Abstract
Objectives
The aim of this retrospective case-control study was to report the efficacy of subcutaneous triamcinolone as part of a regimen for feline eosinophilic keratoconjunctivitis (FEK).
Methods
Records and clinical photographs were reviewed and lesions semiquantitatively graded for cats with cytologically confirmed FEK. Clinical data were compared between a study population of nine cats (11 eyes) treated with, and a reference population of seven cats (eight eyes) treated without, a median of 0.11 mg/kg (range 0.10–0.20 mg/kg) of triamcinolone acetonide subcutaneously.
Results
Breed, sex, age and prevalence of corneal ulceration at presentation; corneal disease severity before and at the initiation of immunomodulation; and duration of antiviral treatment before immunomodulation did not differ significantly between populations (P ⩾0.059). Corneal plaques resolved in five cats each from the study and reference populations (P = 0.366). Median (range) time from immunomodulation to corneal plaque resolution did not significantly differ (P = 0.246) between the study (median 14 days; range 8–38 days) and reference (median 28 days, range 14–46 days) populations. No adverse reactions were attributed to triamcinolone administration, and all corneal ulcers in the study population re-epithelialized within 14 days (range 8–38 days) following triamcinolone injection. Time to corneal ulcer re-epithelialization following triamcinolone injection varied minimally in those receiving antivirals prior to (8 or 30 days until re-epithelialization), simultaneously with (38 days) or after (14 or 24 days) triamcinolone.
Conclusions and relevance
In otherwise healthy cats with FEK, subcutaneous administration of triamcinolone appears to be well tolerated and as efficacious as conventional topical immunomodulatory therapies. It may be especially useful in ulcerated eyes where topical immunomodulation is contraindicated.
Introduction
Feline eosinophilic keratoconjunctivitis (FEK) is typified by vascularized white-to-pink corneal plaques; concurrent or prior corneal ulceration is reported in up to 66.7% of cats.1–4 Cytology reveals mixed populations of mast cells, eosinophils, neutrophils and lymphocytes. 5 An association with feline herpesvirus type 1 (FHV-1) has been proposed,2,6,7 with FHV-1 DNA detected in corneal scrapings from 55–76% of cases,2,8 especially if corneal ulceration preceded the diagnosis of FEK. 2 Therapy includes a variety of topical or systemic immunomodulatory drugs, including corticosteroids,2,9 megestrol acetate, 10 ciclosporin2,11 or non-steroidal agents. 12 However, these drugs may stimulate FHV-1 reactivation from latency or exacerbate recrudescent herpetic disease, 13 and corticosteroid use may be contraindicated with concurrent ulceration. 14 This study reports a treatment protocol in which triamcinolone was administered subcutaneously as the primary method of immunomodulation in cats with FEK so as to defer use of topical immunosuppressive medications in corneas already ulcerated or at high risk of ulceration. Clinical data are compared between this study population and a reference population treated only with topical immunomodulation.
Materials and methods
Cats with cytologically confirmed FEK treated using at least one subcutaneous (SC) injection of triamcinolone acetonide (Vetalog; Boehringer Ingelheim Vetmedica) were identified through retrospective review of electronic medical records at the University of California Davis Veterinary Medical Teaching Hospital (UCDVMTH). Similarly diagnosed cats examined during the same study period of July 2009 to February 2019, but not administered triamcinolone, formed the reference population. Cats were excluded from either population if only the conjunctiva was affected, clinical descriptions or images within the medical record were insufficient for semiquantitative grading of disease severity, or if they were lost to follow-up after the initial visit. Data retrieved for all cats included sex, breed, age, month of presentation, medications administered prior to presentation, clinical and cytologic findings, treatment, and time from initial immunomodulatory medication to re-epithelialization and plaque resolution. One cat in the study population was followed up by the referring veterinarian and was therefore excluded from analyses of time to corneal plaque resolution, ongoing medical management and total examination period at UCDVMTH. Slit-lamp biomicroscopy, binocular indirect ophthalmoscopy and fluorescein staining were performed on all cats. Laboratory testing for FHV-1 was not conducted in any cat. Before, at the time of, and after administration of SC triamcinolone (study population) or topical immunomodulatory drugs (reference population) corneal disease for all eyes was graded by two authors (DRL and KEK) in a standardized manner. Grading was based on clinical images and/or descriptions. Eyes graded as minimally affected had mild or absent superficial corneal vascularization, edema, and/or fibrosis with no visible plaque. In mildly, moderately or severely affected eyes, plaque with or without associated corneal changes occupied ⩽25%, 26–75% or 76–100% of the cornea, respectively. Statistical comparisons between the two populations were performed using the Mann–Whitney U-test for continuous data or Fisher’s exact test for binary data (GraphPad Prism v8.4.1 for Macintosh; GraphPad Software). P values of ⩽0.05 were considered significant. Continuous data are presented as median (range).
