Abstract
Practical relevance:
Abdominal ultrasound plays a vital role in the diagnostic work-up of many cats presenting to general and specialist practitioners. B-mode ultrasonography is likely the most widely used modality for imaging the gastrointestinal (GI) tract in cats, providing information on intestinal wall thickness and the presence or absence of intestinal dilation and peristalsis, among other factors.
Clinical challenges:
Despite ultrasonography being a commonly used modality, many practitioners are not comfortable performing an ultrasound examination or interpreting the resulting images. Even differentiating between normal variations and pathological changes can be challenging for all but the most experienced.
Aim:
This review, part of an occasional series on feline abdominal ultrasonography, discusses the ultrasonographic examination and appearance of the normal GI tract; the diseased GI tract is addressed in an accompanying article in this issue of JFMS. Aimed at general practitioners who wish to improve their knowledge and confidence in feline abdominal ultrasound, this review describes where findings contrast with those typically seen in dogs. It is accompanied by high-resolution images and videos available online as supplementary material.
Equipment:
Ultrasound facilities are readily available to most practitioners, although use of ultrasonography as a diagnostic tool is highly dependent on operator experience.
Evidence base:
Information provided in this article is drawn from the published literature and the author’s own clinical experience.
Scanning the gastrointestinal tract
B-mode ultrasonography is probably the most widely used modality for imaging the feline gastrointestinal (GI) tract. It provides information regarding overall intestinal wall thickness, and the echogenicity and distinctness of individual layers, and permits assessment of luminal content including the presence or absence of intestinal dilation and peristalsis. The adjacent peritoneum and regional lymph nodes can also be evaluated simultaneously. Ultrasound facilities are usually readily available to most practitioners, although the main drawback of ultrasonography is that its use as a diagnostic tool is highly dependent on operator experience.
Where possible, it is usually preferable to fast animals waiting for an elective examination for 12 h prior to ultrasonography, to minimise the volume of ingesta and gas within the GI tract. Although, as a rule, cats often have less intestinal gas than dogs, fasting is nevertheless important because ultrasound cannot penetrate gas and hence structures deep to intestinal gas can become obscured. Water, on the other hand, should not be withheld and can actually improve organ visibility by providing an acoustic window through which to scan.
Some cats may be scanned while conscious and this is preferable if the main purpose of the scan is to assess the degree of intestinal peristalsis present. Notwithstanding, sedation is desirable in most instances since it helps to relax the abdominal wall muscles and immobilise the patient. A drug that minimally affects GI motility, such as acepromazine, can be considered if sedation is necessary yet assessment of peristalsis is important. Preparation prior to scanning involves clipping of the abdominal hair followed by the application of acoustic coupling gel to remove the air gap between the transducer surface and the skin.


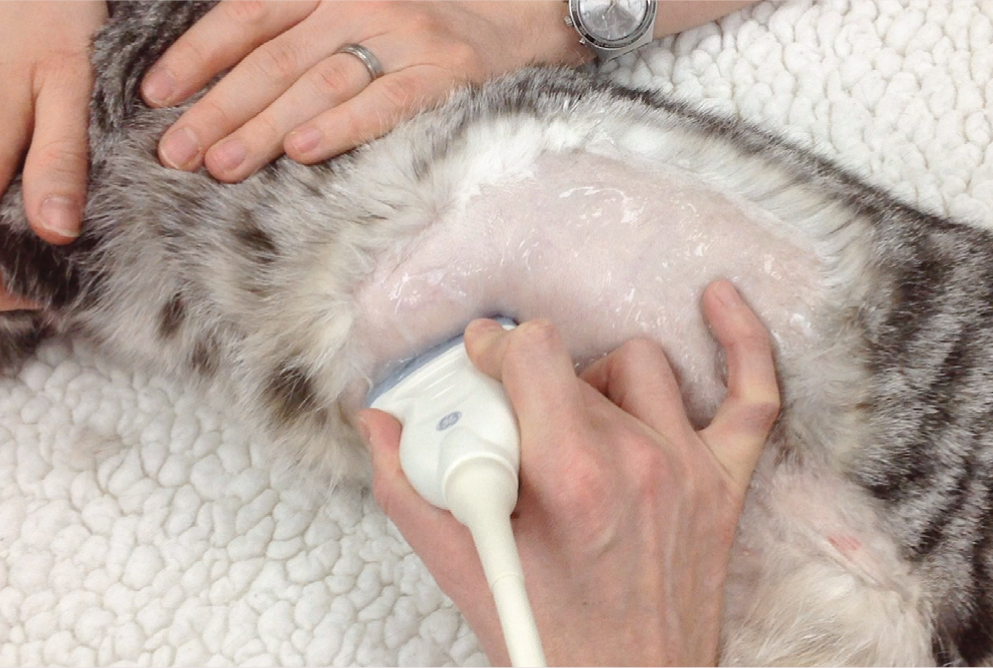
A high frequency linear transducer is preferable for assessing the feline gastrointestinal tract
Normal anatomy
