Abstract
Practical relevance:
Abdominal ultrasound plays a vital role in the diagnostic work-up of many cats presenting to general and specialist practitioners. Ultrasound examination of the adrenal glands can provide important information pertaining to several conditions including hyperaldosteronism and hyperadrenocorticism.
Clinical challenges:
Despite ultrasonography being a commonly used modality, many practitioners are not comfortable performing an ultrasound examination or interpreting the resulting images. Even for the experienced ultrasonographer, differentiating between incidental findings, such as adrenal mineralisation, and clinically significant pathological changes can be challenging.
Aim:
This review, part of an occasional series on feline abdominal ultrasonography, discusses the ultrasonographic examination of the normal and diseased adrenal glands. Aimed at general practitioners who wish to improve their knowledge of and confidence in feline abdominal ultrasound, this review is accompanied by high-resolution images and videos available online as supplementary material.
Equipment:
Ultrasound facilities are readily available to most practitioners, although the use of ultrasonography as a diagnostic tool is highly dependent on operator experience.
Evidence base:
Information provided in this article is drawn from the published literature and the author’s own clinical experience.
Keywords
Imaging the adrenal glands
Unless markedly enlarged or mineralised, the adrenal glands are not visible radiographically; occasionally, however, radiography may provide useful information about the secondary effects of adrenal disease as a consequence of the associated hormonal dysfunction. 1
Ultrasonography is non-invasive, readily available, does not require the use of general anaesthesia and allows direct visualisation of the glands. Despite this, a relatively high level of skill is required to reliably locate the adrenal glands and a good knowledge of the regional anatomy and local landmarks is imperative. Adequate patient immobilisation, and operator patience and persistence, are the keys to success.
Ultrasound scanning technique
Due to their small size, a medium to high frequency (>7 MHz) linear or microconvex transducer is necessary to visualise the glands and, depending upon the exact depth of penetration required, the highest possible frequency should be used to optimise resolution. If an adrenal gland cannot be located initially using a high frequency linear-array transducer, the author uses a lower frequency microconvex transducer to locate the gland before returning to the higher frequency probe for the best resolution.
While there are relatively few requirements to image the adrenal glands in cats, owing to the relative scarcity of adrenal disease in this species, it is nevertheless important to be able to recognise and assess them routinely during an abdominal ultrasound examination. 2 The adrenal glands are paired retroperitoneal organs located either medially or craniomedially to each kidney and can be reliably visualised with ultrasound. 3 Some authors report that identification of the glands is easier in cats than in dogs, although this is subjective.3,4 The author’s preference is to image the left adrenal gland with the cat positioned in right lateral recumbency, and vice versa; although, depending upon personal preference, a ventral approach with the cat in dorsal recumbency is also perfectly acceptable.
Left adrenal gland
The position of the left adrenal gland can be determined by identifying the left kidney in the longitudinal plane and fanning medially. It is usually located in the area bordered by the left renal artery caudally, the coeliac and cranial mesenteric arteries cranially, the left kidney laterally and the aorta medially (Figure 1). The coeliac and cranial mesenteric arteries are the first two arteries to branch off the abdominal aorta. Since they always emerge from the aorta in close proximity to each other, they are easily recognisable landmarks. The left renal artery emerges from the aorta further caudally and forms a characteristic hook-shape as it travels towards the kidney. Typically, only part of the left gland is visible initially with this technique since the long axis of the gland is not necessarily parallel with that of the kidney. Thus the transducer must be rotated approximately 10–15° clockwise for the entire adrenal gland to appear on screen. 5 Once identified, in a similar fashion to that described in an earlier article in this series on the kidneys (see box on page 45), the adrenal glands are scanned in at least two orthogonal planes, usually the transverse plane followed by either the sagittal or dorsal plane.

Images illustrating the various landmarks in relation to the left adrenal gland in three cats. (a) The adrenal gland (short arrow) is visible medial to the kidney, caudal to the coeliac and cranial mesenteric arteries (arrowheads), and cranial to the left renal artery (long arrow), which is only partly visible. The coeliac artery emerges from the aorta cranial to the cranial mesenteric artery. (b) The left renal artery (arrow) forms a hook caudal to the left adrenal gland (between the measuring calipers). (c) The coeliac and cranial mesenteric arteries (arrowheads) are shown in cross section cranial to the adrenal gland (between the measuring calipers). A portion of the left renal artery (long arrow) is visible caudal to the adrenal gland. Two videos showing the normal ultrasonographic appearance of the left adrenal gland are available as supplementary material

Right adrenal gland
The right adrenal gland can be slightly more difficult to visualise and is usually found in close association with the caudal vena cava just caudal to the liver, in the area craniomedial or medial to the right kidney and dorsolateral to the caudal vena cava (Figure 2).4,6 In some cats, the search may need to be widened slightly as either adrenal gland can also be located slightly cranial to their respective kidney. 7 For the right adrenal gland, it is helpful to start with a longitudinal image of the caudal vena cava at the level of the right kidney and fan slowly dorsolaterally. The angle through which the hand rotates to move between the caudal vena cava and adrenal gland is surprisingly small; the gland is easily missed if movements are exaggerated or made too quickly. Care should be taken not to apply too much pressure with the transducer and inadvertently collapse the thin-walled caudal vena cava. Left and right phrenico-abdominal veins, which are small veins originating directly from the caudal vena cava, may be visible immediately ventral to each adrenal gland.4,8 It is important to note that the kidneys may not be visible when the image is optimised for the adrenal glands.