Results
Signalment and history
The study and reference populations contained nine and seven cats, respectively, initially presented over a wide range of months (Figure 1a) between July 2009 and February 2019. No significant difference in age at presentation (P = 0.066; Figure 1b), or proportion of male vs female cats (P >0.999; Figure 1c) or purebred vs domestic cats (P = 0.438; Figure 1d) was detected between the two populations.

Demographic information for cats with cytologically confirmed eosinophilic keratoconjunctivitis treated with (study population, black bars [nine cats]) or without (reference population, gray bars [seven cats]) subcutaneously administered triamcinolone acetonide. (a) Month of presentation. (b–d) Significant differences between the study and reference populations were not detected for (b) age at presentation (P = 0.066), (c) sex distribution (P >0.999) or (d) breed distribution (P = 0.438). All cats were neutered. In (b), boxes represent the range between the 25th and 75th percentiles, whiskers represent the 5th and 95th percentiles and the horizontal line defines the median
Prior to referral, cats had received a variety of anti-inflammatory, antiviral and other medications. Two cats within the study population received immunomodulatory therapy; one received topically administered ciclosporin starting 80 days and ending 2 days before presentation, and the other received topically administered dexamethasone starting 37 days and ending 10 days prior to presentation, as well as an unknown dose of triamcinolone administered subcutaneously 286 days prior to presentation (328 days prior to receiving triamcinolone at the UCDVMTH). No cats in the reference population received immunomodulatory therapy prior to referral. Antiviral medications were administered to two cats in the study population (one topically, and one topically and orally) and three cats in the reference population (one topically and two orally). Antibiotics were administered to eight cats in the study population (seven topically and one topically and orally) and six cats in the reference population (all topically). Additionally, in the study population one cat each received topical serum, 1% polyvinyl alcohol artificial tears solution or oral buprenorphine; and, in the reference population, one cat each received artificial tears solution, hyaluronan-containing ophthalmic lubricating solution or oral buprenorphine.
Examination and laboratory findings
On presentation, all eyes in both populations had characteristic raised pink/white plaques involving various corneal regions (Figure 2). Fluorescein staining of the corneal plaque was apparent in seven eyes (five cats) in the study and four eyes (four cats) in the reference population. In addition, corneal ulcers were detected in a not significantly different (P = 0.362) number of eyes in the study (five eyes of five cats) and the reference (two eyes of two cats) populations. Other major ophthalmic and cytologic examination findings for both populations are shown in Table 1.

Topographic distribution of corneal plaques in cats with cytologically confirmed feline eosinophilic keratoconjunctivitis treated with (study population; nine cats, 11 eyes) or without (reference population; seven cats, eight eyes) subcutaneously administered triamcinolone acetonide. Data are presented as [number of study population eyes, number of reference population eyes].
