Abstract
Objectives
We compared the effects of two alpha (α)2-adrenergic agonists on semen traits.
Methods
In this study, 13 adult domestic cats were divided into two experimental groups, according to the chemical ejaculation protocol used: the first group received medetomidine hydrochloride (100 µg/kg) and ketamine (5000 µg/kg); the second group received dexmedetomidine hydrochloride (25 µg/kg) and ketamine (5000 µg/kg), both by the intramuscular route.
Results
The animals responded positively (P >0.05) to chemical collection. Seminal parameters evaluated included volume, sperm vigor, total motility, progressive motility, sperm concentration, and the structural and functional integrity of the plasma membrane; sperm morphology values did not differ between groups (P >0.05).
Conclusions and relevance
The results indicated that dexmedetomidine is a more viable and economical alternative to medetomidine in domestic cats submitted to semen collection by urethral catheterization. Semen collection by urethral catheterization after using α2-adrenergic agonists is a recently developed technique in feline species that is considered to be quick and highly applicable to assisted reproduction programs in felids.
Introduction
Alpha 2 (α2)-adrenergic agonists have been used to induce ejaculation in both domestic (asinine and equine)1,2 and laboratory animals.3,4 The association with urethral catheterization with α2-adrenergic agonists is a recent technique used for the collection semen in cats, developed by Zambelli et al. 5 This technique is increasingly being used in studies of both domestic and wild felids,5–11 owing to it producing similar quality semen to other techniques (electroejaculation), and also cost advantages and decreased contamination of the semen sample. 10
The urethral sperm collection technique consists of the introduction of a 1.3 mm feline catheter through the penile urethra after sedation of the animal 5 following the administration of an α2-adrenergic agonist (analgesic and sedative), whose mechanism of action involves stimulation of the adrenoreceptors in the deferent duct, thereby releasing semen into the penile urethra.
Generally, two α2-adrenergic agonists are used: medetomidine (MED) and dexmedetomidine (DEX). MED is a potent α2-adrenergic agonist and plays a role in promoting sedation, analgesia and muscle relaxation.12–14 Several authors have reported semen collection by ejaculation in domestic cats after the administration of this agent, using posterior urethral catheterization.7,8,13,15
DEX is the newest and most selective α2-adrenergic agonist with effects such as sedation, analgesia and anxiolysis.16,17 Its efficiency as an inductor of ejaculation in domestic cats has also been demonstrated. 9 This technique offers semen with motility parameters that can reach 90% 9 and progressive motility of 70%, 18 and becomes important when using the domestic cat as a model for assisted reproduction processes in wild cats.
As MED is not commercially available in multiple countries, and with the need for comparative studies between the two drugs regarding collection efficiency, obtained semen quality and comparative cost, the aim of this study was to evaluate the effects of α2-adrenergic agonists (MED and DEX) on chemical ejaculation induction and semen quality, obtained by urethral catheterization in domestic cats.
Materials and methods
Ethical approval
The experimental protocol for this study was approved following the standards recommended by the Ethics Committee on the Use of Animals of the Federal University of Bahia (UFBA; Protocol CEUA EMEVZ-UFBA no. 10/2016).
Animals
The experiments were performed at the Veterinary Hospital Renato Rodenburg de Medeiros Neto of UFBA, in 2018, in the northeastern region of Brazil (Municipality of Salvador, state of Bahia, Brazil). Thirteen male crossbred cats, which were healthy and mature, aged between 1 and 4 years and with a mean ± SD weight of 4.5 ± 0.4 kg, were included in this study. The animals were considered healthy after a clinical and reproductive examination, prior to which they were submitted for hematologic (blood count) and biochemical evaluations (creatinine, urea, alanine aminotransferase and alkaline phosphatase). According to the clinical records of the animals, all the pet cats were owned privately, lived in apartment buildings, were fed commercial diets and did not have contact with other male or female cats.