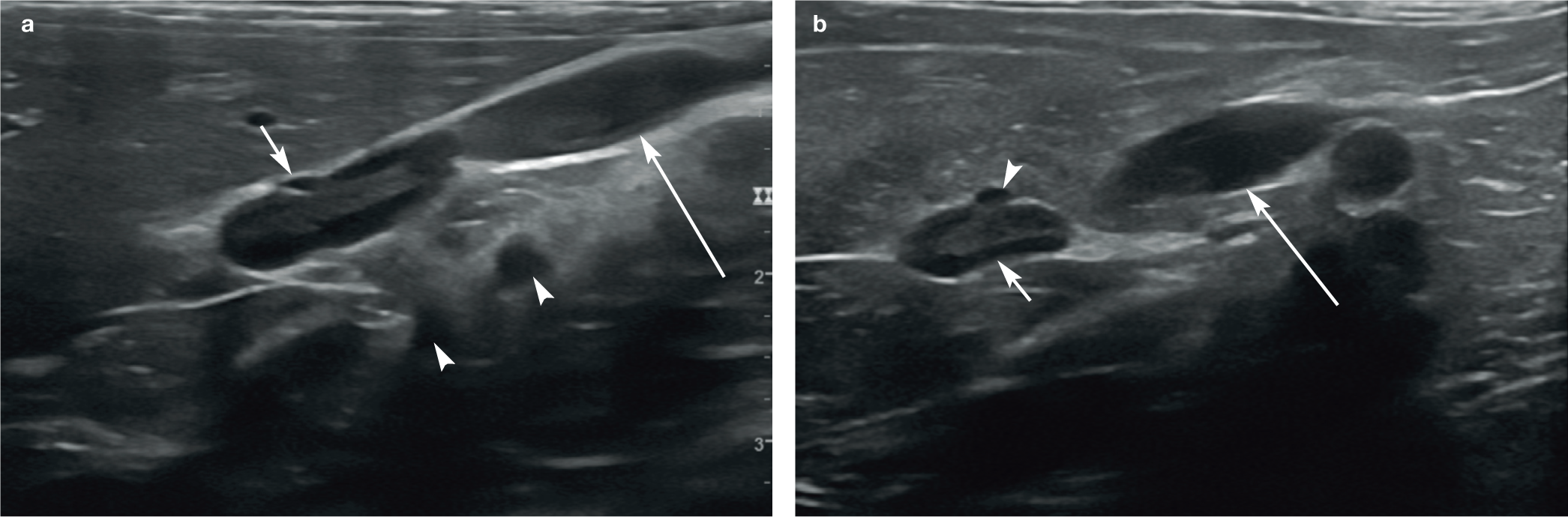
(a,b) Images showing the proximity of the caudal vena cava to the right adrenal gland. Both images were acquired with the cat in left lateral recumbency and the transducer positioned along the right flank, immediately caudal to the rib cage and directed cranially. (a) A portion of liver lobe is present within the top left of the image, while the adrenal gland is visible in the centre of the image. A short arrow points towards a small vessel in cross section located immediately ventral to the adrenal gland – the phrenicoabdominal vein. The large vessel (long arrow) adjacent to the adrenal gland is the caudal vena cava. The coeliac and cranial mesenteric arteries (arrowheads) are also present in the far field in cross section. (b) The right adrenal gland (short arrow) is located centrally within the image. The small vessel (arrowhead) visible in cross section ventral to the adrenal gland is the phrenicoabdominal vein and the larger vessel (long arrow) appearing just caudal to the adrenal gland is a segment of the caudal vena cava. Two videos showing the normal ultrasonographic appearance of the right adrenal gland are available as supplementary material
Normal appearance of the adrenal glands
Unlike in the dog, the left and right adrenal glands of the cat are identical in appearance. 4 Normal feline adrenal glands are most commonly bean-shaped (ie, oval with a small depression across the short axis resulting in a bipolar appearance), well defined and uniformly hypoechoic relative to surrounding fat (Figure 3).2–5 ,7–11 Less frequently, the adrenal gland may appear either oval, with no associated narrowing or depression, or fusiform in shape (Figure 4), and in some cases a thin hyperechoic halo may be visible surrounding the gland.3,11 If imaged with a high frequency linear transducer, it is occasionally possible to discern two distinct concentric layers, an outer hypoechoic layer and an inner hyper-echoic layer (Figure 5a,b). It is not known whether these represent the cortex and medulla.2–4,9 Less commonly, this pattern can be reversed and the central portion of the adrenal gland may appear hypoechoic and the outer layer hyperechoic. This variation was reported by Cartee et al in 1993, 8 and has been encountered by the author (Figure 5c). Details pertaining to the size of the adrenal glands can be found in the box on page 37.