Major clinical and cytologic examination findings on presentation for cats with cytologically confirmed eosinophilic keratoconjunctivitis, treated with (study population) or without (reference population) subcutaneously administered triamcinolone acetonide
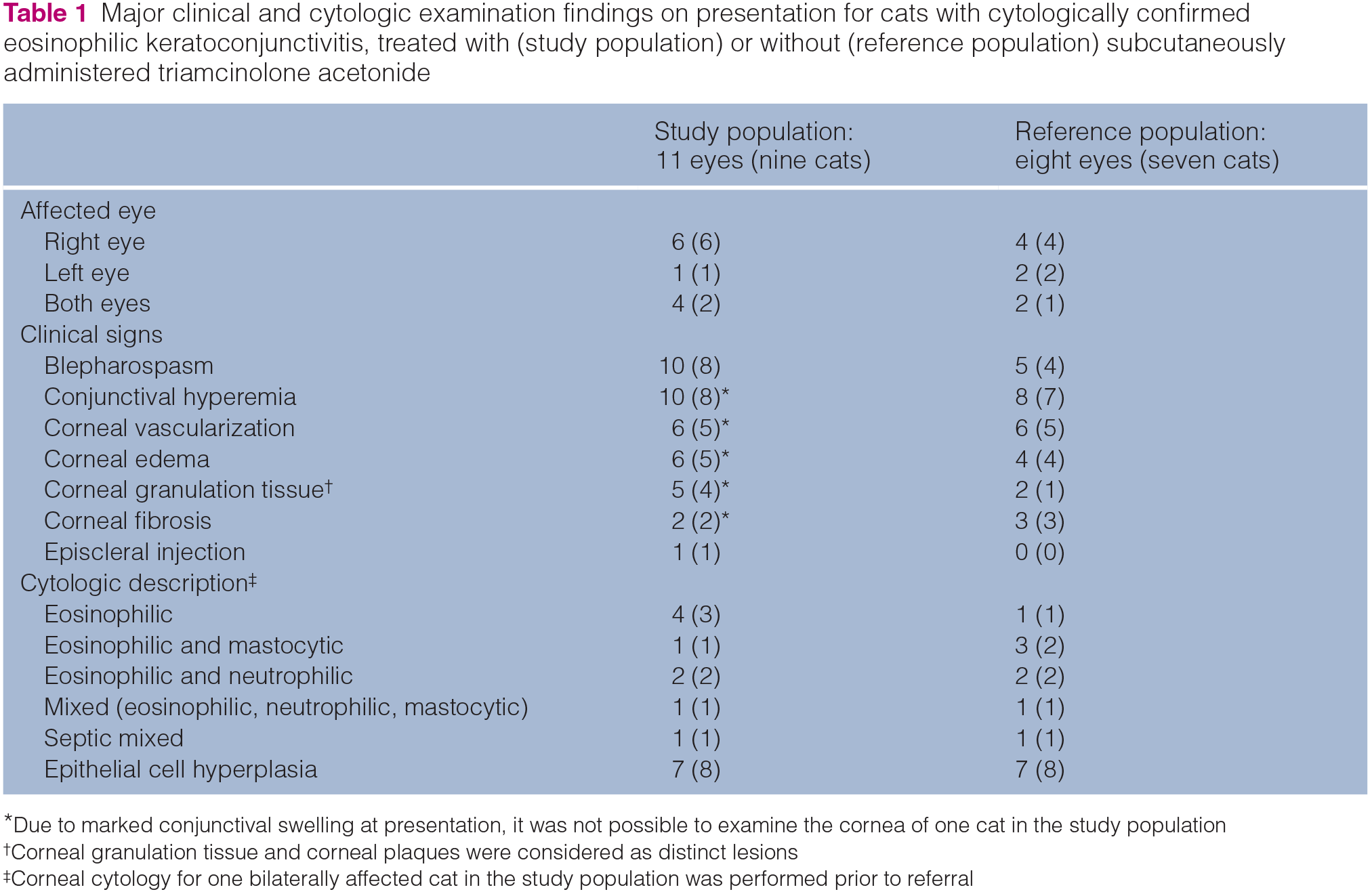
Due to marked conjunctival swelling at presentation, it was not possible to examine the cornea of one cat in the study population
Corneal granulation tissue and corneal plaques were considered as distinct lesions
Corneal cytology for one bilaterally affected cat in the study population was performed prior to referral
Treatment
Following cytologic confirmation of FEK, seven cats in the study population received an orally (four cats) and/or topically (five cats) administered antiviral drug followed (in all nine cats) by 0.11 mg/kg (0.10–0.20 mg/kg) triamcinolone acetonide administered subcutaneously 17 days (0–63 days) after initiation of the antiviral drug. Fifteen days (9–329 days) following triamcinolone administration, topical immunomodulatory drugs were prescribed for eight cats in the study population (Figure 3a). A second SC triamcinolone injection (0.11 mg/kg [0.10–0.17 mg/kg]) was administered to five cats in the study population 15 days (10–289 days) after the first. The rationale for the second triamcinolone injection included difficulty administering topical medications, notable clinical improvement following the first injection or concurrent corneal ulceration. No adverse reactions were attributed to triamcinolone administration in any cats.

