Abstract
Uterine infarctions have not been reported in domestic animals, and there are few reports in the human medical literature. In a retrospective study, uterine infarctions were identified in 9 of 323 (2.8%) female cynomolgus monkeys (Macaca fascicularis) necropsied over a 13-year period. The infarctions were grossly visible, after fixation, on the serosal surface of the uterus in 2 monkeys; the remainder were first recognized in histologic sections. Histologically, the lesions consisted of well-demarcated regions of endometrial and myometrial necrosis and of hemorrhage. All affected monkeys had histologic evidence of a previous pregnancy, which included enlarged myometrial vessels with an expanded perivascular matrix. In all monkeys with uterine infarctions, there was clinical evidence of severe systemic illness, which included trauma, diarrhea, hypovolemia, or septicemia. The major pathologic findings in affected monkeys included cutaneous or skeletal muscle necrosis (n = 5), enterocolitis (n = 4), pulmonary edema or diffuse alveolar damage (n = 3), and intestinal amyloidosis (n = 1). Histopathologic evidence of intravascular fibrin thrombi in multiple organs of 5 monkeys was consistent with a diagnosis of disseminated intravascular coagulopathy (DIC). Based on these findings, it appears that uterine infarction is an uncommon finding in cynomolgus monkeys and may occur secondary to a severe systemic illness, predisposing to DIC.
Uterine infarctions are very uncommon in humans, and a literature search failed to identify a previous report in any animal species. Four reports describe uterine infarction in women: one because of an incomplete prolapse; one because of uterine torsion; one associated with endotoxic shock; and one because of poorly controlled diabetes mellitus, resulting in progressive atherosclerotic disease of the uterine vessels.7,9,12,13 We identified 9 cynomolgus monkeys (Macaca fascicularis) with uterine infarctions in a retrospective analysis of tissues and records from 323 animals over 4 years of age at the time of necropsy and those necropsied during a 13-year period. The purposes of the present study were to document the morphology of uterine infarctions and the frequency of their occurrence, and to describe the clinical and pathologic findings associated with these lesions in cynomolgus monkeys.
Materials and Methods
Necropsy reports on adult female Indonesian cynomolgus monkeys between November 1993 and March 2005 were reviewed (n = 323). The monkeys were subjects in comparative clinical studies at the Comparative Medicine Clinical Research Center (CMCRC) of the Wake Forest University School of Medicine. All animals were housed in social groups of 3 to 6 monkeys, in an effort to provide for environmental enrichment and socialization, in a facility accredited by the Association for the Advancement and Accreditation of Laboratory Animal Care, in experimental protocols approved by the Institutional Animal Care and Use Committee. All animals died or were euthanatized because of clinical illness, and diagnostic necropsies completed included gross and histologic examination of all major organs, including the uterus. Tissues were fixed in 10% neutral buffered formalin and were processed by standard methods, and sections were stained with hematoxylin and eosin. Selected slides were stained with phosphotungstic acid hematoxylin (PTAH) and Masson stains.
Uterine infarctions were defined histologically as sharply demarcated areas of endometrial and/or myometrial necrosis, with no evidence of metritis. These were visible grossly in 2 animals, after fixation in 10% neutral buffered formalin, as sharply demarcated pale, red, or brown areas on the serosal and/or cut surfaces of the uterus; however, gross visibility of the lesion was not a criterion for inclusion in this report.