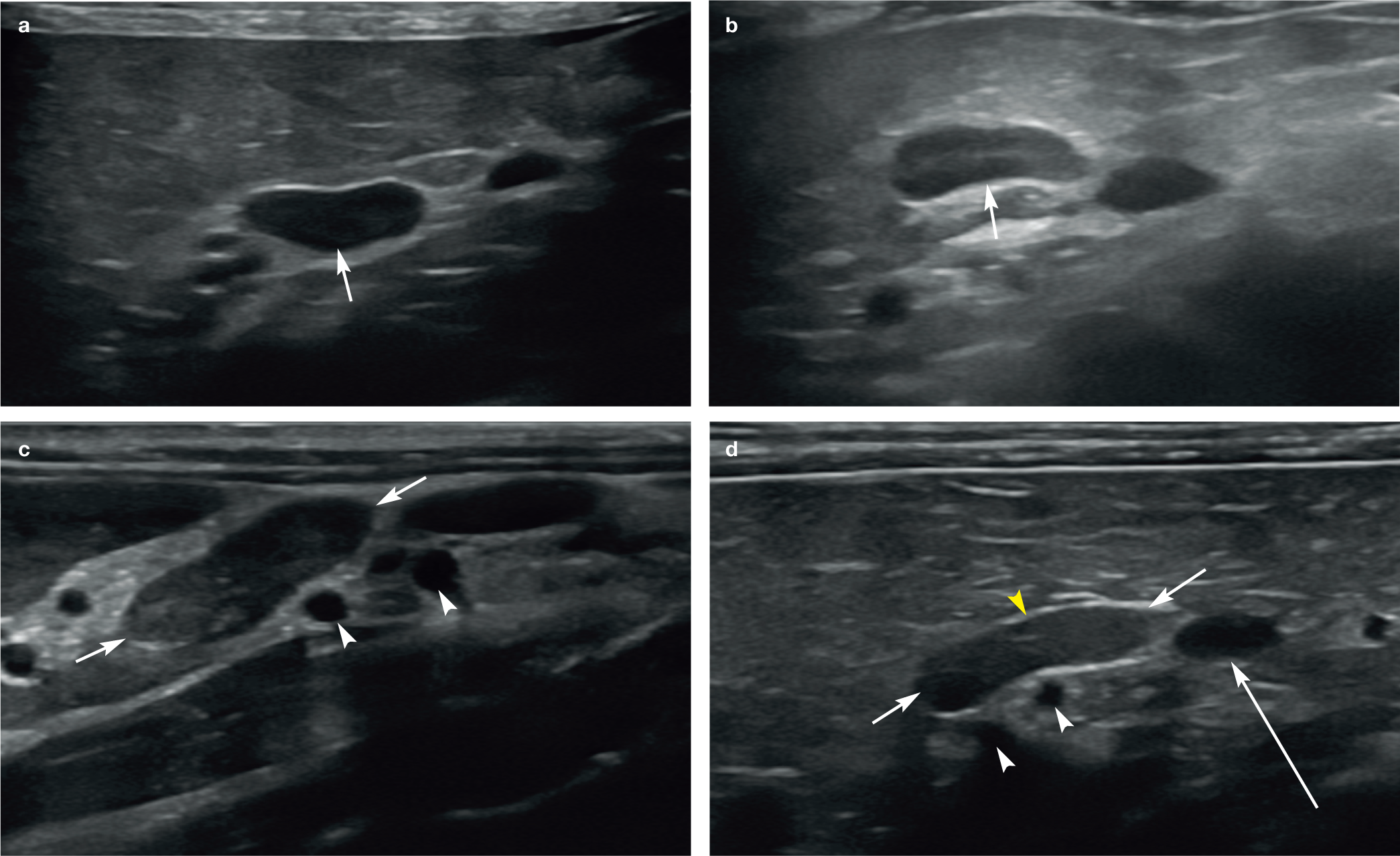
(a–d) Images showing the normal variation in appearance of the adrenal glands (short arrows). Note the small depression across the short axis of the gland in each case, resulting in a bean-shaped or bipolar appearance. In (c) and (d), the coeliac and cranial mesenteric arteries (white arrowheads) are visible in cross section. In (d), the long arrow depicts the left renal artery (only partially visible) and the yellow arrowhead ventral to the adrenal gland shows the location of the phrenicoabdominal vein, which, although small, can be identified in cross section

(a,b) Normal oval-shaped adrenal glands (short arrows) in two cats. In (b), note the coeliac and cranial mesenteric arteries in cross section (arrowheads). A portion of the left renal artery (long arrow) is also visible just caudal to the gland

(a–c) Adrenal corticomedullary definition (short arrows) in three cats using a high frequency linear transducer. All adrenal glands shown are left adrenals. In (a) and (b), note the coeliac and cranial mesenteric arteries in cross section (arrowheads). A portion of the left renal artery (long arrow) is also visible just caudal to the gland

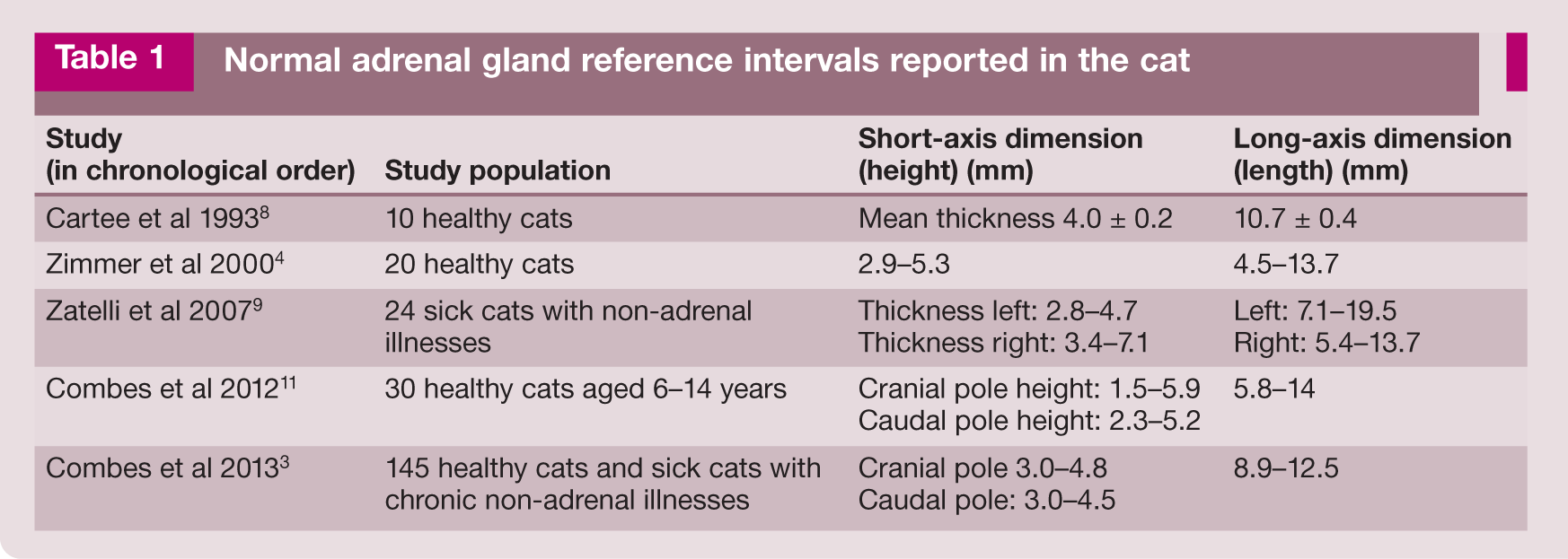
Normal adrenal gland reference intervals reported in the cat
Randomly distributed hyperechoic foci that are associated with distal acoustic shadowing are observed in otherwise hypoechoic adrenal glands in up to 30% of healthy adult cats (Figure 6).4,10,15 These are thought to represent tiny areas of dystrophic calcification, which can occasionally be visible radiographically.10,16 In some cats, non-shadowing hyperechoic foci are present. It has been hypothesised that these may also represent areas of calcification that are simply too small to cast a shadow or, alternatively, they may represent focal areas of either lipid deposition or haemorrhage.3,4,11 In a study examining the histopathological appearance of the adrenal glands in 159 cats, large lipid droplets were identified in 156 cats and areas of dystrophic calcification in 62 cats. 17 Haemorrhage was less commonly observed and identified in only 15 cats. It should be noted that no ultrasound examination was performed in this particular study to directly correlate ultrasonographic with histopathological findings. 17

(a,b) Suspected adrenal mineralisation in two cats presenting for investigation of liver disease. (a) Solitary hyperechoic focal area (arrowhead) within the adrenal gland (arrows) of a 9-year-old neutered female domestic shorthair cat. (b) Multiple small, non-shadowing hyperechoic foci (arrows) within the left adrenal gland of an 11-year-old neutered male domestic shorthair cat. The arrowheads indicate the coeliac and cranial mesenteric arteries (from the viewer’s left to right, respectively) in transverse. (c,d) Mineralised left and right adrenal glands (arrows) in a 16-year-old neutered male domestic shorthair cat. Both glands are irregularly hyperechoic and cast a strong distal acoustic shadow. (e) Same cat as in (c) and (d). Mineralisation of the glands (arrows) was confirmed radiographically. A video showing hyperechoic foci in the left adrenal gland of an adult cat is available as supplementary material
Hyperechoic foci may be more commonly observed in cats with either adrenal or non-adrenal disease compared with healthy cats. 3 However, such foci are not associated with malignancy (unlike in dogs) and, since they are especially common in older cats with no evidence of adrenal disease, they are currently regarded as being incidental.5,7,18
Abnormalities associated with adrenal disease
Most adrenal disease in cats falls into one of three categories: hyperplasia, neoplasia or immune-mediated destruction. The incidence of primary adrenal neoplasia is lower in cats than in dogs 17 and is estimated to account for around 0.2% of all feline neoplasms.19,20 A survey of 3145 feline necropsies conducted over a period of 11 years by The Animal Medical Center, New York, USA, revealed only two adrenal tumours. 21 Tumours of the adrenal gland arise either from the cortex (adenoma or carcinoma) or from the medulla (phaeochromocytoma) and may be functional or non-functional. 19 Functional cortical tumours generally secrete aldosterone, but, less commonly, can produce excess quantities of cortisol or sex hormones (eg, progesterone, oestradiol, testosterone).19,22

Hyperadrenocorticism (hypercortisolism)