Typical treatment protocol for cats with cytologically confirmed eosinophilic keratoconjunctivitis treated (a) with (study population; nine cats, 11 eyes) or (b) without (reference population; seven cats, eight eyes) subcutaneously administered triamcinolone acetonide. Five cats in the study population received a second injection of 0.10–0.17 mg/kg triamcinolone. Cats in both populations also received antiviral drugs and (following resolution of corneal ulceration) topical immunomodulatory agents. Antibiotics were prescribed topically if corneal ulceration was present.
Six cats in the reference population received orally (three cats) and/or topically (six cats) administered antiviral drugs for 19.5 days (0–1687 days) before receiving topical immunomodulatory medications (Figure 3b). No significant difference in duration of antiviral treatment prior to immunomodulation was detected between populations (P = 0.944). In both populations, all cats with corneal ulceration also received various combinations of topical antibiotics, serum and atropine. For all cats, topically administered immunomodulatory medications were selected according to clinician preference and included prednisolone acetate ophthalmic suspension, dexamethasone sodium phosphate ophthalmic solution, ciclosporin ophthalmic ointment, megestrol acetate ophthalmic suspension or neomycin–polymyxin B-dexamethasone ophthalmic suspension.
Clinical response
The total period over which examinations were conducted at the UCDVMTH was not significantly different between the study (149 days [29–1250 days]) and reference (67 days [34–1701 days]; P = 0.589) populations. Although clinical response varied among individual cats in both populations (see Table S1 in the supplementary material, Figures 4 and 5), no significant differences in corneal disease severity between the two populations were identified before (P = 0.098), on the day of (P = 0.059) or after (P = 0.222) SC injection of triamcinolone (study population) or initiation of topical immunomodulatory medications (reference population) (Figure 6). Corneal disease severity grading before and after was performed on presentation to the UCDVMTH and at the first recheck visit following triamcinolone injection (study population) or initiation of topical immunomodulatory medications (reference population), respectively, with the exception of the one cat in the study population whose severe conjunctival swelling precluded grading corneal disease severity until the second visit. Time from initial grading until starting immunomodulation did not significantly differ between the study population (5 days [0–63 days]) and the reference population (25.5 days [14–34 days]) (P = 0.109). Time to grading after initiation of immunomodulation, however, was significantly shorter in the study population (13 days [9–23 days] after triamcinolone injection) than the reference population (28 days [19–35 days] after topical immunomodulatory medications) (Figure 7; P = 0.011).
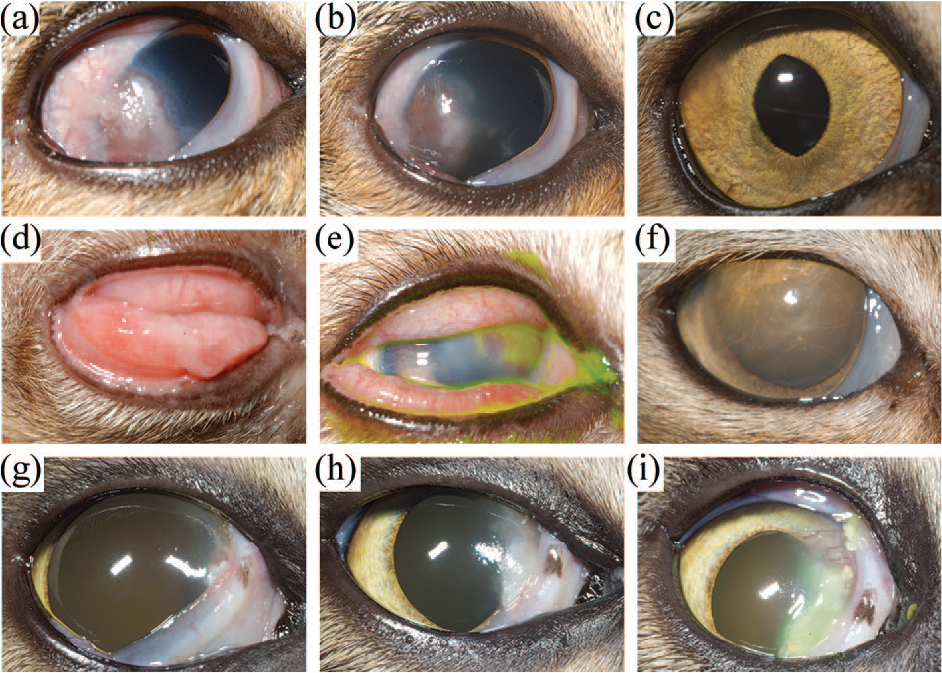
Photographs showing clinical progress of three cats from the study population with cytologically confirmed eosinophilic keratoconjunctivitis treated with subcutaneously administered 0.10–0.20 mg/kg triamcinolone acetonide. (a–c) Right eye of a 4.5-year-old spayed female domestic shorthair (DSH) cat (S1 in Table S1 in the supplementary material) (a) 5 days before, (b) 9 days after and (c) 24 days after triamcinolone injection. (d–f) Right eye of a 5-year-old castrated male DSH cat (S2 in Table S1 in the supplementary material) (d) 47 days before, (e) on the day of and (f) 16 days after triamcinolone injection. (g–i) Right eye of a 2-year-old castrated male DSH cat (S4 in Table S1 in the supplementary material) (g) 42 days before, (h) on the day of and (i) 10 days after triamcinolone injection

