Abstract
Objectives
The objective of this study was to determine if inflammatory markers are associated with antithrombin activity in cats.
Methods
For a retrospective population of 231 cats admitted to a referral hospital, antithrombin activity was classified as decreased (n = 77), intermediate (n = 97) or in the upper quartile (n = 57). Odds ratios (ORs) were calculated for an association between decreased or upper quartile activity and hypoalbuminemia, hyperfibrinogenemia, band neutrophilia and toxic change. Multiple logistic regression was performed to determine if an association between band neutrophilia and decreased antithrombin activity was independent of decreased hepatic synthesis, consumptive coagulopathy or protein loss.
Results
Cats with decreased antithrombin activity were more likely than cats with intermediate-to-upper quartile activity to have band neutrophilia (OR 2.85, P = 0.0050), hypoalbuminemia (OR 12.1, P <0.0001) or toxic neutrophils (OR 4.47, P <0.0001). Cats with antithrombin activity in the upper quartile were less likely than those with intermediate-to-low activity to have hypoalbuminemia (OR 0.31, P = 0.0023) or toxic neutrophils (OR 0.44, P = 0.033). In a regression model that included other mechanisms for decreased antithrombin, band neutrophilia remained associated with decreased antithrombin activity (adjusted OR 2.62, P = 0.013).
Conclusions and relevance
Contrary to previous studies suggesting antithrombin is a feline positive acute phase protein, this study demonstrates an association between decreased antithrombin activity and inflammation. Further studies are needed to determine the mechanistic basis of this association.
Keywords
Introduction
Inflammatory cytokines alter hepatic protein expression, with proteins that show a ⩾25% change in concentration defined as acute phase proteins (APPs). 1 Activity of the endogenous anticoagulant antithrombin has been reported to be normal or elevated in sick cats, including cats affected by disorders that might be predicted to induce consumptive coagulopathies.2–5 This has led to the suggestion that antithrombin is a feline positive APP upregulated by inflammation. 2 This is contrary to findings in other species, in which antithrombin production is reduced by experimental administration of the inflammatory cytokine interleukin (IL)-6, consistent with a negative APP. 6
Antithrombin has numerous anti-inflammatory and anticoagulant activities, including acting as a powerful inhibitor of thrombin and factor Xa. 7 In humans, acquired or congenital antithrombin deficiencies increase the risk of thrombosis,8–10 and in dogs, decreased antithrombin activity is associated with disseminated intravascular coagulation (DIC) and mortality. 11 Acquired antithrombin deficiency can occur as a result of reduced hepatic synthetic capacity, increased consumption or loss; for example, owing to hemorrhage or urinary or gastrointestinal protein loss.7,11 As these mechanisms can be associated with inflammation,12–14 if inflammatory cytokines upregulate antithrombin production in cats, this could limit the sensitivity of decreased antithrombin as a marker of hepatic failure, consumptive coagulopathies or protein-losing disorders in feline patients.
This study aimed to determine if antithrombin activity is positively or negatively associated with markers of acute inflammation in cats, and if any association between decreased antithrombin and inflammation is independent of other mechanisms known to decrease antithrombin concentration or activity.
Materials and methods
Electronic medical records of the Texas A&M University Veterinary Medical Teaching Hospital were searched for cats with at least one antithrombin result reported between 1 January 2009 and 10 April 2019. Cats with antithrombin activity below the institutional reference interval (RI) of 114.0% were paired with two breed-matched controls with antithrombin activity ⩾114.0%. If there were more than two possible controls of the appropriate breed, included controls were selected by a random number generator. If two breed-matched controls were not available, two cats of any other breed were randomly selected. If cats had multiple antithrombin results, including results both above and below 114.0%, the first visit at which antithrombin was decreased was used for data collection. For cats with multiple antithrombin results all of which were ⩾114.0% or all of which were <114.0%, the first visit at which antithrombin was measured was used. Case numbers and cats excluded from each stage of analysis are summarized in Figure 1.

Flowchart summarizing cats included in each phase of the analysis. AT = antithrombin; CBC = complete blood count; RI = reference interval
Using data from the medical record, cats were classified as affected or unaffected by hypoalbuminemia (<2.5 g/dl), hyperfibrinogenemia (>467 mg/dl) and band neutrophilia (>300/µl), as defined by institutional RIs or toxic neutrophils. Toxic neutrophils were considered present if toxic change other than a few Döhle bodies were reported. To further investigate if inflammation was independently associated with decreased antithrombin, cats were classified according to whether established mechanisms for decreased antithrombin were likely present.