The feline stomach is located immediately caudal to the liver in the cranial abdomen. In contrast to the long axis of the canine stomach, which is orientated roughly perpendicularly to the spine when viewed from ventrally, the long axis of the feline stomach is orientated almost parallel to the spine and, for this reason, is often described as being ‘J-shaped’ on ventrodorsal radiographs (Figure 2). The curved portion of the ‘J’ is formed from the pyloric antrum, which travels a short distance cranially before reaching the pyloroduodenal junction. The feline pylorus and descending limb of the duodenum are positioned more closely to the midline than in the dog. 1 The descending limb of the duodenum follows a relatively straight course caudally before entering the caudal duodenal flexure and turning cranially to form a short ascending limb that rapidly becomes the jejunum.

Normal orientation of the feline stomach as seen on a cropped dorsoventral radiograph – unlabelled (a) and labelled (b). F = fundus; B = body; P = pylorus
Scanning technique
There are different techniques for scanning the GI tract and those described here are the ones used by the author. Complete assessment of the stomach requires the use of both longitudinal and transverse scanning planes. The transducer is initially placed along the mid-line at the xiphisternum and orientated longitudinally relative to the long axis of the patient. The fundus is identified to the left of the midline and is followed to the body and then the pylorus before rotating the transducer through 90° to enable the ultrasonographer to scan the stomach in the transverse plane. As previously mentioned, cats tend to have very little GI gas and thus it is often possible to examine the entire gastric wall without difficulty.
The procedure for scanning the small intestine can be likened to the method by which slides are examined under a microscope. To begin with, the animal is positioned in right lateral recumbency (author’s preference) and the transducer is orientated longitudinally relative to the patient and placed on the craniodorsal abdomen. While keeping the orientation of the transducer consistent, the transducer is moved along the flank in a ventral direction using a sliding action along the skin surface. Without losing contact with the surface of the skin, the transducer is repositioned slightly caudally before being slid dorsally, and so on (Figure 3a). Next, the transducer is rotated through 90° and the process is repeated, this time with the transducer moving back and forth in a craniocaudal direction (Figure 3b). Thus, on completion, the small intestine will have been examined in two orthogonal planes. Jejunal loops are usually distributed fairly evenly within the abdomen, although in cats with a large amount of intra-abdominal fat, the intestines can become clumped together within one area (often the right side).

Arrows show the normal direction of travel of the transducer when performing a survey of the small intestine. (a) The transducer is initially orientated longitudinally to the patient and moved in a dorsoventral direction. (b) The transducer is then rotated through 90° and moved back and forth in a craniocaudal direction
The easiest way to assess the large intestine is to locate the descending limb of the colon and follow it orally to the ileocaecocolic junction (ICCJ). An image of the urinary bladder is obtained in the longitudinal plane and, from there, the transducer is fanned slowly dorsally; in doing so, the descending colon should appear on the screen. The descending colon is relatively straight and, with practice, can be followed without too much difficulty cranially to the transverse and ascending portions of the colon and then finally to the ICCJ. The ICCJ lies to the right of the midline in the mid-abdomen. It may also be located more directly by obtaining a sagittal image of the right kidney and then sliding the transducer medially.