Results
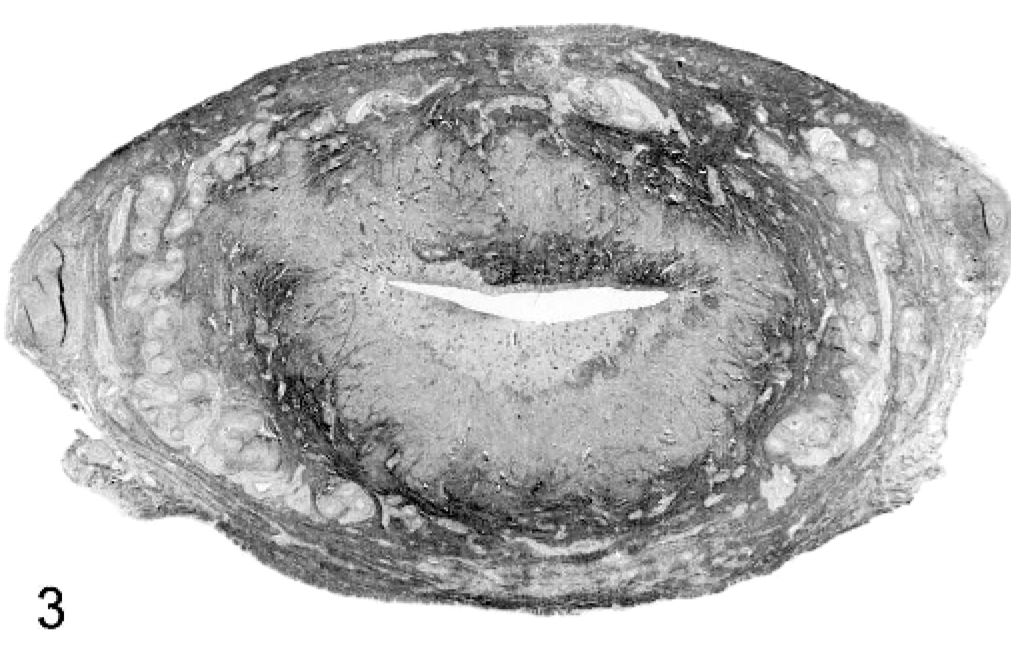
Nine of the 323 adult female monkeys that were necropsied in the 13-year period were identified as having a uterine infarction (Table 1). Three of these monkeys had been born at the CMCRC and had known birth dates; the other 6 had been imported from Indonesia between 2 and 6 years before death. The ages of these latter monkeys were estimated by dentition, by using criteria adapted from those of Bowen and Koch.5 The mean age of the 9 affected monkeys was 10.9 years (range, 7–20 years).
Signalment, clinical, and postmortem features of monkeys with uterine infarctions.
Wake Forest University School of Medicine.
Four of the 9 monkeys with infarctions were known to have had multiple live births, with the period between the last parturition and the diagnosis of the uterine infarction ranging from 1 to 22 months (Table 1). Although the remaining 5 monkeys had no known history of pregnancy, histologic examination revealed enlarged myometrial vessels, with expanded perivascular matrix in all monkeys, indicating that they had experienced a pregnancy.6 Three monkeys had intact ovaries, and 6 monkeys had been ovariectomized for experimental protocols (Table 1).
Uterine infarctions were observed in the context of severe systemic illness. The most common clinical illnesses were severe diarrhea (n = 5; Table 1), with accompanying lethargy and signs referable to hypovolemic shock (dehydration, tachycardia), or severe trauma inflicted by other monkeys because of cage-mate aggression, an occasional and unfortunate consequence of social housing. Limited clinical pathologic data were available from 5 animals. Results were variable and included anemia (4/5), azotemia (4/5), mature neutrophilia (3/5), and hyperproteinemia (2/5). Fibrin degradation products were not measured. Necropsy findings generally reflected the major clinical illness. Lesions associated with diarrhea included enteritis and colitis, often with colonic crypt hyperplasia. In the monkeys that suffered traumatic injuries, necropsy findings included puncture wounds, lacerations, and contusions. Fibrinous thromboemboli were identified in multiple organs of 5 of 9 monkeys examined during initial histologic evaluation. Three monkeys had pulmonary edema or diffuse alveolar injury, and 3 monkeys had hepatic lipidosis. Other lesions that occurred in individual animals included urocystitis, intestinal and splenic amyloidosis, acute tubulointerstitial nephritis, and acute nephrosis (Table 1).
Uterine infarction was recognized grossly, after formalin fixation, in 2 monkeys. The first had paired, 5 × 10-mm, ovoid, circumscribed red–brown foci on the dorsal and ventral serosal uterine surfaces, which, on section, extended as wedge-shaped red regions from the serosa to the uterine lumen, which created an hourglass shape. The second had a 10 × 12-mm, dark, irregular focus on the anterior serosal surface. Sectioning of the uterus revealed dark-brown endometrium.