Secretion of serum testosterone was confirmed by the presence of penile spicules. No penile injuries were detected. Testicle mobility inside the scrotum and consistency were considered normal. Weight and age were homogeneous among the selected 13 animals (P >0.05).
Anesthetic procedures
Prior to semen collection, the animals were randomly divided into two experimental groups. The experiments were performed in series. The first group consisted of seven animals (group 1), which were anesthetized using medetomidine hydrochloride (MED; 100 µg/kg IM [Domitor; Group Orion]) and ketamine (5000 µg/kg IM [Syntec; Pharmaceutical Technology Applied to Veterinary Medicine]). The second group consisted of six animals (group 2), anesthetized with dexmedetomidine hydrochloride (DEX; 25 µg/kg IM [Dexdomitor; Orion Pharma]) together with ketamine (5000 µg/kg IM). The cephalic vein was then cannulated for the administration of fluid with a physiological solution of 0.9% NaCl (5 ml/kg/h). During anesthesia, vital signs such as heart rate, respiratory rate, oxygen saturation, and systolic, diastolic and average blood pressure, as well as temperature, were monitored with the help of a multiparametric monitor (LifeWindow-LW9xVet; Digicare Biomedical Technology). After topical application of 2% lidocaine without epinephrine (0.2 ml [Xylestesin; Cristália]), all cats were incubated with cuffed endotracheal tube size 3–4 to provide a patent airway and supplemental oxygen.
For anesthetic recovery, the α2-adrenergic antagonist was administered (atipamezole 4280 µg/ml; Group Orion) at a dose of 250 µg/kg of body weight. In group 1 (MED), atipamezole:MED at a dose ratio of 2.5:1 was given. 19 In group 2 (DEX), the atipamezole was used as early as 30 mins after sedation, to avoid adverse effects.
Semen collection
Twenty minutes after the administration of the injectable drug, and in an adequate anesthetic state, with visible muscle relaxation and absence of response to manipulation, the penis was cleaned with 0.9% NaCl solution and dried with a paper towel. Immediately, a sterile and lubricated catheter (without a side window, 1 mm in diameter and 13 cm in length [Provar; Provar]) was carefully inserted into the penile urethra to a depth of 7 cm and left there for approximately 40 s. Semen entered into the catheter by capillarity. After obtaining the seminal sample visualized in the interior of the catheter, the catheter was removed from the urethra and the collected semen transferred to a preheated polypropylene tube (1.5 ml) containing an isotonic solution of 50 µl Tris-egg yolk diluent (199.7 mM Tris(hydroxymethyl)aminomethane, 71.4 mM monohydrated citric acid, 69.4 mM glucose, 20% (v/v) egg yolk, 0.5% (v/v) sodium dodecyl sulfate, 134 mg/l gentamicin sulfate and distilled water) at pH 7.4 and an osmolarity of 351 mOs mol/kg (Figure 1). All reagents, with the exception of gentamicin sulfate (13.4 mg; Novafarma Pharmaceuticals) were produced by the Cromaline laboratory. The use of an isosmotic medium for the semen was necessary to extend the volume of the samples, especially those of very low volume, to ensure that all samples were subjected to all experimental analyses and processes; this would not have been possible if the aliquots of semen were being handled in a non-laboratory environment.

(a) Penile spicule appearance, (b) penile catheterization and (c) collection of semen sample in tom cats
After semen collection, the animals received tramadol (2.0 mg/kg IV) (Tramadon; Cristalia) and underwent scrotal orchiectomy.
