Abstract
Objectives
The aim of this study was to assess the methodology of guaiac-based fecal occult blood (FOB) testing in healthy cats, and to examine the effect of feeding selected gastrointestinal diets (canned and dry) on FOB results.
Methods
Stool obtained from three cats was mixed with incremental quantities of EDTA whole blood. FOB tests were performed in triplicate and interpreted at 5 mins, and at 24, 48 and 72 h post-test set-up. Ten cats were fed their normal diets, a dry hydrolyzed protein diet, a dry gastrointestinal diet and then transitioned back to their normal diet over a 7-week period. A subsequent study was repeated with similar hydrolyzed protein and gastrointestinal canned diets. Two fecal samples were tested per cat each week using human point-of-care guaiac tests.
Results
Ten microliters of whole feline blood could be reliably detected in feline stool at all time points evaluated. There was no evidence of an association between the dry hydrolyzed and dry gastrointestinal diets and a positive FOB test result (P = 0.33). Cats fed a canned gastrointestinal diet had a significantly higher probability of producing a positive FOB result than when on their normal diet (P <0.01). Feeding the canned hydrolyzed diet was not associated with an increased probability of a positive FOB test vs the normal diet (P = 0.94).
Conclusions and relevance
Small amounts of blood in feline stool can be reliably and repeatedly detected as early as 5 mins post-FOB test set-up, making this a user-friendly bench-top screening test in a veterinary setting. Cats fed a single dry hydrolyzed, dry gastroenteric or canned hydrolyzed diet in this study had a low chance of false-positive FOB test results, while those fed the canned gastrointestinal diet had a significantly greater likelihood of producing false-positive results.
Introduction
Guaiac-based fecal occult blood (FOB) testing is used in people as a diagnostic aid to monitor patients for gastrointestinal bleeding, and as an early test for gastrointestinal disease and colorectal cancer screening.1–5 Other human stool-based occult blood tests include heme-porphyrin and fecal immunochemical tests (FITs). Commercially available and commonly used guaiac-based tests rely on the oxidation of guaiac acid by hydrogen peroxide catalyzed by the heme of hemoglobin, which has peroxidase activity.5,6 In the presence of heme in the stool, oxidation occurs within 60 s, resulting in a blue color change. In humans, several factors are known to interfere with FOB test results, including hydration of the stool sample, the administration of non-steroidal anti-inflammatory medications, and red meat and plant peroxidases in the diet.5,7 To avoid the interference of plant peroxidases in fruits and vegetables in the diet of individuals undergoing testing, the manufacturer of a common human FOB test kit (Hemoccult Single Slides; Beckman Coulter) recommends waiting to interpret the FOB test until 72 h post-test set-up.
Several studies have evaluated the use of these human point-of-care FOB test kits in dogs, with subsequent information extrapolated to cats.8–10 There is limited information on using human point-of-care FOB tests in our feline patients.11,12 Studies in dogs have evaluated the use of FOB testing with chronic kidney disease, and whether or not diet should be controlled for during initial FOB screening.8,9,13 To our knowledge, there have only been two studies published regarding feline FOB testing in the past two decades – neither evaluated the reliability of the FOB testing methodology.11,12
Early detection of gastrointestinal bleeding is important; however, causes of the bleeding vary according to the species studied. True-positive FOB results can be caused by varying etiologies based on patient species. Cats are unique in that they are even less likely to develop gastric bleeding as a result of kidney disease than dogs, and they are not typically on long-term non-steroidal anti-inflammatory drugs, unlike dogs and humans.14–16 However, cats are commonly afflicted with many gastrointestinal problems, which may result in microscopic gastrointestinal bleeding, including – but not limited to – inflammatory gastroenteropathies, gastrointestinal neoplasia, polyps, gastrointestinal foreign bodies and gastrointestinal infections.17–19 In fact, feline gastrointestinal disease may be quiescent for a period of time and blood may go unnoticed by a caregiver or veterinarian, especially as feline stool is often covered in cat litter. FOB testing for cats warrants further investigation as a potentially inexpensive, fast, non-invasive screening tool for a number of feline gastrointestinal pathologies.