Reduced hepatic synthesis was considered likely if there was an increase above the RI in at least one of alanine aminotransferase, alkaline phosphatase or gamma-glutamyl transferase and at least three of the following: increased bilirubin; increased ammonia; decreased albumin; decreased urea; decreased cholesterol; or decreased glucose. If cats met the criteria for both consumption and reduced hepatic synthesis, they were classified as affected by reduced hepatic synthesis only, unless clinical information provided evidence of concurrent thrombosis.
Consumption was considered likely if at least three of the following were present: prothrombin time (PT) and/or activated partial thromboplastin time (aPTT) prolonged by >25% of the upper RI; fibrinogen below the lower limit of the RI; D-dimers increased above the RI; or platelets below the lower limit of the RI without reported clumping on blood smear examination.
Antithrombin loss was considered likely if there was evidence of severe hemorrhage or protein-losing disease. Severe hemorrhage was considered present if there was reported clinical evidence of bleeding and at least one transfusion of whole blood, donor packed red blood cells or oxyhemoglobin during the same hospitalization as antithrombin measurement. Protein-losing disorders were considered likely if albumin was below the RI and there was histologically confirmed gastrointestinal disease, or an increased urine protein:creatinine ratio (UPC), without an active sediment.
For biochemistry, urinalysis and gastrointestinal histopathology, results from the same visit as the antithrombin measurement were included. For coagulation parameters, results generated by analysis of the same sample as the antithrombin measurement were included. For hematology parameters, any complete blood count performed within 24 h of antithrombin measurement was included. Band neutrophils and platelet clumping were assessed during microscopic blood smear examination performed by a clinical pathologist, clinical pathology resident, medical technologist or trained student worker at the time of sample submission and were not re-reviewed for the purposes of this study. Band neutrophil percentage was determined by a 100–200-cell microscopic leukocyte count. Toxic neutrophils are a criterion for pathologist review in our laboratory, so all cases of toxic change were confirmed by a board-certified clinical pathologist or clinical pathology resident.
Total leukocyte count and platelet concentration were measured using a Cell-Dyn 3500 (Abbott) or an Advia 2120 (Siemens). Biochemistry analyses were performed using a Vitros 4600 chemistry system (Ortho Clinical Diagnostics). Coagulation assays were performed using an ACL-TOP CTS 9000 or ACL-TOP CTS 300 (Instrumentation Laboratories) coagulation analyzer and the following reagents: HemosIL RecombiPlasTin 2G (PT); HemosIL SynthASil APTT reagent (aPTT); HemosIL QFA Thrombin (Bovine; fibrinogen Clauss assay); HemosIL D-Dimer HS 500 (D-Dimer); HemosIL Liquid Antithrombin (antithrombin). The antithrombin activity assay is chromogenic and results are expressed as a percentage of normal pooled plasma. All analyzers underwent daily quality control and regular maintenance throughout the years included in the study. To the best of our knowledge, analytical sensitivity and specificity for feline antithrombin has not been confirmed for any commercially available antithrombin assay.
Correlations between antithrombin activity and albumin, fibrinogen and band neutrophil concentrations were assessed by Pearson’s R if normally distributed or Spearman’s rho if one or both datasets were non-normal based on a D’Agostino–Pearson test. Antithrombin concentration was compared between cats with and without toxic change using a Mann–Whitney U test.
For categorical analysis, antithrombin concentrations below the lower limit of our institutional RI (114.0%) were considered to be low. At our institution, an upper reference limit is not provided for antithrombin, because elevated values are not considered clinically relevant. Therefore, to explore the possibility that antithrombin is a positive APP, we defined cats with antithrombin concentrations in the upper quartile (⩾153.0%) for the study population as having high antithrombin concentrations. Antithrombin concentrations of 114.0–152.9% were considered intermediate. Odds ratios (ORs) were calculated for below the RI antithrombin or antithrombin in the upper quartile of the study population between cats affected or unaffected by each inflammatory marker. ORs for death or euthanasia during the same hospitalization as antithrombin measurement were calculated for cats with vs without below the RI or upper quartile antithrombin.
