Abstract
Objectives
Venlafaxine, a specific inhibitor of both noradrenaline and serotonin, is commonly used in human medicine to treat depression, anxiety and social phobia. Its formulation in small granules renders it interesting to test on cats, which are usually reluctant to take medication. Venlafaxine was administered at 1 mg/kg for 60 days, using a double-blind, placebo-controlled protocol, to cats aged ⩾6 months exhibiting aggressiveness, fear or house-soiling.
Methods
After one cat’s withdrawal, 21 cats were included in the study: 11 in the venlafaxine group and 10 in the placebo group. Three consultations were conducted, on day 0, day 30 and day 60. Each visit consisted of (1) veterinarian- and owner-based scoring of the cat’s behavioural improvement; (2) scoring of the cat’s compliance with removal from its carrier and compliance with manipulation; and (3) owner scoring of the ease of administration and recording of the potential adverse effects of the treatment.
Results
Improvement was significantly higher in the venlafaxine group; as early as day 30, according to the veterinarian scoring, and at day 60, according to both the veterinarian and owner scoring. In contrast, neither the removal nor the manipulation scores were significantly different between the two treatment groups. Venlafaxine seemed to improve all three studied signs, as early as day 30 for fear and aggressiveness, and at day 60 for house-soiling. The adverse effects of venlafaxine were limited to drowsiness in one cat. All cats tolerated the treatment well during the 60-day period.
Conclusions and relevance
These results suggest that venlafaxine is efficient in treating several behavioural problems and is easy to administer. More studies should be conducted to explore its effects at different dosages on specific diagnoses.
Introduction
In veterinary medicine, few psychotropic drugs are available to treat companion animals, especially cats.1,2 However, many behavioural disorders cannot be treated without the help of psychotropic drugs given daily for long periods and combined with behavioural modification. In cats, very few drugs are available with official market authorisation; many of those that are available are challenging to administer. The difficulty in administering treatments is such a problem that it often leads veterinarians to try other options such as alpha (α)-casozepine 3 and pheromones to treat behavioural issues in cats. 4 However, in many cases these options are not sufficient to manage the cat.
Venlafaxine is a dual serotonin (5-HT) and noradrenaline reuptake inhibitor (NRI); it combines the properties of a selective serotonin reuptake inhibitor (SSRI) and an NRI. Venlafaxine comprises a racemic mix of two enantiomers. 5 Its plasma concentration is similar for both enantiomers in humans and dogs.6,7 Venlafaxine is absorbed by the gastrointestinal tract and biotransformed in the liver by cytochrome 2D6 (CYP2D6) to generate two main metabolites: an active metabolite called O-desmethylvenlafaxine (desvenlafaxine) 6 and a secondary metabolite called N-desmethylvenlafaxine. 1 Other metabolites seem to play a minor role in the mechanism of action of venlafaxine. 8
The serotonergic effects of venlafaxine occur at low doses, while its noradrenergic effects are progressively enhanced as the dose is increased. Like venlafaxine, desvenlafaxine inhibits reuptake of 5-HT (serotonin reuptake inhibitor [SRI]) and NRI, but its NRI actions are greater than its SRI actions. 9 In plasma, desvenlafaxine is usually twice as concentrated as venlafaxine, depending on the genetic profile of the patient with regard to the expression of CYP2D6. Because its action is more noradrenaline-based, desvenlafaxine has been purified to produce a selective NRI (SNRI) that is more noradrenaline-oriented than the venlafaxine one. 9
To illustrate these specificities, Katofiasc et al compared the use of SNRIs (duloxetine and venlafaxine) with the simultaneous use of an SSRI (norfluoxetine) and an NRI (thionisoxetine) to increase bladder capacity and urethral sphincter electromyographic activity in cats. 10 They concluded that ‘there are unexplained pharmacological differences between the effects of single compounds that exhibit dual NE [noradrenaline] and 5-HT reuptake inhibition and a combination of compounds that exhibit selective NE and 5-HT reuptake inhibition’.