The purpose of our study was to investigate FOB testing methodology and to determine if FOB test results are affected by selected veterinary-prescribed gastrointestinal diets (dry and canned) utilizing a common and commercially available guaiac FOB test kit.
Materials and methods
Ethical approval
This pilot study research protocol was approved by the Washington State University Institutional Animal Care and Use Committee (IACUC #04919-002 and IACUC #04919-006).
Stool samples for methodology study
The stool samples used for the detection of feline whole blood were obtained via free catch from the litter boxes of three healthy, privately owned indoor cats aged 1–5 years. The purpose of using three separate cat stools as the negative control was to allow for enough stool to be tested throughout the study. All cats were deemed healthy, with no history of systemic disease based on a physical examination and bloodwork (complete blood count and serum chemistry) and were dewormed (20 mg/kg by mouth once; pyrantel pamoate [Columbia Laboratories]), 1 week prior to the study. The cats were fed various dry commercial diets (Friskies Indoor Delight; Purina Pro-plan Focus Weight Management; and Purina Pro-plan Chicken and Rice [all Nestlé Purina]) for at least 1 month prior to commencement of the study. Two fresh (within 24 h) stool samples from various days from each of the three cats were kept at room temperature and tested in triplicate with a FOB test kit (Hemoccult Single Slides) to ensure consistency of negative stool results. Each sample day these stools were verified as negative at 5 mins, and at 24, 48 and 72 h by the same observer (KES).
Source of whole blood
Whole blood in EDTA used for this portion of the study was obtained from one healthy cat submitted to our veterinary teaching hospital’s clinical pathology laboratory that had enough blood in the EDTA tube for the duration of the study. The packed cell volume on the whole blood sample was 32%, hemoglobin was 11.1 g/dl and total protein was 6.6 g/dl.
Methodology testing procedure
Two grams of stool from each of the three cats were weighed on a gram scale. These quantities were then combined for a total of 6 g. Stool was mixed with a wooden tongue depressor in a small plastic container and then re-weighed. Incremental quantities (1, 2, 5, 10, 20, 50, 100 and 200 µl) of whole feline blood in EDTA from one cat were added and mixed into the stool samples (6 g) using a wooden tongue depressor. Incremental quantities of whole blood were applied directly to stool samples rather than fed to cats as a way of controlling for blood volume and assessing test sensitivity. This method does not reflect the effects of upper gastrointestinal bleeding but does mimic hematochezia.
A small amount of the stool mixture was applied to the guaiac paper test card using the provided wooden spatula. Each test card was labeled with the quantity of whole blood in the stool and the time when the test should be interpreted: at 5 mins, and at 24, 48 and 72 h post-test set-up. All tests were performed in triplicate for each blood volume measured and testing was performed by applying two drops of the provided hydrogen peroxide solution within the test kit to the guaiac paper test card as directed by the manufacturer (Hemoccult Single Slides). All tests were interpreted by the same observer (KES). The entire experiment was repeated using the same whole blood and the same cats’ stools in order to show reliability of the results.
Diet (dry and canned) study stool source
Ten clinically healthy, privately owned indoor cats aged 1–8 years enrolled and completed each 7-week portion of the study (dry and canned) (Files 1 and 2 in the supplementary material). The population of cats was not the same for each portion of the study due to owners’ availability and interest. Four of the cats were the same and participated in each aspect of the study. All cats were free of previous or evident disease and were not on any current (within 1 month) topical or systemic antibiotics or any other medications. A thorough history was obtained, and a physical examination, including body condition score, complete blood count, serum chemistry, urinalysis and deworming (pyrantel pamoate 20 mg/kg PO) were performed prior to enrollment (File 3 in the supplementary material). Each study (dry and canned diet trial) lasted 7 weeks (Figure 1). Stool for all enrolled cats was collected fresh (within 12 h) as free catch from the litter box by the owners. The submitted stool samples were obtained from day 2 to day 6 each week of the study and remained at room temperature. All stool samples received a fecal score (fecal scoring chart; Purina Pro Plan Veterinary Diets [Nestlé Purina]). All FOB tests were performed in triplicate and interpreted by a single observer (KES).