Hyperadrenocorticism (HAC), while defined as the excessive secretion of any adrenocortical hormone, in veterinary medicine is typically used to describe hypercortisolism.24,25 The zona fasciculata and zona reticularis of the adrenal cortex are responsible for producing cortisol. 25 In contrast to dogs, spontaneous feline HAC or Cushing’s syndrome is an uncommon endocrine disorder, typically affecting middle-aged to older cats, with a mean age at diagnosis of 10 years and no purebreed predisposition.3,6,25,26 A unique and common feature of HAC in cats is extreme skin fragility. Therefore, particular care should be taken when restraining and clipping cats for ultrasound to avoid iatrogenic skin tears.6,16,25,27–31 In some cases of endocrine-related alopecia, clipping may not be necessary but, if performed, owners should be notified that hair may fail to regrow after clipping.18,25,32,33
Similar to their canine counterparts, around 80% of feline cases are pituitary-dependent and the remaining 20% are due to a functional tumour of the adrenal cortex.6,27 Around 50–60% of adrenal tumours are functional adenomas and the remainder are malignant cortical adenocarcinomas.18,25
Although endocrine testing is typically used for the initial diagnosis of HAC, ultrasound is a valuable diagnostic aid for differentiating between cats with pituitary-dependent hyperadrenocorticism (PDH) and those with adrenal-dependent hyperadrenocorticism (ADH).6,27 In one study of 30 cats with HAC, ultrasound was successfully used to differentiate between PDH and ADH in 28 cats, yielding a sensitivity of 93%. 34
Of 27 cats with PDH in the above-mentioned study, 34 22 had bilateral adrenomegaly (defined as an adrenal gland width >6 mm based on a previous study 11 ), consistent with the expected presentation of this form of the disease. Three cats had normal-sized adrenal glands bilaterally and two had unilateral adrenomegaly. Of the two cats with unilateral adrenomegaly, the contralateral gland was normal in size in one cat and unidentifiable in the second cat. Further studies have similarly demonstrated that while bilaterally symmetrical adrenomegaly is the most common finding in cats with PDH, a significant proportion will have adrenal glands within the normal reference interval for size; moreover, in a minority, unilateral adrenomegaly may be present, which could potentially lead the ultrasonographer to incorrectly suspect primary adrenal disease.3,6,16,28,32,34–46 In cats with PDH, enlarged adrenal glands usually remain bipolar or oval in shape and have a hypo-echoic parenchyma relative to surrounding fat, possibly with hyperechoic foci or a hyper-echoic medulla. 3
In ADH, the most frequently identified abnormality on ultrasound is a unilateral adrenal mass or adrenomegaly and a small or unidentifiable contralateral gland.18,25,34 In the presence of a unilateral cortisol-producing tumour, adrenocorticotropic hormone (ACTH) secretion by the pituitary gland is chronically suppressed, causing atrophy of the contralateral adrenal gland. 30 Occasionally, potentially misleading results are obtained, such as a normally sized contralateral gland or bilateral adrenomegaly.6,34 Additional ultrasonographic findings reported in cats with HAC and excess cortisol production include hepatomegaly, a diffuse increase or decrease in hepatic echogenicity and increased amounts of intra-abdominal fat.16,25,34,36,37,39,47
It should be noted that the majority (around 80%) of cats with HAC also have diabetes.3,18,25,27,34–37,40,41 However, diabetes mellitus does not appear to cause a measurable increase in adrenal gland size according to a study conducted in 2007. 42
In dogs, the appearance of the adrenal glands following medical treatment (specifically trilostane) has been reported; typical findings include an increase in size of the glands and alterations in parenchymal echogenicity. 49 To the author’s knowledge, the post-treatment appearance of the feline adrenal glands has not been described, perhaps due to the fact that there are only a limited number of reports documenting the medical treatment of cushingoid cats.24,34,36,44,46
Excess sex hormone production
Despite being uncommon in both cats and dogs, the prevalence of adrenal sex steroid-producing tumours is higher in cats 24 and a number of cases have been documented in this species.22,26,29,50–56
Excessive adrenocortical production of oestradiol and testosterone has been reported in a 14-year-old neutered female cat displaying signs of aggression and male-type behaviour. 24 Abdominal ultrasound revealed bilateral adrenomegaly (left and right adrenal glands measured 10 mm x 13.7 mm and 12 mm x 15mm, respectively) and splenomegaly. While the cause of the splenomegaly was not determined, it is known that the majority of ferrets with excess adrenocortical sex steroid production also have idiopathic splenomegaly.24,57 Concurrent hyperprogesteronism and hyperaldosteronism have also been described in two elderly cats, one with a large, lobulated, heteroechoic left adrenal mass (46 mm diameter) and a small right adrenal gland (1.8 mm diameter), 54 and the other with a 20 mm x 25 mm hypo-echoic right adrenal mass confirmed to be an adrenocortical carcinoma. 53

