = action unit is absent; 1 = moderate appearance of the action unit, or uncertainty over its presence or absence; and 2 = obvious appearance of the action unit. Adapted from Evangelista et al (2020) 29
Limitations and factors affecting pain assessment/recognition
The advent of novel pain assessment systems has improved our ability to recognise and treat pain in cats. However, these instruments have not fully eliminated potential confounding factors and bias. Temperament (fearful, shy, etc), the administration of some drugs (eg, sedatives or tranquillisers) and illness can still pose challenges during pain assessment.34–37 Fear and anxiety may bias pain assessment due to changes in posture, comfort and facial expressions. 38 For example, pain assessment can be a challenge in unsocialised cats in trap–neuter–return programmes (Figure 7). The administration of ketamine-based protocols produces a confounding effect early in the recovery period and may falsely increase pain scores, which could lead to unnecessary administration of rescue analgesia. 39 Residual anaesthetic and drug effects may also bias pain assessment. Pain may be difficult to differentiate from drug-induced dysphoria (see box).

(a,b) Means of pain assessment can be limited in trap-neuter-return programmes or when working with other unsocialised cats. However, cats may still be evaluated using the Feline Grimace Scale, with observation of posture, comfort and facial expressions. Treatment of pain should be performed using a multimodal approach, based on the observer’s interpretation of how painful the cat’s condition could be, since wound palpation, for example, may not be possible. As with all patients, these individuals should be handled with empathy and care. Images courtesy of Sheilah Robertson

Pain scores may be influenced by the gender, training, cultural background and language of the observer, demonstrating that pain assessment is not an exact science and differences may be expected.34–36 It has been found that females and those with more training will provide higher pain scores than males and those with minimal training.13,34,36 Feline pain assessment should be integrated into the veterinary curriculum in conjunction with cat friendly handling techniques.
Cat carers should be considered part of the ‘team’ when it comes to pain management. Client education regarding pain behaviours (what to expect and how to proceed) is important and could lead to early intervention by prompting the client to seek veterinary attention. 5 For example, cat owners can identify specific behavioural alterations up to 3 days after castration or ovariohysterectomy. 42 A decrease in overall activity level and playfulness, an increase in the amount of time spent sleeping, and altered locomotion were reported by these owners. 42 ISFM has produced a Cat Carer Guide to acute pain in cats (see page 30). Finally, the Feline Grimace Scale can be used reliably by cat owners, veterinary nurses and students, providing the first feline acute pain assessment tool that may be used in the home environment 33 and by non-veterinarians, thereby paving the way towards better feline welfare.

Treatment of pain – principles and considerations
Failure to recognise and adequately treat acute pain is known as oligoanalgesia. The lack of analgesic administration to a painful cat may lead to the development of peripheral and central sensitisation,16,43 which in turn can reduce the nociceptive threshold to non-noxious and noxious stimuli (Table 4). 44 In humans, inadequate treatment of pain following surgical procedures including, but not limited to, amputation, thoracotomy, mastectomy 45 and caesarean section 46 can result in a transition to chronic or maladaptive pain states (ie, persistent postoperative pain), leading to disability and a negative impact on quality of life. A similar scenario may occur in cats; 43 for example, after onychectomy (Figure 8).
Glossary of terms used in the neurophysiology of pain