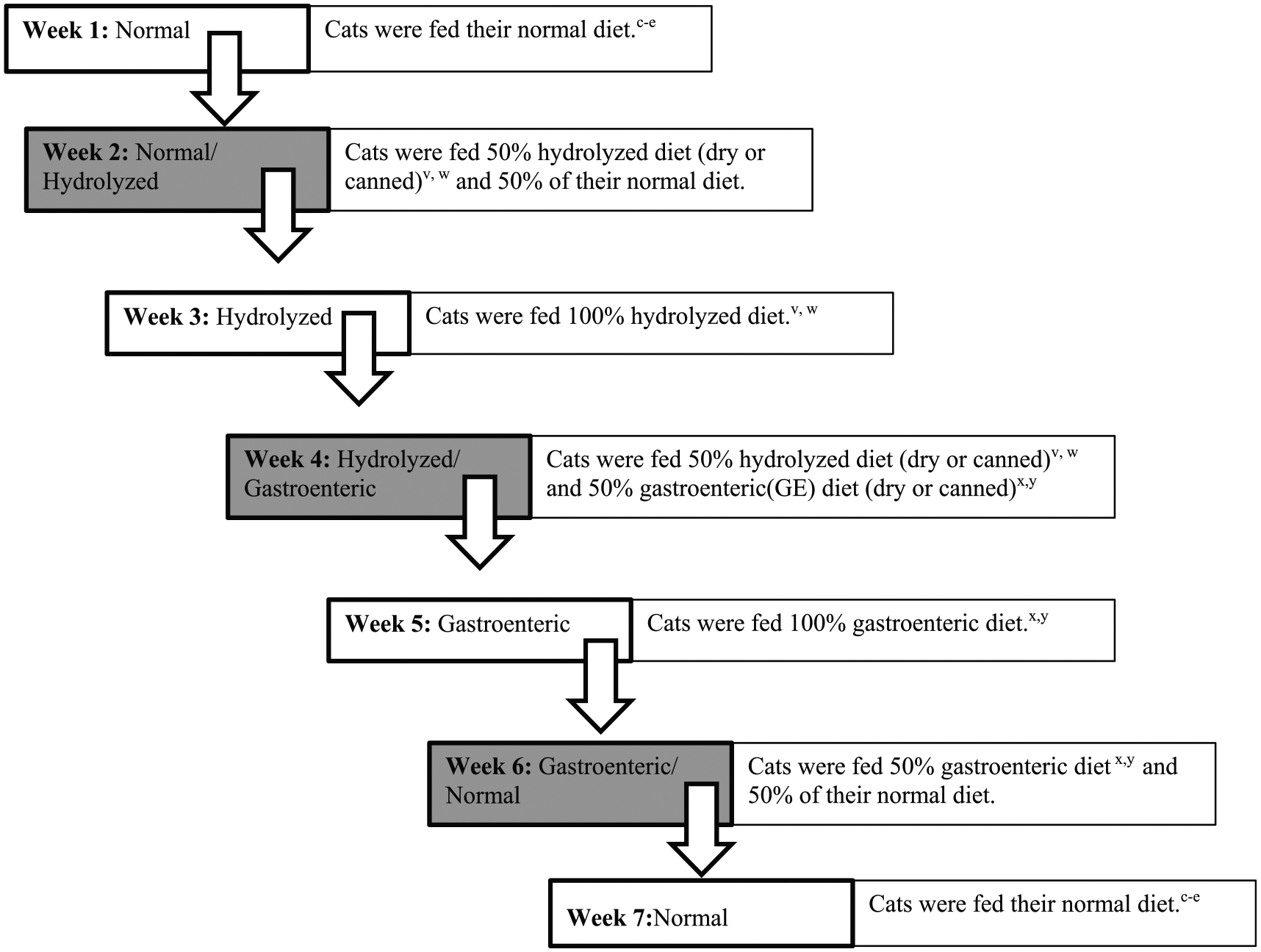
Diet study (dry and canned) feeding schedule. cFriskies Indoor Delight (Nestlé Purina); dPurina Pro-plan Focus Weight Management (Nestlé Purina); ePurina Pro-plan Chicken and Rice (Nestlé Purina); vHA Hydrolyzed Protein Pro-plan Veterinary Dry Diet (Nestlé Purina); wHill’s z/d Feline (Hill’s Pet Nutrition); xEN Gastroenteric Pro-plan Dry Diet (Nestlé Purina); yEN Gastroenteric Pro-plan Veterinary Canned Diet (Nestlé Purina)
Testing procedure
All stool samples were fresh (collected according to owners from the day/evening before) and kept at room temperature. Tests were set up according to manufacturer recommendations (Hemoccult Single Slides), as described in the methodology testing procedure, and only interpreted at 5 mins post-test set-up (based on repeatability of methodology study results). Control stool samples containing 50 µl of whole blood were used to verify positive FOB test interpretations each week during the study. Fecal scores were assigned to each stool sample (File 4 in the supplementary material).