Hyperaldosteronism
Hyperaldosteronism, also known as Conn’s syndrome, was first described in cats by Eger et al in 1983. 58 Since then, many more cases have been reported and, although it still remains relatively under-diagnosed,23,59,60 it is probably the most common adrenocortical disorder in the cat. 23 Mineralocorticoids are produced in the zona glomerulosa and secretion of aldosterone specifically is regulated via the renin–angiotensin–aldosterone axis.25,61 The normal function of aldosterone is to regulate fluid balance and blood pressure, thereby preventing hyperkalaemia and hypotension. 61 By increasing the activity of the sodium–potassium pumps within the distal renal tubules, sodium and water reabsorption and potassium secretion are promoted. 61 Hyperaldosteronism primarily affects geriatric cats and refers to a disorder characterised by excessive, autonomous secretion of aldosterone by the adrenal cortex, leading to sodium retention and systemic hypertension. 62
Hyperaldosteronism is classified as either primary or secondary. 6 The underlying cause of primary hyperaldosteronism is usually an aldosterone-secreting adrenal tumour within the zona glomerulosa of the adrenal cortex (an adenoma or carcinoma); much less commonly, it may be due to uni- or bilateral idiopathic hyperplasia.61,63–65 In fact, most primary adrenal tumours in cats secrete aldosterone and, as a result, the condition is more common in cats than in dogs.22,30 Approximately 50% of tumours are adenomas and 50% are carcinomas;61,64,65 unilateral disease is also more common than bilateral disease.64,65 Secondary hyperaldosteronism is caused by conditions such as chronic kidney disease that enhance renin concentrations. 6
Abdominal ultrasound can be very useful when presented with a cat with unexplained hypertension. 6 The most common ultrasonographic presentation in cats with primary hyperaldosteronism due to an adrenocortical tumour is a unilateral adrenal mass, often 1–5 cm in diameter, in association with a small/unidentified, or occasionally normal, contralateral gland (Figure 7).59,62,65–67 Bilateral adrenal tumours are uncommon. Occasionally, the adrenal glands may appear unremarkable or have only very minor changes such as mild thickening or a focal change in echogenicity, particularly in cats with nodular hyperplasia of the zona glomerulosa.23,62,63

(a–d) Adrenal nodules in three cats with hyperaldosteronism. (a,b) A well-defined 1.33 cm x 1.7 cm hypoechoic nodule (between the measuring calipers in [a]) distorts the cranial pole of the right adrenal gland in a 16-year-old neutered female domestic shorthair cat with hypokalaemia (potassium 2.9 mmol/l; reference interval [RI] 3.3–5.5 mmol/l) and an elevated plasma aldosterone level (>4000 pmol/l; RI 195–390 pmol/l). The measuring calipers in (b) indicate the short-axis diameter of the left adrenal gland, which is also visible. (c) A 2 cm diameter adrenal mass (arrows) within the left adrenal gland of a 13-year-old neutered male domestic shorthair cat with hindlimb weakness, hypokalaemia, systemic hypertension and proteinuria. Plasma aldosterone level was >5000 pmol/l (RI 87–224 pmol/l). (d) A 2.7 cm diameter left adrenal mass (between the measuring calipers) in a 10-year-old neutered male domestic shorthair cat presenting with a history of persistent hypokalaemia, generalised weakness and an intermittent stiff gait, and systemic hypertension. Histopathology confirmed the mass was an adrenal adenocarcinoma. Images (a) and (b) courtesy of Andrew Kent, Willows Veterinary Centre and Referral Service, UK
In a report on six domestic shorthair cats with hyperaldosteronism, the median age at diagnosis was 12.5 years (range 11–17 years) and the most common ultrasonographic finding was a unilateral adrenal mass affecting either gland. 3 The mass was homogeneously hypoechoic in three cats, heterogeneously hyperechoic in two cats and target-like (hyperechoic centre with a hypoechoic rim) in the final cat. The authors were unable to distinguish between benign and malignant masses based on their size, shape, echogenicity or homogeneity on ultrasound. 3 These findings are supported by another study of 13 cats with primary hyperaldosteronism in which there was significant overlap in the size of carcinomas and adenomas, the largest (measured) mass of the study being an adenoma and the smallest being a carcinoma. 65
