Abstract
Objectives
Malodors stemming from soiled cat litter are a major frustration for cat owners, despite the widespread use of absorbent litters with claims of odor control. Technologies for effective litter odor control have not been rigorously evaluated. Here, we report on the effectiveness of a novel litter formulation of 1-monochlorodimethylhydantoin (MCDMH)-modified clinoptilolite zeolite (MCDMH-Z) to control the odors of 3-mercapto-3-methylbutanol (3M3MB) and ammonia, the principal products generated by the enzymatic breakdown of felinine and urea, respectively.
Methods
The efficacy of MCDMH-Z for the odor control of 3M3MB was determined by solid-phase microextraction and gas chromatography mass spectrometry analysis, colorimetric analysis and a sensory panel. Enzyme inhibition was monitored by a colorimetric coupled assay for ammonia. The antimicrobial properties were measured by a reduction in colony-forming units (CFUs).
Results
3M3MB proved highly susceptible to modification by MCDMH-Z granules. Headspace above litter exposed to MCDMH-Z showed no detectable 3M3MB; levels >59 ng were detected in commercially available products. Urease activity decreased by >97% after incubation with MCDMH-Z to 0.14 mg/ml. Cat litter F showed comparable inhibition (0.13 mg/ml); others showed less inhibition, producing up to 4.8 mg/ml of ammonia. MCDMH-Z reduced the CFUs of Proteus vulgaris by six log reduction values in 30 mins; in the same amount of time, no reduction was seen with commercial products tested. The odor control capability of the MCDMH-Z granules was further supported by a sensory panel scoring 3M3MB-spiked litters.
Conclusions and relevance
Samples of commercially available litter products showed an effect on malodor, or inhibition of urease, or contained antimicrobial activity; no samples were capable of accomplishing these concurrently. In contrast, MCDMH-Z granules were effective in all three test categories. Control of felinine-derived odors, in particular, has the potential to improve cat owner satisfaction, and may beneficially affect cat behaviors provoked by pheromonally active sulfurous metabolites deposited in the litter.
Introduction
The majority of domestic cats are confined indoors throughout their lives, and deposit their daily urine and feces output in litter containers. 1 In the USA approximately five million tons of absorbent granules, largely inorganic, are consumed yearly for this purpose, and more than 3500 metric tons of feces and 9600 m3 of urine are voided into indoor litter containers every day.2,3 These deposits generate offensive odors that permeate household air, adversely affecting the quality of life of cat owners, and seriously affecting their enjoyment of cats as household companions.1,3 Despite the importance of the issue there are few published reports on either the scale and social impact of the malodor problem, or on the effectiveness of litter media in counteracting it.4–6 Only one peer-reviewed report on comparative aspects of commercial cat litter products appears to have been published in the past 25 years. 7
Microbial enzymatic breakdown of cat waste metabolites is largely responsible for generating malodors in the air above soiled litter beds. Ammonia produced by the action of microbial ureases on urea is a well-recognized example; most types of litter lay claim to mitigate the problem, largely by masking it with fragrances or using absorbent litter materials (eg, charcoal and bentonite).8–13 The notoriety surrounding cat litter odor production stems from urinary and fecal excretion of the thioether felinine (2-amino-7-hydroxy-5,5-dimethyl-4-thioheptanoic acid). Subsequent microbial modifications of this compound lead to the formation of thiol-containing compounds, of which 3M3MB is a major product.10,11,14 Thiol-containing volatiles produced from the breakdown of felinine are challenging to mitigate as the human odor detection threshold for 3M3MB and other sulfur-containing compounds is very low. 15 Currently, there are no data available on technological approaches that might allow the malodors produced from the breakdown of sulfurous metabolites deposited in cat litter to be overcome.