This dual action of the SNRI family can also be compared with that of tricyclic antidepressants. SNRI molecules have fewer adverse effects as they do not block α1-noradrenergic, histaminic or cholinergic receptors. 11
Venlafaxine was the sixth most prescribed antidepressant in humans in the USA in 2007, with 17.2 million prescriptions, 1 and the second-most prescribed in 2008. 12 It has been proven efficient in several mental disorders such as anxiety, depression, social phobia and panic attacks. 1 Several genetic variants of CYP2D6 have been identified, leading to different effects of venlafaxine.5,13 These different profiles explain the failure of some treatments and the variation of the dose needed from one individual to another. The doses commonly used range from 1 to 2 mg/kg.
In pets, only five publications in cats and seven in dogs have investigated the effects of venlafaxine, whereas 68 publications about cats and 66 about dogs investigated the effects of fluoxetine, one of the most commonly used psychotropic drugs in pets. In dogs, venlafaxine is indicated for the treatment of neuropathic pain at a dose of 4 mg/kg and narcolepsy at a dose of 2.5 mg/kg.14,15
In cats, venlafaxine has been used for the treatment of idiopathic cystitis at doses ranging from 1 to 2 mg/kg PO,2,16 and refractory misdirected play and impulse-control aggression at a dose of 1.1 mg/kg PO. 17 Moreover, two studies investigated the toxicity of venlafaxine. Bjorvatn et al showed that full inhibition of the neurons of the dorsal raphe only occurs at doses of at least 5 mg/kg IV. 11 Pugh et al showed that in 12 cats that ingested their owners’ venlafaxine at doses ranging from 5 to 30 mg/kg, six were asymptomatic, while the other six recovered from adverse effects (sedation, depression, arousal, gastrointestinal disorders, tachycardia and hypertension) with fluid therapy. 12
In addition, venlafaxine is marketed (under the name Effexor) in the form of pills containing very small granules, which are easy to administer to cats. We therefore assessed the effects and, because cats are known to be very reluctant to take treatments, also the ease of administration of venlafaxine was also assessed. Thus, we investigated the possible efficacy of venlafaxine in cats presenting with a range of problem behaviours namely aggression, house-soiling and fear, in order to clarify possible indications for venlafaxine in cats to be explored in future studies.
Materials and methods
Subject recruitment and venlafaxine dosage
The subjects of our study were healthy, adult domestic cats (Felis silvestris catus) of both sexes and neuter status, which exhibited excessive fear reactions (ie, recurrent tremors, sialorrhoea, anisocoria or attempts to escape and/or hide in response to a stimulus), exaggerated aggressive behaviours (ie, growling, hissing or attempts to scratch and/or bite in the presence of humans or cats) and/or house-soiling. In other words, cats were included in our study if – and only if – they exhibited one or several of the aforementioned behavioural signs, were ⩾6 months of age and had not been receiving medication other than vaccines and deworming treatments for at least 15 days.
Data were collected in a total of 22 cats that had been recruited during routine visits by the clinical investigator (ie, the first author) at her own clinic with a goal of enrolling at least 20 cats. All the owners of the cats meeting the inclusion criteria were informed of the trial and asked to re-contact the clinical investigator if they wanted to participate. Interested owners were then given all the necessary instructions and gave their informed consent for participation.
The study was performed in compliance with the French government guidelines for research on animals (www.enseignementsup-recherche.gouv.fr).
Analyses were conducted on data from 21 cats. The owner of one of the cats withdrew from the trial after around a month, because the behavioural disorder (house-soiling) of her 7-year-old neutered female cat persisted despite the treatment, which turned out to be the placebo. This cat was subsequently treated with venlafaxine (Effexor), which resolved its behavioural disorder.
Prior to the start of the study and because there were no available data on the dose of venlafaxine that could be adapted to cats besides the two studies mentioned above,2,16 the clinical investigator explored the effects of this treatment at a single dose of 1 mg/kg q24h PO for 2 months on two of her own cats, a 5-year-old neutered male displaying fear reactions in the presence of other cats, strangers and noises, and a 4-year-old neutered female displaying aggressive behaviours towards other cats. The results of this preliminary test, which showed an improvement in both cats’ behavioural disorders with no adverse effects other than transient drowsiness in the female cat, led us to use this dose of venlafaxine in our study. Moreover, while our experiment was being conducted, there was a further report published in which a dose of 1.1 mg/kg was used in a cat, 17 which was in line with the doses chosen for our study.