ORs for decreased antithrombin were calculated for cats classified as likely affected or unaffected by reduced hepatic synthetic capacity, increased consumption or protein loss. Reduced hepatic synthesis, consumption, loss and band neutrophils were then included as independent variables in a multivariate logistic regression analysis to determine adjusted ORs for decreased antithrombin. The rationale for including band neutrophils, rather than all four inflammatory markers, as components in the multivariate regression analysis was to avoid collinearity. Band neutrophils were selected as likely more specific for inflammation than hypoalbuminemia and possibly less subjective and thus less susceptible to inter-observer variation than assessment of toxic change. 15 Statistical analysis was performed using MedCalc Statistical Software version 19.1.3 and Stata 16.
Results
Demographics
There were 77 cats with antithrombin <114%, including 53 domestic shorthairs, six domestic longhairs, six domestic mediumhairs, two Russian Blues, two Siamese and one each of Angora, Maine Coon, mixed breed, Oriental Shorthair, Persian, Ragdoll, Siberian and Tonkinese. The 154 cats with antithrombin ⩾114% were composed of 107 domestic shorthairs, 19 domestic longhairs, 13 domestic mediumhairs, five Siamese, two Maine Coons, two mixed breeds, two Persians and one each of Russian Blue, British Shorthair, Oriental Shorthair and Ragdoll. The median age was 9 years (range 1–18 years) for cats with antithrombin <114% and 9 years (range 0.5–19 years) for cats with antithrombin ⩾114%. For cats with antithrombin <114%, there were 44 castrated males and 33 spayed females. For cats with antithrombin ⩾114%, there were 87 castrated males, 57 spayed females, seven intact females, two entire males and one cat of unrecorded sex.
Association between antithrombin and outcome
Below RI antithrombin was associated with increased odds of death or euthanasia during the same hospitalization as antithrombin measurement (OR 2.46, 95% confidence interval [CI] 1.37–4.42; P = 0.0026). Antithrombin activity in the upper quartile was not significantly associated with death or euthanasia (OR 1.00, 95% CI 0.52–1.92; P = 0.99). The diagnostic category for cats with below RI, upper quartile and intermediate antithrombin activities are provided in Supplementary Table 1 in the supplemenary material.
Associations between antithrombin and inflammatory markers
Albumin, fibrinogen and band neutrophil concentrations are summarized in Table 1. Antithrombin activity was positively correlated with albumin concentration (Pearson’s R 0.51, 95% CI 0.40–0.60) (Figure 2) and negatively correlated with band concentration (Spearman’s rho −0.32, 95% CI −0.44 to −0.20) (Figure 3). Fibrinogen concentration was not significantly correlated with antithrombin activity (Spearman’s rho 0.11, 95% CI −0.02 to 0.23) (Figure 4). Antithrombin was significantly lower in cats with toxic change (median 110.0%, interquartile range [IQR] 20.7) compared with those without (median 137.8, IQR 44.0; P <0.0001).
Concentrations of inflammatory markers in cats with below reference interval (RI), intermediate and upper quartile antithrombin concentrations


Antithrombin activity and albumin concentration. The horizontal dotted line defines the lower limit of the reference interval (RI) for albumin (2.5 g/dl) and the vertical dotted line defines the lower limit of the RI for antithrombin activity (114%)

Antithrombin activity and band neutrophil concentration. The horizontal dotted line defines the upper limit of the reference interval (RI) for band neutrophils (300/µl) and the vertical dotted line defines the lower limit of the RI for antithrombin activity (114%)

Antithrombin activity and fibrinogen concentration. The horizontal dotted line defines the upper limit of the reference interval (RI) for fibrinogen (467 mg/dl) and the vertical dotted line defines the lower limit of the RI for antithrombin activity (114%)
Hypoalbuminemia, band neutrophilia and toxic neutrophils were associated with significantly increased odds of antithrombin <114% (Table 2). Toxic neutrophils and hypoalbuminemia were associated with decreased odds of antithrombin activity in the upper quartile (Table 3).