Heterocyclic N-halamine compounds such as 1-monochlorodimethylhydantoin (MCDMH) that stabilize and display oxidative chlorine have opened up new opportunities to use these compounds for cat litter odor control. This family of heterocyclic compounds can provide not only inhibition of microbial activity in litter, but also the direct interaction with odor-causing compounds, including volatile sulfur-containing malodorants, converting them into less repugnant reaction products. Here, micronized particles of MCDMH were used to modify the surface of clinoptilolite zeolite (MCDMH-Z) in order to examine the efficacy of the modified granules in mitigating odor from 3M3MB and other byproducts of cat litter deposits. We also measured the impact of MCMDH-Z on the enzymes and microbes responsible for their production. The results were compared with the efficacy of commercially available litter products, and demonstrate that this novel approach offers high level performance with likely benefits to household management of soiled cat litter.
Materials and methods
Reagents and test samples
Urea, 3-mercapto-3-methylbutanol (3M3MB), urease, 5,5′-dithiobis(2-nitrobenzoic acid) (DTNB), ethanol and the ammonia assay kit were purchased from Sigma-Aldrich. 2-(4-Morpholino)ethane sulfonic acid (MES) and beta-mercaptoethanol (βME) were purchased from Fisher Scientific. Cultures of urease-positive Proteus vulgaris (ATCC 8427) were obtained from the American Type Culture Collection (ATCC). Normal cat urine was withdrawn from the bladders of cats submitted for necropsy at the School of Veterinary Medicine and pooled, courtesy of Dr Bruce Hammerberg, North Carolina State University, College of Veterinary Medicine. Pooled material was shipped to Washington on ice. Additional normal cat urine was collected by extraction from pooled clumps of urine in bentonite in litter boxes used by the pets of authors affiliated with this study.
Commercial cat litter products were purchased at retail outlets in the US and UK. These consisted of granular mixtures of undisclosed composition, most likely blends of bentonite, zeolite and other inorganic particulates, though one contained only nuggets of hydrated calcite. Some products claimed to have antimicrobial activity. Examples of eight major brands were included, and identified herein only as samples A–H. Not all sample types were used in every experiment, although the packaging on all the litters sampled claimed that they were effective for odor control. N-halamine-modified clinoptilolite zeolite granules (MCDMH-Z) were provided by OxiScience, as well as untreated identical zeolite (Z) to use as a control. Clinoptilolite zeolite granules (mesh size 14/40) were purchased from KMI Mines. MCDMH was purchased from Lonza. Milling to microparticle dimensions and particle size analysis was conducted at CMC Milling, Fleetwood, PA, USA, using a Horiba LA-910 Particle Size Analyzer.
MCDMH-Z was prepared by exposing zeolite granules to a liquid dispersion equivalent to 1.75% of the weight of the granular substrate, and consisting of 40% micronized MCDMH (~4–6 µm) microparticles in isopropyl myristate (IPM). MCDMH-Z samples were examined in a JEOL model 6610LV scanning electron microscope at an accelerating voltage of 20 kV to image and collect elemental data. Energy dispersive X-ray spectroscopy (EDS) was performed with an Aztec System (Oxford Instruments).
While there are no published data on MCMDH toxicity in felids, the safety of all halohydantoins in vertebrates and invertebrates has been comprehensively reviewed by the United States Environmental Protection Agency. 16 The amounts incorporated into MCMDH-modified zeolite (approximately 0.2%) are unlikely to pose toxicological risks to cats using the material as litter.
Scanning electron microscopy and EDS
To examine the outer surface of the modified zeolite, granules were placed on aluminum scanning electron microscopy (SEM) stubs covered with high-vacuum carbon tabs (SPI Supplies), and then carbon coated in a carbon string evaporator (Ernest F Fullam). These samples were examined in a JEOL model 6610LV scanning electron microscope at an accelerating voltage of 20 kV to image and collect elemental data. EDS was performed with an Aztec System (Oxford Instruments), software version 2.1a and 20 mm2 Silicon Drift Detector.
To examine the interior surface of the granules they were placed on high-vacuum carbon on the surface of aluminum SEM stubs, and cut using a double-sided razor blade. The cut surfaces were not further coated before being examined in the JEOL 6610LV scanning electron microscope at an accelerating voltage of 12 kV using the variable pressure mode and a sample chamber pressure of 50 Pa. Imaging was carried out using the backscattered mode to determine differences in average atomic weight of the sample components. Elemental analysis was performed with the Aztec System (Oxford Instruments).