General procedure
The study was designed as a double-blind, placebo-controlled clinical trial in which 21 cats were assigned to either the venlafaxine group (n = 11) or the placebo group (n = 10).
For 60 days, all cats were administered by their owners a single 4 mg, 5 mg or 6.5 mg capsule q24h PO (depending on the cat’s weight) of either venlafaxine (for the venlafaxine group) or placebo (for the placebo group), without the cats’ owners or the clinical investigator being aware of the treatment allocation. To ensure blinding, the clinical investigator asked a pharmacy (Delpech Marseille) to compound venlafaxine capsules from commercially available granules (Effexor) and placebo capsules from cellulose powder, then asked a colleague to covertly rename these overtly identical capsules as ‘capsules A’ and ‘capsules B’. Moreover, cats’ owners were asked not to provide any information to the clinical investigator about the aspect of the content of the capsules ‘A’ or ‘B’ that had been provided blindly. The clinical investigator’s colleague did not unblind the trial until the analyses were complete.
One strong motivation for starting this clinical trial was the apparent ease of administration of venlafaxine. To ensure that cats could spontaneously take the treatment, the owners were asked to use a small amount of wet food or treat loved by their cat to administer it.
The study comprised three test phases or visits at the clinical investigator’s clinic that took place 1 day before (D0), 30 days after (D30) and 60 days after (D60) starting the treatment. Each visit consisted of (1) a clinical examination of the cat leading to scoring of its compliance with removal from the carrier (‘removal score’) and scoring of its compliance with being handled (‘manipulation score’); (2) a veterinarian-based evaluation (‘veterinary score’) and an owner-based evaluation (‘owner score’) of the cat’s behavioural improvement; and (3) the owner’s recordings of both the ease of administration (converted into an ‘administration score’) and the potential adverse effects of the treatment. The owner score was considered to be 0 on day 0 and administration scores were not recorded on day 0.
For ethical reasons, the authors decided not to let the cats that would receive the placebo to go without any treatment at all. Consequently, during the first consultation, the owners of each cat, regardless of its treatment group (placebo or venlafaxine), were given both general and specific advice to remedy their animal’s behavioural signs.
For all the presented complaints, owners were asked neither to reprimand nor punish their cat. The veterinarian insisted on the fact that, if the cat seemed scared when it was reprimanded, this reprimand was received as a punishment by the cat and would only render it more anxious or aggressive. 18
The specific advice given for house-soiling consisted of using at least one litter tray per cat plus using one that was large enough and long enough (one times the width of the cat and one and a half times the length of the cat), putting the tray in a calm area, using a substrate that the cat was not reluctant to use, 19 removing stools and urine waste once daily and cleaning the litter tray completely once a week. 20
The specific advice given for dealing with aggressive behaviours consisted of moving away from the cat and letting it calm down, or interrupting the behaviour (by saying ‘no’ or ‘stop’) and redirecting it to play, depending on the specific cases encountered by the authors. For example, a cat chasing a person’s foot had to be redirected onto a toy, and a cat being aggressive towards another cat in the household had to be interrupted before or as soon as it started chasing the other, and taken away by the owners if no risk of redirected aggression was assessed by the veterinarian, to isolate it and let it settle down. The choices for both interrupting stimuli and redirecting behaviour were chosen individually for each case by the veterinarian, based on an interview with the owners.
Finally, the specific advice concerning fear was to never force or block the cat, but rather to let it explore at its own pace. Total avoidance of problematic situations (ie, isolating the cat in a room away from the stimulus eliciting fear) was discouraged. Instead, owners were asked to desensitise their cat’s phobia with a standard procedure: they were to bring the cat into the room containing the stimulus eliciting fear (vacuum cleaner, strangers, etc), to place it where it usually likes to be (eg, the top of the cat tree or on a piece of furniture) and let it explore. Owners were asked to observe the cat’s attitude and not to put it in uncomfortable situations (as evidenced by mydriasis, tremors, aggressive attempts to run away, etc). After repeated attempts, depending on the cat’s degree of relaxation, their owners could move one step further (eg, by asking the strangers to sit a bit closer to the cat). Owners were asked to give their cats special treats each time it voluntarily approached the stimulus that elicited fear. The veterinarian insisted on the key point of the procedure being to look at the cat’s postures; to bring it out of its comfort zone but never to let it panic was the common thread of the desensitisation therapy. In the case where the first step of the procedure could not be achieved, the owners were asked to do nothing and wait for the cat to be able to stay in the room without the eliciting stimulus and make new attempts every day.