Normal appearance of the gastrointestinal tract
Stomach
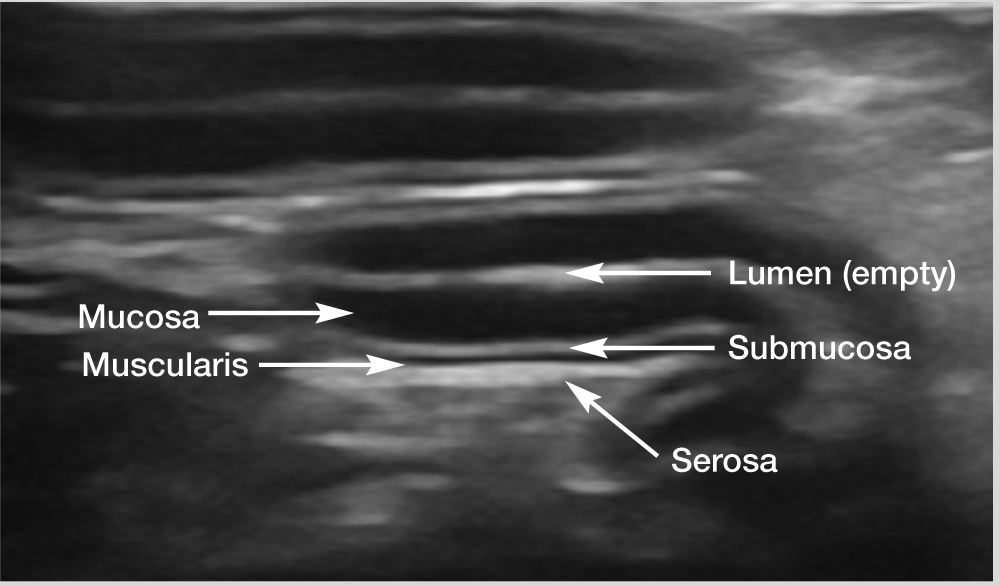
When empty, the normal feline stomach has an appearance that is often likened to that of a wagon wheel, owing to the prominent rugal folds of the fundus that project into the lumen, thereby mimicking the spokes of a wheel (Figure 4). Five distinct layers may be distinguished within the gastric wall with ultrasound. The order and appearance of these layers is repeated throughout the intestinal tract (see box), with some minor differences, which are described below.

(a,b) Normal ultrasonographic appearance of the empty gastric fundus in two cats. Note the prominent rugal folds projecting into the lumen, giving rise to the characteristic ‘wagon wheel’ appearance. In (a), measuring calipers indicate the inter-rugal fold thickness of the gastric wall. A video showing the ultrasonographic (ie, classic wagon wheel) appearance of the normal empty gastric fundus in a cat is available as supplementary material
The mucosa has been reported to be consistently the thickest layer regardless of location within the feline GI tract, 2 although a second study found that the mucosa was thickest only in the duodenum and jejunum. 3 Discrepancies in methodology may account for the differing results between the two studies, although the findings of the second study are more consistent with the experience of the author. The muscularis layer has been reported to be of equal or greater thickness than the submucosal layer in the stomach, duodenum, jejunum and ileum in a study of 38 healthy cats 2 (although no histopathology was performed to prove normality of the intestine), and it naturally thickens at the pylorus to form the pyloric sphincter. 4 In the author’s experience, the muscularis can also be thinner than the adjacent submucosa in cats without clinical signs of GI tract disease and, therefore, there is a range of normal variation. A peculiarity of the gastric submucosa is the presence of a layer of hyperechoic fat that creates a stripy echogenic appearance; this may also be visible on radiographs as an intramural radiolucent band within the gastric wall. 5 The typical inter-rugal gastric wall thickness in cats is around 2 mm and the rugal fold thickness has been reported to be 4.4 mm. 6