It should be noted that where bilateral disease is present, this may not be apparent on ultrasound. 65 There are several case reports documenting unilateral adrenal gland changes on ultrasound in cats that were ultimately diagnosed with bilateral disease. In one cat described in a 13-cat case series, ultrasonography revealed a unilateral mass of the right adrenal gland; at post-mortem examination performed 13 days later, bilateral adrenocortical adenomas were identified on histopathology. 65 In a separate study, another cat had thickening of the caudal pole of the left adrenal gland visible on ultrasound, prompting unilateral adrenalectomy. Histopathology confirmed the presence of left adrenal nodular hyperplasia of the zona glomerulosa. However, hypokalaemia returned just 2 months after surgery, leading the authors to conclude progressive hyperplasia of the contralateral adrenal gland as the most likely cause. 63 Therefore, in the light of the above, it is important when surgery is undertaken that both adrenal glands are examined even if pre-operative imaging suggests unilateral disease. 65
Furthermore, pathology can be present even when no abnormalities at all are detected on ultrasound. This was demonstrated in two cats with normal adrenal glands on ultrasound, but with bilateral adrenocortical nodular hyperplasia subsequently detected by histopathology during post-mortem examination. 63 Where an adrenal nodule or mass is identified, the remainder of the abdomen and, in particular, the liver and retroperitoneum should be assessed ultrasonographically for evidence of metastatic disease. 6
Haemorrhage
Acute adrenal haemorrhage due to neoplasia has been reported in three cats. Two cats had unilateral aldosterone-secreting adrenocortical carcinomas. In one of these cats, a large haematoma, which presented as a 5 cm diameter heterogeneous area adjacent to the tumour, and haemorrhage into the retroperitoneal space were identified on ultrasound. 68 In the third cat, the tumour type was not specified and the imaging findings pertaining to this case were not described. 22
Vascular invasion
Invasion of local vasculature by malignant adrenal masses, while uncommon, has been reported in cats and, depending upon the vessel involved, can have important implications with regard to prognosis.22,59,61,69–71 The most common observation is for caval invasion to occur via the phrenicoabdominal vein. One report describes the identification of caval invasion on ultrasound in a 13-year-old cat with a 22 mm diameter adrenocortical carcinoma causing primary hyperaldosteronism. 69 At surgery, the mass was revealed to be invading the caudal vena cava via the phrenico-abdominal vein. Readers should also be aware that the apparent absence of vascular invasion on ultrasound does not exclude the possibility, as illustrated by several cases where invasion was missed during preoperative imaging and only found during surgery.3,53
The sensitivity of ultrasound for the detection of vascular invasion in the cat has yet to be determined, although a sensitivity and specificity of 80% and 90%, respectively, have been documented in the dog. 72 Contrast-enhanced CT has been reported to have an even better sensitivity of 92% and specificity of 100% for the detection of vascular invasion, but these figures are similarly derived from dogs rather than cats. 73
Primary hypoadrenocorticism
Primary hypoadrenocorticism (Addison’s disease) is exceptionally rare in cats. Although the exact pathogenesis in this species is unknown, autoimmune destruction of the adrenal cortex is suspected in most cases, resulting in both glucocorticoid and mineralocorticoid deficiency.74–78 The condition usually arises in middle-aged cats, although it has been reported in the young and elderly (8 months to 14 years).77,79–86 There is no known breed or sex predilection,74–77,78–81,85,86 but most reports involve domestic shorthair and longhair cats. 6 Clinical signs appear to be similar to those in dogs, being non-specific and vague including anorexia, vomiting, lethargy and weight loss, which should respond to glucocorticoid therapy.74,77,83,86–88 Radiographic changes resemble those reported in the dog and include microcardia and pulmonary hypoperfusion.74,79,85,86
As anticipated, descriptions of the ultrasonographic appearance of the adrenal glands in cats with hypoadrenocorticism are rare and limited to a handful of isolated case reports. Where descriptions are available, the adrenal glands have been reported to be normal in size, although in one cat the right adrenal gland could not be identified.77,81,86,87 The adrenal glands may also appear normal in cats with Addison’s disease due to disease of the pituitary gland.83,84 This is somewhat unexpected – in dogs with this condition, the adrenal glands are typically small and frequently difficult to identify. 89 Therefore, the presence of normal-sized adrenal glands on ultrasound does not preclude a diagnosis of hypoadrenocorticism in the cat. On a single occasion, the author has identified a small adrenal gland in one cat with confirmed hypoadrenocorticsm (Figure 8).