Adapted from Steagall et al (2021) 44

Persistent postoperative pain in a 7-year-old female cat after onychectomy. The cat persistently keeps the left paw lifted while sitting, to avoid bearing weight on it, and also resents being touched on that paw. Image reproduced from Monteiro BP and Steagall PV. Chronic pain in cats: recent advances in clinical assessment. Journal of Feline Medicine and Surgery 2019; 21: 601–614
Understanding the principles of acute pain management aids in the development of an effective analgesic strategy and can mitigate or prevent oligoanalgesia and adverse outcomes in cats, a species in which analgesic administration has historically been neglected.
Feline drug metabolism and elimination: impact on analgesia
Drug metabolism and elimination are different in cats compared with other species. This has a significant influence on drug choice, dose and dosing intervals of analgesics. Drugs that are eliminated by metabolic conjugation (ie, glucuronidation, sulfation and/or glycosylation) are usually excreted more slowly in cats than in humans or dogs. 47 Examples include acetylsalicylic acid, acetaminophen (paracetamol), carprofen, ketoprofen and morphine. Deficient glucuronidation has significant effects on phenolic compounds such as acetaminophen, resulting in life-threatening methaemoglobinaemia due to oxidative injury to erythrocytes, anaemia and Heinz body formation.47,48 The clearance of carprofen is significantly slower in cats than in dogs (ie, the drug has a much longer elimination half-life in cats). 49 The deficient UDP-glucuronosyltransferase (UGT) pathways in cats may also be responsible for reduced morphine glucuronidation in cats. Cats eliminate morphine at a similar rate to dogs; however, they produce lower concentrations of morphine-3-glucuronide, and do not produce morphine-6-glucuronide, an active metabolite responsible for analgesia in humans. 50
Conversely, drugs that primarily undergo oxidation are often eliminated more rapidly in cats compared with dogs. Examples include meloxicam, piroxicam, buprenorphine and meperidine (pethidine). The major route of elimination of meloxicam in cats is via the gut (faeces), which is similar to dogs; the main pathway of meloxicam biotransformation is oxidation.51,52
Drugs that are excreted unchanged into urine and/or bile such as gabapentin have similar elimination half-lives in dogs and cats. 47
Feline comorbidities
The presence of comorbidities must be considered in the treatment of pain. As many as 40% of cats over 10 years of age have chronic kidney disease (CKD). 53 Drugs or metabolites that require healthy kidney function for excretion may accumulate in cats with CKD. Therefore, doses and dosing intervals may require adjustment and this will be based on clinical experience, the patient’s response and pain assessment. The effects of drugs may not be predictable because, in addition to decreased renal elimination in the face of CKD, azotaemia increases blood–brain barrier permeability, which can result in a more enhanced drug effect. A classic example is the metabolism of ketamine by the liver into the active metabolite norketamine, which is excreted by the kidneys in its active form along with ketamine itself. Ketamine and nor-ketamine accumulation may occur, with potential prolonged anaesthetic effects in cats with urethral obstruction or kidney dysfunction. Metabolic acidosis in cats with CKD can alter the biodisposition of ketamine, resulting in accumulation. Outcomes may depend on dose since accumulation does not appear to be a problem with the administration of subanaesthetic (anti-hyperalgesic) doses of ketamine given by infusion (10 µg/kg/min) as a component of a postoperative analgesic regimen after feline subcutaneous urethral bypass when renal function is improving. 27
Overall, best practice with respect to anaesthesia and analgesia is particularly important in cats with comorbidities. The administration of fluid therapy will correct dehydration and electrolyte and acid-base abnormalities while maintaining adequate renal blood flow during anaesthesia and supporting drug excretion. In some cases, the administration of a minimum effective dose of an NSAID can be considered for normovolaemic cats with stable renal disease (International Renal Interest Society [IRIS] stage I or II) undergoing surgery with a strong inflammatory component (eg, multiple dental extractions, fracture repair, large excisions) once blood pressure is controlled and there is no risk of intraoperative haemorrhage. Ideally, the cat should be eating and drinking after anaesthetic recovery before the administration of an NSAID. 54

Building an analgesic plan
Building an effective analgesic plan relies on two fundamental principles: preventive analgesia and multimodal analgesia (see box above).
The first step of acute pain treatment involves the administration of opioids, local anaesthetics and NSAIDs. The second step of treatment involves the administration of adjuvant analgesics. 57 Further therapy should focus on analgesia and comfort, while minimising adverse drug reactions and preventing deleterious endocrine and physiological responses. Additional steps of equal importance include anxiolysis and muscle relaxation, antiemetics and non-pharmacological therapy, which should be incorporated when needed (Figure 9). Maropitant and ondansetron reduce perioperative vomiting in cats,58,59 and gabapentin or trazodone are often administered to reduce anxiety; for example, during hospitalisation.60,61 Non-pharmacological therapies should be incorporated when possible and include appropriate bandaging (wound care), cold therapy 62 (Figure 10), nursing (ie, patient positioning, fluid therapy, nutrition, cleaning and grooming), environment (ie, dry, calm, quiet and comfortable) (see Figure 2) and an area for cats that is separated from dogs (eg, as included in the requirements for ISFM Cat Friendly Clinic accreditation and the AAFP Cat Friendly Practice Program).17,63

Successful management of acute pain does not just include use of analgesic agents – a combination of components is fundamental to a successful pain management plan