Ultrasound image of a jejunal loop, illustrating the order of the wall layers within the gastrointestinal tract
Duodenum
As mentioned above, the pylorus and descending limb of the duodenum are positioned more closely to the midline in the cat than in the dog, and the pyloroduodenal junction forms a focal narrowing within the GI tract (Figure 6). The major duodenal papilla is more prominent in the cat than in the dog and, with practice, can be identified as a focal soft tissue bulge in the wall of the proximal descending duodenum. 7 The papilla is reported to range from 2.9–5.5 mm in width and to have a maximum diameter of 4.0 mm in the transverse plane. 8 In cats, the distal portions of the common bile and pancreatic ducts merge before entering the duodenum at the duodenal papilla. 9 This is in contrast to the dog where the point of entry of the accessory pancreatic duct into the duodenum, at the minor duodenal papilla, is not commonly seen on ultrasound. 9

Normal appearance of the pyloroduodenal junction. The pylorus, which contains a small amount of ingesta, is on the right side of the image and the empty duodenum is on the left
In cats, duodenal wall thickness has been reported to vary from approximately 2–4 mm and can be slightly thicker than the jejunal wall, which measures around 2–3 mm in width.6,8 In the author’s experience, however, and in keeping with other references including a more recent publication on the subject, wall thickness in normal cats tends to be towards the lower end of these ranges (around 1.8–2.7 mm; average 2.2 mm for both the duodenum and jejunum).3,10 Small intestinal wall layering is as described earlier, with the mucosa being the thickest layer in both the duodenum and jejunum (Figure 7a), where it can contribute more than half of the overall thickness of the intestinal wall. 3
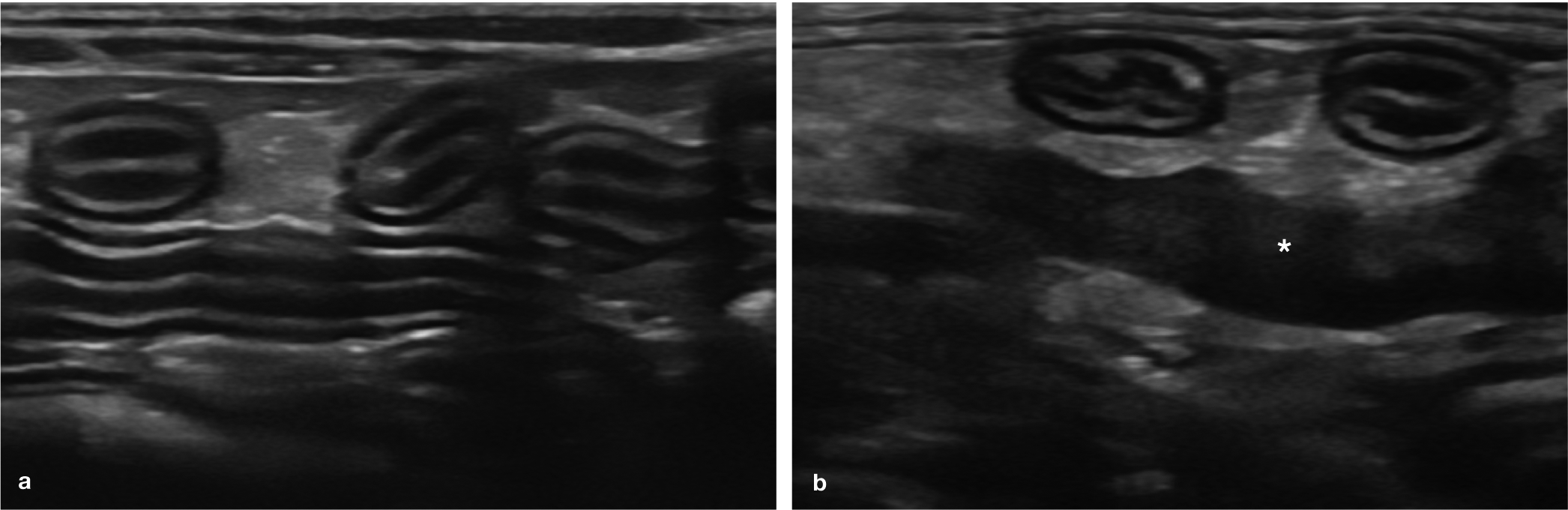