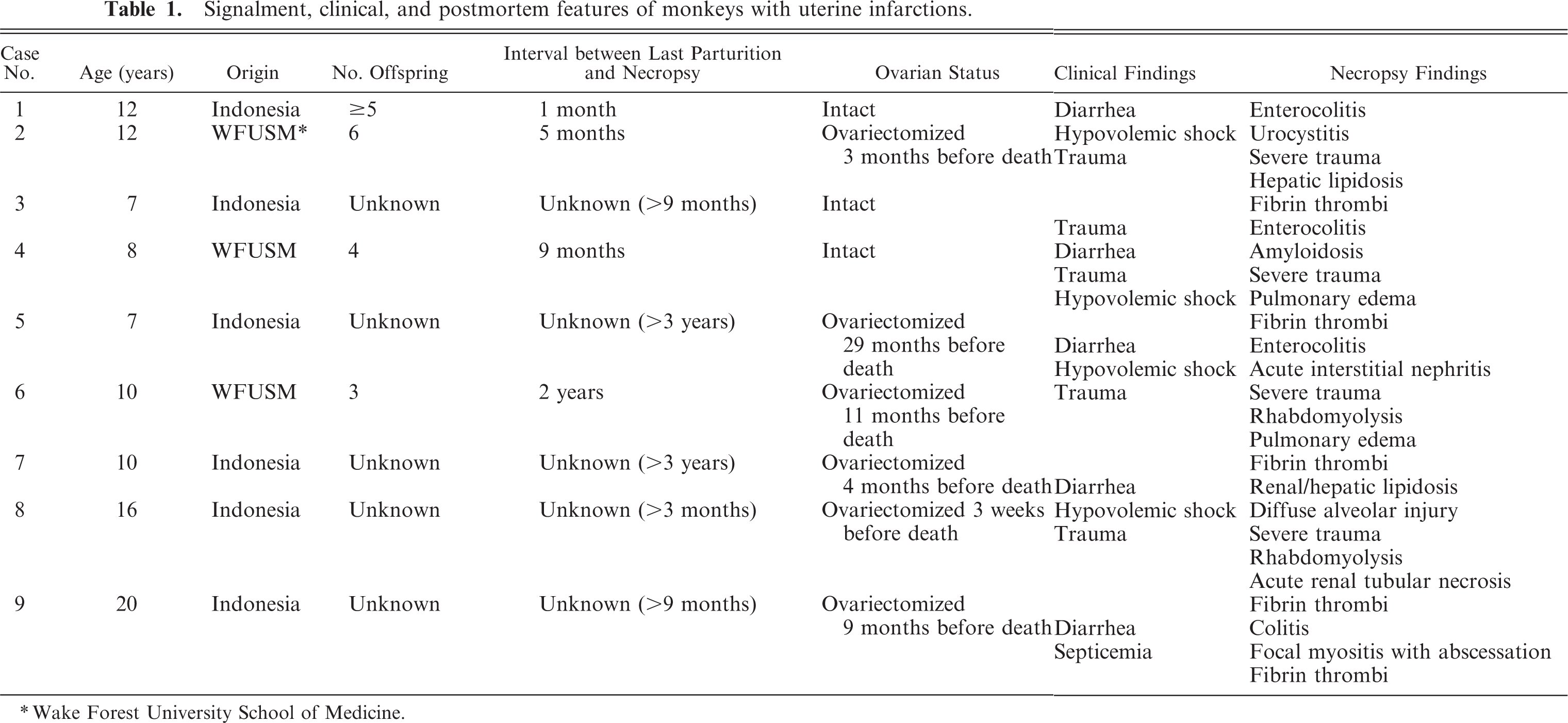
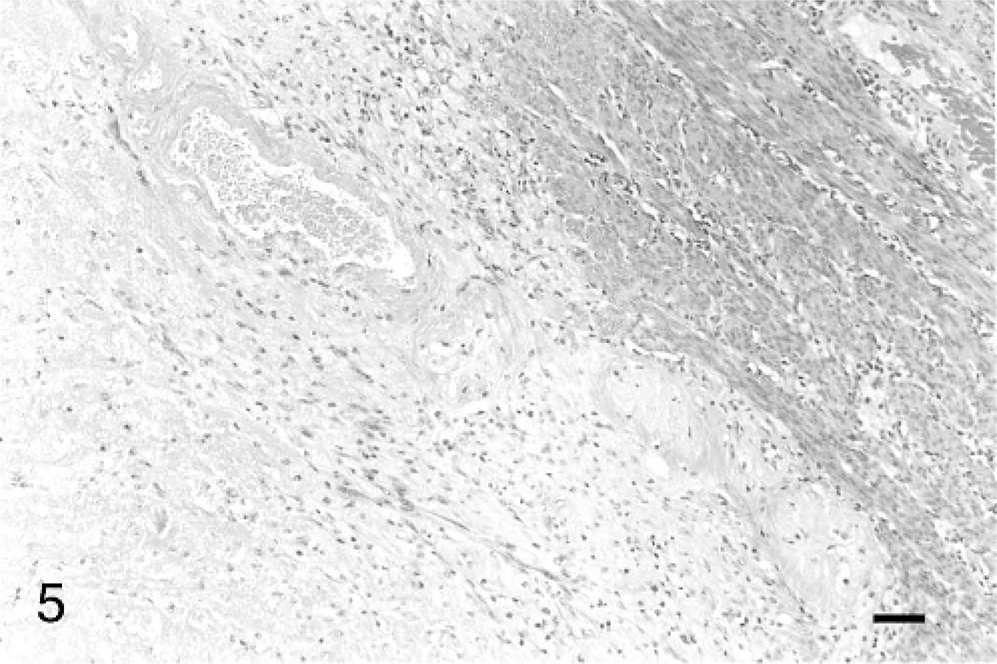
Histologically, the uterine infarctions involved both the endometrium and the myometrium, and were characterized by well-demarcated regions that were pale eosinophilic and either lacked nuclei or had pyknotic nuclei. Patterns of necrosis are illustrated in Figs. 1 –4. The extent of involvement of the endometrium and the myometrium varied among monkeys, and ranged from approximately 25 to 50% of the area of sections examined. The infarctions were generally bilaterally symmetrical and were centered on the midline of the uterus; they most frequently involved the inner myometrium and endometrium. In some cases, there was sparing of periarterial tissue within the myometrium (Fig. 4). There was hemorrhage and fibrin deposition, with little to no