Abstract
Practical relevance:
Non-regenerative anemia, or anemia with reticulocytopenia, is a daily diagnosis in feline practice.
Clinical challenges:
The disease processes underlying non-regenerative anemia are many and diverse. A major diagnostic evaluation may be required to correctly diagnose and treat the underlying cause.
Audience:
All veterinarians caring for cats will face the diagnostic and therapeutic challenge of non-regenerative anemia. Readers will benefit from the review of diagnostic testing and therapeutic options for non-regenerative anemia.
Evidence base:
This review summarizes the currently available literature informing diagnostic and treatment recommendations related to non-regenerative anemia. The evidence available to support the recommendations in this review is graded as low and includes predominantly expert opinion, case reports and cases series, on which the authors’ interpretation/consensus is based.
Keywords
Non-regenerative anemia, or anemia with reticulocytopenia, is frequently encountered in feline practice. The reason for the high frequency of non-regenerative anemia in the cat is unknown, but is most likely because cats develop anemia more commonly than dogs, and cats suffer from a number of chronic diseases that contribute to the feline propensity for anemia (see box). 1 A wide variety of diseases may underlie feline non-regenerative anemia. Review of all causes and therapies is beyond the scope of this article and the reader is referred to other reviews in this journal for further information (see box on page 616). The two main pathologic mechanisms leading to non-regenerative anemia are decreased or ineffective erythropoiesis and decreased red blood cell (RBC) lifespan.


Pathogenesis
Non-regenerative anemia in cats can occur either at the level of the bone marrow secondarily to decreased and/or ineffective erythropoiesis or as a sequela to decreased RBC lifespan once the mature RBCs are in circulation.
Decreased and ineffective erythropoiesis
Erythropoiesis can be decreased due to an absolute or a relative lack of erythropoietin, or secondary to a decreased bone marrow response to erythropoietin. Erythropoietin is produced primarily by the kidney in the peritubular interstitial cells of the inner renal cortex and outer medulla. Renal hypoxia is the main driving factor stimulating erythropoietin synthesis. Erythropoietin production is decreased by both acute and chronic causes of kidney disease, and chronic kidney disease is known to be a common cause of non-regenerative anemia in the cat.27–29 Production of neutralizing antibodies against erythropoietin has occasionally been associated with the administration of ESAs, and infrequently results in a relative deficiency of erythropoietin and non-regenerative anemia in cats.7,8,30
Ineffective erythropoiesis can be secondary to absolute deficiencies in the nutrients essential for hemoglobin or RBC biosynthesis, such as iron. Deficiencies of vitamin B12 can also cause non-regenerative anemia in the cat due to inhibition of purine and thymidylate synthases, which impairs DNA synthesis within erythroblasts and causes erythroblast apoptosis.12–14 Ineffective erythropoiesis in the cat also occurs as a result of cytokine abnormalities seen in various states of inflammation, and is commonly designated anemia of chronic disease.
Primary bone marrow disorders, including PRCA, NRIMHA and AA, as well as myelophthisis, can cause both decreased and ineffective erythropoiesis in the cat.
Decreased RBC lifespan
Feline RBCs have an expected survival of 73 days, shorter than that of dogs which is 100–115 days. 31 In circulation, the RBC must retain the ability to deform in order to deliver oxygen to tissues. With age, RBCs accumulate cellular injuries, triggering the removal of these senescent RBCs from the circulation by the reticuloendothelial system (RES).
RBC oxidative damage is one of the principal mechanisms leading to RBC senescence. Because cats have higher numbers of oxidizable sulfhydryl groups in comparison to many other species, feline RBCs are more susceptible to oxidative injury, commonly manifested as Heinz body formation.2,32 Feline RBCs also possess a lower intrinsic antioxidant capacity than many other species, such as humans, rabbits and mice, 33 which further increases the risk of Heinz body formation. 34 Feline RBCs lack N-acetyl transferase 2 (NAT2) and its absence results in a futile metabolic pathway where the reactive compound p-aminophenol is produced, leading to perpetuation of methemoglobin production. 35 Clinically healthy, non-anemic cats may have small, single Heinz bodies found on a minority of RBCs due to closed splenic circulation, increased feline RBC sulfhydryl groups and reduced feline RBC reductive capacity.32,36,37 However, the presence of Heinz bodies on the majority of RBCs, large Heinz bodies or multiple Heinz bodies per RBC should alert the feline practitioner to underlying pathology.36,37
Oxidative stress, mechanical stress, complement-induced injury, rearrangement of membrane phospholipids, contact with cationic proteins released from activated neutrophils, Heinz body formation and hemotropic parasites all alter cytoplasmic viscosity, resulting in impaired deformability, which targets RBCs for early removal from circulation. 38 Similarly, hereditary erythrocyte defects such as membrane protein abnormalities, erythrocyte enzyme deficiencies, hemoglobinopathies and increased osmotic fragility lead to shortened RBC lifespan. Excessive activation of the RES, as seen with certain immune-mediated, infectious, inflammatory and paraneoplastic conditions, can also stimulate early removal of RBCs from the circulation. In general, disorders resulting in decreased RBC lifespan cause regenerative anemia in cats; however, this is variable and non-regenerative anemia can also be seen (Table 1).
‘Regenerative’ anemias that do not always play by the rules

