Abstract
Practical relevance:
Abdominal ultrasound plays a vital role in the diagnostic work-up of many cats presenting to general and specialist practitioners. Ultrasound examination of the lymph nodes, peritoneal cavity and abdominal vasculature provides important information useful in the investigation of a number of conditions, including aortic thromboembolism.
Clinical challenges:
Despite ultrasonography being a commonly used modality, many practitioners are not comfortable performing an ultrasound examination or interpreting the resulting images. Even for the experienced ultrasonographer, differentiating between incidental findings such as Bates bodies and pathological changes can be challenging.
Aim:
This review, the last in an occasional series on feline abdominal ultrasonography, discusses ultrasound examination of the abdominal lymph nodes and peritoneal cavity, and also the role of the technique in identifying thromboembolism of the aorta. Aimed at general practitioners who wish to improve their knowledge of and confidence in feline abdominal ultrasound, this review is accompanied by high-resolution images and videos available online as supplementary material.
Equipment:
Ultrasound facilities are readily available to most practitioners, although the use of ultrasonography as a diagnostic tool is highly dependent on operator experience.
Evidence base:
Information provided in this article is drawn from the published literature and the author’s own clinical experience.
Abdominal lymph nodes
With advances in the quality of ultrasound equipment, many normal lymph nodes are now visible during the standard abdominal scan. Due to their small size, a high frequency transducer providing high spatial resolution is usually required to image feline abdominal lymph nodes satisfactorily. 1
Not all normal abdominal lymph nodes are consistently identified in all cats during routine ultrasound examination.2,3 This may be due to a genuine absence of certain nodes in some cats, 4 lack of contrast between normal nodes and the surrounding tissue and, in some cases, gas within the intestinal tract interposed between various nodes and the transducer. 2 However, as is the case in dogs, 5 certain lymph nodes, such as the medial iliac and jejunal lymph nodes, are recognised in the vast majority of cats. In a study by Schreurs and colleagues, involving 10 clinically normal, adult domestic shorthair cats, these nodes were identified in 10/10 and 9/10 of the animals, respectively. 2 In the same study, the hepatic nodes were visible in 7/10 of the cats, the aortic lumbar, pancreaticoduodenal and splenic nodes were each identified in 6/10 cats and the colic and ileocaecal lymph nodes were each seen 5/10 cats. The ileocaecal lymph nodes are more easily identified in cats than in dogs and, as the name suggests, are located at the ileocecocolic junction, embedded within the ileocaecal fold adjacent to the caecum. 2 The renal, gastric, sacral and caudal mesenteric nodes are observed less commonly (in 4/10, 3/10, 2/10 and 1/10, respectively, of the cats in Schreurs et al’s study). 2
Good anatomical knowledge is a prerequisite when searching for lymph nodes and it is useful to bear in mind that because of their function as part of the lymphatic system, lymph nodes are closely associated with the abdominal blood vessels. There is usually overlap of drainage between different lymph node groups, such that each node may drain several organs and each organ may be served by more than one group of nodes. For example, the pancreas is drained by the hepatic, splenic, pancreaticoduodenal and jejunal lymph nodes and the jejunal nodes drain the pancreas, jejunum and ileum.
Anatomical location and normal appearance
Normal lymph nodes are elongated structures with a fine echotexture that are uniformly isoechoic to slightly hypoechoic to the adjacent mesenteric fat and have a smooth contour.6–8 In some cases, a thin hyperechoic capsule surrounding the node may be identified. 7 When measuring the size of a lymph node, the maximum width in the short axis is considered to be more reliable than the length. 7 The normal ultrasonographic appearance of various abdominal lymph nodes is shown in Figures 1–3. In addition to these, the reader should be aware of the lumbar aortic nodes, which encompass a series of nodes situated along the abdominal aorta and caudal vena cava from the diaphragm to the deep circumflex iliac arteries. 2 Of these nodes, up to four are associated with the renal vessels and are known as the renal lymph nodes. 2 The splenic lymph nodes are positioned adjacent to the spleen at the splenic hilus. The sacral lymph nodes are located at the junction of the median sacral and internal iliac arteries but may not be visible if intrapelvic.
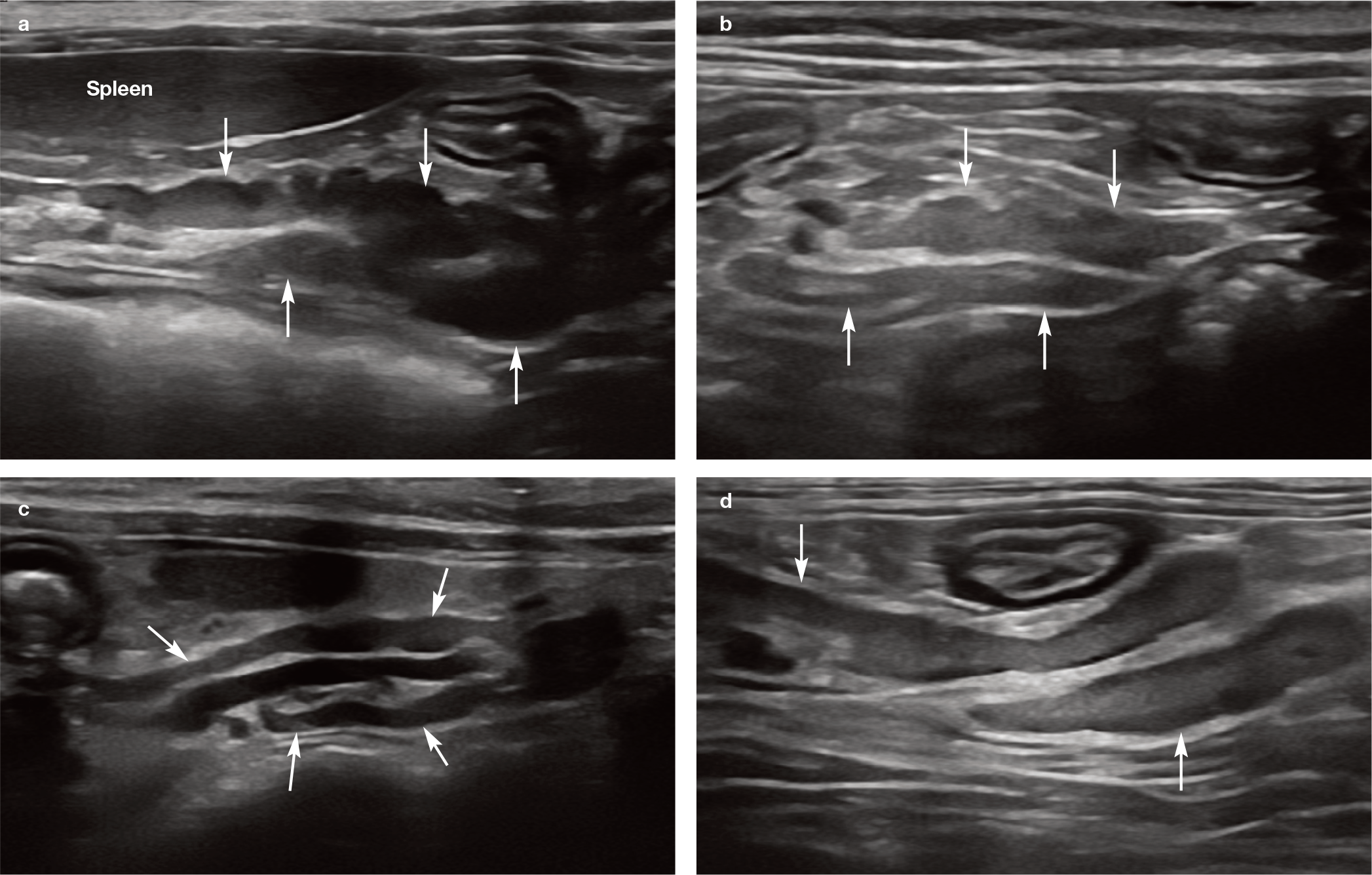
Normal appearance of multiple jejunal lymph nodes (arrows) in (a) a kitten and (b–d) three adult cats. The jejunal lymph nodes are located at the root of the mesentery, adjacent to the mesenteric artery and at the origin of the jejunal arteries. 1 In (c), note the anechoic mesenteric artery between the lymph nodes. In some cats, lymph nodes may also be located along the jejunal arteries more distally within the mesentery, close to the jejunum and ileum 2