Normal ultrasonographic appearance of the jejunum and the ileum. (a) Several loops of normal jejunum are visible in the near field and mid-field of the image. Note the thick anechoic mucosal layer that is characteristic of both the duodenal and jejunal wall. A video showing the ultrasonographic appearance of normal feline jejunal loops is available as supplementary material. (b) A loop of ileum in transverse orientation (left) is visible in the near field of the image, adjacent to a loop of jejunum (right), also in transverse orientation. Note the thick hyperechoic submucosa of the ileum, allowing it to be differentiated from jejunum. The solid, relatively hypoechoic structure in the mid-field of the image is a slightly enlarged jejunal lymph node (asterisk)
Ileum
The ileum is easily recognised and distinguished from the jejunum by a prominent hyperechoic submucosal layer and a prominent anechoic muscularis layer (Figure 7b). 3 The latter may be explained by the important sphincter-like role played by the ileum in preventing colonic material from passing orally back into the small intestine. 11 It is also the thickest intestinal segment, with an average thickness of 3 mm. 3 In a similar fashion to the rugal folds of the stomach, the folds of an empty terminal ileum project into the lumen on cross-sectional images and, as such, are also sometimes described as resembling the spokes of a wagon wheel. 10 It may be easier to obtain measurements of the ileum with the wall in the transverse rather than longitudinal plane, owing to potential loss of distinction of wall layering that can result from the presence of these folds in the longitudinal plane. 3 Ileal wall thickness across folds is in the range of 2.5–3.6 mm and between folds is much lower, at around 1.7–2.3 mm. 3
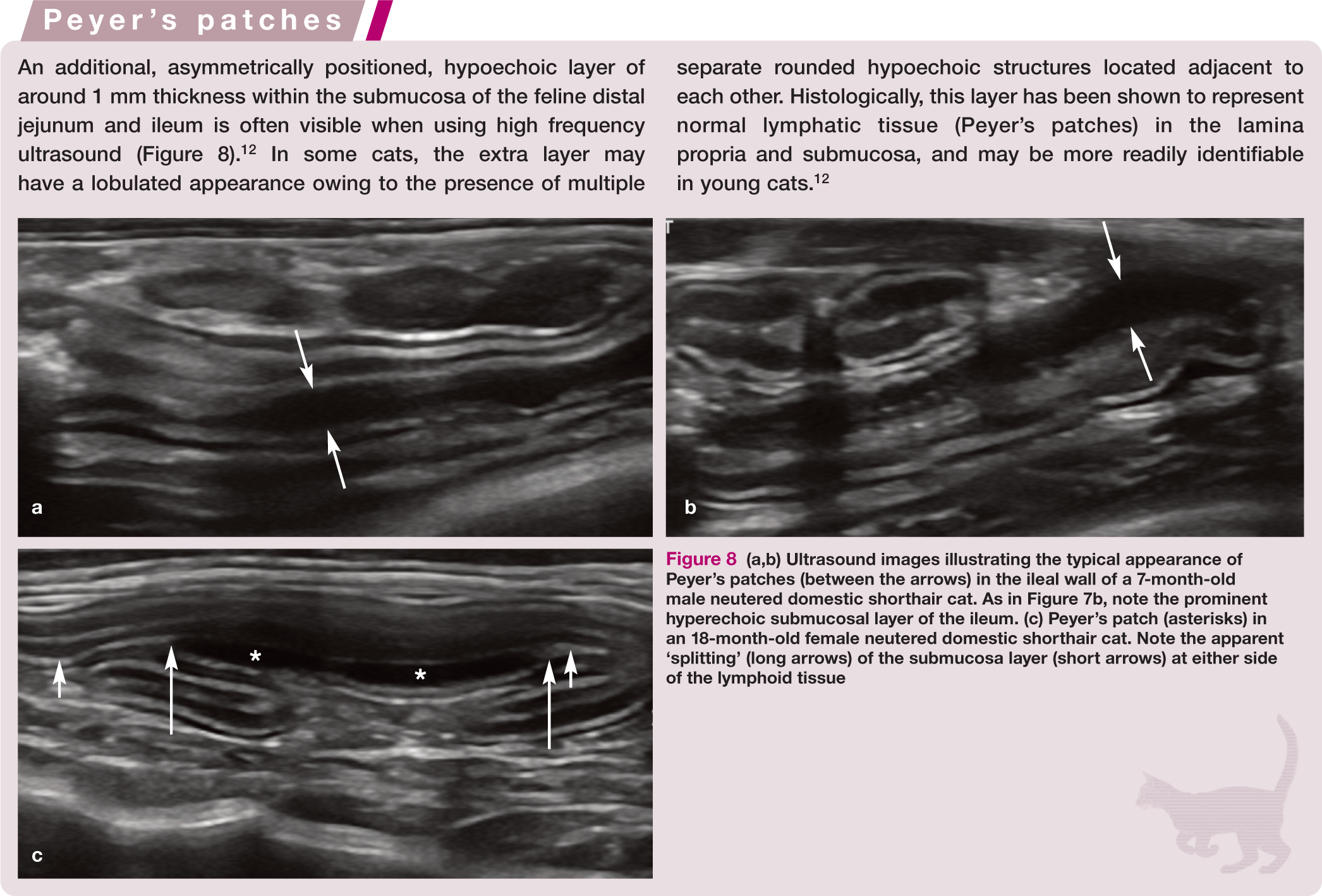

(a,b) Ultrasound images illustrating the typical appearance of Peyer’s patches (between the arrows) in the ileal wall of a 7-month-old male neutered domestic shorthair cat. As in Figure 7b, note the prominent hyperechoic submucosal layer of the ileum. (c) Peyer’s patch (asterisks) in an 18-month-old female neutered domestic shorthair cat. Note the apparent ‘splitting’ (long arrows) of the submucosa layer (short arrows) at either side of the lymphoid tissue