inflammatory response, along the margins of the infarctions (Fig. 5). There were vessels within the necrotic region that exhibited fibrinoid degeneration within their walls in 3 of 9 uteri examined (Fig. 5). Several of the vessels along the margins of the infarctions appeared to contain fibrin thrombi, but the majority of these thrombi were PTAH negative. All of the lesions appeared to be relatively acute, with little evidence of chronic inflammation or fibroplasia, as evaluated by Masson stain.
Uterus; macaque. Margin of the myometrial portion of the infarction, case 1. HE stain. Bar = 100 μm.
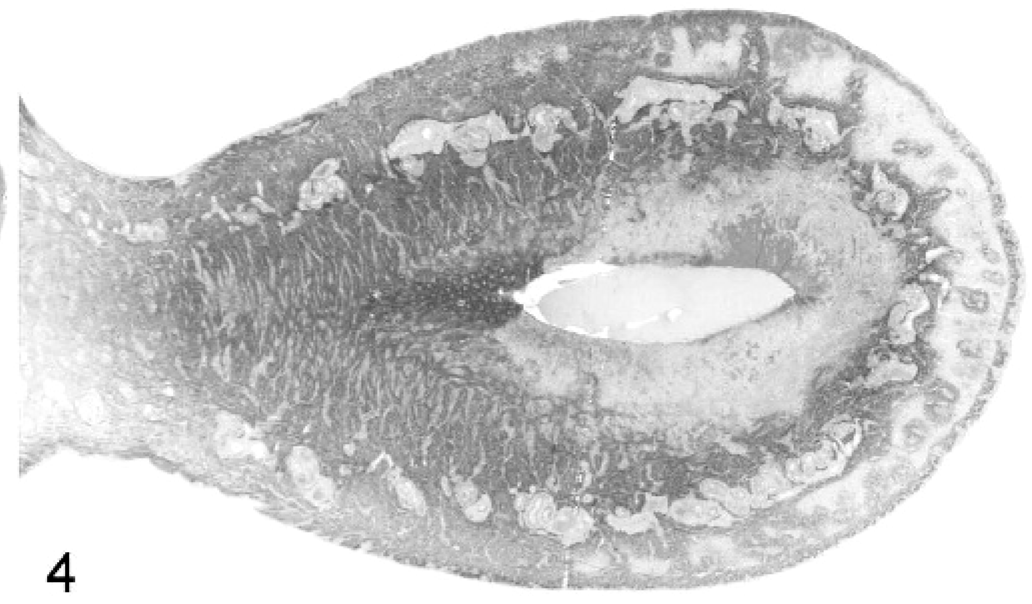
Uterus; macaque. Sagittal section of the uterus from case 5, showing necrosis of the fundic endometrium and myometrium with sparing of perivascular tissues in the mid-myometrium. HE stain. Bar = 5 mm.