(a–c) Normal appearance of medial iliac lymph nodes (arrows) in three adult cats. The medial iliac lymph nodes are located either side of the aorta at the level of the aortic trifurcation, cranial to the external iliac artery (EIA) and caudal to the deep circumflex iliac artery and vein. 2 A video showing the normal medial iliac lymph node in the cat in (a) is available as supplementary material

Normal appearance of an ileocaecal lymph node (short arrow) adjacent to the ileocolic junction (long arrow) in an adult cat
The normal ultrasonographic dimensions of the more common abdominal nodes of the 10 healthy, adult, domestic shorthair cats investigated by Schreurs et al are presented in Table 1. 2 There are further, sporadic reports detailing the presence and ultrasonographic appearance of feline abdominal nodes. In one such report describing the normal appearance of the feline pancreas and associated landmarks, the gastric lymph nodes were identified in 6/20 cats, cranial and ventromedial to the pyloroduodenal junction. 3 The gastric lymph nodes receive afferent lymphatic drainage from the oesophagus, diaphragm, liver, stomach and peritoneum and were described as being asymmetrical in shape due to a larger caudal pole in 5/6 of the cats, having a hypoechoic outer zone surrounding a hyper-echoic central region in all six cats, and measuring up to 10 mm in length and 6 mm in width. 3
Ultrasonographic measurements (mean [range]) of various feline abdominal lymph nodes*