Determination of 3M3MB in aqueous solution after contact with litter samples
The cat litters were evaluated on their ability to modify 3M3MB in solution. Aliquots (1.5 g) of each sample were added to 5 ml of 1 mM 3M3MB in 50 mM MES buffer containing 150 mM KCl at pH 6.2. The suspensions were mixed well before serial sampling of the supernatants obtained at each time point by centrifuging the mixtures at 5724 g for 20 s. Supernatants (50 μl) were added to cuvettes containing 50 μl of 20 mM DTNB solution and 900 μl of 50 mM MES buffer at pH 6.8. The absorbance of TNB- was measured at 412 nm. A calibration curve in 50 mM MES buffer at pH 6.8 had been prepared, giving an extinction coefficient of 13986 M-1cm-1. 17
Determination of 3M3MB in cat urine after contact with litter samples
The influence of the intrinsic thiol content of cat urine on the interaction between litter and 3M3MB was measured by using the above protocol. In this case, litter samples (0.28 g or 0.56 g of each) were added to 500 µl of pooled cat urine containing 1.7 mM free thiols (3M3MB plus the urinary thiol background). At each time point tested an aliquot of the suspension was centrifuged at 5724 g for 20 s, and 30 µl of supernatant added to a cuvette containing 50 µl of 20 mM DTNB solution and 920 µl of 50 mM MES buffer at pH 6.8. The absorbance of TNB- was measured at 412 nm, as described above.
Headspace analysis of 3M3MB above litter samples exposed to 3M3MB
A solution (2 ml) containing 100 ppm 3M3MB and 200 ppm βME in 50 mM phosphate buffer (pH 7.0) was added to 500 mg of each litter in a 10 ml Supelco glass headspace vial. Headspace analysis was performed using a Supelco solid-phase microextraction (SPME) polydimethylsiloxane/Carboxen 75 µm fiber and an adsorption time of 10 mins following modified literature procedures.18,19 This fiber was chosen based on the molecular weight and polarity of the compounds analyzed. Before use and in between each type of litter, a fiber blank was performed in order to clear the fiber of potential residues from earlier runs. An Agilent 6850 series Gas Chromatograph with a 0.75 × 6.35 × 78.5 mm inlet liner was used. The oven settings were programmed to 40°C for 5 mins, and then ramped up to 125°C at 8.5°C/min for a total of 15 mins. The column had a flow rate of 1.2 ml/m and the detector temperature was set at 300°C. The inlet temperature was set to 250°C with the pressure at 9.14 psi. The mass spectrophotometer used was an Agilent 5973 Network Mass Selective Detector. The headspace was analyzed four times for each litter sample and the controls, and the ratio of the 3M3MB to βME peak areas, and the ratios of the oxidized 3M3MB to βME peak areas were averaged, and a standard deviation calculated for each test condition. 3M3MB routinely gave rise to two peaks in the chromatogram, corresponding to the intact compound (retention time = 10.06 mins) and one byproduct of its reaction after adsorption and de-adsorption on the SPME fiber (retention time = 11.91 mins). This peak was only present in headspace analysis experiments.
Determination of the effects of litter samples on urease activity
A solution containing Canavalia ensiformis urease (10 U/ml) and 200 mM phosphate buffer (pH 7.0) was incubated with 100 mg of each litter and the positive and negative zeolite controls at room temperature. After 5 mins, the samples were centrifuged for 60 s at 9240 g, and the supernatant was removed and urea (15 mM) was added. This solution was diluted 1:20, and 100 μl of the diluted solution was assayed following the Sigma Aldrich Ammonia Assay Kit protocol (AA0100). Briefly, the 100 μl aliquot was added to the Ammonia Assay Reagent and incubated for 5 mins. Then, 10 μl of L-glutamate dehydrogenase was added and the initial absorbance at 340 nm was recorded. The final absorbance was measured after 5 mins.