Once identified, the ileum can be traced to the ICCJ, which usually lies to the right of the midline, ventromedial to the right kidney. In some cats, the ICCJ can be located more cranially, close to or underneath the 13th rib. 13 The ileum enters the ascending colon from the left side, just distal to the caecocolic junction. 14 The ileocolic sphincter results in a focal intestinal constriction (Figure 9) and the ileocolic papilla bulges distally into the colon. 15
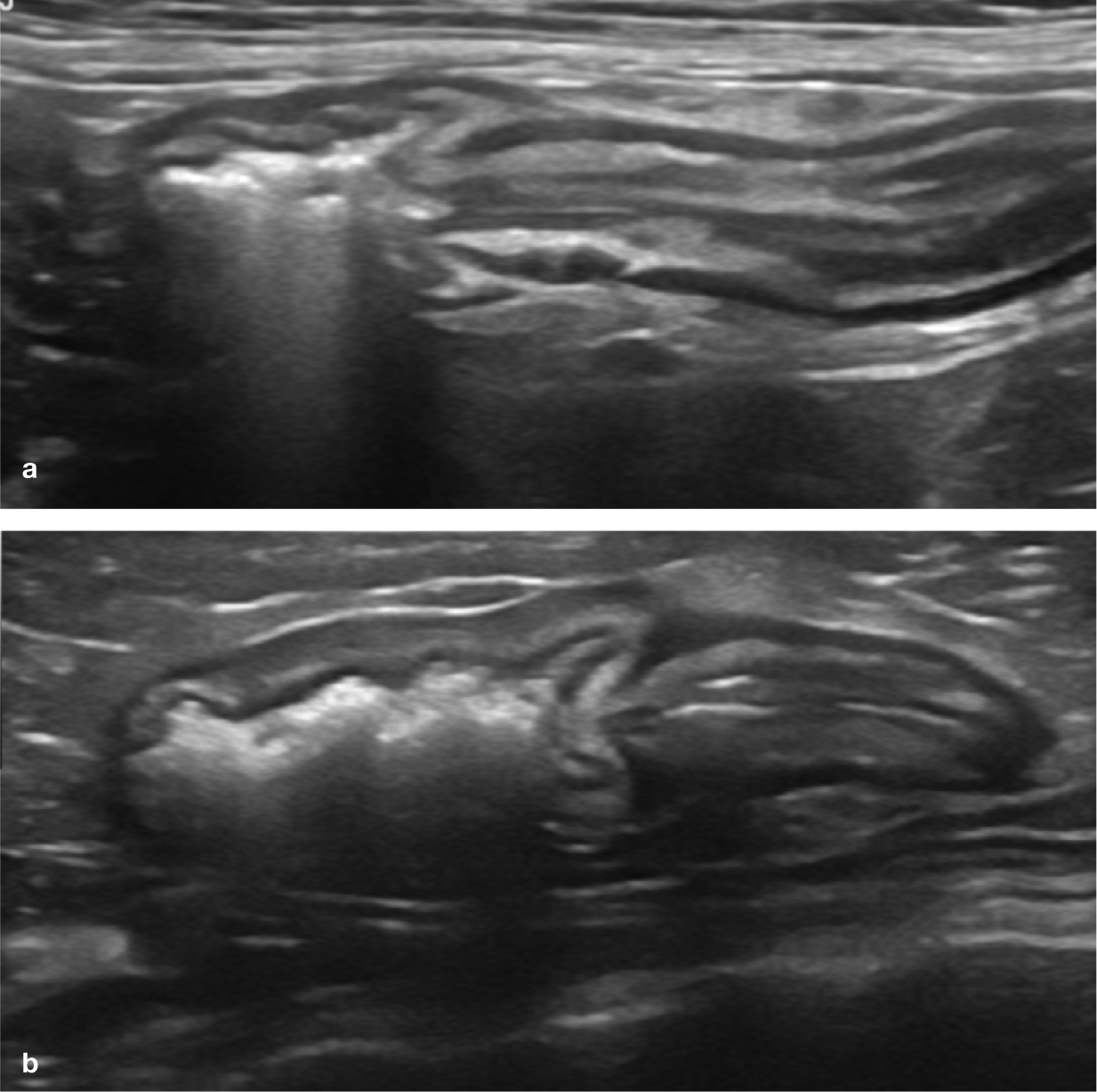
(a,b) Ultrasound images of the ileocolic junction; the ileum is to the right and the colon is to the left. The natural narrowing between them represents the ileocolic sphincter. In (b), the colonic wall is mildly thickened due to inflammatory disease. Videos showing the ultrasonographic appearance of the ileocolic junction are available as supplementary material
Caecum
Despite being smaller than the canine caecum, the feline caecum is more consistently recognised on ultrasound as it is often either empty or contains only a small volume of gas.13,16,17 It is identified as a comma-shaped, blind-ending pouch measuring approximately 2–3 cm in length. 15 The distal cul-de-sac portion of the caecum is somewhat thicker (mean 3.1 mm; range 2.4–4.1 mm) than the more proximal portion (mean 2.5 mm; range 1.7–3.6 mm). 13 Ultrasonographically identifiable wall layering within the caecum comprises the mucosa, submucosa and muscularis, which are reported to be of similar thickness, and a serosa, although the wall of the thicker distal portion of the caecum may appear hypoechoic with an apparent lack of distinct layering if a relatively low frequency transducer is used.13,15
A hypoechoic nodular layer arising from the submucosa, which also overlaps the mucosa, may be visible within all portions of the feline caecum and corresponds to the presence of multiple coalescing lymphoid follicles (Figure 10). 13 In a small study population of 20 asymptomatic cats, this follicular layer had a mean thickness of 1.8 mm (range 1.3–2.9 mm) in the proximal part of the caecum and 2.4 mm (range 1.5–3.2 mm) in the distal part. 13

(a,b) Normal ultrasonographic appearance of the caecum in two cats. The hypoechoic regions (arrows) within each caecal wall correspond to the multiple coalescing lymphoid follicles within the mucosa and submucosa