From Schreurs et al 2
Abnormalities of the lymph nodes
An alteration in lymph node appearance can occur if the node becomes reactive or infiltrated by neoplastic cells, although differentiation between the two is not usually possible based on grey-scale ultrasound alone. 9 Lymphadenopathy may arise due to a number of diseases, both infectious and non-infectious, and is especially common in cats with lymphoma.10–15 Of the infectious causes, feline infectious peritonitis can result in particularly severe lymphadenopathy that can mimic masses. 16 While much of the published information regarding the ultra-sonographic appearance of lymph node abnormalities pertains to dogs or humans, most descriptions can, in the author’s experience, be broadly extrapolated to the cat.
Abnormal lymph nodes typically become enlarged, hypoechoic and potentially rounded to varying degrees (see Figures 4–14).1,5,8,10,17–20 It should be noted that younger cats typically have larger abdominal lymph nodes and this should not automatically be assumed to represent pathology. 21 In dogs, echogenicity is a poor discriminator of benign and malignant lymph nodes.8,18,19 A relatively recent study by Dave et al, evaluating both feline and canine lymph nodes, supports previous studies in finding that the majority of both benign and neoplastic lymph nodes were hypoechoic. 22 Although heterogeneity has been shown to be significantly associated with malignancy in the dog, no such association has been identified in cats and it is thus considered to be a non-specific finding in this species. 9 The study by Dave et al found that although echogenicity of perinodal fat is a non-specific finding in the cat, abdominal lymph nodes with hyperechoic perinodal fat are less likely to be reactive (than malignant) and thus potentially warrant sam-pling. 22 The same study also reported that a greater maximum long-axis lymph node diameter and a higher overall number of abnormal lymph nodes were significantly associated with malignancy in those nodes that had hyperechoic perinodal fat. 22 As a general guide, lymph node size and shape changes tend to be more marked with neo-plasia than as a result of reactive change. However, despite attempts by various authors to differentiate neoplastic from non-neoplastic lymph nodes, including with the use of elastography (see box on page 839), there is undoubtedly significant overlap in their ultrasonographic appearance, such that sampling either by ultrasound-guided fine-needle aspiration or surgical biopsy remains necessary to achieve a definitive diagnosis.

Presumed reactive hepatic lymph nodes. (a) Enlarged hepatic lymph node (between the measuring calipers) in a 10-year-old neutered female domestic shorthair cat with hepatitis. The node retains an elongated shape and near-normal echogenicity. (b) Enlarged hepatic lymph nodes (between the measuring calipers) in a 4-year-old neutered female domestic shorthair cat with cholangiohepatitis. (c) Reactive hepatic lymph node (between the measuring calipers) in a 12-year-old neutered female domestic shorthair cat with triaditis. The node is mildly enlarged, slightly rounded but normal in echogenicity. (d) Mildly enlarged hepatic lymph node (arrow) of normal echogenicity adjacent to the portal vein in a 16-year-old neutered female domestic longhair cat with extrahepatic biliary obstruction. (e) Hepatic lymph node (arrows) in a 7-year-old neutered male domestic shorthair cat with a diagnosis of acute neutrophilic cholangiohepatitis based on cytology of liver aspirates and bile culture. The lymph node is mildly enlarged, irregularly rounded and hypoechoic. A video showing the presumed reactive hepatic lymph node in the cat in (e) is available as supplementary material