Statistics
For statistical analysis, the statistical program R was used (R Core Team). The transition weeks between the diets were removed from analysis so as not to degrade the power of any post-hoc tests. To test for an association between diet and the presence of positives, a χ2 test was performed. Additionally, a logistic mixed-effects regression was performed to relate the number of positives for a sample to the type of diet. A likelihood ratio test was performed to test the main effect of treatment. Lastly, a Wilson score interval was performed to demonstrate the confidence intervals (CIs) for the uniform dry diet weeks. A P value of <0.05 was considered significant.
Results
Methodology
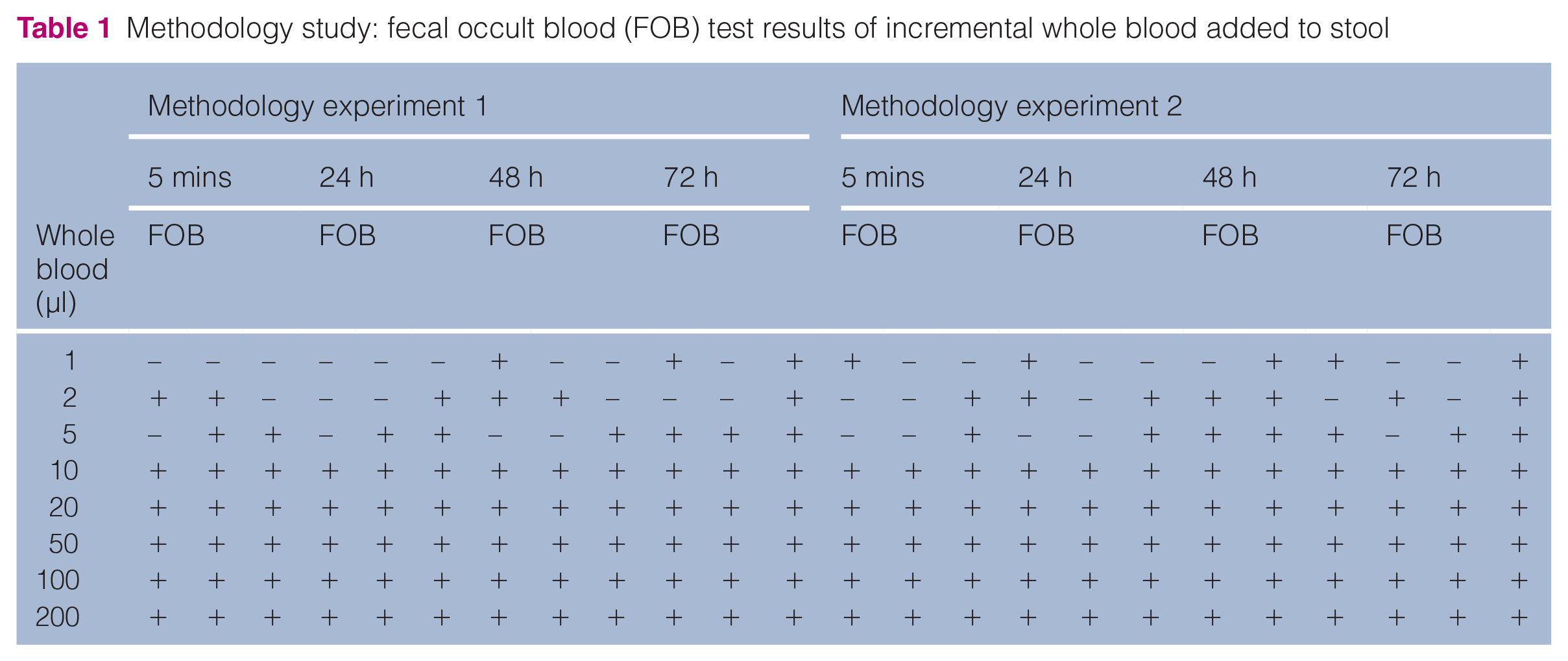
Ten microliters of whole feline blood in feline stool could be detected at all time points evaluated (at 5 mins and at 24, 48 and 72 h post-test set-up) in two separate experiments with exactly the same stool control, technique and observer (Table 1).