A cat undergoing ice therapy after total ear canal ablation. Cold therapy is an example of a non-pharmacological tool that provides effective analgesia, and is readily available. Image courtesy of Paulo Steagall
Treatment will be determined by the type, location, severity and expected duration of stimuli, and whether the cat is an inpatient or outpatient. The analgesic plan will be based on these key concepts, which can be referred to using the acronym ‘TELLS’ (Table 5 and Figure 11). For example, acute visceral pain associated with elective procedures (eg, feline neutering) may be treated effectively with NSAIDs, opioids, alpha-2 agonists64,65 and local anaesthetic techniques (eg, intratesticular, incisional, intraperitoneal or transversus abdominis plane [TAP] block). Acute somatic or neuropathic pain associated with orthopaedic or vertebral surgery/spinal cord injury may require the addition of N-methyl D-aspartate (NMDA) receptor antagonists (eg, ketamine, amantadine) and gabapentinoids.
Key concepts (TELLS) used for pain management,* with specific examples

These concepts help determine the dose, frequency, type and route of analgesic drug(s) to be provided

Suggestions for acute pain management in patients with specific types of pain. NSAID = non-steroidal anti-inflammatory drug; NMDA = N-methyl D-aspartate
The expected duration and severity of pain also determines which analgesic drugs are best suited for each individual patient. In cats with prolonged moderate to severe pain, pain management can be performed with intermittent doses of buprenorphine 66 or methadone. Alternatives include constant rate infusions (CRIs) of opioids and/or ketamine. Specific classes of analgesics are discussed below.
The inpatient or outpatient nature of the treatment may affect the frequency and route of administration. Hospitals without after-hours or overnight staffing that either discharge patients or leave them unattended overnight may rely on long-lasting oral, sustained-release or transdermal analgesic drugs. However, some patients may require intense postoperative monitoring and analgesia, and should be transferred, if possible, to clinics that can provide such care (eg, CRIs of analgesics, fluid therapy, wound care and monitoring).
Further practical considerations when building an analgesic plan in cats are outlined in the box above.
Treatment of pain – analgesic drugs
Opioids
Opioids are commonly used analgesics for acute pain in cats.14,69 By binding to opioid receptors located within the peripheral and central nervous system they decrease the release of excitatory neurotransmitters, hyperpolarise the neuronal post-synaptic cell membrane,70–73 inhibit Ca2+ influx in afferent sensory neurons, and activate the descending inhibitory pathways. The net result is an antinociceptive effect, thus producing analgesia.

The popularity of opioids is attributable to a remarkable safety profile, potential reversibility and analgesic efficacy. Opioids can be given by a variety of routes and can reduce the minimum alveolar concentration (MAC) of volatile anaesthetics, potentially improving cardiopulmonary function during general anaesthesia. 74 However, their ability to reduce MAC in cats when administered parenterally75,76 or epidurally is lower than in dogs. For example, high doses of remifentanil reduce isoflurane requirements by a maximum of 25% in cats,77,78 compared with 79% in dogs. 79 In cats, additional anaesthetic sparing can be achieved with the inclusion of ketamine infusions. 80
At effective opioid doses, mydriasis, purring, kneading and euphoria will occur (see box on page 14).
Table 6 presents suggested dosage regimens for commonly used opioid analgesics, with comments on their clinical relevance, advantages and disadvantages. Choosing an opioid analgesic is dependent on the patient’s signalment, temperament, comorbidities, degree of pain intensity and required sedation level, and is also determined by the planned procedure, the pharmacology of the opioid itself (onset, magnitude and duration of action) and what the clinician has access to in their location. Kittens and young cats may require more frequent opioid dosing. One study showed that age can influence thermal antinociception produced by opioids in cats. 88 Specifically, hydromorphone provided a shorter duration and smaller magnitude of antinociception in cats of 6 months of age than in the same individuals at 9 and 12 months of age. 88
Commonly used opioid analgesics in cats, with suggested doses and routes of administration* (continued on page 16)