Neoplastic gastric lymph node (arrows) in a 13-year-old neutered female domestic shorthair cat with lymphoma. The node is markedly enlarged, hypoechoic, rounded and surrounded by hyperechoic fat. Cytology following ultrasound-guided fine-needle aspiration of the node revealed a population of large lymphoid cells with a high mitotic rate indicative of lymphoma. A video showing neoplastic lymph nodes in a cat with gastric lymphoma is available as supplementary material
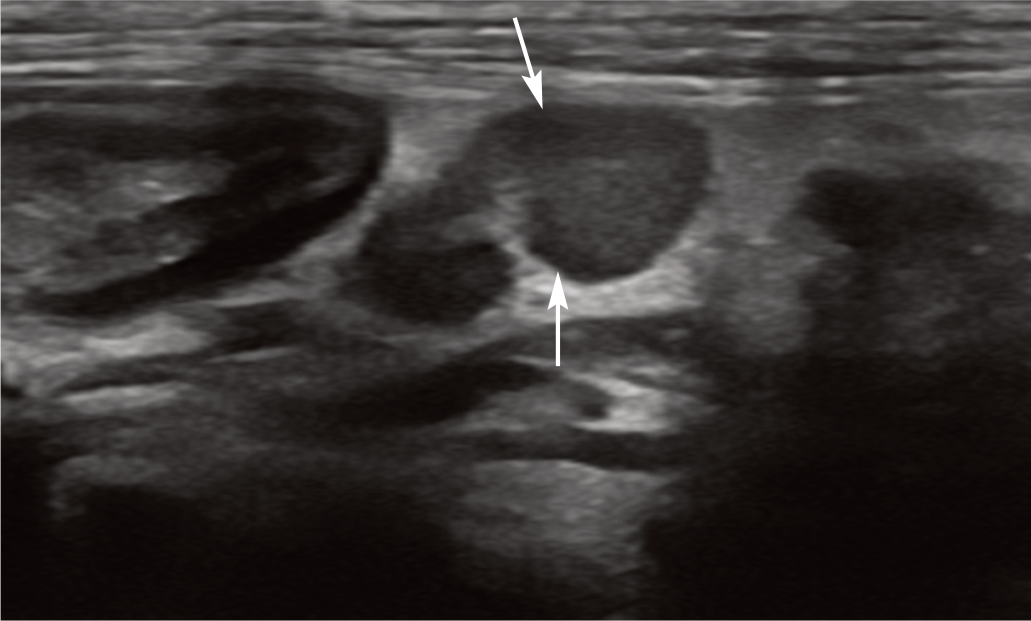
Reactive pancreaticoduodenal lymph node (arrows) in a 4-year-old neutered male Maine Coon with enteritis. The node is mildly enlarged and hypoechoic. Following ultrasound-guided fine-needle aspiration of the node, cytology revealed reactive lymphoid hyperplasia and probable neutrophilic lymphadenitis. A video showing a reactive pancreaticoduodenal lymph node is available as supplementary material

Neoplastic pancreaticoduodenal lymph node (arrows) in a 13-year-old neutered female domestic shorthair cat with lymphoma. The node is markedly enlarged, heterogeneously hypoechoic and rounded. The diagnosis was confirmed following ultrasound-guided aspiration of the lymph node

Reactive jejunal lymph nodes (presumed reactive in [a–c]). (a) Jejunal lymphadenopathy in a 12-month-old neutered male domestic shorthair cat with feline infectious peritonitis. The node (between the measuring calipers) is mildly enlarged, hypoechoic and slightly rounded. (b) Mildly enlarged jejunal lymph nodes (arrows) adjacent to the spleen in a 5-month-old entire male kitten with a chronic linear foreign body. (c) Jejunal lymph node (arrows) in a 7-year-old neutered male domestic shorthair cat with a diagnosis of acute neutrophilic cholangiohepatitis based on cytology of liver aspirates and bile culture. The lymph node is mildly enlarged and hypoechoic. (d) Markedly enlarged jejunal lymph node (arrows) in a 9-year-old neutered male Maine Coon due to severe necrotising and neutrophilic lymphadenitis, confirmed by histopathology performed on a surgical biopsy of the node. The node is irregularly marginated, rounded, diffusely hypoechoic and surrounded by hyperechoic reactive fat. (e) Markedly enlarged jejunal lymph node (arrows) measuring around 2 cm in diameter in a 5-year-old neutered male domestic shorthair cat with lymphoma. The node is abnormally hypoechoic and displaces adjacent intestinal loops. Histopathology was performed following surgical biopsy of the node and revealed lymphoid hyperplasia.

Neoplastic jejunal lymph nodes. (a) Jejunal lymph node (arrows) in a 2-year-old neutered male domestic shorthair cat with diffuse thickening of the small intestinal muscularis layer owing to lymphoma. The node is enlarged, rounded and diffusely hypoechoic. Cytology from ultrasound-guided lymph node aspiration revealed evidence of a population of large lymphocytes with morphology highly suggestive of lymphoma. (b) Enlarged, heterogeneous jejunal lymph nodes in a 10-year-old neutered male Exotic Shorthair cat with intestinal lymphoma (measuring calipers span the uppermost node). Cytology from ultrasound-guided aspirates of one of the nodes revealed an expanded population of small to intermediate lymphocytes, with a monomorphic appearance and accompanying necrosis and numerous free nuclei, raising suspicion of small cell lymphoma infiltration. Subsequent PCR for antigen receptor rearrangements (PARR) testing was supportive of lymphoma. (c,d) Jejunal lymphadenopathy (short arrows) in a 14-year-old neutered male domestic shorthair cat with small cell lymphoma. Jejunal vessels (long arrows) are visible in cross section in (d). Fine-needle aspiration of the jejunal lymph nodes under ultrasound guidance revealed an atypical lymphoid population suggestive of early small cell lymphoma