Antibacterial properties of cat litter samples
In the antibacterial study, P vulgaris (ATCC 8427) was used as an example of Gram-negative bacteria. To prepare the suspensions, P vulgaris 8427 was grown in broth at 37°C for 24 h. Cells were harvested by centrifuge, washed twice with sterile phosphate buffered saline (PBS), and then re-suspended in sterile PBS to 108–109 colony-forming units (CFUs)/ml.
Microbial tests were performed in a Biosafety Level 2 hood. One milliliter of the suspension was pipetted onto 5 g of each cat litter sample (E, MCDMH-Z and Z control) in a 1 l vessel. After measured contact times 20 lm 0.03 wt% sodium thiosulfate aqueous solution was added to quench any active chlorine, and the mixtures vortexed for 1 min and then sonicated for 5 mins. An aliquot of the supernatant solution was serially diluted, and 100 µl of each dilution was plated for colony counting after 24 h at 37°C.
Sensory evaluation of cat litter challenged with 3M3MB
Three cat litter samples (A, B, MCDMH-Z plus Z control) were used in a sensory evaluation of their respective efficacies in reducing the odor of 3M3MB. To 125 ml of each sample in a 1.25 l glass vessel 1 ml 100 ppm of 3M3MB was added, before being sealed with plastic film. Glass vessels were labelled with random numbers and presented to 10 judges in random order. Vessels were securely covered and allowed to equilibrate for 1 h prior to subsequent evaluation. Judges scored the sensory attributes as good, neutral, bad and other comments. All judges participated in a training session so as to understand the study objectives and to practice the evaluation protocol with nonchallenged samples in flasks.
Results
SEM (Figure 1a) shows the external surface Z (untreated clinoptilolite granules). Direct visualization of the 5 µm MCDMH-modified particles by SEM proved difficult because the irregularly shaped particles were not clearly distinguishable from the microtopography of the zeolite surface. To visualize the delivery of solid 5 µm MCDMH-particles to the external surface, we first used insoluble latex spheres (5 µm) dispersed in IPM and blended with zeolite granules (Figure 1b). EDS revealed the presence of chlorine (Cl) on zeolites subjected to MCDMH dispersion in IPM, as compared with the control (Figure 1c).

Surface modification of clinoptilolite zeolite granules using dispersions of solid microparticles in isopropyl myristate (IPM). (a) Scanning electron microscopy image of zeolite granules exposed to IPM. (b) Surface of granules exposed to dispersion of 5 µm latex spheres in IPM. (c) Energy dispersive X-ray spectroscopy spectrum of unmodified (top) and MCDMH-Z granules (bottom). The presence of chlorine in the MCDMH-Z granules is outlined in blue
Analysis of 3M3MB in aqueous solution and in cat urine
The cysteine thioether felinine (Figure 2a) can be broken down enzymatically by β-lyases yielding the odorant 3M3MB as a major reaction product. 10 3M3MB was therefore chosen as the thiol of interest to determine the extent to which litter samples could modify or retain the compound upon incubation of the mixtures. The results of the aqueous solution litter assays are shown in Figure 2b. Cat litter sample E was the most effective of the branded litters in impacting on the persistence of the thiol in solution, reducing the detectable levels by 82.2% over 93 mins. By contrast, cat litter samples A and H absorbed or modified the thiol only to the extents of 8.9% and 4.8% of the starting amounts, respectively. Thiol concentrations in the presence of MCDMH-Z fell by 99.5%, almost immediately.