Methodology study: fecal occult blood (FOB) test results of incremental whole blood added to stool
Dry diet study
The data were summarized by tabulating the number of cats that had at least one positive FOB test for each uniform diet at weeks 1, 3, 5 and 7 (Table 2). There was no evidence of an association between the diet and a positive test result (P = 0.33). The Wilson score interval for the proportion of cats that had a positive FOB test was 0.108–0.603 (11–60%) during the uniform diet weeks. Two of 10 cats were positive on their regular/normal food diet (Hill’s Science Diet Adult Light Liver and Chicken entrée canned [Hill’s Pet Nutrition]) at week 1 (20%; 95% CI 5.7–51%), and one cat was positive in week 5 (10%; 95% CI 1.8–40.4%) when incidentally given the canned enteric diet (Friskies Seafood Sensations; Nestlé Purina) instead of the dry enteric diet (Hill’s Metabolic Feline; Hill’s Pet Nutrition) by the owner owing to palatability issues (Table 2). There were two other cats that produced positive FOB test results during the study; this occurred during the transition diet week 4 (Table 2).
Dry diet study: fecal occult blood (FOB) test results of the 10 cats during uniform diet weeks and transitional diet weeks

nrm = normal diet; HA = Purina HA dry hydrolyzed protein diet; EN = Purina EN dry gastroenteric diet
Transition weeks
Bold indicates when cat 5 was fed canned EN by owner instead of dry
Canned diet study
A likelihood ratio test was performed to test the effect of the canned diet on FOB test results and strong evidence was found to suggest that the canned diets fed differed in the likelihood of positives (P = 0.004). When cats were fed a uniform diet of canned hydrolyzed protein diet during week 3 of the study, three cats produced positive results (Table 3). The probability of a positive FOB result from cats fed the canned hydrolyzed protein diet was not significantly different from those fed their normal diet (P = 0.935). This is in contrast to week 5 of the study, when 8/10 cats had positive FOB results while eating a uniform diet of canned gastroenteric food (Table 3). Cats fed the canned gastroenteric diet were significantly more likely to produce a positive FOB test result than when fed their normal diet (P <0.01). In fact, the greatest number of FOB-positive stool samples occurred during week 6 of the study when 10/10 cats produced positive results (Table 3).
Canned diet study: fecal occult blood (FOB) results of the 10 cats during uniform diet weeks and transitional diet weeks

nrm = normal diet; zd = Hill’s z/d canned hydrolyzed protein diet; EN = Purina EN canned gastroenteric diet
Transition weeks
Discussion
To our knowledge, this is the first study to evaluate the effect of time on FOB testing methodology in cats and the first to evaluate the effects of currently available veterinary gastrointestinal feline diets on feline FOB testing.