Doses, drugs and routes of administration presented in this table are suggestions and not exhaustive. Individual variability should be appreciated and opioids are better administered as part of multimodal analgesia. The analgesic effects may be variable depending on the type of pain, age, breed, surgical procedure, anaesthetic duration and route of administration. Clinical judgement is important. Veterinarians should consult drug labels if a veterinary product is available
NA = not applicable; IV = intravenous; IM = intramuscular; SC = subcutaneous; OTM = oral transmucosal; NSAID = non-steroidal anti-inflammatory drug; NMDA = N-methyl D-aspartate; FDA = Food and Drug Administration
Routes of administration
Traditionally, opioids are administered by the intramuscular (IM) or intravenous (IV) route. The onset, magnitude and duration of analgesia are influenced by the route of administration, which should be chosen on an individual basis. 89 The IV route provides reliable analgesia and a rapid onset of action. The subcutaneous (SC) route of administration provides less pain on injection than the IM route; 90 however, depending on the drug, dosage and formulation, large variations in the magnitude and duration of antinociception may be observed.82–84 For example, analgesia may be suboptimal after the SC administration of recommended doses of buprenorphine or hydromorphone in cats. 84 In contrast, a high-concentration formulation of buprenorphine (1.8 mg/ml; Simbadol, Zoetis) is approved by the US Food and Drug Administration and provides up to 24 h of mild to moderate pain relief following SC administration (Table 6). 85 This formulation is also effective when given to cats by the IV or oral transmucosal (OTM) route (off-label administration), both providing up to 8 h of analgesia. 85 The OTM route is a non-invasive alternative to parenteral administration. It bypasses the first-pass effect in the liver, leading to higher bioavailability when compared with oral administration. Variations in analgesia may still occur via the OTM route, and thus the IM or IV route is preferred when possible for acute postoperative pain management. 82
In cats, plasma drug concentrations following application of a transdermal fentanyl patch (a human product) are highly variable and may fail to produce analgesia.91,92 A trans-dermal matrix patch of buprenorphine failed to produce thermal antinociception, with high plasma concentration variability of the drug, in a preliminary study of pharmacokinetics in cats. 93
Epidural administration of opioids alone (eg, morphine or buprenorphine 94 ) provides effective control of visceral 95 and somatic 96 pain, with few adverse effects97–99 and without changes in proprioception. The bladder should be gently expressed before anaesthetic recovery to avoid the rare but reported side effect of urinary retention after the administration of epidural morphine.96,100 Regardless of which analgesic drugs are administered, an empty bladder may improve comfort during the early recovery period.
Adverse drug reactions
Some opioids may produce adverse effects, which are drug-, dose- and route-dependent (Table 6). Nausea and vomiting (Figure 12), gastro esophageal reflux, hyperthermia, dysphoria, mydriasis (Figure 12), sedation, vagally induced bradycardia, and respiratory depression with mild hypercapnia may be observed following the administration of opioids. Antiemetics (maropitant, ondansetron), prokinetics (metoclopramide), and acupuncture 101 can minimise or treat some of the adverse gastrointestinal (GI) effects associated with the administration of opioids.102–105 These GI adverse effects can be avoided by choosing methadone, fentanyl or buprenorphine. The administration of epidural opioids provides effective long-term analgesia with few side effects.98,99 Naloxone or butorphanol may be administered to effect when antagonising opioid-induced adverse effects. Analgesia may also be decreased or antagonised in this scenario and other analgesic techniques should be in place or considered.

Mydriasis and hypersalivation in a cat following the intravenous administration of hydromorphone. Image courtesy of Bradley Simon
Non-steroidal anti-inflammatory drugs
NSAIDs are used extensively in cats for their analgesic, anti-inflammatory and antipyretic effects, and are crucial in the control of postoperative pain. After any contraindication for their use has been ruled out (GI disease, NSAID intolerance, unmanaged renal disease, hepatic disease, coagulopathies, hypovol-aemia and hypotension, concurrent NSAID or corticosteroid administration), NSAIDs are highly effective for the treatment of acute pain in cats as part of a multimodal approach.106–114 Recommended NSAID regimens for management of acute pain in cats are summarised in Table 7.
Common NSAIDs used in cats for management of acute pain*