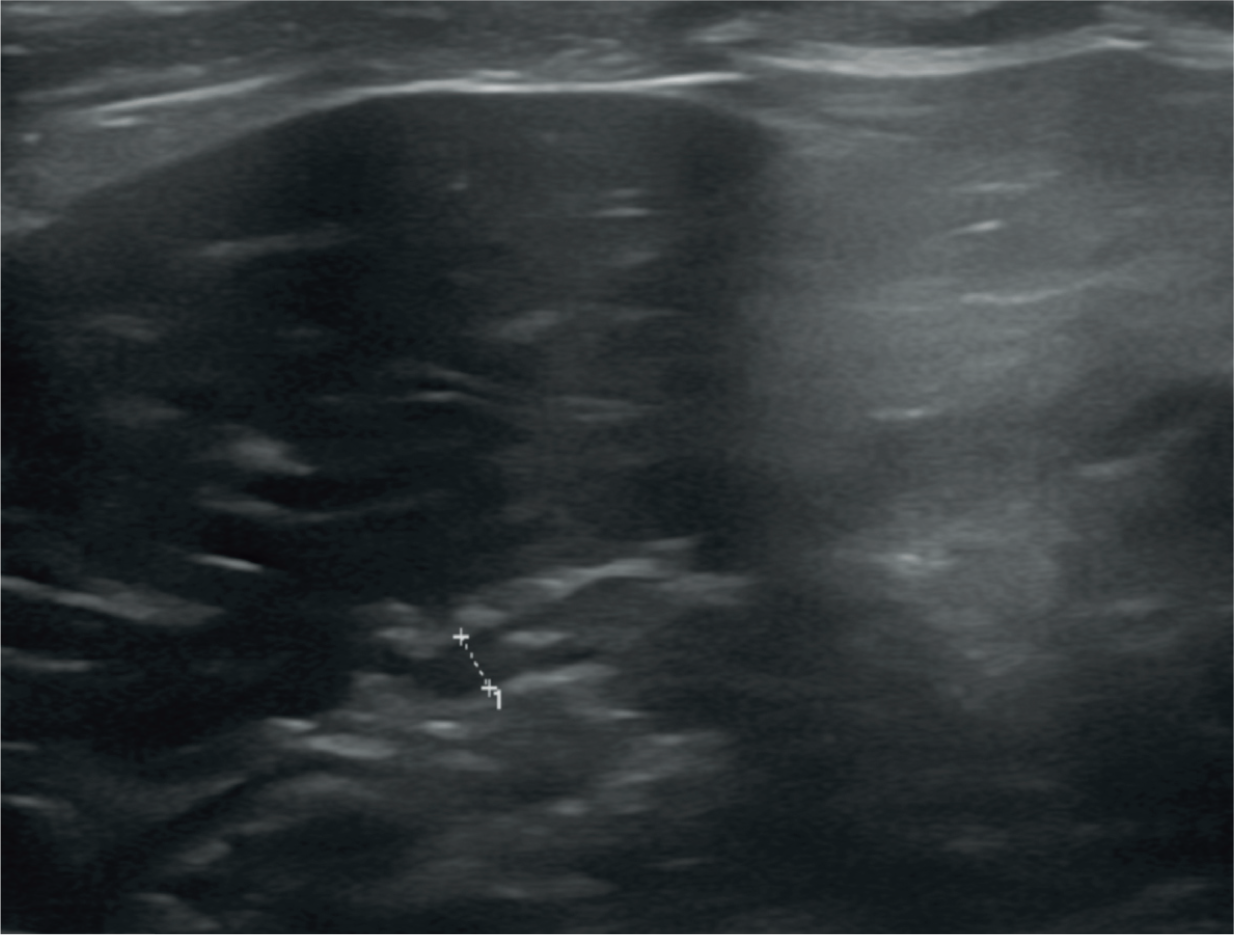
Adrenal gland of a 5-year-old neutered male Tonkinese cat with hypoadrenocorticism. The short-axis diameter of the cranial pole of the right adrenal gland (between the measuring calipers) measures 2.2 mm. With the exception of the study by Combes et al 2012, 11 this value falls outside reported reference intervals. The left adrenal gland could not be located in this cat
There have also been three reported cases of hypoadrenocorticism in adult cats caused by bilateral lymphomatous adrenal infiltration.76,78 In the most recent case, the authors describe bilateral and progressive adrenomegaly, the left gland reaching 2.4 x 2.6 x 2.9 cm in size and the right 2.8 x 2.6 x 2.8 cm. The glands were also reported to be abnormally hypoechoic and rounded with complete loss of normal architecture and were surrounded by mildly hyperechoic tissue. 78 No evidence of infiltration of either the liver or spleen was found during ultrasonography or following fine-needle aspiration and there was no ultrasonographic evidence of lymphadenopathy.
Phaeochromocytomas
Phaeochromocytomas are catecholamine-secreting tumours arising from the chromaffin cells of the adrenal glands.90,91 Extraadrenal sites, such as sympathetic paraganglia, are also possible and are called paragangliomas.90,92,93 Phaeochromocytomas are frequently malignant and are associated with a median survival time of 20 weeks. 22 They are uncommon in the cat and, to date, only a handful of cases have been reported.22,90,94–98 In a retrospective study of 33 cats with adrenal masses, only three cats were diagnosed with phaeochromocytomas. 22 Most of the case reports have involved older (>7 years), neutered domestic shorthair cats. Males and females are fairly equally represented. Clinical signs are similar to those reported in the dog and typically non-specific and vague, including weakness, lethargy, anorexia, weight loss, polyuria and polydipsia, which may be paroxysmal due to the episodic nature of catecholamine release.22,31,91,94,96,98
While the clinical history and laboratory results may be indicative of a phaeochromocytoma, imaging (usually ultrasound in the cat) is required to demonstrate the presence of an adrenal mass. 95 To the best of the author’s knowledge, the ultrasonographic appearance of adrenal phaeochromocytoma has only been described in five cats.90,95–98 Where size was given, tumours ranged in diameter from 7.6 mm to 22 mm and they varied from normal to hypo-echoic or heterogeneous in echogenicity. In 4/5 cases, the left adrenal gland was involved.90,95,96,98 The ultrasonographic and intraoperative appearance of the adrenal glands in one cat were suggestive of generalised bilateral adrenal enlargement. However, histopathology later revealed the presence of a phaeochromocytoma in the left adrenal gland and an adrenocortical adenoma in the right gland. The apparent absence of focal nodular enlargement within either gland was attributed to the position of the tumours within the glands and the small size of the glands (which were only a few millimetres thick, despite being midly enlarged) such that gross changes were difficult to detect. 95 Thus, failure to detect an adrenal mass does not rule out the diagnosis.
While tumour thrombus formation has not been reported in association with adrenal phaeochromocytoma in the cat, it appears to be a common occurrence in the dog.72,99 Therefore, it may well be prudent to check for evidence of vascular involvement in cats suspected of having a phaeochromocytoma.
Adrenal metastases
Metastases to the adrenal glands are rare in cats and usually occur in the later stages of disease. In a retrospective study of 81 cats, 12 (14.8%) were identified with metastatic disease to the adrenal glands based on postmortem examination. 100 Metastases arose from transitional cell carcinoma, melanoma, thyroid follicular carcinoma and fibrosarcoma, as well as being due to multicentric involvement in cats with lymphoma. 100 Unfortunately, no ultrasound descriptions were included in the report, although metastases were noted to be bilateral in 50% of the cases.