Odds ratios (ORs) for occurrence of inflammatory markers in cats with antithrombin activity below the reference interval (RI; <114.0%) vs cats with within RI antithrombin (⩾114%)

Albumin concentration was not available for 7/231 cats
P <0.05
Fibrinogen was not measured in 4/231 cats
Complete blood count was not performed in 21/231 cats
CI = confidence interval
Odds ratios (ORs) for occurrence of inflammatory markers in cats with antithrombin activity in the upper quartile (>153.0%) vs cats in the lower three quartiles (<153%)
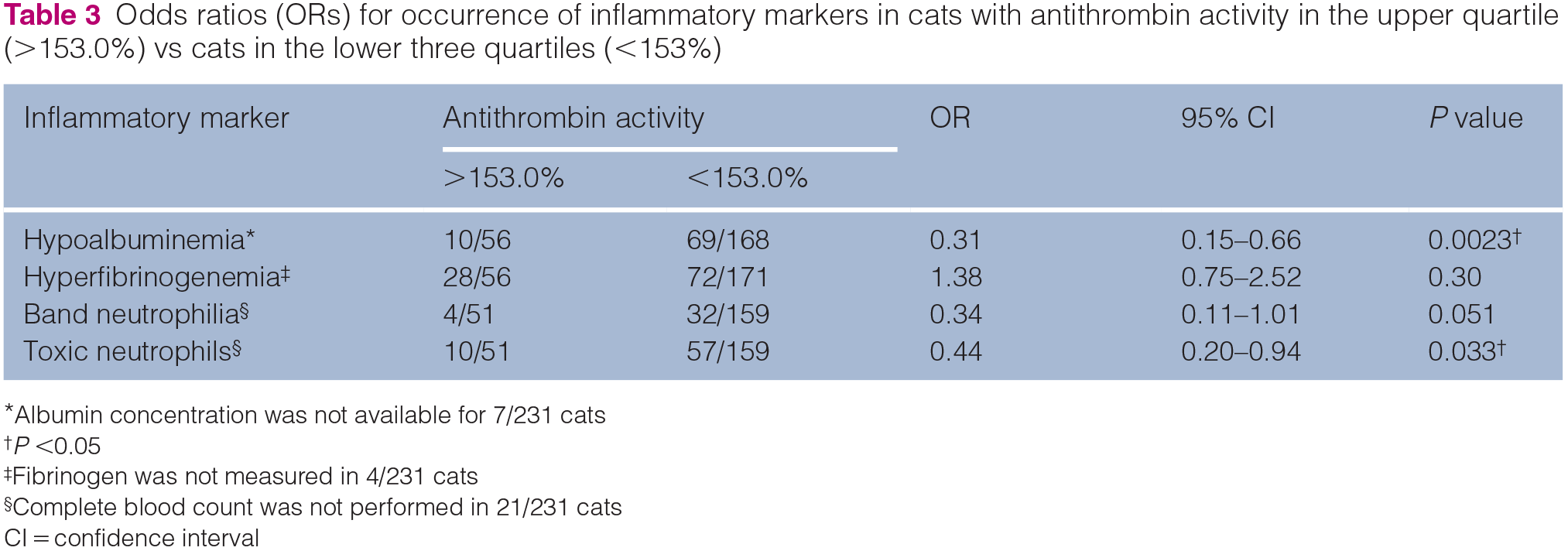
Albumin concentration was not available for 7/231 cats
P <0.05
Fibrinogen was not measured in 4/231 cats
Complete blood count was not performed in 21/231 cats
CI = confidence interval
Mechanisms associated with decreased antithrombin
For 210 cats with sufficient data to allow inclusion in the regression analysis, numbers meeting the criteria for likely reduced hepatic synthesis, consumptive coagulopathy or protein loss are shown in Table 4. Platelets were clumped in 48/77 cats with antithrombin <114% and 93/137 cats with antithrombin ⩾114%, and consumption was scored without including platelet count for these cases. Details of coagulation findings in cats meeting criteria for consumptive coagulopathy are provided in Supplementary Table 2 in the supplementary material. Of the 13 cats with decreased antithrombin and meeting the criteria for increased protein loss, eight had evidence of severe hemorrhage, four were suspected to have gastrointestinal protein loss and one had both protein-losing enteropathy and blood loss. Of the 22 control cats meeting the criteria for protein loss, nine had evidence of blood loss and 13 met the criteria for gastrointestinal or urinary protein loss. A UPC was available for only 2/210 cats, and clinically relevant urinary protein loss was assumed to be absent in the remaining cats.
