Abstract
Objectives
The aims of this study were to determine if stable chronic kidney disease (CKD) cats and uremic crisis cats have altered platelet function, and to determine the prevalence of positive fecal occult blood in CKD cats.
Methods
Platelet function in normal cats, clinically stable International Renal Interest Society (IRIS) stage 2–4 CKD cats and CKD cats experiencing a uremic crisis were evaluated using impedance aggregometry. Area under the curve (AUC) at 6 mins was calculated for saline, adenosine diphosphate (AUCADP) and arachidonic acid (AUCASPI). The AUC in addition to hematocrit, platelet count and mean platelet volume (MPV) were compared between groups using the Kruskal–Wallis test followed by Dunn’s post-hoc analysis. Guaiac fecal occult blood tests were performed on fecal samples and results were compared between groups using a χ2 for trend test.
Results
AUCADP (P = 0.04) and AUCASPI (P = 0.05) were significantly higher in uremic crisis cats compared with normal cats at 6 mins. Hematocrit was significantly higher in normal cats when compared with IRIS stage 3 and 4 (P = 0.002) and uremic crisis (P = 0.0008) cats, with no difference among groups for platelet count or MPV. The proportion of cats with positive fecal occult blood samples was significantly different between groups (P = 0.0017); 50% uremic crisis cats, 33% IRIS stage 3 and 4 cats, and 10% IRIS stage 2 cats were positive, while no normal cats were positive. The proportion of cats with platelet clumping was significantly different between groups (P = 0.03).
Conclusions and relevance
Platelet hyper-reactivity may be occurring in CKD cats experiencing a uremic crisis. The etiology of positive fecal occult blood samples in CKD cats is unclear and did not appear to be related to decreased platelet function as measured in this study and requires further investigation.
Introduction
The propensity for bleeding in uremic human patients is a well-documented observation, occurring in 40–50% of patients with chronic renal failure or those receiving hemodialysis.1,2 The underlying mechanism is multifactorial; however, abnormalities in platelet function are the most prominent feature, which has led to the widely used term of uremic thrombocytopathy. Clinical signs of uremic thrombocytopathy in humans may include mucosal, gingival and gastrointestinal bleeding, as well as epistaxis.1–3 More serious complications can occur, including spontaneous bleeding, prolonged bleeding from surgical sites and intracranial bleeding.1–3 The pathogenesis of this hemostatic disorder is multifactorial and includes effects of circulating toxins on platelet adhesion and aggregation, alterations of the vessel wall, anemia, beta lactam drugs and disturbance of the platelet-dense granules, which contain the intracellular platelet agonists adenosine diphosphate (ADP) and serotonin.1–8 Owing to this complexity, no single test can reliably predict a patient’s bleeding risk. In addition, the severity of platelet dysfunction appears to be related to the degree of uremia. The correction of azotemia with hemodialysis typically decreases uremic bleeding but fails to correct it completely. 4
In contrast to human medicine, uremic thrombocytopathy is poorly documented in veterinary medicine, although it is frequently referenced as the cause of bleeding tendencies in uremic veterinary patients (ie, gastro-intestinal bleeding, bleeding from surgical sites). Although there is a lack of studies documenting increased bleeding propensities during invasive procedures for uremic patients, anecdotally it has been observed that severely uremic patients are prone to prolonged bleeding or excessive bruising. Additionally, chronic kidney disease (CKD) is commonly associated with non-regenerative anemia, and although lack of erythropoietin production is most commonly implicated as a contributing cause, the possibility of gastrointestinal bleeding cannot be ruled out. In cats with CKD, gastric ulceration has been found to be less common than other species, 5 and thus it should be considered if uremic thrombocytopathia contributes to the anemia seen in CKD patients via subclinical gastrointestinal bleeding.