Uterus; macaque. Transverse sections of uteri from case 8, showing pale regions that represent endometrial and myometrial necrosis. HE stain. Bar = 5 mm.

Uterus; macaque. Transverse sections of uteri from case 4, showing pale regions that represent endometrial and myometrial necrosis. HE stain. Bar = 5 mm.
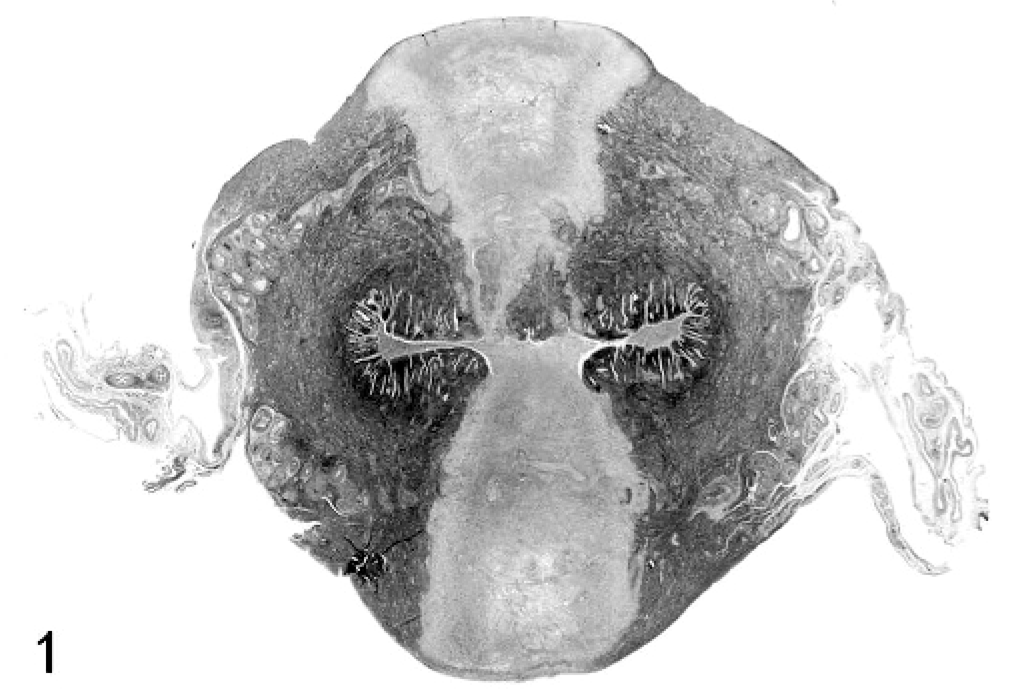
Uterus; macaque. Transverse sections of uteri from case 1, showing pale regions that represent endometrial and myometrial necrosis. HE stain. Bar = 5 mm.