Investigated items and statistical analyses
The effect of the administered treatment (placebo or venlafaxine) on the cats’ behaviour was first and foremost assessed by the statistical comparison of the veterinary and owner scores of cats in the venlafaxine group with those in the placebo group.
The veterinary score, detailed in Table 1, ranging from 0 (ie, no behavioural disorder) to 70 (ie, severe behavioural disorder), corresponds to the sum of the answer to the six possible choices (0, 2, 3, 5, 7, 10) for each of the seven questions. This clinical questionnaire was completed by the veterinarian based on a 1 h interview with the cat’s owners and direct clinical observations. Because a cat’s behaviour can be altered in an unfamiliar environment, the practitioner always probed what was observed and what the owners described, by asking them whether the cat would behave the same way at home. This regular, complete behavioural consultation included an estimate of the number and severity of episodes to provide a point of comparison for follow-up consultations. All the cat’s behaviours were reviewed to establish the diagnosis (see Table 2) and the table was completed. This clinical questionnaire was used to assess the cat’s emotional status. It screened several behavioural axes (affiliation behaviour, fear, aggression, etc) and was not designed to establish a diagnosis, but rather to detect a deviation from normal behaviour. The greater the score the more severe the deviation was. This questionnaire was completed by the clinical investigator by conducting a detailed interview with the owner, like any behaviour consultation, and interpreting it alongside the clinical observations.
Veterinary score items

Main signs and emotional status diagnosis

P = placebo; V = venlafaxine
For the two follow-up visits, the veterinarian completed the table based on shorter interviews. The point of this procedure was to compare the cat’s behaviour on separate visits based on quantitative values.
The owner score, ranging from 0 (no positive effect) to 10 (maximum positive effect), corresponded to the owner’s evaluation of the cat’s response to treatment.
The effect of the administered treatment (placebo or venlafaxine) on the cats’ behaviour was secondarily assessed by statistical comparison of the removal and manipulation scores of cats in the venlafaxine group and those in the placebo group (veterinarian-assessed). The removal score (see Table 3) corresponded to the cat’s degree of compliance with removal from the carrier, while the manipulation score (see Table 4) corresponded to the cat’s degree of compliance with being handled by the clinical investigator, based on Kassler and Turner’s (1997) Cat Stress Score. 21 This handling first consisted of making contact with the cat, by stroking it, on the consultation table. If this contact was accepted, the veterinarian proceeded with a quick clinical examination, with abdominal palpation, simple manipulation of the limbs and the head (examining the ears, if possible) and a cardiac auscultation. Finally, the veterinarian let the cat move around on the table, explore the room or hide or return to its carrier.
Scoring of cat’s compliance with removal from the carrier

Scoring of cat’s compliance with handing (manipulation)

Finally, we compared the administration scores, ranging from 0 (impossible to administer) to 10 (taken spontaneously) and the number of potential adverse effects of placebo and venlafaxine.
Because all the aforementioned scores were ordinal data, all were analysed with non-parametric tests conducted with R statistical software (https://www.rproject.org). A significance threshold of 0.05 was adopted for all statistical analyses.
Results
Demographics
Of the 21 cats that completed the trial (see Table 5), nine displayed exaggerated fear reactions, six displayed aggressive behaviours and six displayed house-soiling. The proportions of cats displaying each of these behavioural disorders were identical in the two treatment groups except for fear (five cats in the venlafaxine group). The veterinary scores at D0 (ie, before starting the treatments) were significantly higher (two-tailed Mann–Whitney test: U[11,10] = 23.5; P = 0.029) in the venlafaxine group (mean 34.8 ± 8.0) than in the placebo group (mean 24.3 ± 9.1).