Ileocaecal lymphadenopathy (short arrows) (a,b) in a 14-year-old neutered male domestic shorthair cat with small cell lymphoma. (a) Rounded, hypoechoic ileocaecal lymph nodes are surrounded by hyperechoic fat. The ileum is visible in cross section in the centre of the image. (b) An enlarged, rounded hypoechoic ileocaecal lymph node is visible adjacent to the caecum (long arrow). (c) Reactive ileocaecal lymph node (between the measuring calipers) in a 12-year-old neutered male domestic shorthair cat with inflammatory bowel disease. The node is enlarged, rounded, hypoechoic and surrounded by hyperechoic reactive fat. Lymph node cytology from ultrasound-guided fine-needle aspiration yielded a diagnosis of reactive lymphadenopathy with possible mild suppurative lymphadenitis. No neoplastic cells were identified.
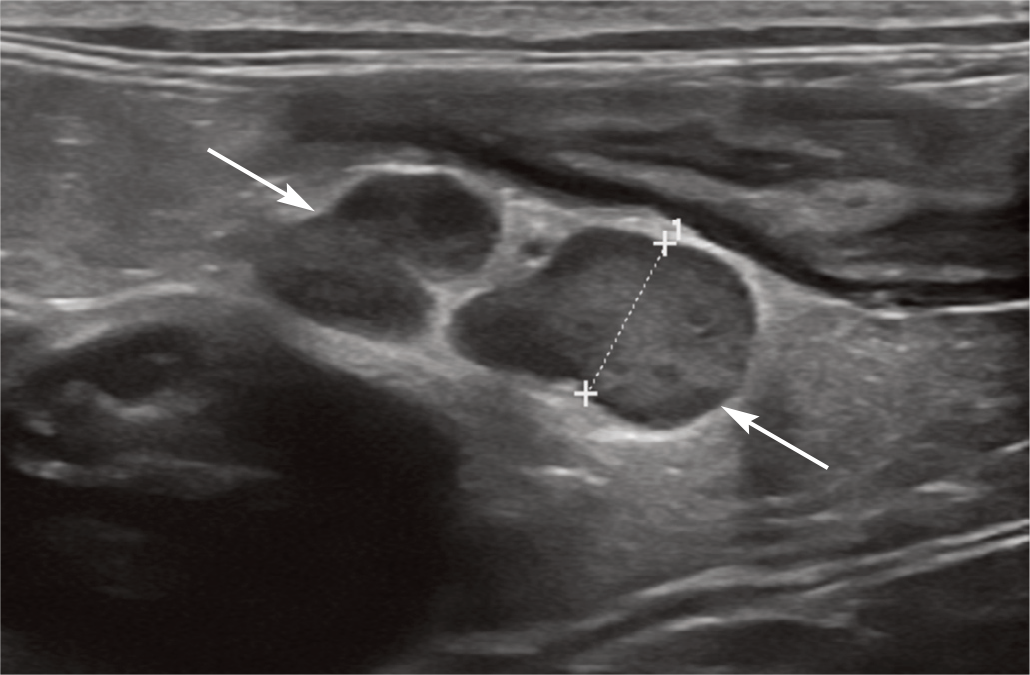
Colic lymph nodes (arrows and measuring calipers), presumed reactive, in a 12-year-old neutered male domestic shorthair cat with inflammatory bowel disease and colonic involvement. The colon is visible in the near field

Presumed reactive caudal mesenteric lymph node (arrows) in a 7-year-old neutered male domestic shorthair cat with a diagnosis of acute neutrophilic cholangiohepatitis based on cytology of liver aspirates and bile culture. The lymph node is mildly enlarged, rounded, hypoechoic and surrounded by hyperechoic fat. A video showing the presumed reactive caudal mesenteric lymph node in this cat is available in the supplementary material

Presumed reactive medial iliac lymphadenopathy. (a) Enlarged, rounded and hypoechoic medial iliac lymph node (between the measuring calipers) in a 12-month-old neutered male domestic shorthair cat with feline infectious peritonitis. (b) Reactive medial iliac lymph node (arrows) of unknown aetiology in an 11-year-old neutered male domestic shorthair cat. The node is mildly enlarged and retains an elongated appearance. Multiple other organs and nodes were sampled within the abdomen, yielding reactive change only. (c) Presumed reactive medial iliac lymph node (arrows and measuring calipers) of unknown aetiology in a 3-year-old neutered male domestic shorthair cat. The node is abnormally hypoechoic but only mildly enlarged and retains a normal elongated appearance

Presumed neoplastic medial iliac lymph node. Markedly enlarged medial iliac lymph node (between the measuring calipers) in a 12-year-old neutered female domestic longhair cat with carcinomatosis. Free peritoneal fluid is visible towards the top left of the image

Peritoneal cavity
Peritoneal fluid
Peritoneal effusions, even when small in volume, are easily detected on ultrasound with practice. In dogs, ultrasound is more sensitive than radiography for the detection of free peritoneal fluid 29 and the same is likely to be true in cats. While free fluid can collect anywhere in the peritoneal cavity, pockets are often best appreciated between the liver and the diaphragm or between liver lobes, adjacent to the spleen, around the urinary bladder and in the dependent portion of the abdomen generally (Figure 15).