Veterinarians should consult drug labels in their respective countries before administering NSAIDs
COX-2 preferential = greater suppression of COX-2 than COX-1; COX-2 selective = virtually no COX-1 suppression
COX = cyclooxygenase; OVH = ovariohysterectomy; SC = subcutaneous; PO = oral
Adverse effects (GI irritation, protein-losing enteropathy and nephrotoxicity) are reported when recommended doses or dosing intervals are not adhered to, or if NSAIDs are administered in combination with corticosteroids. Anorexia, inappetence, diarrhoea, vomiting and depression are usually the first signs of toxicity, and treatment should be stopped immediately if any of these occur. However, omitting NSAIDs because of fear of adverse effects is not recommended and may seriously compromise pain management.
Dosing should be based on lean body weight and cats should be normovolaemic before drug administration. Some postoperative patients may benefit from prolonged NSAID administration and this should be considered on a case-by-case basis. 115
Meloxicam and robenacoxib have good palatability, which facilitates treatment compliance,113,117 and once a day dosing also increases the likelihood of the cat receiving the prescribed medication. Injectable formulations are used in the perioperative period and can be administered IV (off-label use) or SC, depending on the drug used and country. Oral administration usually follows in the postoperative period and home environment. Many studies have shown that different perioperative NSAIDs usually perform similarly in terms of analgesic efficacy and duration of effect (ie, non-inferiority or comparison trials).110,111,113
The timing of NSAID administration is a controversial subject. NSAIDs can generally be administered preoperatively for routine neutering of healthy cats, especially if blood pressure is monitored and cats are normo-volaemic. For longer procedures, fluid therapy may be indicated, blood pressure should be monitored, and the risks and benefits related to the timing of NSAID administration need to be taken into consideration. 107 The administration of NSAIDs in patients with comorbidities such as CKD was discussed in the earlier section ‘Feline drug metabolism and elimination: impact on analgesia’.
Local anaesthetics
Local anaesthesia provides excellent analgesia by preventing transduction and transmission of nociceptive signals from the periphery to spinal and supraspinal centres. 118 Compared with other antinociceptive drugs, local anaesthetics are unique in providing complete analgesia. Locoregional anaesthesia is an important part of multimodal analgesia and should be incorporated into a pain management plan whenever possible. Anaesthetic maintenance and recovery are usually smooth when these techniques are integrated into the plan. Other advantages of locoregional anaesthesia include low cost, accessibility in all countries, muscle relaxation, decreases in injectable and volatile anaesthetic requirements, and, in turn, improved cardiopulmonary function.119–122 Additionally, the effects of long-acting local anaesthetics extend into the postoperative period, and reduce the requirements for other drugs.121,123 Recently, a liposomal bupivacaine formulation was approved for use in cats undergoing onychectomy in the USA and may provide postoperative analgesia for up to 3 days. 124 This formulation has been used off-label for incisional anaesthesia (see later). 124
Commonly used local anaesthetics are presented in Table 8.118,125 Local anaesthetic toxicity can result in severe neurological signs, cardio-respiratory collapse and death. Therefore, calculation of doses, which must always be accurately drawn up into syringes, is particularly important in cats due to their small size, especially with bupivacaine due to its cardiotoxicity. Accidental intravascular administration can be avoided by applying negative pressure to the syringe plunger before injection118,125 and repositioning the needle if blood is obtained. The most serious adverse effect reported for local anaesthetics is cardiac arrest and intravenous administration of lipid emulsion may reverse bupivacaine-induced toxicity. 127
Commonly used local anaesthetics for locoregional anaesthesia in cats

The information in this table represents a consensus of opinion of the Guidelines panel members
The duration of a block may vary depending on the block, technique, site of injection, anaesthetic concentration and volume, and in terms of sensory versus motor block
Lumbosacral epidural administration of opioids combined with local anaesthetics is sometimes used for the treatment of feline perioperative pain, particularly during orthopaedic procedures or hindlimb amputation. In cats, the spinal cord and dural sac terminate at the first (S1) and third (S3) sacral vertebra, respectively, while the end of the conus medullaris is located within the sacral canal (Figure 13). 128 These anatomical features make the risk of intrathecal drug administration more likely compared with dogs. 128

Magnetic resonance image of a feline lumbar and sacral spinal column. Note the spinal cord (SC) extending beyond the 7th lumbar (L7) vertebra. Image courtesy of Wade Friedeck, Texas A&M University College of Veterinary Medicine & Biomedical Sciences
Sacrococcygeal epidural anaesthesia has been used as an alternative to lumbosacral epidural injections for cats with urethral obstruction (Figure 14).129,130 A sacrococcygeal technique using bupivacaine or bupivacaine/morphine reduced propofol requirements and extended the time to rescue analgesia in these cats. 129 Anatomical and ultrasonographic studies have shown that the depth of needle insertion for sacrococcygeal epidural anaesthesia is approximately 0.6 cm in cats, whereas the distance between skin and the ligamentum flavum is 1 cm at the lumbosacral space. 131 The epidural space is small in cats but the technique can provide excellent perioperative analgesia and be performed safely. Appropriate anatomical landmarks should be considered and an aseptic technique used.

Sacrococcygeal epidural administration of local anaesthetics can be used in cats with urethral obstruction as an adequate means of pain management. Leakage of spinal fluid is less likely at this location but can occur, as shown in this image. Image courtesy of Paulo Steagall
Selected local and regional anaesthetic techniques
Several local anaesthetic techniques have been reported in cats, including sophisticated ultrasound-guided techniques, allowing a better understanding of anatomical landmarks, and improved safety and efficacy. These Guidelines provide an overview of easy-to-use, selected techniques with immediate application in feline practice (Table 9).
Summary of common local anaesthetic techniques used in cats