The few veterinary studies investigating platelet function in kidney disease have only been performed in dogs and have yielded conflicting results. Buccal mucosal bleeding time has been repeatedly documented to be prolonged in both experimental and naturally occurring CKD despite a normal platelet count.6,7 Platelet aggregation can be increased, decreased or normal in dogs with naturally occurring CKD depending on the methodology utilized. For example, platelet aggregation was delayed in CKD dogs in one study using a different platelet function analyzer. 8 Using the Born method, aggregation was either increased or decreased depending on the concentration of agonist used, 9 and no difference in platelet aggregation was found when using a whole-blood impedance method in naturally occurring CKD dogs. 10 Additionally, a recent study in dogs with acute kidney injury utilizing impedance platelet aggregometry demonstrated decreased platelet aggregation to collagen (but not to ADP or arachidonic acid [ASPI]), as compared with healthy dogs, but was still within reference intervals. 11 It is perhaps not surprising that results in dogs vary, as not all previous studies have characterized the specific disease process that may yield different results depending on the type of kidney disease (eg, protein-losing nephropathy, renal dysplasia and acute kidney injury).
There are currently no studies evaluating platelet function in cats with CKD, so the incidence of uremic thrombocytopathy and how it correlates with clinical bleeding is unknown. Given the prevalence of anemia of CKD combined with the increasing utilization of procedures such as placement of subcutaneous ureteral bypass systems and dialysis for cats with acute uremic crises, assessment and recognition of decreased platelet function in this population has the potential to improve patient care and clinical outcomes.The primary aim of this study was to compare platelet function measured by impedance aggregometry in cats with naturally occurring stable CKD, CKD cats experiencing an acute uremic crisis and healthy cats. Our hypothesis was that platelet function would decrease as renal function decreased. A secondary aim of this study was to determine the prevalence of positive fecal occult blood samples among these groups as a potential indicator of subclinical gastrointestinal bleeding.
Materials and methods
Case selection
This prospective, cross-sectional study was performed at the Colorado State University Veterinary Teaching Hospital. Normal cats from the Colorado State University Veterinary Teaching Hospital student and employee population were recruited to participate. The study was approved by the Institutional Animal Care and Use Committee and the Clinical Review Board at Colorado State University, and all owners signed informed consent forms prior to participation.
To assess health status, all cats were screened with clinical history, dietary history, physical examination, complete blood count (Siemens; ADVIA 120 Hematology System), serum biochemistry (Roche Cobas C501; Diamond Diagnostics), urinalysis, urine protein:creatinine ratio, blood pressure (Doppler with sphygmomanometer), and total thyroxine for cats >8 years old at enrollment. All diagnostic tests were performed at the Colorado State University Veterinary Teaching Hospital (CSUVTH) Diagnostic Laboratory.
CKD cats and CKD cats experiencing a uremic crisis were recruited from the patient population at the CSUVTH. Cats were considered to have CKD based on persistently elevated creatinine >1.6 mg/dl and low urine specific gravity (USG) <1.035. Cats were categorized as International Renal Interest Society (IRIS) stage 2, 3 or 4, based on IRIS guidelines, and determined to be clinically stable if their creatinine had not increased by >0.5 mg/dl or they required additional medical intervention within the preceding 2 weeks. The uremic crisis group was defined as cats that presented with clinical signs of illness that had (1) a previous history of CKD with an acute rise in creatinine from the individual’s historical baseline creatinine by more than 0.5 mg/dl, or (2) severe azotemia (creatinine >5 mg/dl) with evidence of chronic renal degeneration on ultrasonographic imaging.
Healthy cats were recruited to participate from the CSUVTH student and employee population. Cats were considered healthy based on normal examination and creatinine <1.6 mg/dl with USG >1.035 on diagnostic testing.
Exclusion criteria for all cats included history of gastrointestinal disease, cardiac disease requiring medical management, uncontrolled endocrinopathies (diabetes mellitus, hyperthyroidism), consumption of a non-commercial or raw diet, administration of clopidogrel, prednisolone, fish oil, non-steroidal anti-inflammatory drugs, erythropoietin products and antimicrobials within the preceding 2 weeks.