Free anechoic peritoneal fluid. (a) A small volume of anechoic fluid (asterisk) is visible between liver lobes. The echogenic line (arrow) in the far field represents the lung–diaphragm interface. (b) Free peritoneal fluid surrounding the liver (single asterisks) and caudal to the stomach (double asterisk) in a 2-year-old neutered male domestic shorthair cat with hypoalbuminaemia owing to a protein-losing nephropathy. (c) Same cat as in (b). A moderate volume of free fluid (asterisk) is visible adjacent to the spleen. A video showing ascites in the cat in (b,c), with the liver floating in a large volume of anechoic peritoneal fluid, is available as supplementary material

The echogenicity of an effusion will often provide an important clue as to the potential nature of the fluid present, although a definitive diagnosis requires abdominocentesis and analysis of the resulting sample. For example, transudates, modified transudates and urine leakage are often anechoic. 10 As described in puppies, very small volumes of anechoic fluid serving as a lubricant can be normal in kittens in the author’s experience.7,30 Echogenic effusions, on the other hand, should always be regarded with suspicion and considerations include chronic inflammatory exudates and carcinomatosis (Figure 16).7,10 Haemorrhage is also usually echogenic and swirling of the effusion may be observed if bleeding is still active at the time of examination. 31 In cats with feline infectious peritonitis, the effusion can be small or large in volume and anechoic or echogenic. 16

Free echogenic peritoneal fluid in a 9-year-old neutered male Ragdoll representing an exudate due to pyogranulomatous inflammation. A video showing the copious volume of peritoneal fluid in this cat is available as supplementary material
Free peritoneal gas
The most common causes of free peritoneal gas are perforation of the gastrointestinal tract and abdominal wall puncture, either through trauma or iatrogenic (eg, abdominocentesis, aspiration of an organ or recent abdominal surgery including spaying). Gas introduced during surgery can be considerable and persist for several weeks. Free gas is recognised ultrasonographically by the presence of an echogenic line associated with reverberation artefact that is located outside the gastrointestinal tract; often this is observed adjacent to the non-dependent abdominal wall unless the gas is trapped between viscera (Figure 17). Large amounts of free gas can hamper an ultrasonographic examination of the abdomen by reflecting the ultrasound beam and preventing visualisation of structures deep to the gas. In such cases, scanning can be attempted from the dependent side; if unsatisfactory, consideration can be given to the use of an alternative imaging modality such as CT.

Pneumoperitoneum owing to gastrointestinal tract perforation. Note the echogenic interface (arrows) deep to the body wall and reverberation artefact (arrowheads). A video showing pneumoperitoneum due to gastrointestinal rupture is available as supplementary material
Steatitis
Sterile pansteatitis involving the omental, thoracic and subcutaneous fat has been reported in a 13-year-old neutered female Siamese cat with severe hypocalcaemia. 32 Ultrasonographically, the omental fat appeared hyperechoic with scattered hypo-echoic foci and was accompanied by moderate peritoneal effusion, confirmed to be a modified transudate. The cause of the pansteatitis was not determined.
Isolated abdominal fat tissue inflammation and necrosis has also been described in a 4.5-month-old female domestic shorthair cat. 33 On ultrasound examination, the affected fat was described as forming a well-defined, heterogeneous mass of mixed echogenicity in the cranioventral abdomen, ventral to the stomach. The authors speculated that the fat necrosis may have arisen as a consequence of recent abdominal surgery or possibly chemical peritonitis due to localised bile leakage, and may have been the cause of the observed vomiting due to local mechanical compression of the stomach and duodenum.
Nodular fat necrosis
Focal regions of fat necrosis, known as Bates bodies, are frequently seen in the abdominal or, less commonly, thoracic cavity and are considered to be benign and incidental. 7 Although also reported in dogs, in the author’s experience they are more common in cats, particularly in elderly and/or obese animals. 34 They are most easily recognised on radiographs within the peritoneal or retroperitoneal fat as well-defined, circular or oval structures associated with a distinct eggshell-like rim of mineralisation (Figure 18).7,34 More than one may be present in a single individual and should not be mistaken for neoplasia, abscessation, foreign material or uro- or ureterolithiasis. On ultrasound examination, Bates bodies appear as hyperechoic interfaces associated with strong clean distal acoustic shadowing within abdominal (or thoracic) fat (Figure 19). 34 A hypoechoic centre may be present in some cases. 34