The information in this table represents a consensus of opinion of the Guidelines panel members
Refer to Table 8 for the maximum recommended dose of the particular drug being used, to avoid toxicity OVH = ovariohysterectomy
Topical local anaesthesia
The administration of topical anaesthesia is simple and effective. In one study of cats undergoing radical mastectomy, topical application of 2 mg/kg bupivacaine in 30 ml of saline 0.9% via impregnated gauze decreased pain scores at 4 h postoperatively. 133 Although placing an intravenous catheter may seem a minor procedure, it should be remembered that each painful procedure a cat undergoes can have an additive effect and, therefore, every opportunity should be taken to decrease pain and stress. Topical local anaesthetic cream (lidocaine-prilocaine) (Table 9) reduces pain and stress during intravenous puncture or catheteristion (Figure 15), and is not systemically absorbed, thus methaemoglobinaemia or other adverse effects are avoided.126,134–136 In addition, the success rate for catheterisation is improved. 128


A eutectic mixture of local anaesthetics (lidocaine and prilocaine cream) is used to desensitise the skin before venous catheterisation. The area is covered with an occlusive bandage for 20-30 mins before venepuncture. Adverse reactions have not been reported in cats. Image courtesy of Paulo Steagall
Incisional and intraperitoneal analgesia
The World Small Animal Veterinary Association (WSAVA) Global Pain Council recently published an evidence-based review on incisional (Figure 16) and intraperitoneal (Figure 17) techniques. 137 Readers are referred to that resource for a detailed discussion of indications and recommendations for use, as well as limitations. These techniques should be incorporated into multimodal pain management.132,138–143

(a,b) Incisional anaesthesia is performed before or at the end of surgery by deposition of local anaesthetic subcutaneously at the incision line. Image (a) courtesy of Yael-Shilo Benjamini; image (b) courtesy of Paulo Steagall

Intraperitoneal administration of local anaesthetic is performed before ovariectomy/ovariohysterectomy and other abdominal procedures. Local anaesthetics with a longer duration of action (ie, bupivacaine) are used, and the solution is divided into three parts, which are ‘splashed’ over the right and left ovarian pedicles and at the caudal aspect of the uterine body (arrows) using a 3 ml syringe. The solution is quickly absorbed by the peritoneum. A technique involving injecting bupivacaine (2 mg/kg) at the ovarian suspensory ligaments and vessels, uterine body and incisional subcutaneous tissues has also been described; 132 it resulted in lower postoperative pain scores compared with a control group and was most beneficial in larger cats (>2.7 kg). Image courtesy of Paulo Steagall
Intratesticular block
Intratesticular anaesthesia is a simple technique that may decrease sympathetic and haemodynamic responses during feline castration (Figure 18).120,144,145 It can be combined with incisional anaesthesia for complete antinociception (Figure 18b [needle B]). This technique should be performed under general anaesthesia or deep sedation, as injection and distension of the testicle can be painful.

Intratesticular block. The needle is inserted into the centre of the testis and 0.1-0.25 ml of local anaesthetic is injected per testicle (a; b [needle A]). During injection the testis will become firm. The intratesticular injection can be combined with subcutaneous incisional infiltration, which is applied bilaterally at the scrotum (b [needle B]). The higher volume is used for larger testes while the lower volume is administered in kittens (c). Images (a) and (c) courtesy of Paulo Steagall; image (b) courtesy ofYael-Shilo Benjamini
Dental blocks
There are many blocks that can be used for a variety of dental and oral procedures including extractions, oral mass removal, biopsies, maxillectomy and mandibulectomy. Injections are performed using 25–27 G needles and volumes of lidocaine of 0.1–0.3 ml at each nerve;25,125,146 however, the total dose guidelines (mg/kg, Table 8) must be adhered to. The final injectate volume will depend on the number of dental blocks to be performed. Care must be taken to carefully track the amount of drug administered, especially when injecting at multiple sites.
The infraorbital nerve block is shown in Figure 19 (needle A) and Figure 20. The maxillary nerve courses through the inferior periorbita and enters the infraorbital canal, where it becomes the infraorbital nerve.147,148 The needle should be inserted with caution into the infraorbital canal, no further than 4–5 mm, to avoid the risk of penetration of the eyeball, especially in brachycephalic cats. 149 The efficacy of this nerve block for caudal teeth (ie, feline premolar 3 and molar 1) anaesthesia has not been established in cats. 147 A maxillary nerve block has been performed as an alternative for anaesthesia of the caudal teeth, but there are reports suggesting an increased risk of penetrating the globe and, therefore, it is no longer recommended.150,151

Locations for the infraorbital nerve block (needle A) and inferior alveolar (mandibular) nerve block (needle B). The techniques are illustrated photographically in Figures 20 and 21, respectively. Image courtesy ofYael-Shilo Benjamini