The junction between the feline caecum and the colon is large since there is no sphincter present between the two. 15 Several lymph nodes, known as the ileocaecal lymph nodes, may be visible at this location and are situated within a fold of peritoneum, the ligamentum ileocaecalis, positioned between the ileum and the caecum. 18 The ascending mesocolon also contains between one and five colonic lymph nodes. 14

Colon
The ultrasonographic appearance of the feline colon mirrors that of the dog and may be distinguished from the small intestine by its much thinner wall, measuring around 1–2.5 mm (average 1.5 mm) in width (Figure 11).6,10 Unlike the small intestine, the colon is frequently gas-filled, which results in poor visibility of the far wall, although all portions of the wall may be appreciated when the lumen is empty or contains fluid.

Ultrasound image of the normal colon. The colonic wall (arrows) in the near field is much thinner than the wall of the small intestine. Acoustic shadowing distal to the colonic wall is common due to the presence of gas and/or faeces within the lumen. In such cases, the far wall of the colon is not seen. A video showing the ultrasonographic appearance of the colon is available as supplementary material
Key Points
Use of a high frequency transducer is important for distinguishing individual intestinal wall layers in cats.
The feline stomach is orientated differently to that of the dog and this should be taken into consideration when scanning.
As in the dog, the feline duodenum and jejunum are easily recognised by their thick mucosal layer.
In contrast, the ileum has prominent submucosal and muscularis layers and, in the transverse plane, ileal folds may resemble the spokes of a wagon wheel.
The feline caecum is small relative to that of the dog. Hypo- to anechoic lymphoid tissue may be visible in both the caecum and the ileum, and occasionally within the distal jejunum.
The colon has a distinctive thin wall allowing it to be differentiated from the small intestine, which has a comparatively thicker wall.

Footnotes
Conflict of interest
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