Characteristics and scores of the cats in the placebo (P) and venlafaxine (V) groups on day 0 (D0)

DSH = domestic shorthair; B = Birman; BS = British Shorthair; MN = male neutered; FN = female neutered; FI = female intact; HS = house-soiling; F = fear; A = aggression
The 21 cats were of three different breeds (one Birman, two British Shorthair and 18 domestic shorthair cats); the proportions of cats of each breed (except for the Birman) were identical in the two treatment groups.
The 21 cats included 13 females (10 neutered) and eight males (all neutered); the proportions of cats of each sex and neuter status were not significantly different between the two treatment groups (Freeman–Halton extension of the Fisher exact probability test for a two rows by three columns contingency table: P = 0.50).
Finally, the 21 cats were aged 1–14 years (mean 5.6 ± 4.4) and there was a significant difference (two-tailed t-test for independent samples with unequal variances: t(12,5) = 2.92; P = 0.01) between the age of the cats in the venlafaxine group (8.15 ± 4.85 years) and those of the placebo group (mean 3.24 ± 2.27). Thus, this difference will be accounted for in subsequent analyses.
Effects of the treatments
All scores at D30 and D60 are presented in Table 6. The differences (see Figure 1) between the veterinarian scores at D30 and at D0 (delta [Δ] veterinarian scores at D30) were significantly higher (two-tailed Mann−Whitney test: U[11,10] = 22; P = 0.022) in the venlafaxine group (mean 13.4 ± 10.7) compared with the placebo group (mean 5.6 ± 6.4).
Scores at day 30 (D30) and day 60 (D60), in comparison with day 0, of the cats in the placebo (P) and venlafaxine (V) groups


Delta veterinarian scores at day 30 (D30) and day 60 (D60) in the two treatment groups
Similarly, the differences (see Figure 1) between the veterinarian scores at D60 and at D0 (Δ veterinarian scores at D60) were significantly higher (two-tailed Mann−Whitney test: U[11,10] = 12.5; P = 0.003) in the venlafaxine group (mean 21.8 ± 10.7) than in the placebo group (mean 7.9 ± 5.4).
Moreover, there was no significant difference between Δ veterinarian scores at D30 and Δ veterinarian scores at D60 in the placebo group (two-tailed Wilcoxon test: W = −15; P >0.05), whereas veterinarian scores at D60 were significantly higher compared with veterinarian scores at D30 in the venlafaxine group (two-tailed Wilcoxon test: W = −66; P = 0.004).
Finally, a series of two-tailed Spearman correlation tests showed that neither the Δ veterinarian scores at D30 nor the Δ veterinarian scores at D60 were significantly associated with the cats’ ages, whether the treatment groups were considered together (at D30: r[19] = −0.32; [P = 0.16]; at D60: r[19] = −0.42 [P = 0.06]) or separately (venlafaxine group at D30: r = 0.42 [P >0.20]; placebo group at D30: r = 0.61 [P = 0.06]; venlafaxine group at D60: r[9] = 0.39 [P = 0.23]; placebo group at D60: r[8] = −0.60 [P = 0.07]).
The owner scores at D30 (see Figure 2) were not significantly different (two-tailed Mann−Whitney test: U[11,10] = 29.5; P = 0.08) between the venlafaxine group (mean 7.2 ± 1.3) and the placebo group (mean 6.2 ± 0.9).

Owner scores at day 30 (D30) and day 60 (D60) in the two treatment groups
In contrast, the owner scores at D60 (see Figure 2) were significantly higher (two-tailed Mann−Whitney test: U[11,10] = 4; P <0.001) in the venlafaxine group (mean 7.8 ± 1.6) compared with the placebo group (mean 4.0 ± 1.2).
The differences between the cats’ removal scores at D30 and at D0 (Δ removal scores at D30, as shown in Figure 3) were not significantly different (two-tailed Mann−Whitney test: U[11,10] = 64; P = 0.55) between the venlafaxine group (mean −0.1 ± 0.8) and the placebo group (mean 0.2 ± 0.4). Similarly, the differences between the cats’ removal scores at D60 and at D0 (Δ removal scores at D60, as shown in Figure 3) were not significantly different (two-tailed Mann−Whitney test: U[11,10] = 62; P = 0.65) between the venlafaxine group (mean 0 ± 1.1) and the placebo group (mean 0.2 ± 0.4).