Multiple-channel electrical impedance platelet aggregometry
Multiple-channel electrical impedance platelet aggregometry (Multiplate 5.0 Analyzer, Diapharma Group) was performed in all enrolled cats. Heparinized blood (1.3 ml) was collected (lithium heparin micro tube; Sarstedt), kept at room temperature and analyzed within 40 mins of blood collection for a total run time of 12 mins. Before each analysis, the test cells were warmed to 37°C within the aggregometer and an automated pipette was used to decrease pre-analytical variability. Heparinized blood samples (300 µl) were diluted with 300 µl of diluent and incubated for 3 mins as recommended by the manufacturer. ADP and ASPI (Diapharma Group) were used as platelet agonists and the area under the curve (AUC) for ADP (AUCADP) and for ASPI (AUCASPI) was recorded at 3 mins, 6 mins and 12 mins. As recommended by the manufacturer, the final concentration of ADP was 6.5 µM and the final concentration of ASPI was 0.5 mM. To serve as a control and to evaluate for spontaneous platelet aggregation, an identical volume of saline was used in place of the agonists (AUCsaline). The reagents were reconstituted and stored according to the manufacturer’s recommendations in 60 µl aliquots.
Fecal occult blood
Voided fecal samples were collected by owners or Colorado State University staff for hospitalized cats for fecal occult blood testing. Fecal samples were screened for helminths using a modified double centrifugal fecal floatation technique with Sheather’s sugar solution on 3 g of feces. Identification of occult blood was determined by using the Hemoccult test (Hemoccult; Beckman Coulter), which is based on oxidation of guaiac and was performed based on manufacturer’s recommendations.
Statistical analysis
All data were analyzed with Prism software (GraphPad Software). Platelet aggregometry AUC data at 6 mins and clinicopathologic data were assessed for normality with D’Agostino and Pearson test, and found to not be normally distributed; therefore, multiple comparison analysis between the different groups (normal, IRIS stage 2, IRIS stage 3 and 4 CKD, and uremic crisis) was performed using a non-parametric Kruskal–Wallis test with Dunn’s post-hoc analysis. The AUC data (saline, ADP and ASPI) from normal cats at each time point (3, 6 and 12 mins) were assessed for normality with the D’Agostino and Pearson test and found to not be normally distributed. Therefore, for each data set (saline, ADP and ASPI), AUC data at each time point were compared with a Friedman test to determine whether the AUC results were comparable when measured at 3, 6 and 12 mins. The χ2 test for trend was used both for a comparison of positive fecal occult blood results and to compare the presence of platelet clumping. For all analyses, a P value ⩽0.05 was considered to be statistically significant.
Results
Age and clinicopathologic data for all groups are presented in Table 1.
Summary of age and clinicopathologic data for the normal, International Renal Interest Society (IRIS) stage 2 chronic kidney disease (CKD), IRIS stage 3 and 4 CKD, and uremic crisis CKD cats

Data are median (range) unless otherwise indicated
Significant difference between normal and IRIS CKD stage 2 cats
Significant difference between normal and IRIS CKD stage 3 and 4 cats
Significant difference between normal and uremic crisis cats
Normal cats
Twenty-one apparently healthy cats were enrolled, including 15 male cats and six female cats, all of which were neutered. Breeds represented were domestic shorthair (DSH; n = 13), domestic mediumhair (DMH; n = 1), domestic longhair (DLH; n = 4) and one of each of Siamese, Russian Blue and Ragdoll. The median creatinine was 1.2 mg/dl (range 0.8–1.8 mg/dl). The cat with a creatinine of 1.8 mg/dl was a 3-year-old castrated male cat with a USG of 1.068, a hematocrit of 47% and normal kidney palpation. Unfortunately, a symmetric dimethylarginine was not available.
IRIS stage 2 CKD cats
Eleven cats with IRIS stage 2 CKD were enrolled, including nine spayed female cats and two castrated male cats. Breeds represented included eight DSHs and three DLHs. Median creatinine was 2.3 mg/dl (range 1.5–2.8 mg/dl). The cat with a creatinine of 1.5 mg/dl had a creatinine of 2.0 mg/dl 1 month prior to enrollment, moderate degenerative changes on renal ultrasound and moderate muscle wasting that likely was influencing the creatinine.