This study demonstrates that 10 µl of whole feline blood can be reliably detected in feline stool at all time points evaluated (at 5 mins, and at 24, 48 and 72 h). Based on this information, there does not have to be a delay of 3 days to interpret FOB tests in cats. Perhaps the reason for this difference is owing to lower plant peroxidase activity in commercial feline diets vs human diets. This information increases the usefulness of FOB testing in a hospital setting to make clinical decisions. Despite the convenience of FOB testing in veterinary medicine, the clinical significance of performing these tests in pets has been previously questioned.8,10–12 Perhaps the perceived obsolescence of FOB testing in veterinary medicine is due to the lack of knowledge about the tests. Preceding this report, there had been no known published data evaluating testing methodology (effect of timing of test interpretation) of the human point-of-care guaiac-based FOB tests in veterinary patients. In addition, there has been a paucity of information regarding the effect of different diets on feline FOB testing, limited to just one paper published 18 years ago. 12
Although FOB testing is not commonly performed in veterinary medicine, it is still used in human medicine, as several studies have shown that early screening using FOB testing for patients at risk of developing colorectal cancer reduces mortality, especially if timely FOB testing is pursued.4,5,20 Although colonoscopy is the most effective screening method in human medicine for colorectal cancer, both the FIT and the guaiac FOB test have been proven to be effective screening modalities.4,20 In veterinary medicine, an abdominal ultrasound, endoscopy of the upper and lower gastrointestinal tract, and/or an exploratory laparotomy with biopsies may not be financially feasible for every patient with gastrointestinal disease. Therefore, using a human FOB test to screen cats for such disease may be clinically useful. Fortunately, the hemoglobin molecule is preserved among mammalian species and allows for the use of the human test in cats. 21
A recently published study investigating the sensitivity of FOB testing detected occult melena in cats fed canine packed red blood cells at a mg of hemoglobin per kg of body weight dose (~1.5 ml of blood); this differs from our methodology study, which relied on the addition of feline whole blood to stool samples, with as little as 10 µl of blood detected, mimicking hematochezia. 11 Identifying melena and or hematochezia in our feline patients is equally valid; the type of gastrointestinal diseases resulting in gastrointestinal bleeding, such as inflammatory bowel disease and neoplasia, may vary in severity, location and presentation, and may result in both melena and hematochezia.22–24 Differences in FOB sensitivity based on the location of gastrointestinal bleeding may occur as a result of the presence of bacteria and digestive enzymes, which degrade hemoglobin. Administering incremental volumes of whole blood directly to stool samples, we were able to assess test sensitivity for varying levels of hematochezia. Our research expands on the previous study in which cats were fed packed red blood cells by demonstrating test sensitivity for lower gastrointestinal bleeding rather than for proximal gastrointestinal tract bleeding (hematochezia vs melena).
Controlling for diet when interpreting FOB tests is a widely supported recommendation.9,12,25,26 Diet trials are often performed in small animals with suspected gastrointestinal disease;27–29 therefore, we chose two popular veterinary gastroenteric diets (Hill’s Science Diet Adult Urinary Hairball Control and Hill’s Metabolic Feline; both Hill’s Pet Nutrition) to increase the current clinical relevance of this study. Based on our results, if a cat has a positive FOB test when fed a hydrolyzed diet or dry gastroenteric diet, then further diagnostic testing or treatment may need to be pursued, such as an abdominal ultrasound, an endoscopic evaluation or gastrointestinal protectants.
The two cats with positive FOB results during week 1 of the dry diet trial were likely the result of their normal diet (Hill’s Science Diet Adult Light Liver and Chicken entrée canned; Hill’s Pet Nutrition), which was canned food with a large liver-based ingredient content. In human medicine, liver is known to cause false-positive FOB results by the addition of excess amounts of non-human hemoglobins.9,13,26 These two cats did not have positive FOB test results during week 7, when they were transitioned back onto a normal diet, because the owner eliminated the canned liver-based diet and fed the cats an exclusive dry diet (Hill’s Science Diet Adult Indoor; Hill’s Pet Nutrition).
Overall, there was a low number of positive tests produced during weeks 1 and 7 of the dry diet study, when cats were fed their normal diets. This is suspected to be due to low dietary interference. None of the normal diets contained ingredients expected to contain high amounts of heme (such as liver) or peroxidase activity, except for the two cats eating the liver-based diet. A similar reasoning can be made for the other positive FOB test results in week 5 of the dry diet trial when one cat was fed the canned version (Friskies Seafood Sensations; Nestlé Purina) of the gastroenteric diet instead of the dry (Hill’s Metabolic Feline; Hill’s Pet Nutrition). The canned enteric food used in the study has large amounts of meat by-products; liver is listed as a main ingredient. This may have led to the false-positive results for this same cat in weeks 4 and 6, as the owner of this cat used the canned version as enticement to help in the food transition (Table 2). The other cats’ positive FOB tests occurred in week 4, during the transition week between the hydrolyzed protein and the gastroenteric diet.