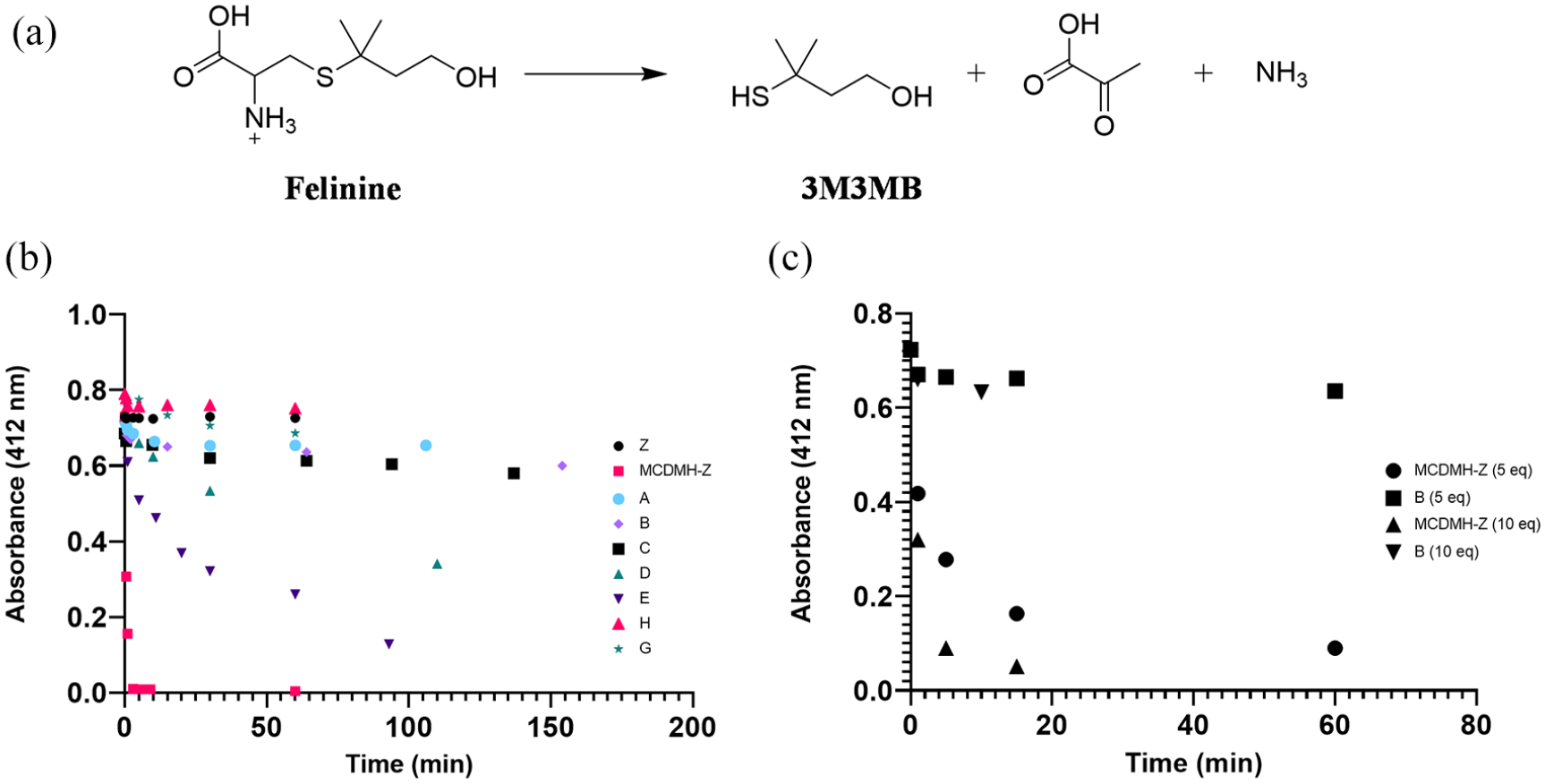
Effects of various cat litter samples on the concentration of 3-mercapto-3-methylbutanol (3M3MB) in solution. (a) Structure of felinine and the initial enzyme degradation products. (b) One mM solution of 3M3MB in 50 mM MES buffer (pH 6.2) was assayed for free thiols using DTNB at various time points in the presence of various cat litter samples. (c) DTNB assay for thiols in cat urine containing 3M3MB (total thiol concentration 1.7 mM) in the presence of 0.58 g cat litter B and MCDMH-Z
Although cat litter B showed only <17% decrease of 3M3MB in solution, it was chosen as a comparison with MCDMH-Z to determine if the effects were altered in the presence of cat urine owing to its claim for basic odor control in the absence of a claim as an antimicrobial. The same masses were used for both samples, calculated based on 5 and 10 equivalents of the loading capacity of the MCDMH-Z. With 5 equivalents, 91.5% and 22.5% of the free thiols (intrinsic cat urine thiol plus the added 3M3MB) remained in solution in the presence of cat litter B and MCDMH-Z, respectively, after 15 mins. With 10 equivalents of MCDMH-Z only 7.0% of the free thiols in solution remained after 15 mins. When cat litter B was tested with the 10 equivalents it could not be measured at 15 mins because of the high level of fluid absorption, but at 10 mins, 87.6% of the free thiols remained in the supernatant (see Figure 2c).