IRIS stage 3 and 4 cats
Ten IRIS stage 3 cats and two IRIS stage 4 cats were enrolled, including eight spayed female cats and four castrated male cats. Owing to an insufficient number of stable IRIS stage 4 cats, these cats were combined with the IRIS stage 3 cats for analysis. Breeds represented included DSH (n = 9) and one each of the following: DMH, DLH and Siamese.
Uremic crisis cats
Six CKD cats experiencing a uremic crisis were enrolled and included five spayed female cats and one castrated male cat. Breeds represented included DSH (n = 3), Siamese crosses (n = 2) and Abyssinian (n = 1). Prior to the uremic crisis, two cats were considered IRIS stage 2, two cats were considered IRIS stage 3 and two cats did not have previous blood work to determine IRIS stage but had ultrasound findings consistent with CKD. The etiologies resulting in uremic crisis included suspected pyelonephritis (n = 2) and unknown cause (n = 4).
Clinicopathologic data
Normal cats had significantly higher hematocrit than the IRIS stage 3 and 4 cats (P = 0.002) and uremic crisis cats (P = 0.0008). There was no difference in hematocrit between normal cats and IRIS stage 2 cats, or between the three CKD cat groups (IRIS stage 2, IRIS stage 3 and 4, and uremic crisis). Serum creatinine concentration was significantly lower in the normal cats compared with IRIS stage 2 cats (P = 0.03), IRIS stage 3 and 4 cats (P <0.0001), and uremic crisis cats (P <0.0001). No significant difference in serum creatinine concentration was found between the three CKD cat groups. No significant difference in platelet count or mean platelet volume (MPV) was found any group comparison; however, normal cats had the highest percentage of platelet clumping (38% of samples) observed on blood smear (Table 1).
Multiple-channel electrical impedance platelet aggregometry
When AUC data (saline, ADP, ASPI) from the normal cats were compared from each time point (3, 6 and 12 mins), all time points were significantly different from each other for AUCsaline (3 mins vs 6 mins [P = 0.003], 3 mins vs 12 mins [P <0.0001], 6 mins vs 12 mins [P = 0.01]), AUCADP (3 mins vs 6 mins [P = 0.004], 3 mins vs 12 mins [P <0.0001], 6 mins vs 12 mins [P = 0.004]) and AUCASPI (3 mins vs 6 mins [P = 0.004], 3 mins vs 12 mins [P <0.0001], 6 mins vs 12 mins [P = 0.004]).
No significant difference in AUCsaline was found between groups (normal, IRIS stage 2, IRIS stage 3 and 4, uremic crisis; Figure 1) at the 6 min time point. Uremic crisis cats had significantly higher AUCADP (P = 0.04) and AUCASPI (P = 0.05) compared with the normal cats (Figures 2 and 3) at the 6 min time point. No other significant differences among groups were found (Table 2).

Impedance platelet aggregometry area under the curve for saline control at 6 mins performed in normal, stage 2 chronic kidney disease (CKD), stage 3 and 4 CKD, and uremic crisis cats. There was no significant difference between groups

Impedance platelet aggregometry area under the curve for platelet agonist adenosine diphosphate (ADP) at 6 mins performed in normal, stage 2 chronic kidney disease (CKD), stage 3 and 4 CKD, and uremic crisis cats. There was a significant difference between normal cats and CKD cats experiencing a uremic crisis (P = 0.04)