Nodular fat necrosis (arrows) within the ventral (a) and caudal (b) abdomen in two cats. Image (a) courtesy of Paul Mahoney, IDEXX UK. Image (b) courtesy of Frederike Schiborra, University of Liverpool Small Animal Teaching Hospital

(a) Nodular fat necrosis in a 4-year-old neutered female domestic shorthair cat. A 5.3 mm diameter structure (between the measuring calipers) with an echogenic interface and distal acoustic shadowing is visible within the retroperitoneal fat, caudal to the left kidney. The lesion is not associated with any other structures and is consistent with a Bates body. (b) Nodular fat necrosis in a 2-year-old neutered male domestic shorthair cat. An 8.5 mm diameter mineralised peritoneal body (short arrows) is visible adjacent to the colon (long arrow). Note the strong distal acoustic shadowing
Peritonitis
Peritonitis may be localised to a single region of the abdomen (eg, surrounding the pancreas in a cat with pancreatitis), or it can be generalised, affecting most or all of the abdomen. Radiographically, the hallmark of peritonitis is a non-specific regional or diffuse reduction in serosal detail that may be accompanied by a mass effect and/or pneumoperi-toneum, depending on the cause. 35 Peritonitis results in inflammation of mesenteric and/or omental fat, which is appreciated ultra-sonographically as an increase in echogenicity and thickening. 7 Inflamed fat can also develop a hazy, less well-defined appearance and mass effect displacing adjacent viscera, and is usually accompanied by a peritoneal effusion of variable volume and echogenicity depending upon the underlying aetiology. 7 Intestinal corrugation and secondary ileus may additionally accompany peritonitis, although these are non-specific findings and the former has also been reported in association with enteritis, neoplasia and ischaemia of the bowel wall.35,36 In instances where peritonitis is focal, the search for a cause, such as pancreatitis or a ruptured gastric ulcer, should be focused on the immediate vicinity of the observed changes.
Carcinomatosis
Carcinomatosis refers to the widespread dissemination of neoplasia throughout a body cavity (peritoneal or, less commonly, pleural or cerebrospinal) and encompasses metastatic spread from epithelial (carcinomatosis), mes-enchymal (sarcomatosis) and haematopoietic (lymphomatosis) tumours.37–42 Based on two studies comprising a total of 24 cats with abdominal carcinomatosis, primary tumours most commonly originated in the pancreas (n = 9), hepatobiliary system (n = 7) or small intestine (n = 5).37,41 The vast majority of these cats had carcinomatosis due to spread of a primary epithelial tumour (n = 21). Two cats were diagnosed with lymphomatosis (ie, metastatic spread of lymphoma to the peritoneum) and a single cat with abdominal sarcomatosis owing to metastatic spread of haemangiosarcoma. 37 Across these studies, the median age of the cats was 11 years, both males (n = 14) and females (n = 10) were represented, and purebreed as well as cross-breed varieties were included. In the more recent of the two studies, the most common presenting complaint was inappetence, whereas abdominal pain, a relatively common finding in dogs with carcinomatosis, was not documented in any cat. 41 On ultrasound examination, multiple, variably sized, but often small, hypo-echoic nodules were present throughout the connecting peritoneal cavity, mesentery and omentum, associated with a variable volume of either anechoic or echogenic peritoneal effusion (Figure 20).37,41 Primary or metastatic masses and nodules within solid organs, visceral and parietal peritoneal masses, lymph node enlargement and pleural effusion were also recognised.