Associations between decreased antithrombin activity and occurrence of reduced hepatic synthetic capacity, consumptive coagulopathy, protein-losing disease or band neutrophilia

To assess for collinearity, variance inflation factors were calculated and were as follows: reduced hepatic synthesis 1.02, consumptive coagulopathy 1.04, loss 1.01 and band neutrophilia 1.02 (mean 1.02); 21/231 cats were excluded because insufficient information was available to determine if the cat was affected by one or more condition
P <0.05
OR = odds ratio; CI = confidence interval; adjusted OR = OR from multiple logistic regression analysis
Reduced hepatic synthesis and consumption were associated with increased odds of decreased antithrombin. These mechanisms and band neutrophilia were identified as independent predictors of decreased antithrombin in a multivariate logistic regression analysis (Table 4). Loss was not significantly associated with decreased antithrombin based on absolute or adjusted ORs.
Discussion
Toxic neutrophils and hypoalbuminemia were less common in cats with higher antithrombin concentrations, and there were no significant associations between higher antithrombin concentrations and other inflammatory markers. These findings do not support the hypothesis that antithrombin is a positive APP in cats. Instead, we identified associations between three inflammatory markers, hypoalbuminemia, 16 band neutrophilia16–18 and toxic change, 17 and reduced antithrombin activity.
The suggestion that antithrombin is a positive APP originates from radioactive tracer studies that reported increased antithrombin synthesis in response to inflammation. These include two studies that induced inflammation by infusing dogs with endotoxin or injecting rabbits with turpentine.19–21 In both studies, antithrombin synthesis increased, as measured by incorporation of the radioactive tracer. However, there was a net reduction in plasma antithrombin concentration in the canine study and a marginal increase in the rabbit model, rather than the ⩾25% increase in concentration, which defines a positive APP.19,21 Thus, these studies could be consistent with an appropriate upregulation of antithrombin production in response to accelerated consumption or clearance, rather than providing evidence that inflammatory cytokines upregulate antithrombin production as part of an acute phase response. A later study treated cultured human hepatocytes and baboons with IL-6, a key mediator of the acute phase response, resulting in decreased antithrombin expression, consistent with a negative APP. 6
The continued suggestion that antithrombin is likely a positive APP in cats is based on reports of normal or elevated antithrombin concentrations in sick cats.2–5,22 This included cats with cardiac disease or DIC, two scenarios associated with decreased antithrombin in dogs.11,23 These studies did not specifically investigate associations between antithrombin and inflammatory markers, but it was hypothesized that the lack of an expected decrease in antithrombin could be consistent with a positive APP.2,4,24
However, there are other possibilities that might explain the lack of decreased antithrombin in these studies. One consideration is that inflammatory cytokines are not the only potential regulators of antithrombin expression. The promoter region of the antithrombin gene includes a hormone response element and upregulation of antithrombin occurs in response to a range of activators, including thyroid hormones, retinoic acid and glucocorticoids. 25 In one study reporting increased antithrombin activity in cats with cardiac disease vs healthy controls, 9/11 cats were hyperthyroid. 4 A similar association between elevated thyroid hormone concentrations and increased antithrombin has been reported in women. 26
Another consideration is variability in assay performance, as studies of cats with DIC have conflicting results, with some authors reporting frequent normal to increased antithrombin concentrations and others reporting that antithrombin is relatively commonly decreased in DIC or inflammatory disease.2,5,27–30 This could reflect differences between study populations and the criteria used to define DIC, but variability in the sensitivity of assays for decreased antithrombin activity in feline plasma is also a possibility. Measurement of antithrombin activity in veterinary samples relies on adapting assays developed for human plasma. Confirming analytical sensitivity and specificity for feline antithrombin is challenging as a purified source of feline antithrombin is not readily available, and assays are typically adopted if biologically plausible results are generated for pooled normal feline plasma. 2 Comparison of performance of commercially available assays for feline plasma has not been published, but it is noticeable that studies reporting that decreased antithrombin is uncommon in sick cats have frequently used the same commercial chromogenic assay.2,5,22 Studies that have commonly found decreased antithrombin have used a variety of other reagents, including other reports using the same reagent as the current study.27,28 More detailed assay validation is required to better interpret differences between published feline case series regarding the diagnostic value of decreased antithrombin activity for DIC or other hypercoagulable states.