Effects of litter exposure on headspace concentrations of 3M3MB
The effectiveness of MCDMH-Z and cat litters G and B in decreasing the detectable amounts of 3M3MB in the headspace was measured by SPME and gas chromatography mass spectrometry analysis. Cat litters G and B were selected because they are branded versions of litters that claim odor control as a feature. Each sample was incubated in a buffered solution containing 3M3MB (100 ppm) together with βME as an internal standard. The ratio of the areas under the curves of 3M3MB and βME was determined. Ratios for cat litters G and B were 2.2 ± 0.05 and 4.7 ± 0.39, respectively (see Figure 3), which corresponded to 59 ng (cat litter G) and 110 ng (cat litter B) of 3M3MB in the headspace adsorbed onto the SPME fiber, respectively, as determined from a standard curve of 3M3MB. No detectable 3M3MB or βME was measured in the headspace above MCDMH-Z.

Effects of MCDMH-Z and cat litters G and B on the headspace analysis of 3M3MB. The ratio of 3M3MB to β-mercaptoethanol after 10 mins of incubation with the SPME fiber is shown. **Not detected
Effect of litter exposure on urease activity
Jack bean urease activity was used as an indicator of inhibitory effects on enzymes generating ammonia from urinary substrates after exposure to litter samples. The results are shown in Figure 4. Urease activity was least affected by cat litters G, H, B and C. The most significant decrease in urease activity occurred with cat litter F and MCDMH-Z, reducing ammonia production to 0.14 and 0.13 mg/ml, respectively.

Effect of cat litter samples on urease activity. Urease was incubated with the various cat litter samples and then tested by monitoring the production of ammonia from urea hydrolysis
Antimicrobial efficacy
The antibacterial efficacy of cat litter E containing boric acid, a known antimicrobial, vs P vulgaris was evaluated on contact. 20 The results are shown in Table 1. Cat litter E caused a reduction in the number of CFUs of bacteria of <1 log (ie, <90%) after 300 mins of exposure to the inoculum. By contrast, exposure to MCMDH-Z produced a log reduction value of 6 (ie, 99.9999%) at all time points tested.
Reduction of colony-forming units (CFUs) of Proteus vulgaris microbial cells after different periods of contact with Z-negative control, MCDMH-Z and cat litter E

Data represent the average log reduction value of the number of CFUs of the bacteria. P vulgaris test suspensions contained 108 CFUs/ml
Sensory evaluation of litter efficacy vs 3M3MB
Sensory evaluation scores are shown in Figure 5. All judges found the thiol odor in the negative control to be offensive; seven judges also scored the samples incubated with either cat litter B (with deodorizing claims) or cat litter F (contained a fragrance) offensive. In contrast, only one judge found the sample incubated with MCDMH-Z offensive. No intermediate scores were assigned by any of the judges.

Histogram of sensory scores recorded by a panel of 10 untrained judges after evaluation of headspace odor above litter samples to which 3M3MB had been added 4 h previously
Discussion
Cat litter purchases make up a large proportion of consumer spending on domestic cat care, yet there are few published data on the efficacy of commercially available cat litters in the scientific literature.7,9 Some patent disclosures reveal the presence of antimicrobial additives or malodor absorbents, but overall marketplace formulations are of unspecified compositions, and their functions are described in package labels and marketing materials rather than as a result of independent experimental analysis. Our studies on a novel granular litter medium based on the integration of a heterocyclic N-halamine, MCDMH, into an aluminosilicate substrate enabled us to focus not only on important sources of nuisance malodor in cat-soiled litter, but also to characterize how well an array of commonly used litter products compare in this regard.