Photographs showing the clinical progress of two cats from the reference population with cytologically confirmed eosinophilic keratoconjunctivitis treated with topically administered immunomodulatory medications. (a–c) Right eye of a 7-year-old castrated male domestic shorthair (DSH) cat (R4 in Table S1 in the supplementary material) (a) 23 days before, (b) on the day of and (c) 35 days after initiating treatment with 1% prednisolone acetate ophthalmic suspension 1 gtt q12h. Topical prednisolone acetate had not yet been tapered at the time image (c) was collected. (d–f) Left eye of a 16.7-year-old spayed female DSH cat (R3 in Table S1 in the supplementary material) (d) 16 days before, (e) on the day of and (f) 19 days after initiating treatment with 1% prednisolone acetate ophthalmic suspension 1 gtt q12h. Topical prednisolone acetate had not yet been tapered at the time image (f) was collected

Corneal disease severity for cats with cytologically confirmed eosinophilic keratoconjunctivitis treated (a) with (study population; nine cats, 11 eyes) or (b) without (reference population; seven cats, eight eyes) subcutaneously administered triamcinolone acetonide. Closed circles represent individual cats and horizontal lines represent medians. Grading criteria are described in the materials and methods. Corneal disease severity did not significantly differ between the two populations at any time point (P = 0.059–0.222)

Time to corneal disease grading (a) before and (b) after initial immunomodulatory therapy in cats with cytologically confirmed eosinophilic keratoconjunctivitis treated with (study population; nine cats, 11 eyes) or without (reference population; seven cats, eight eyes) subcutaneously administered triamcinolone acetonide. Closed circles represent individual cats and horizontal lines represent medians. *Cats in the reference population received immunomodulatory medications for significantly longer before the final semiquantitative grading of clinical disease severity than did cats in the study population (P = 0.011)
At the first recheck following immunomodulation, corneal plaques had resolved in four eyes and improved in five eyes of the study population, and had resolved in four eyes and improved in one eye of the reference population. Time to corneal plaque resolution from onset of clinical signs did not significantly differ (P = 0.841) between the study (128 days [59–784 days]) and reference (176 days [57–2608 days]) populations. Time to corneal plaque resolution from initiation of immunomodulation also did not significantly differ (P = 0.246) between the study (14 days [8–38 days]) and reference (28 days [14–46 days]) populations. Ultimately, corneal plaques resolved in five cats from the study population and five cats from the reference population; the proportion of cats in which corneal plaques ultimately resolved did not differ between populations (P = 0.366). Clinical evidence of FEK was considered resolved by the last visit at UCD in a similar number of cats for which follow-up information was sufficient in the study (3/5 cats) or reference populations (5/7 cats; P >0.999).