Discussion
Our findings indicate that uterine infarctions are an uncommon finding in cynomolgus monkeys and appear to be restricted to parous animals. In the present study, 9 monkeys were found to have locally extensive uterine infarction during routine diagnostic necropsy, which represented 2.8% of all adult females examined for diagnostic necropsy during the 13-year period. There was clinical evidence of severe systemic illness in all the monkeys that had uterine infarctions, with histopathologic evidence of intravascular fibrin thrombi in multiple organs of 5 monkeys. Although confirmatory clinical pathologic data were not available, the histopathologic findings were consistent with a diagnosis of disseminated intravascular coagulation (DIC) in these 5 animals. Clinical signs were nonspecific (depression, diarrhea, dehydration) and were interpreted to be because of the primary disease process (e.g., enteritis). No specific clinical signs could be attributed to the uterine infarctions. Similarly, clinicopathologic changes also appeared to reflect the underlying disease processes rather than being specifically indicative of the uterine lesions. Major lesions associated with uterine infarctions included severe trauma that involved skin, subcutaneous fat, and skeletal muscle (5/9), severe enterocolitis (5/9), and pulmonary edema and/or diffuse alveolar injury (3/9).
To our knowledge, uterine infarctions have not been reported in nonhuman primates and are not commonly recognized in humans. Leiomyomas appear to be the most common underlying factor in human cases. Uterine infarction may occur either because of degeneration and necrosis of the tumor or secondary to tumor-associated uterine torsion.7 Although leiomyomas were described in uteri of macaques, associated uterine infarction has not been reported.3 Leiomyomas were not seen in any of the monkeys in this report. Iatrogenic uterine infarction also was reported to occur after caesarian section in humans,11 and there is 1 human case that occurred postpartum because of an incomplete prolapse of the corpus, with subsequent compression of the uterine blood supply and resulting infarction.12 Uterine infarction also was reported to occur in association with endotoxic shock in a human female.9 Degeneration and necrosis of a large fibroid (leiomyoma) led to a syndrome similar to endotoxic shock. Finally, in 1 human case, uterine infarction and subsequent rupture were reported to be the result of progressive atherosclerotic disease associated with poorly controlled diabetes mellitus.13 As far as we are aware, there are no reports of uterine infarction in humans that occurred secondary to hypovolemic shock or disseminated intravascular coagulopathy, as appears to be true in the monkey cases described in the present report.
Although previous pregnancies had been documented in only 4 of the 9 monkeys, we believe that all 9 had been previously pregnant based on the presence of blood-vessel enlargement and altered perivascular matrix within the walls of the uterine arteries and veins.6 Physiologic alterations are known to occur in the uterine vessels of primates during pregnancy. These changes in the arterial wall have a fibrinoid appearance histologically and result in a low-resistance, high-conductance blood supply to the placenta.4 This vascular alteration occurs as the trophoblasts migrate and invade the uterine arteries and veins, and subsequently modify the matrix, physiologically altering the uterine blood flow for the pregnancy.1,2 This altered appearance of the uterine vessels was present in all 9 monkeys described in this report. An examination of a series of monkey uteri with known reproductive histories indicated that the matrix alterations in the uterine vessels persist for at least 1 year after parturition.6 These changes also occur in humans, and a study of placental site involution in humans suggests that the remaining hyaline change in these vessels is permanent.1 Uterine infarctions were not detected in nulliparous monkeys at the CMCRC. All cases of uterine infarction were found in parous animals, thus the relationship between uterine infarction and pregnancy suggests that prior pregnancy may predispose the uterus to infarction. In monkeys with a known reproductive history, the length of time between parturition and diagnosis of the uterine infarction was variable. A common postpartum complication in humans is hemorrhage because of failure of normal placental-site involution;10 however, this has not been described in nonhuman primates.
The stomach has been described as a preferential site of thrombosis in conditions that cause DIC in the cynomolgus monkey.8 Gastric thrombosis was not observed in any of the cynomolgus monkeys in this report. The pathogenesis of the uterine infarction remains unknown; however, past pregnancy appeared to be a predisposing factor. The matrix alteration in the vessel walls may predispose this site to injury and subsequent thrombosis in situations such as endotoxemia or severe hypovolemic shock.
In summary, uterine infarction was detected in 9 monkeys with severe systemic illnesses or trauma, with evidence of DIC in 5 of the 9 monkeys. The uterine vessels in postpartum females may be a previously unrecognized site for thrombosis because of DIC in the cynomolgus monkey.