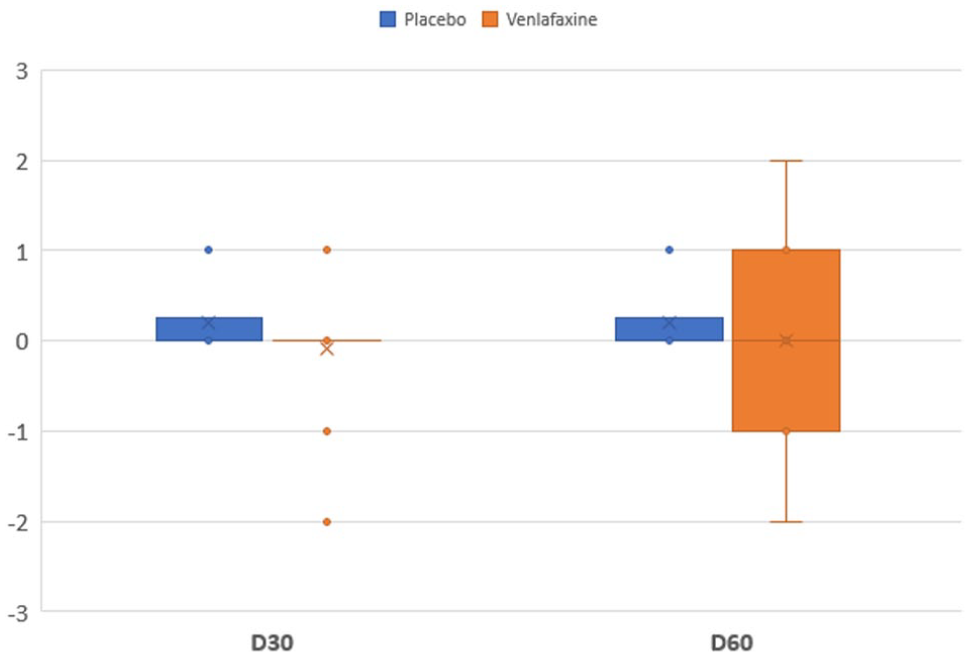
Delta removal scores at day 30 (D30) and day 60 (D60) in the two treatment groups
The differences (see Figure 4) between the cats’ manipulation scores at D30 and at D0 (Δ manipulation scores at D30) were not significantly different (two-tailed Mann−Whitney test: U[11,10] = 34.5; P = 0.16) between the venlafaxine group (mean 1.0 ± 1.3) and the placebo group (mean 0.6 ± 0.7). Similarly, the differences (see Figure 4) between the cats’ manipulation scores at D60 and at D0 (Δ manipulation scores at D60) were not significantly different (two-tailed Mann−Whitney test: U[11,10] = 29.5; P = 0.08) between the venlafaxine group (mean 1.5 ± 1.1) and the placebo group (mean 0.8 ± 0.6).

Delta manipulation scores at day 30 (D30) and day 60 (D60) in the two treatment groups
Unfortunately, the sample size was too small to statistically compare the effects of the two treatments on each of the cats’ signs (ie, on fear reactions, aggressive behaviours and house-soiling), but the Δ veterinary subscores for each of these signs at D30 and D60 (ie, the differences between the veterinarian subscores for each sign at D0 and at D30 or D60) could nevertheless be numerically compared between the two treatment groups.
At D30 (see Figure 5), the Δ veterinarian subscores for fear reactions were much higher in the venlafaxine group (mean 15.4 ± 5.5) than in the placebo group (mean 3.0 ± 7.3) and for aggressive behaviours were much higher in the venlafaxine group (mean 19.3 ± 10.1) than in the placebo group (mean 7.0 ± 7.2), but the Δ veterinarian subscores for house-soiling were much lower in the venlafaxine group (mean 4.0 ± 14.7) than in the placebo group (mean 7.7 ± 5.1).