Semen analysis
Semen volume was quantified with the use of a micropipette and obtained by the difference of the initially added extender (>50 µl). Microscopy was performed with a phase-contrast microscope (Olympus); the samples were prepared on a pre-warmed plate at 37°C (98.6°F). Sperm vigor was estimated subjectively based on cell movement (scale of 0–5). Characteristics such as total motility (TM) percentage and progressive motility (PM) were determined without sample staining and based on cell movement (0–100% scale in increments of 5%; 0, no cell movement; 100, all cells showed fast and progressive movements) and estimated using a phase-contrast microscope (× 100). The sperm concentration was obtained with a Neubauer chamber. The structural integrity of the membrane was evaluated with the supravital test (EOS), using eosin dye in a light microscope at × 40 magnification, 20 allowing for the separate classification of the percentage of folded tails. The functional integrity of the plasma membrane was estimated using the hypo-osmotic test (CH).21,22 Sperm morphology analysis was performed using buffered saline solution. 21 Morphological alterations were observed by phase-contrast microscopy (× 100) and were classified as either minor or major defects. 23
Statistical analysis
Data were analyzed for normality and homozygosity using the Shapiro–Wilk and Levene tests, respectively. Differences between percentages of response to the evaluated drugs were estimated between groups using the χ2 test. Comparisons of the averages of normally distributed variables were performed between groups using the t-test for independent samples, with a significance index 5%. Comparisons of the averages of the non-normally distributed variables were performed using the Mann–Whitney test. All statistical analyses were performed with SPSS, version 13.0 (IBM).
Results
Semen samples were obtained in 85.7% of the group 1 cats and 66.7% of the group 2 cats; these values did not differ (P >0.05) between the evaluated groups. The results of our study confirmed that the seminal parameters analyzed showed no significant differences (P >0.05) regarding volume variables, vigor, total and progressive motility, the structural and functional integrity of the plasma membrane, sperm concentration and the percentage of morphologically normal spermatozoa between the evaluated groups. The descriptive statistics of the semen characteristics according to the anesthetic protocol used are described in Table 1. The behavior of the variable sperm concentration per ejaculation is presented in Figure 2.
Evaluated semen parameters of fresh semen samples from domestic cats treated with ketamine associated with medetomidine (group 1) or dexmedetomidine (group 2)

Variable with non-normal distribution (by Shapiro–Wilk test)
TM (%) = total motility percentage; PM (%) = progressive motility percentage; Mspa/ml = million of spermatozoa/ml; EOS (%) = structural integrity of the membrane (supravital test); CH (%) = functional integrity of the plasma membrane; SMN (%) = percentage of morphologically normal spermatozoa
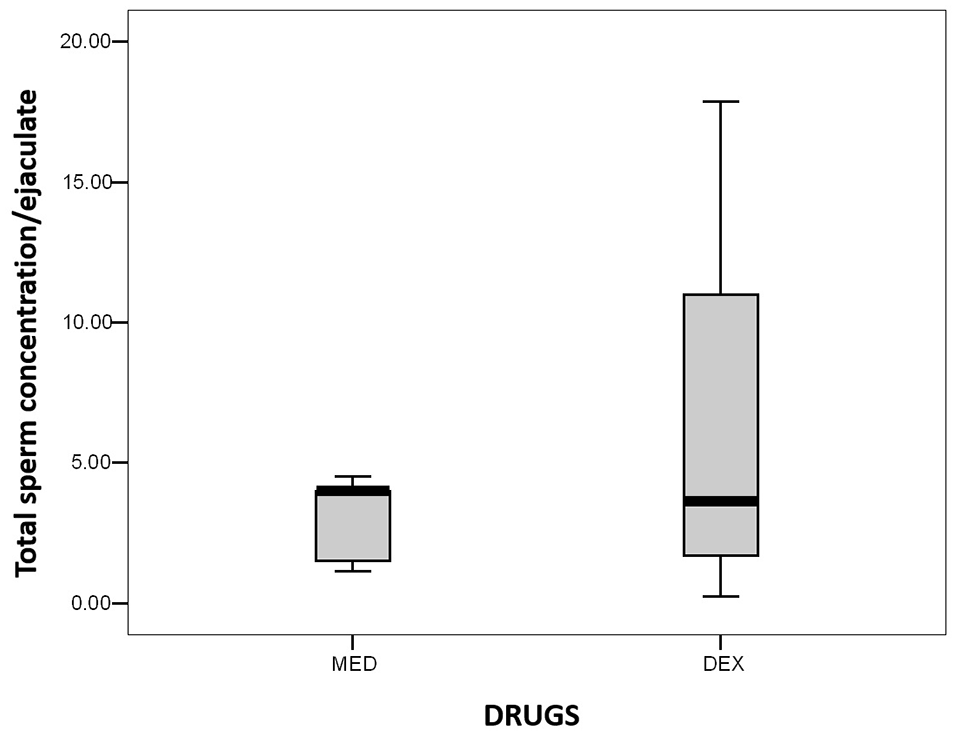
Estimation of total sperm number obtained by urethral catheterization of domestic cats using two different α2-adrenergic agonist drugs (in millions of spermatozoa).