In the canned food diet trial, many cats produced positive FOB results during the uniform canned gastroenteric diet week and transitional weeks (Table 3). As mentioned, the ingredient content may have contributed to these results, but the stress of diet transition must also be considered, as all cats enrolled normally ate a dry diet. It is possible that disruption to normal gastrointestinal mucosa may have occurred during transition weeks due to stress as a result of diet change; however, markers of stress were not measured in these cats. Waiting to test the stool later in the week, such as on day 6 or day 7, may have helped to reduce the chances of positive results during the diet transition weeks. Therefore, as diet transition weeks could have caused clinical gastrointestinal changes/stress in the cat participants, we elected to perform our statistics on only uniform diet weeks as a means to try and eliminate the potential of false-positive results.
The FOB test is likely to be used in cats with clinical signs of intestinal disease, which were not represented in the study population. The limitations of this study include, first, the small sample size, as only 10 cats were evaluated, although to date this is the largest study to evaluate FOB test results in cats. Second, all participant cats were presumed healthy and, despite reasonable screening of study participants, the cats did not have abdominal ultrasounds, extensive gastrointestinal biomarker analyses, a fecal analysis (although deworming was performed in all cats) or endoscopies performed to rule out underlying gastrointestinal pathology. Third, we only tested a limited number of veterinary diets in an at-home setting, and we cannot be certain that treats or food scavenging by the cats at home did not occur, which may have affected FOB results. Fourth, only two stool samples from each cat were evaluated at various times each week, which means we may have missed intermittent gastrointestinal bleeding, although sample collection at two time points during the week was selected to increase owner compliance. Increasing the number of stool samples tested for each patient in general practice may increase the likelihood of detecting blood. Lastly, the Hemoccult slides used in this study caused a bright blue color change and the observer (KES) did not have difficulty interpreting the tests; however, it is possible that tests may have been misinterpreted. Future studies could include other guaiac-based tests, such as the Hemoccult SENSA tests, which feature a developer enhancer to increase the sensitivity of the test.
Conclusions
A small amount of blood (10 µl) can be reliably detected as early as 5 mins post-test set-up in feline stool using a human guaiac-based FOB test. Cats fed an exclusively veterinary dry or canned hydrolyzed protein diet, or a dry gastroenteric diet, are unlikely to produce false-positive FOB results. Evaluating a larger study population and additional diets would be needed to ensure this is a repeatable finding.
Supplemental Material
File 1
Client consent form (dry study)
Supplemental Material
File 2
Client consent form (canned study)
Supplemental Material
File 3
Body condition score data of diet study cats
Supplemental Material
File 4
Fecal score of cats
Footnotes
Acknowledgements
We thank Dr Julianne Hwang and Dr Nicolas Villarino for their help with data interpretation. We are also grateful to the owners of the feline participants who remained so attentive and committed throughout the study. The authors would also like to thank Nestlé Purina for providing the diets to all study participants.
Author note
The methodology results were presented at the 2017 ECVIM Congress and the dry diet trial results were presented as an abstract by the primary author at the 2018 American College of Veterinary Internal Medicine (ACVIM) Forum. The results of the canned diet trial were presented as a poster at the 2019 ACVIM Forum.
Supplementary material
The following files are available online:
File 1: Client consent form (dry study)
File 2: Client consent form (canned study)
File 3: Body condition score data of diet study cats
File 4: Fecal score of cats
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
All work for this project was performed at Washington State University, Pullman WA. This study was supported by a 2017 Nestlé Purina Summer Student Fellowship grant and the Washington State University Research Scholars program.
Ethical approval
This work involved the use of non-experimental animals only (owned or unowned), and followed established internationally recognised high standards (‘best practice’) of individual veterinary clinical patient care. Ethical approval from a committee was therefore not necessarily required.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work for the procedure(s) undertaken. No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