Clinoptilolite zeolite has intrinsic absorptive capacity that makes it a useful constituent of many commercial litter products, especially in ammonia odor mitigation.3,21 The demonstrable presence of chlorine atoms on the surface of MCDMH of zeolite by SEM and EDS (Figure 1) proved capable of powerful enzyme inhibition, potent antibacterial efficacy and rapid modification of 3M3MB, all of which are likely to contribute to effective odor control, to the benefit of pet owners. For example, daily felinine production from neutered domestic cats is 36 and 41 µmol/kg body weight for females and males, respectively. 14 The laboratory studies were scaled to use up to 1:100 of the amount of 3M3MB from the complete breakdown of that daily felinine output. The amount of litter in a typical cat litter box (approximately 500 g) would therefore likely contain sufficient active chlorine to mitigate the odor produced from that total. Commercial litter samples included in our study showed slight changes to measurable amounts of 3M3MB in solution and in the headspace. Several displayed inhibitory effects on ammonia-generating urease, a major source of ammonia odor in cat litters (Figure 4) and only one cat litter (Table 1) showed any antimicrobial activity, albeit with a slow onset and low efficacy. However, none of the cat litters was able to affect all aspects of odor-generation mechanisms tested simultaneously. Taken together, a reduction in 3M3MB may occur by direct modification of the compound and by inactivation of the microbes and enzymes involved with felinine breakdown. Thiol-containing compounds such as 3M3MB, a major product of the enzymatic breakdown of felinine, are similar to odoriferous compounds with potential pheromonal characteristics, including those produced from thioethers in the human axilla.10,22–24 The components of feline waste and anal gland secretions have been characterized in recently published work, in attempts to understand how felinine is produced in cats, and whether it might function as a pheromone affecting cat behavior.25,26
While previous studies demonstrated felinine only in cat urine, it has recently been detected in feces.25 This indicates that solid and liquid waste may contribute to the presence of 3M3MB in cat litter and that it may be responsible for the distinct odor of cat feces.10,11,14,25,26 There are higher concentrations of both testosterone and felinine in body fluids and urine of intact males than in females, and the amounts of both analytes decline after castration.27,28 These characteristics support the idea that felinine is a hormonally controlled pheromone with a role in sex recognition. Short-chain fatty acids (eg, propionic acid) released from anal sacs, in contrast, appear to be sex-independent markers, and may be involved in individual animal recognition. 26
Conclusions
The presence of felinine, and ultimately 3M3MB, in litter might therefore affect animal behavior in households with multiple cats. Modification of 3M3MB and other sulfur-containing waste products by cat litter in the ways described above may not only provide for more effective malodor control, as our experience with the sensory perception test suggests, but also alter undesirable cat behaviors (eg, aggression and aberrant elimination behavior) arising from responses to pheromonal signals from the litter box. 29
Footnotes
Acknowledgements
We would like to thank Dr Jeffrey Williams, Chief Technology Officer of OxiScience, for his helpful review of this manuscript.
Conflict of interest
Dr Jeffrey Williams, Chief Technology Officer of OxiScience, provided insight for the potential applications of oxidative chlorine activity (eg, odor control, enzyme inhibition and antimicrobial activity) but was not involved with the design of the experiments.
Funding
This study was supported by a gift from OxiScience LLC to the University of Washington Bothell STEM Biochemistry Research Fund.
Ethical approval
This work involved the use of non-experimental animals only (including owned or unowned animals and data from prospective or retrospective studies). Established internationally recognized high standards (‘best practice’) of individual veterinary clinical patient care were followed. Ethical approval from a committee was therefore not specifically required for publication in JFMS.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedure(s) undertaken (either prospective or retrospective studies). No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