Infraorbital nerve block. The needle is inserted from a cranial to caudal direction at the level of premolar 3, approximately 4–5 mm into the infraorbital canal. Image courtesy of Paulo Steagall
The inferior alveolar (mandibular) nerve (Figure 19 [needle B] and Figure 21) enters the mandibular canal via the mandibular foramen on the medial surface of the mandibular ramus.121,148 Cardiovascular collapse was reported in a cat with a mandibular mass following inferior alveolar (mandibular) anaesthesia using bupivacaine. 152 Injection in close proximity to the tumour may have resulted in rapid bupivacaine absorption and toxic peak plasma concentrations. 152

Inferior alveolar (mandibular) nerve block using an extraoral approach. The needle is inserted, perpendicularly to the skin surface, at the lower angle of the jaw, and advanced approximately 5 mm against the medial side of the mandible in the direction of the mandibular foramen. Image courtesy of Yael-Shilo Benjamini
Adjuvant analgesic drugs
Transdermal administration of drugs is an attractive alternative to oral dosing in cats. The choice of base used to mix drugs with is important since it must be able to permeate feline skin. One study showed no significant transdermal absorption of gabapentin when compounded with ethoxydiglycol solvent and a transdermal base (Lipoderm; Professional Compounding Centers of America). 161 However, another study showed that gabapentin in the proprietary base Lipoderm did result in measurable plasma concentrations. 163 The authors of the latter study caution that their investigation involved a small number of cats and, because grooming was allowed, uptake may have occurred transmucosally. More studies using different transdermal creams, concentrations, doses and dosing intervals are needed.
There is limited information on the use of gabapentin for acute pain management.35,55,164 In one study, gabapentin (50 mg, PO, administered 12 h and 1 h before surgery) in combination with buprenorphine produced similar postoperative analgesia when compared with meloxicam and buprenorphine in cats undergoing ovariohysterectomy. 35 Further studies are thus still required to investigate the analgesic properties of gabapentinoids in cats. Gabapentin (50 or 100 mg, PO) is important in attenuating stress and fear responses in client-owned and cage-trap community cats undergoing transportation and veterinary consultation, and remains a valuable component of a holistic approach to feline pain management.60,165
Based on the pharmacokinetics of tramadol in cats, a dosage of 4 mg/kg q6h PO has been recommended as part of a multimodal analgesic plan. 169 However, tramadol has failed to produce clinical analgesia in some studies, and pain assessment should always be performed. 167 The risk of serotonin toxicity is a concern when tramadol is administered with serotonin inhibitors (eg, fluoxetine and trazodone), monoamine oxidase inhibitors (eg, selegiline) and tricyclic antidepressants (eg, clomipramine). Clinical signs of serotonin syndrome include increased neuromuscular activity, tachycardia, fever, tachypnoea and agitation. 170
Analgesic shortages and unavailability: opioid-free or opioid-sparing techniques
Opioids are included in the WSAVA list of essential medicines for cats and dogs, and considered ‘core medicines’. 171 Nevertheless, veterinarians commonly face opioid shortages or do not always have access to this class of drug due to strict regulatory and bureaucratic constraints, and/or where medicines labelled for human use are rarely allowed to be administered to veterinary patients. These constraints affect the ability of veterinarians to treat feline pain, and this could represent a major issue for feline welfare if surgical and medical procedures are performed with limited or no perioperative analgesia.
‘Opioid-free anaesthesia’ (OFA) is a practice that excludes perioperative administration of opioids and uses non-opioid, multimodal analgesic techniques. ‘Opioid-sparing anaesthesia’ is a less restrictive technique where small amounts of opioids are used (ie, single and/or low doses of opioids perioperatively). Both techniques are now being studied in human medicine to avoid or reduce opioid-induced adverse effects, including respiratory depression and death.172–174 Although adverse effects are different between humans and cats, the study of feline OFA is integral to overcoming opioid shortages in the future and reducing opioid-induced adverse effects (vomiting, dysphoria, hyperthermia, etc). There is also an urgent need to provide humane solutions for pain management in sterilisation programmes, which have often been performed without the use of analgesics.
The veterinary literature on the subject is scant, particularly in cats. In one study, an injectable anaesthetic protocol using ketamine-midazolam and dexmedetomidine, intraperitoneal bupivacaine and postoperative administration of meloxicam did not provide optimal postoperative analgesia in more than half of the studied population. 175 Side-by-side comparisons were not performed between an opioid-based analgesic group and an OFA group, 175 and the question of ‘can we attenuate or ameliorate acute pain without opioids?’ remains. Certainly, opioid analgesics with market authorisation for cats are critical to avoid opioid drug shortages and improve feline welfare. This issue is best addressed by involving multiple stakeholders including, but not limited to, governmental bodies, industry, regulatory agencies and veterinary professionals.