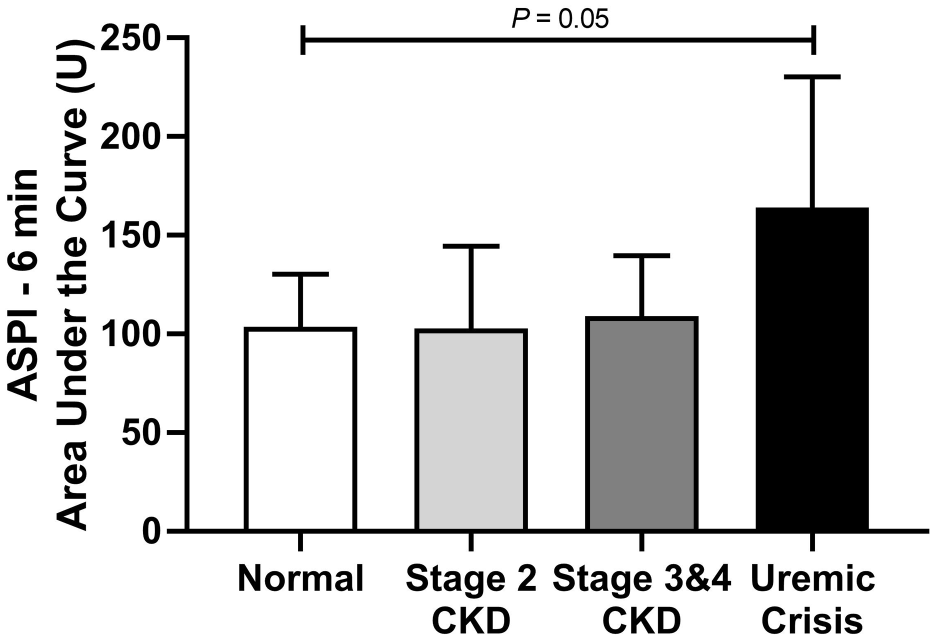
Impedance platelet aggregometry area under the curve for platelet agonist arachidonic acid (ASPI) at 6 mins performed in normal, stage 2 chronic kidney disease (CKD), stage 3 and 4 CKD, and uremic crisis cats. There was a significant difference between normal cats and CKD cats experiencing a uremic crisis (P = 0.05)
Summary of platelet aggregometry results presented as median (range) of area under the curve (AUC) at 6 mins for saline (AUCsaline) and platelet agonists adenosine diphosphate (AUCADP) and arachidonic acid (AUCASPI)

Significant difference between normal and uremic crisis cats
IRIS = International Renal Interest Society; CKD = chronic kidney disease
Fecal occult blood
Fecal samples were provided for 19/21 normal cats, 10/11 IRIS stage 2 CKD cats, 9/12 IRIS stage 3 and 4 CKD cats, and 4/6 uremic crisis cats. There was a statistically significant difference in the proportion of cats with positive samples with more positive samples in the uremic crisis and IRIS stage 3 and 4 cats (P = 0.0017; Table 1). No sample had evidence of parasites on fecal float.
Bleeding complications
One IRIS stage 4 cat was observed to bruise significantly following a clean venipuncture. No other overt evidence of bleeding was appreciated during the study.
Discussion
Evaluation of platelet function by using whole-blood multiple-channel electrical impedance platelet aggregometry demonstrated an increase in platelet aggregation in CKD cats experiencing a uremic crisis, as evident by a significantly higher AUC for both agonists compared with normal cats. This finding is in contrast to our proposed hypothesis that platelet function would decrease as renal function decreased. Increased platelet reactivity has been documented in human studies where CKD patients can have increased baseline surface expression of activated GPIIb/IIIa (platelet receptor for fibrinogen and von Willebrand factor), enhanced response to ADP and ASPI, and decreased response to antiplatelet therapy. 12 Another study in human dialysis-naive CKD patients evaluated platelet function with whole-blood impedance aggregometry and identified no specific trends in response to low-dose collagen, high-dose collagen or ADP, but 80% of the patients had increased aggregation in response to a low-dose ristocetin agonist. 13 Some theorize that platelet aggregation may only be defective in uremic patients in high-shear environments and may not be detected within the artificial environment of the aggregometer. 14 Whole-blood electrical impedance aggregometry does not evaluate the platelet’s interaction with the vessel wall (platelet adhesion and/or secretion), which is suspected to be an important defect in uremic patients that promotes bleeding tendencies. 15 Therefore, electrical impedance may not be the best way to detect platelet dysfunction in CKD cats and an alternative method, such as PFA-100, that evaluates platelet adhesion under high-shear environments should also be explored.