(a–d) Carcinomatosis lesions in three cats. (a) Several well-circumscribed, hypoechoic nodular lesions (arrows) are present within the peritoneum of a 12-year-old neutered male Bengal with small intestinal adenocarcinoma. (b) Carcinomatosis in a 12-year-old neutered female domestic longhair cat. Multiple poorly defined hypoechoic nodules of varying diameters are scattered throughout the mesenteric fat. A moderate volume of echogenic peritoneal fluid is also present (note that the echogenicity is difficult to appreciate on this image). Additionally, a large colonic mass, suspected to be adenocarcinoma, and marked medial iliac lymphadenopathy were also present. (c) A subtle hypoechoic nodule (arrow) is visible along the serosal surface of a small intestinal loop in a 14-year-old neutered female domestic shorthair cat with suspected pancreatic adenocarcinoma. A moderate volume of fluid (later confirmed to be a modified transudate) is also present. (d) Same cat as in (c). Multiple subtle hypoechoic nodules are also present throughout the mesenteric fat (arrows). Videos showing carcinomatosis and a splenic mass, and a solitary metastatic nodule along the parietal peritoneum are available as supplementary material
In two further studies, the ultrasonographic findings in five cats with lymphomatosis secondary to alimentary lymphoma were reported.43,44 In all cats, the mesenteric fat was variably increased in echogenicity and a small volume of free anechoic peritoneal fluid was present. In each case, the primary gastric, small or large intestinal lesion was identified and found to be typical in appearance for neoplasia. Discrete-to-coalescing plaques of thickened, hypoechoic tissue were present throughout the mesentery or omenta in all cats and, in three cats, multiple small hypoechoic nodules were scattered throughout the parietal and/or visceral peritoneal surfaces. 43 In the fifth cat, direct extension from a lymphomatous colonic mass was suspected, leading to localised mesenteric involvement. 44 As with the other cases, small multifocal islands of nodular hypoechoic tissue were visible within the mesenteric fat surrounding the colonic mass. 44 Additional findings included renomegaly and regional lym-phadenopathy in two cats each, due to concurrent neoplastic infiltration.
In all four studies, ultrasound-guided fine-needle aspiration of visceral masses, peritoneal nodules and/or fluid was often helpful in confirming the diagnosis and ruling out differentials such as feline infectious peritonitis, pancreatitis, chronic peritonitis and mesothelioma.37,41,43,44
Encapsulating peritoneal sclerosis
There are occasional case reports in the veterinary literature documenting encapsulating peritoneal sclerosis (EPS) in cats, also known as ‘sclerosing encapsulating peritonitis’, ‘abdominal cocooning’ or ‘peritonitis chronica fibrosa incapsulata’.45–47 EPS is a rare, chronic inflammatory condition resulting in the progressive transformation of the peritoneum into a thick, fibrotic layer and subsequent encasement of the intestines and other abdominal organs. 46 Chronic intestinal obstruction and ascites are common sequelae, and consequently clinical signs such as anorexia, vomiting and abdominal distension often result.45–47 Reported inciting causes in cats include previous ovariectomy 46 and pansteatitis. 45
Ultrasonographic descriptions of the condition in this species are limited to a single case report. 46 A copious volume of peritoneal effusion was identified in addition to rounded hepatic margins associated with a thick, echogenic capsule. The intestines were clustered together within the dorsal aspect of the abdomen and distended with normal content. Abdominocentesis revealed serosanguineous fluid of low viscosity and low cellularity, containing occasional macrophages and erythrocytes.
Exploratory coeliotomy typically reveals a thickened, discoloured peritoneum covering the abdominal organs.45–47 Small intestines may be tightly coiled and multiple adhesions are present between intestinal loops. Of the five cats in which this condition has been reported, one was euthanised at surgery on account of an inability to resect the adhesions, three survived between 3 and 57 days post-surgery and the remaining cat was alive 11 months after diagnosis but no further follow-up was reported.45–47
Aortic thromboembolism
Thromboembolism of the aorta is a common complication of myocardial disease and thrombus formation in the left atrium. 48 Embolisation of the thrombus results in partial or complete vascular occlusion, most commonly within the terminal abdominal aorta at the level of the aortic trifurcation (saddle thrombus), leading to pelvic limb paresis/paralysis. Cats with hyperthyroidism, supravalvular mitral stenosis and pulmonary neoplasia may also be at risk. 48
Acute thrombi may be almost anechoic and are very difficult to identify.7,10 It may be possible to differentiate an immature thrombus from anechoic blood by increasing the gain. 10 Chronic thrombi are usually echogenic relative to blood and frequently visible on standard B-mode ultrasound (Figure 21a). 7 Some thrombi may be uniformly hyperechoic, whereas others can appear heterogeneous due to lysis of the blood clot and subsequent recanalisation. 7 Absent or reduced flow within the vessel can be identified on colour, power or spectral Doppler confirming the diagnosis (Figure 21b). Some aortic thromboemboli extend into the external iliac vessels and hence these should also be thoroughly examined, particularly if clinical signs (cold limb, lack of femoral pulse) are unilateral.

Extensive chronic aortic thrombus in a 10-year-old neutered female Greyhound. (a) B-mode ultrasound image showing abnormal echogenic material (the thrombus) within the aorta at the level of the aortic trifurcation. Although this particular image is from a dog, a similar appearance would be expected of a chronic thrombus in a cat. (b) Colour Doppler image confirming flow cranial to the thrombus and an absence of flow at the level of the thrombus itself. Videos showing these findings from this case, both with B-mode ultrasonography and colour Doppler, are available as supplementary material


Footnotes
Conflict of interest
The author declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
Ethical approval
This work did not involve the use of animals and therefore ethical approval was not specifically required for publication in JFMS.
Informed consent
This work did not involve the use of animals (including cadavers) and therefore informed consent was not required. No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