In the study population, all five corneal ulcers seen at presentation had re-epithelialized, prior to starting topical immunomodulation, by 14 days (8–38 days) following triamcinolone injection 30 days (16–54 days) after the initial visit. Time to corneal ulcer re-epithelialization following triamcinolone injection varied minimally among those receiving antiviral drugs prior to (8 or 30 days until re-epithelialization), simultaneously with (38 days) or after (14 or 24 days) triamcinolone. Considering the two ulcers seen at presentation in the reference population, one had re-epithelialized by 19 days following initiation of topical immunomodulation (35 days after the initial visit). The other was not re-assessed until the cat was re-presented at 754 days. At that time the cat still had evidence of FEK but was not ulcerated.
At the last follow-up examination, antiviral therapy was ongoing in five cats each in the study and reference populations. For cats in which antiviral treatment was stopped, overall treatment duration was 42.5 days (30–105 days) in the study population and 92 days (82–102 days) in the reference population. Neither duration of antiviral therapy (P = 0.533) nor number of cats receiving ongoing treatment (P = 0.633) was significantly different between populations.
Discussion
This study described a series of cats in which the initial immunomodulatory treatment for FEK was subcutaneously administered triamcinolone in conjunction with antiviral drugs. Immunomodulatory medications were administered topically once corneal plaques improved and/or corneal ulceration resolved (Figure 3). A reference population with the same diagnosis and treated similarly but without the use of triamcinolone revealed that triamcinolone was well tolerated, and differences in clinical outcomes were not identified between cats in the two populations. Despite the reference population in this retrospective study being selected post-hoc, no significant differences were identified between the two populations with respect to signalment; corneal disease severity before or at the time immunomodulatory medications were administered; or time from initial grading of corneal disease severity to immunomodulation. In addition, cats in this study were of similar breed,2–4,7,9,11 age2,4,7,8,11 and sex1,4,7,11 distribution as in other studies of FEK. Interestingly, as described by Morgan et al, 7 Spiess et al 11 and Paulsen et al, 1 castrated males predominated in both populations of the present study. Given the similarities in demographics between the two populations of this study and those of other published studies, the data presented here are likely broadly applicable.
Cats in the present study were evaluated over many years by multiple clinicians. Like all retrospective studies, therefore, therapeutic regimens varied according to clinician preference, especially the topical immunomodulatory drug selected and its frequency of administration. However, the major and consistent treatment difference between populations was SC administration of triamcinolone to cats in the study population only. Therefore, it would not have been possible to definitively or causally associate SC triamcinolone administration with any differences in clinical response noted between populations. Importantly, though, no significant differences in outcome were detected in the present study, suggesting that triamcinolone is largely equivalent to conventional therapy. Specifically, no significant difference was detected between populations for corneal disease severity after immunomodulation, time to plaque resolution from the onset of clinical signs or initiation of immunomodulation, and time to corneal ulcer re-epithelialization. The only significant difference detected between the two populations was that cats in the reference population received immunomodulatory treatment for significantly longer before the final grading of corneal disease severity than did cats in the study population. Despite this, there was no significant difference in the severity of corneal disease between the two populations at that time point. Though not statistically significant, trends for cats receiving triamcinolone to have higher clinical disease grades at the time of initial immunomodulation yet faster time to corneal plaque resolution than those treated with topical immunomodulation may indicate clinical relevance. This indicates that cats in the study population improved equivalently to cats in the reference population despite a shorter treatment period, and further supports the hypothesis that triamcinolone is as effective as topical immunomodulation in controlling clinical signs of FEK. It will be important to assess this assertion more accurately using prospective, case-controlled studies with standardized topical medication regimens.
Triamcinolone acetonide is an intermediate-acting glucocorticoid with negligible mineralocorticoid activity, labeled for treatment of inflammation and related disorders in dogs, cats and horses. The labeled dose for cats is 0.11–0.22 mg/kg intramuscularly or subcutaneously, with repeat injection appropriate if clinical remission is not seen within 7–15 days. Cats treated with systemic corticosteroids are at risk for herpetic reactivation, as exemplified by the use of intramuscular methylprednisolone acetate (5 mg/kg) to induce FHV-1 shedding and identify carriers in a feline colony. 14 In the present study, therefore, antiviral medications were recommended for 1–2 weeks prior to triamcinolone so as to limit herpetic reactivation and recrudescence. Despite the proposed association between FHV-1 and FEK,2,6,7 and not testing cats in the present study for FHV-1, no signs of herpetic reactivation were observed at any examination in any cat. Additionally, all cats in the study population were systemically well and no adverse reactions were attributed to triamcinolone. Taken together, these preliminary data suggest that triamcinolone may be a safe and effective treatment for FEK. However, verification of these data in a larger population, and monitoring for glucocorticoid-mediated side effects or herpetic reactivation is recommended.