Hyperthyroidism
It has been suggested that hyperthyroidism may lead to abnormally high levels of responsiveness of the adrenal cortex through stimulation of the hypothalamic–pituitary–adrenocortical axis. 11 To date, the limited studies investigating the size of the adrenal glands in cats with hyperthyroidism have produced conflicting results. In one study, the authors found no statistically significant difference in size of the adrenal glands of hyperthyroid cats vs healthy controls, although the hyperthyroid cats were significantly older than the healthy cats and had notably higher cortisol levels pre- and post-ACTH stimulation. 101
In a second study, the adrenal glands were found to be significantly larger in both the craniocaudal and dorsoventral axes in a cohort of 23 hyperthyroid cats when compared with 30 healthy controls. 11 The mean difference between the hyperthyroid and healthy cats was given as 1.6 and 1.7 mm (length), 0.8 and 0.9 mm (cranial pole height) and 0.4 and 0.9 mm (caudal pole height) for the left and right adrenal glands, respectively. 11 Broadly speaking, these figures equate to a 20% increase in size. 11 Adrenal gland shape and background parenchymal echogenicity remained unchanged between the two groups, but a much higher proportion (39.1%) of hyperthyroid cats (especially those untreated) had hyperechoic foci within one or both adrenal glands compared with only 6.7% of healthy cats. 11 Medical treatment with antithyroid drugs did not appear to have a significant effect on adrenal gland size, although this is perhaps to be expected since no treatment was administered within 2 weeks of ultrasonography and the mean residence time of oral methimazole ranges from 92 to 345 mins following administration.11,102 Therefore, as the authors of the study on methimazole pharma-cokinetics point out, cats that had previously received methimazole could effectively be considered as being equivalent to untreated cats. 102
In summary, it is important for ultrasonographers to be aware of the potential for mild adrenomegaly in cats with hyperthyroidism, particularly when evaluating hyperthyroid cats for suspected adrenal disease. 11 The degree of enlargement observed in hyperthyroid cats overlaps that seen in healthy cats and those with pituitary-dependent HAC. Since a substantial proportion of adrenal tumours are unilateral and measure >2 cm in the short axis, it is unlikely that this would be confused with adrenal enlargement due to hyperthyroidism.22,24,26,40,53,54,56,58,61,65,70,95,103,104
Acromegaly
The term acromegaly describes a syndrome resulting from the excess production of growth hormone (hypersomatotropism) by a pituitary tumour. 27 Older neutered male cats are most commonly affected and cats often present with insulin-resistant diabetes. Studies have shown that this disease may be far more common than previously thought, with its prevalence in UK diabetic cats being estimated to be around 1 in 4.105–107 While most cats with hypersomatotropism do not exhibit classic phenotypical acromegalic signs, such as broad facial features, many will have important changes present on abdominal ultrasound, which, if recognised, can raise suspicion for acromegaly. 105
In 24 acromegalic cats, adrenal gland thickness ranged from 3.2 to 8.8 mm, compared with 2.3–5.3 mm in a healthy control group of the same size. 108 Bilateral adrenomegaly was considered to be present in 7/13 (54%) of the acromegalic cats in which both adrenal glands were measured (based on published reference intervals determined using 20 healthy cats 4 – left adrenal gland thickness 3.0–5.3 mm, right adrenal gland thickness 2.9–4.5 mm). Of the cats with acromegaly, the left and right adrenal glands were enlarged in 7/16 (44%) and 11/13 (85%) cases, respectively. 108 The author has also come across bilateral adrenomegaly in an acromegalic cat (Figure 9). Additional reported ultrasonographic findings include measurably enlarged kidneys and pancreas, and a subjectively enlarged liver. 108 Pancreatic abnormalities, such as diffuse hypoechogenicity or the presence of nodules, were also documented in 88% of the acromegalic cats compared with only 8% of cats in the control group. 108

(a) Left adrenal gland in a 16-year-old neutered female domestic shorthair cat with poorly controlled diabetes mellitus and acromegaly. The short-axis dimension (between the measuring calipers) is 5.4 mm and the gland is 15 mm in length. (b) The short axis of the right adrenal gland of the same cat falls within the reported reference interval; however, the length is also 15 mm, suggesting enlargement. The small intestinal loop seen in cross section at the top of the image is descending duodenum

Adrenal size in cats with idiopathic cystitis
The adrenal glands (specifically the cortex) may be significantly reduced in size in cats with idiopathic cystitis compared with healthy cats, although the total number of cats upon which this statement is based is small (21). 109 No definitive cause was identified for this apparent association, although congenital or developmental hypoplasia was suggested. 109

Amyloidosis
Adrenal deposition of amyloid has been reported in three cats, although only one cat suffered from severe generalised amyloidosis. 17 No information regarding the ultrasound appearance of the adrenal glands was given in that study.

Key Points
The adrenal glands in cats should be assessed routinely during abdominal ultrasound examination.
Mineralisation of the adrenal glands is currently considered to be incidental.
Most cats with PDH have bilateral symmetrically enlarged adrenal glands. However, normal adrenal glands do not exclude the possibility of PDH.
The most common finding in cats with ADH or hyperaldosteronism is a unilateral adrenal mass and a small contralateral adrenal gland.
In the presence of a unilateral mass, a normal-appearing contralateral gland does not negate the possibility of bilateral benign or malignant adrenocortical infiltration.
The adrenal glands may appear normal or have only subtle changes in cats with hyperaldosteronism due to adrenocortical hyperplasia.
Mild to moderate adrenomegaly can be present in cats with hyperthyroidism, PDH, hyperaldosteronism and acromegaly.
Footnotes
Conflict of interest
The author declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
Ethical approval
This work did not involve the use of animals and therefore ethical approval was not specifically required for publication in JFMS.
Informed consent
This work did not involve the use of animals and therefore informed consent was not required. No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