Our findings could suggest that antithrombin is a negative APP, with reduced expression in response to inflammatory cytokines. However, decreased antithrombin activity was associated with only one of the two other APPs included in this study, that is, albumin. Albumin’s value as a negative acute phase marker in cats is questionable. Although decreased albumin has been reported in cats with a variety of inflammatory disease, its sensitivity for distinguishing cats with inflammatory disease from healthy controls is low. 31 The specificity of decreased albumin is also likely to be limited, particularly in this population of cats, as protein-losing disorders or hepatic dysfunction can decrease both albumin and antithrombin.11,32 We did not find a significant association between increased fibrinogen and decreased antithrombin activity. This could argue against a role for antithrombin as a negative APP, as fibrinogen is reported to have high sensitivity and specificity for distinguishing cats with inflammatory disease from healthy controls. 31 However, a relatively high proportion of cats in our population had evidence of reduced hepatic synthetic capacity or consumptive coagulopathy, which could have reduced the sensitivity of hyperfibrinogenemia as a marker for inflammation.12,30 Ideally, we would have included a feline major APP (eg, serum amyloid A), but concentrations were not available for this retrospectively identified population.14,33
In our multivariate regression analysis, the included inflammatory marker, band neutrophilia, was independently associated with decreased antithrombin. However, there are limitations to our analysis. There is subjectivity in identification of band neutrophils, as highlighted by an equine study, which reported only moderate agreement between clinical pathologists for identification of bands. 15 Ideally, to increase consistency, we would have obtained band percentages from leukocyte differentials performed by a single observer, but it was not feasible to retrieve the original slides for large numbers of the cases included in the current study owing to the relatively long timespan for case inclusion. Retrospective classification of cats as affected or unaffected by hepatic failure, consumptive states or protein-losing disorders was likely imperfect. This was particularly true for protein-losing disorders, as only two cats had UPC data available and histopathology was not consistently performed in cats with gastrointestinal signs. The lack of association between decreased antithrombin and protein loss likely reflects these limitations, rather than a difference between pathogenesis of antithrombin deficiency in cats vs other species. Identification of consumptive coagulopathies is also challenging, as demonstrated by a study in which 37.5% of cats with multiorgan fibrin thrombi at necropsy did not have ante-mortem laboratory findings consistent with DIC, and 21% of cats that met laboratory criteria for DIC did not have histological evidence of thrombotic DIC at necropsy. 12 While this may partly reflect the potential for microthrombi to break down during post-mortem examination, it is likely a consequence of the limited sensitivity and specificity of laboratory markers for DIC.12,34 In cats, laboratory diagnosis of consumption is a particular challenge, both because of the difficulty of obtaining accurate platelet counts,12,34 and the low specificity of D-dimers. 29 It is also possible that there are additional factors not included in the regression analysis that could influence antithrombin concentration. For example, our regression model did not include thyroid hormone concentration,25,26 endogenous or exogenous glucocorticoid exposure 20 or hypertension,35,36 all of which merit further investigation.
Conclusions
In cats treated at a veterinary referral hospital for a variety of clinical conditions, decreased antithrombin was associated with inflammation. Further work is needed to determine the mechanism underlying this association, but these results suggest that decreased antithrombin in a cat with inflammatory disease should not necessarily be considered evidence of reduced hepatic synthetic capacity, DIC or protein-losing disease.
Supplemental Material
Supplementary Table 1
Diagnostic categories for cats with below reference interval ((<114%), intermediate (114–152.8%) or upper quartile ≥153%) antithrombin activity
Supplemental Material
Supplementary Table 2
Criteria leading to classification of cats as affected by consumptive coagulopathy
Footnotes
Acknowledgements
The authors would like to thank Dr Nick Jeffery for statistical advice.
Author note
This study was presented by Prudence Sun as a poster at the American College for Veterinary Pathology’s 2019 meeting, San Antonio, TX, USA.
Supplementary material
The following files are available online:
Supplementary Table 1: diagnostic categories for cats with below reference interval (<114%), intermediate (114–152.8%) or upper quartile (⩾153%) antithrombin activity.
Supplementary Table 2: criteria leading to classification of cats as affected by consumptive coagulopathy.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Prudence Sun was supported by the Veterinary Medical Student Research Training Program, funded by the College of Veterinary Medicine and Biomedical Sciences, Texas A&M University.
Ethical approval
This work involved the use of non-experimental animals only (including owned or unowned animals and data from prospective or retrospective studies). Established internationally recognised high standards (‘best practice’) of individual veterinary clinical patient care were followed. Ethical approval from a committee was therefore not necessarily required.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedure(s) undertaken (either prospective or retrospective studies). No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