Whether corneal ulceration in FEK occurs secondary to FHV-1, eosinophilic inflammation or other cytokine-induced cytolysis, topical immunosuppression is typically avoided with corneal ulceration so as to reduce bacterial superinfection, corneal protease potentiation and keratomalacia, and exacerbation or reactivation of herpetic disease. It is also possible that systemically administered corticosteroids may reach the cornea via the tear film. In dogs, prednisolone is detectable in the tear film following oral prednisone administration, but at concentrations unlikely to affect corneal wound healing. 15 However, to our knowledge, similar data are not available for cats receiving triamcinolone. Here, all five cats of the study population with corneal ulceration at presentation healed within a median period of 14 days, despite triamcinolone administration. Interestingly, in the study population, time to corneal ulcer re-epithelialization following triamcinolone injection did not significantly differ between those receiving antiviral drugs prior to (8 or 30 days until re-epithelialization), simultaneously with (38 days) or after (14 or 24 days) triamcinolone. This may suggest that inflammatory mediators, rather than FHV-1, are ulcerogenic in FEK. Although the data reflect few cats, they may support simultaneous administration of an antiviral drug and triamcinolone on the day of cytologically confirmed FEK diagnosis, irrespective of corneal ulceration. If true, this would likely reduce time to disease resolution, cost and number of recheck visits.
Conclusions
This study describes an otherwise healthy population of FEK-affected cats in which initial immunomodulatory treatment with triamcinolone was typically associated with notable improvement in clinical signs, especially plaque reduction and epithelialization of ulcerated corneas permitting initiation of topical maintenance immuno-therapy. No significant differences were identified between the study and reference populations for any parameters of disease resolution, suggesting that triamcinolone may be safe and approximately as efficacious as topical immunomodulatory therapy for treatment of FEK. Based on these preliminary data, we propose a SC triamcinolone injection in conjunction with antiviral drugs as a useful means of gaining relatively rapid control of corneal disease in cats affected with FEK, especially cats affected with concurrent corneal ulceration or cats in which topical medication is difficult. Emphasis on the importance of re-evaluation at 2 weeks following triamcinolone injection to evaluate for corneal ulceration and to begin appropriate maintenance topical immunomodulatory therapy is imperative.
Supplemental Material
Table S1
Clinical disease severity in 16 cats (19 eyes) with cytologically proven eosinophilic keratoconjunctivitis prior to, at the time of and at the first recheck following subcutaneous injection of 0.10–0.20 mg/kg triamcinolone acetonide (study population; nine cats [S1–S9], 11 eyes) or topical administration of immunomodulatory medications (reference population; seven cats [R1–R7], eight eyes). Grading criteria are described in the methods
Footnotes
Acknowledgements
The authors thank Drs Ann Cooper, Sydney Edwards, Michelle Martin de Bustamante and Bret Moore for management of some cats in this study, and Dr Chrisoula Toupadakis Skouritakis for assistance with the figures.
Author note
This paper was presented, in part, at the 12th Comparative Ocular Surface Diseases Workshop, Niseko, Japan, February 2020.
Supplementary material
The following file is available online:
Table S1: Clinical disease severity in 16 cats (19 eyes) with cytologically proven eosinophilic keratoconjunctivitis prior to, at the time of and at the first recheck following subcutaneous injection of 0.10–0.20 mg/kg triamcinolone acetonide (study population; nine cats [S1–S9], 11 eyes) or topical administration of immunomodulatory medications (reference population; seven cats [R1–R7], eight eyes).
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work involved the use of non-experimental animals only (including owned or unowned animals and data from prospective or retrospective studies). Established internationally recognized high standards (‘best practice’) of individual veterinary clinical patient care were followed. Ethical approval from a committee was therefore not necessarily required.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedure(s) undertaken (either prospective or retrospective studies). No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