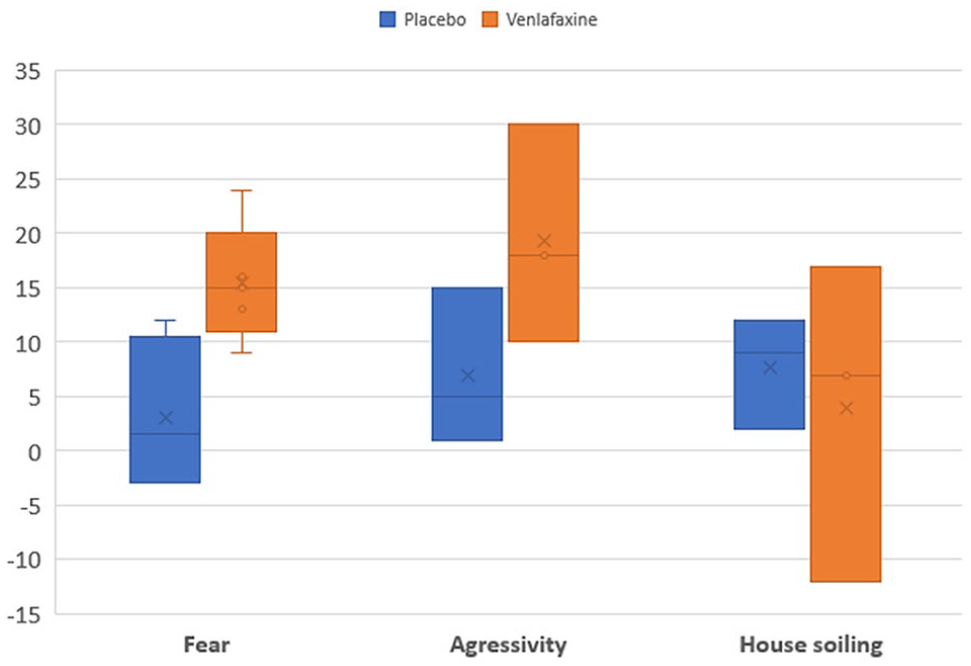
Delta veterinarian subscores at day 30 (D30) in the two treatments groups
At D60 (see Figure 6), the Δ veterinarian subscores for fear reactions were much higher in the venlafaxine group (mean 25.2 ± 6.6) than in the placebo group (mean 8.5 ± 5.5), the Δ veterinarian subscores for aggressive behaviours were much higher in the venlafaxine group (mean 23.7 ± 13.9) than in the placebo group (mean 7.3 ± 7.8), and the Δ veterinarian subscores for house-soiling were much higher in the venlafaxine group (mean 14.3 ± 13.3) than in the placebo group (mean 7.7 ± 5.1).

Delta veterinarian subscores at day 60 (D60) in the two treatment groups
Administration scores and adverse effects
The administration scores at D30 (see Figure 7) were not significantly different (two-tailed Mann−Whitney test: U[11,10] = 57; P = 0.91) between the venlafaxine group (mean 7.7 ± 2.6) and the placebo group (mean 8.0 ± 2.6). Similarly, the administration scores at D60 (see Figure 7) were not significantly different (two-tailed Mann−Whitney test: U[11,10] = 47; P = 0.60) between the venlafaxine group (mean 8.4 ± 1.9) and the placebo group (mean 7.4 ± 2.9).

Administration scores at day 30 (D30) and day 60 (D60) in the two treatment groups
In the placebo group, two cats experienced vomiting during the trial, whereas in the venlafaxine group, one cat experienced drowsiness during the first month of treatment. The dose of venlafaxine was not changed because, according to the owner, the reported effect was mild and resolved at the second appointment, 1 month after the start of the treatment.
Discussion
The results of this pilot study show that venlafaxine is significantly more efficacious than placebo in reducing a wide range of problem behaviours in cats, according to both the veterinarian and the owners. This effect seems to increase progressively, as the difference between the venlafaxine and placebo groups was more significant at D60 than at D30. This is consistent with the several weeks needed to witness the therapeutic effects of all anxiolytic drugs owing to the down-regulation process. 9
The fact that the score used to assess the cats’ behaviour is not validated is a limitation of the study. However, the purpose of the study was to have a simple way of scoring the cats, which could be applied to both venlafaxine and placebo. Significantly different results were seen in our two treatment groups. Nevertheless, the study should be replicated using different scoring systems.