Knowledge gaps and challenges
There is still a need for better understanding of feline behaviour related to pain as well as techniques for long-term pain management in the acute setting so that cats can be discharged to their owner with analgesics that continue to work in the home environment and are easy for owners to administer. Opioid-free anaesthetic techniques and the role of gabapentinoids are important to investigate. Little is known about paediatric analgesic needs, warranting further studies in this age group. There is also a need for safety and efficacy studies of in vivo loco-regional anaesthetic techniques to bridge the gap between in vitro studies and clinical practice. A major gap remains to be filled in the veterinary curriculum regarding feline analgesia and there must be a concerted effort to implement the use of pain assessment tools in feline practice.
Summary Points
✜ The 2022 ISFM Consensus Guidelines on the Management of Acute Pain in Cats provide up-to-date information on the unique features of feline anatomy, physiology and pharmacology in relation to analgesia, as well as on pain assessment tools and novel analgesic drugs and techniques.
✜ Principles of pain management include preventive analgesia and multimodal analgesia, incorporating pharmacological and non-pharmacological options. The use of cat friendly handling techniques, anxiolysis and nursing care are central to appropriate pain management.
✜ In addition to local anaesthetics, NSAIDs and opioids, adjuvant analgesics should be considered depending on the individual and its needs.
✜ The future is bright for feline pain management with the potential automation of acute pain assessment tools that could. I allow cat owners to detect pain in the home environment, the licensing of new analgesic drugs for perioperative pain f control and studies on novel analgesic drugs and techniques.
✜ Gaps in knowledge and challenges remain, but the good news is that these can be addressed with research support, educational opportunities and an enthusiastic group of feline advocates.
Supplemental Material
sj-pdf-1-jfm-10.1177_1098612X211066268 – Supplemental material for 2022 ISFM Consensus Guidelines on the Management of Acute Pain in Cats
Supplemental material, sj-pdf-1-jfm-10.1177_1098612X211066268 for 2022 ISFM Consensus Guidelines on the Management of Acute Pain in Cats by Paulo V Steagall, Sheilah Robertson, Bradley Simon, Leon N Warne, Yael Shilo-Benjamini and Samantha Taylor in Journal of Feline Medicine and Surgery
Supplemental Material
sj-pdf-2-jfm-10.1177_1098612X211066268 – Supplemental material for 2022 ISFM Consensus Guidelines on the Management of Acute Pain in Cats
Supplemental material, sj-pdf-2-jfm-10.1177_1098612X211066268 for 2022 ISFM Consensus Guidelines on the Management of Acute Pain in Cats by Paulo V Steagall, Sheilah Robertson, Bradley Simon, Leon N Warne, Yael Shilo-Benjamini and Samantha Taylor in Journal of Feline Medicine and Surgery
Supplemental Material
sj-pdf-3-jfm-10.1177_1098612X211066268 – Supplemental material for 2022 ISFM Consensus Guidelines on the Management of Acute Pain in Cats
Supplemental material, sj-pdf-3-jfm-10.1177_1098612X211066268 for 2022 ISFM Consensus Guidelines on the Management of Acute Pain in Cats by Paulo V Steagall, Sheilah Robertson, Bradley Simon, Leon N Warne, Yael Shilo-Benjamini and Samantha Taylor in Journal of Feline Medicine and Surgery
Footnotes
Appendix: Cat Carer Guide
Acknowledgements
ISFM is grateful to Dr Sabrine Marangoni for technical help with formatting the Guidelines, and to Nirit Roth-Benyamini for help in creating Figures 16a, 18b and ![]() .
.
Supplementary material
The following files are available online:
✜ Troubleshooting dysphoria algorithm
✜ Cat Carer Guide: ‘Recognising and managing acute pain in cats: information for owners/caregivers’ (see Appendix, page 30).
Conflict of interest
Paulo V Steagall has provided consultancy services to, and received honoraria from, Boehringer Ingelheim, Dechra, Elanco, Nexyon, Vetoquinol and Zoetis. Sheilah Robertson has provided consultancy services to, and received honoraria from, Elanco and Zoetis. The other members of the Panel have no conflicts of interest to declare.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work did not involve the use of animals and, therefore, ethical approval was not specifically required for publication in JFMS.
Informed consent
This work did not involve the use of animals (including cadavers) and, therefore, informed consent was not required. For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