In the sperm morphology analysis, approximately 9.7% of all evaluated cells presented with major defects, while 19.2% had minor defects (Table 2).
Minor and major defects in sperm morphology in both experimental groups and total analyzed cells

Group 1 cats were treated with ketamine associated with medetomidine and group 2 cats were treated with ketamine and dexmedetomidine
Discussion
An important contribution of the present study to the scientific literature is the fact that it compares the use of MED and DEX for the urethral collection of semen in cats, as well as the influence of these drugs on semen quality. Our results confirmed that the use of MED and DEX for semen urethral harvest is efficient.
Semen collection has been performed using urethral catheterization after the administration of an α2-adrenergic agonist in cats, but medetomidine drugs are emphasized in several studies; their effectiveness for this procedure is referenced in both domestic and wild cats.8–24 MED is a selective α2-adrenergic agonist composed of the enantiomers DEX and levomedetomidine. 24 DEX is the newest and most selective α2-adrenergic agonist used with effects such as sedation, analgesia and anxiolysis. 17 Its efficiency as an ejaculation inductor has also been demonstrated in domestic cats. 9
Considering that studies performed on domestic cats contribute to the application of the sperm collection by urethral catheterization technique in wild species, 11 these results regarding the effectiveness of the two drugs generate valuable knowledge, especially in countries that lack the license to commercialize MED. Moreover, another advantage is that our results suggest that a reduced dose of DEX may be required per animal as it has the same anesthetic effects with half the dose needed of the racemic mixture.25,26 As such, our results show that, for semen collection, just one-quarter of the MED dose is sufficient, thereby reducing the procedure cost. Based on an average weight of 4.5 kg per animal, a MED vial (10 ml [1000 µg/ml]) contains a drug amount sufficient for 22 doses, while a vial of DEX (10 ml [1000 µg /ml]) is the equivalent of 44 doses. The differential dose cost is an important factor when it comes to large cats. As shown by Swanson et al, 9 based on a 100 kg cat, the cost for one dose of DEX for one individual would be US$60 vs US$120 for a dose of MED.
Regarding semen traits described in previously published studies, different volumes of semen have been collected by urethral catheterization in domestic cats using MED (ie, 20.9 ± 15.1 µl 15 and 21.1 ± 31.9 µl), 25 similar to the volumes obtained in this study (~25 µl). With the use of DEX, the reported values 9 were between 12 µl and 46 µl of semen, within the minimum and maximum range for seminal volume found in this study (25–45 µl).
Sperm vigor has not been intensively studied following urethral semen collection. However, the verified vigor of all samples in this study ranged between 3.0 and 4.0. Similar results have been shown for feline spermatozoa retrieved from the epididymis of individuals anesthetized with MED associated with ketamine. 27
The total semen motility reported in previously published studies for semen collected by urethral catheterization after MED administration has been 59.5% ± 21.1%, 25 71.5% ± 9.0% 28 and 75.0%, 8 lower values than those obtained in this study (81.6% ± 9.3%). However, the verified range of TM percentage (70–95%) is close to the parameters reported by Prochowska et al (25–90%). 8 These authors also reported that feline semen motility should be >60%, as verified in this study. For semen collection by urethral catheterization after DEX use, the only reference found mentioned a concentration of 89%, 9 close to the TM range obtained with the administration of DEX (70–90%).