Platelet hyper-reactivity has also been documented in human patients with CKD in association with the uremic toxin indoxyl sulfate. Indoxyl sulfate is a protein-bound uremic toxin derived from the amino acid tryptophan and is produced by intestinal bacteria. 16 Indoxyl sulfate has been shown to promote platelet hyper-reactivity as demonstrated by an enhanced response to platelet agonists thrombin and collagen, increased P-selectin expression and release of platelet microparticles. 16 The platelet hyper-reactivity induced by indoxyl sulfate has been shown to induce thrombosis in a mouse model. 17 Thromboembolism itself has a two-fold greater risk in patients with advanced kidney disease and is a common complication for patients on dialysis. 1 The incidence of thrombosis in feline CKD patients has not been investigated but is a potential area of interest given recent descriptions of increased indoxyl sulfate levels in cats with CKD.18–21
The number of positive fecal occult blood tests increased with the severity of CKD despite a lack of platelet dysfunction identified on aggregometry. Additionally, the single cat that was observed to bruise after venipuncture did not show evidence of decreased platelet function. Reasons for the lack of platelet dysfunction noted in our study, despite some clinical evidence of bleeding, may be that whole-blood electrical impedance failed to detect platelet dysfunction in this study population, platelet dysfunction was not the etiology of gastrointestinal bleeding in these cases or there were false-positive fecal occult blood results. The majority of CKD cats were fed a commercial renal diet; however, a thorough diet history was not obtained to exclude the possibility of treats or other sources of food that are high in peroxidases (fruits, vegetables, red meat, tuna) that could cause a false-positive result. 22 Gastrointestinal ulceration would be the most straightforward explanation for the positive fecal occult blood test results; however, McLeland et al 5 did not observe any signs of gastric ulceration on histopathology of 37 CKD cats, which suggests bleeding would have to be occurring at a different location in the intestinal tract, or there is an alternative mechanism for blood loss. 5 For example, a defect in the mucosa may not be necessary for diffuse mucosal bleeding to occur in the intestinal tract. 2 Fecal samples were not obtainable from all cats enrolled in the study and therefore more robust conclusions about the prevalence of positive fecal occult blood in CKD cats cannot be made.
Limitations of the study include an inability to find cats with stable IRIS stage 4 CKD for enrollment. Hematocrit could have influenced the platelet aggregometry results as adequate red blood cell concentrations help propel platelets radially toward damaged vessel walls in laminar flow conditions and ADP in red blood cells is a potent stimulator of platelet responses. 23 Additionally, it has been shown that higher hematocrit levels can result in lower aggregometry values, which has been suggested to be related to scavenging by erythrocytes. 24 While there was a difference in platelet aggregometry and hematocrit between the normal cats and uremic crisis cats, there was no difference in platelet aggregometry between the normal cats and IRIS stage 3 and 4 cats, despite a difference in hematocrit. An additional limitation is that some cats may have had an occult cardiomyopathy, which could have had an effect on platelet aggregometry results. Previous studies in cats using different methodologies for platelet function assessment have shown that cats with heart disease can have increased platelet aggregation in response to ADP.25,26
Conclusions
Decreased platelet function assessed by whole-blood electrical impedance aggregometry was not apparent in IRIS stage 2–4 CKD cats with stable disease, or in CKD cats experiencing an acute uremic crisis. Platelet hyperreactivity may be occurring in cats experiencing a uremic crisis. The etiology of increased positive fecal occult blood samples in CKD cats is unclear and did not appear to be related to decreased platelet function as measured by the methodology used in this study and requires further investigation.
Footnotes
Acknowledgements
The authors would like to thank Dr Christine Olver for assay technical assistance, the clinical pathology technicians for assistance in sample processing, the internal medicine technicians for assistance in sample collection and all of the owners and cats who participated in the study.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by Buttons Fund for Feline CKD Research.
Ethical approval
This work involved the use of non-experimental animals only (including owned or unowned animals and data from prospective or retrospective studies). Established internationally recognized high standards (‘best practice’) of individual veterinary clinical patient care were followed. Ethical approval from a committee, while not specifically required for publication in JFMS, was nonetheless obtained, as stated in the manuscript.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedure(s) undertaken (either prospective or retrospective studies). No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