The therapeutic effects of venlafaxine were slower to appear in our study than in that of Hopfensperger, 16 where 62% of the cats had full resolution of their urinary house-soiling signs in a week. Several hypotheses can explain this finding. First, the cats in our study displayed different behavioural signs, and venlafaxine could be more effective in treating house-soiling than fear reactions or aggressive behaviours. However, in our study, the veterinarian scores for each of the cats’ signs (ie, on fear reactions and aggressive behaviours as well as on house-soiling) were numerically better in the venlafaxine group than in the placebo group, and house-soiling is the sign that venlafaxine seems the least successful in treating. Secondly, the dose of venlafaxine used in our study was close to 1 mg/kg, whereas the doses used in Hopfensperger’s study ranged from 1 to 2 mg/kg. 16 The onset of the effects of venlafaxine may therefore be more rapid at these higher doses, but urine retention − an expected adverse effect of any SNRI − could also account, in part, for the results of Hopfensperger. Thirdly, unlike the study by Hopfensperger, 16 our study implemented a behavioural modification for each cat. This may explain not only why we observed so much improvement in the placebo group, but also why the venlafaxine effect was mildly visible at the second visit. However, more importantly, its effects might be hidden by those of the behavioural modification. This phenomenon, if confirmed by future studies, underlines the importance of behavioural modifications without detracting from the importance of medication to treat behavioural disorders in cats, as behavioural modification alone was not sufficient in the placebo group to achieve a satisfactory level of improvement. The same was observed in the study by Simpson et al on fluoxetine in dogs. 22
In contrast, the results of our study suggest that venlafaxine does not improve cats’ compliance with removal from their carriers or handling by the veterinarian. However, several studies on medications used to assist cats during transport to the veterinary clinic or their medical examination used these medications at high doses. For example, gabapentin improves the fear responses of cats during veterinary visits, but the dose administered is up to 10 times the usual dose. 23 A future study should thus investigate whether a higher dose of venlafaxine lowers fear responses during the veterinary visit or during transport. Adverse effects might prevent the use of such a dose, but the margin for manouevre with venlafaxine is comfortable, as shown by a study of toxicosis in cats. 12
Another major result of this study was that the administration of venlafaxine is remarkably easy. All cats readily accepted the medication (venlafaxine or placebo) which is notable for an animal as difficult to medicate as the cat. Two things contributed to this: the granules had a neutral taste rather than a repellent or even an appealing taste that would not suit all cats; and the quantity needed for a cat per day is very small. Combined with the absence of noticeable adverse effects at 1 mg/kg, these results should encourage more studies on venlafaxine in cats.
Finally, the small number of cats in this pilot study did not allow investigation of whether the efficacy of venlafaxine is higher for certain behavioural disorders than others. In the future, the respective effects of venlafaxine on each should be investigated separately. In addition, the effects arising from the complex pharmacological mechanisms of action of venlafaxine require many doses to be tested for each indication, as different effects can be expected at different doses. For example, to control aggression, we could expect effects of serotonin even at low doses, but higher doses will increase this effect and the NRI effects acting as catalyst of the SSRI effect might also be needed to obtain better control. Clarifying the effects of venlafaxine thus requires a lot of future work on larger populations of cats, and precise doses and diagnoses to allow better use of this drug.
Conclusions
This pilot study showed that venlafaxine is effective in treating behavioural disorders in cats without side effects. Its excellent ease of administration makes it even more promising for treating cats with behavioural disorders that are reluctant to take medication.
Further studies are needed to narrow down the range of doses that can be used safely, what indications venlafaxine can be used for and what the adverse effects may be.
Footnotes
Acknowledgements
The authors would like to thank all the cats and cat owners that agreed to participate in this study.
Conflict of interest
The authors whose names are listed above certify that they have no affiliations with or involvement in any organisation or entity with any financial interest (such as honoraria, educational grants, participation in speakers’ bureaus, membership, employment, consultancies, stock ownership or other equity interest, and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this paper.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work involved the use of non-experimental animals only (including owned or unowned animals and data from prospective or retrospective studies). Established internationally recognised high standards (‘best practice’) of individual veterinary clinical patient care were followed. Ethical approval from a committee was therefore not specifically required for publication in JFMS.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedure(s) undertaken (either prospective or retrospective studies). No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