The PM observed in the semen samples obtained by urethral catheterization with MED associated with ketamine ranged from 12.2% to 73.9%.25,29 The maximum reported value in previous studies 25 is similar to the PM value obtained in this study (75%). When using DEX, a PM of 68.9% ± 2.6% 9 was reported. However, this information was obtained from a study including only three animals, which makes it harder to establish associations between the obtained data in the article and the current study.
For semen collection via the urethra, Jelinkova et al 18 showed that the sperm concentration per ejaculate ranged between 0.0 and 26.8 million spermatozoa, with a range of sperm per unit volume of 0.01–1850, similar to that observed in our study. Sperm concentration analysis in group 2, when compared with the only study found in the literature, was similar for both total quantity and sperm quantity per milliliter. 9
The sperm concentration per ejaculate of cats in group 2 presented a wider distribution than in the ejaculates of the group 1 cats (Figure 2). However, there were no statistically significant differences between groups (P >0.05). This is important in the conservation of genetic resources and the commercialization of elite male semen samples. Owing to increasing sperm concentration ranges with adequate kinetic and viability sperm parameters, there is an increase in the number of straws subjected to cryopreservation per animal, the number of straws to be produced and the number of inseminated females.
For the supravital test (EOS), samples directly collected from the epididymis of animals anesthetized with MED presented an average of 83.7% ± 7.5% cells with an intact membrane, 27 while those from urethral collection, also anesthetized with MED, presented an average of 74.0% ± 11.8%. 29 The observed EOS percentage for both groups in this study was greater than those described for spermatozoa collection with urethral catheterization and lower than those reported for epididymal spermatozoa. However, the values were within the range of 64–90% reported in a previous study. 18
The functional integrity of the membrane of spermatozoa collected by electroejaculation and evaluated via osmotic shock (CH) has been described in domestic cats by Comercio et al, 21 with averages varying from 67% to 98%. For spermatozoa obtained from the epididymis of animals anesthetized with MED, >80% of ejaculated spermatozoa presented a CH of 71–93%. 27
The morphologically normal spermatozoa percentages for both treatments were above the values reported for spermatozoa also collected via the urethra. 20,28,29 However, Jelinkova et al 18 reported that spermatozoa collected from the urethra presented a morphological normality range of 1–82%, which is in agreement with the results of our study.
Head defects are more common for epididymis-collected spermatozoa than for those obtained via urethral collection. 30 However, Jelinkova et al 18 reported that such abnormalities may be present at low levels in cat semen samples. Tail abnormalities were the defects that occurred with the highest frequency in urethral spermatozoa (39% [range 10–76%]).18,28 This is similar to the data obtained in this study. Finally, the percentage of isolated normal heads in samples from group 1 was similar to that reported by Prochowska et al. 8
Conclusions
The results of this study show that MED and DEX are effective anesthetic drugs for urethral semen collection, showing no differences with regard to semen quality in the samples obtained. Moreover, DEX offers economic advantages over MED due to its lower cost per dose, and represents an alternative drug for countries in which MED is not commercialized.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Fundação de Amparo à Pesquisa do Estado da Bahia (FAPESB) resources approved in edict 04/2014. This study was financed, in part, by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, Brasil (CAPES), Finance Code 001.
Ethical approval
This work involved the use of non-experimental animals (owned or unowned) and procedures that differed from established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient. The study therefore had ethical approval from an established committee as stated in the manuscript.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedure(s) undertaken (either prospective or retrospective studies). No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