While typically associated with regenerative anemia, these diseases may cause a non-regenerative anemia in a proportion of cases
Breeds where ‘normal’ may not be the norm

Breeds that have been shown to have a reference interval (RI) that differs from other published feline RIs for hematocrit and reticulocyte count
The proposed RI for hematocrit in Maine Coons is narrower than most other published feline RIs
Diagnosis
Reticulocyte counts
A reticulocyte count is required to differentiate regenerative from non-regenerative anemia and an elevated reticulocyte count indicates a regenerative anemia. The definition of reticulocytosis in the cat ranges from >0.045 x 1012/l to >0.060 x 1012/l.45–47 However, interpreting reticulocyte counts is more complex than just assessing the absolute number of reticulocytes. Some authors advocate using a four-tiered system for assessing reticulocytes: strongly, moderately and weakly regenerative and non-regenerative based on reticulocyte counts of >0.2 x 1012/l, 0.06–0.19 x 1012/l, 0.016–0.05 x 1012/l and <0.015 x 1012/l, respectively. 48 Cats produce two types of reticulocytes: punctate and aggregate. Aggregate reticulocytes are released by the bone marrow and then mature into punctate reticulocytes while in circulation. Aggregate reticulocytes are those considered to represent the regenerative response. Punctate reticulocytes are not considered when differentiating regenerative from non-regenerative anemia.
Feline reticulocytes can be counted using manual and automated techniques. Although reticulocyte counts are slightly higher using automated techniques, results obtained from different automated hematology analyzers (Sysmex XT-2000iV, ADVIA 2120, Cell Dyn and ProCyte Dx) are similar and do correlate with manual aggregate reticulocyte counts.49–51 Reference laboratories typically report reticulocyte numbers based on automated counts unless manual counts are specifically requested or if there is sample interference, such as with autoagglutination. Because reticulocytes continue to mature ex vivo, reticulocyte counts should be performed as soon as possible after blood sample collection in order to obtain an accurate count. 49
Reticulocyte percentages
The reticulocyte percentage and corrected reticulocyte percentage are additional parameters used to quantify regeneration. These methods of assessing regeneration consider bone marrow production of reticulocytes and the peripheral RBC count by incorporating these numbers into the calculation. The formula for corrected reticulocyte percentage also considers the severity of the anemia. A recent study shows reticulocyte percentage may discriminate between regenerative and non-regenerative anemia and can be used when evaluating anemic cats. 52 Recommended cut-offs for a lack of regeneration are a reticulocyte percentage <1.70 for manual counts and <3.06 for automated counts. 52

Differential diagnosis
The differential diagnosis list for causes of non-regenerative anemia in cats is extensive, although some causes are more common than others (see box on page 615). Differential diagnoses should be prioritized based on patient signalment, detailed history and clinical presentation. Use of an algorithm to guide diagnostic recommendations will help to minimize unnecessary testing while simultaneously maximizing the chances of obtaining a diagnosis (Figure 1).
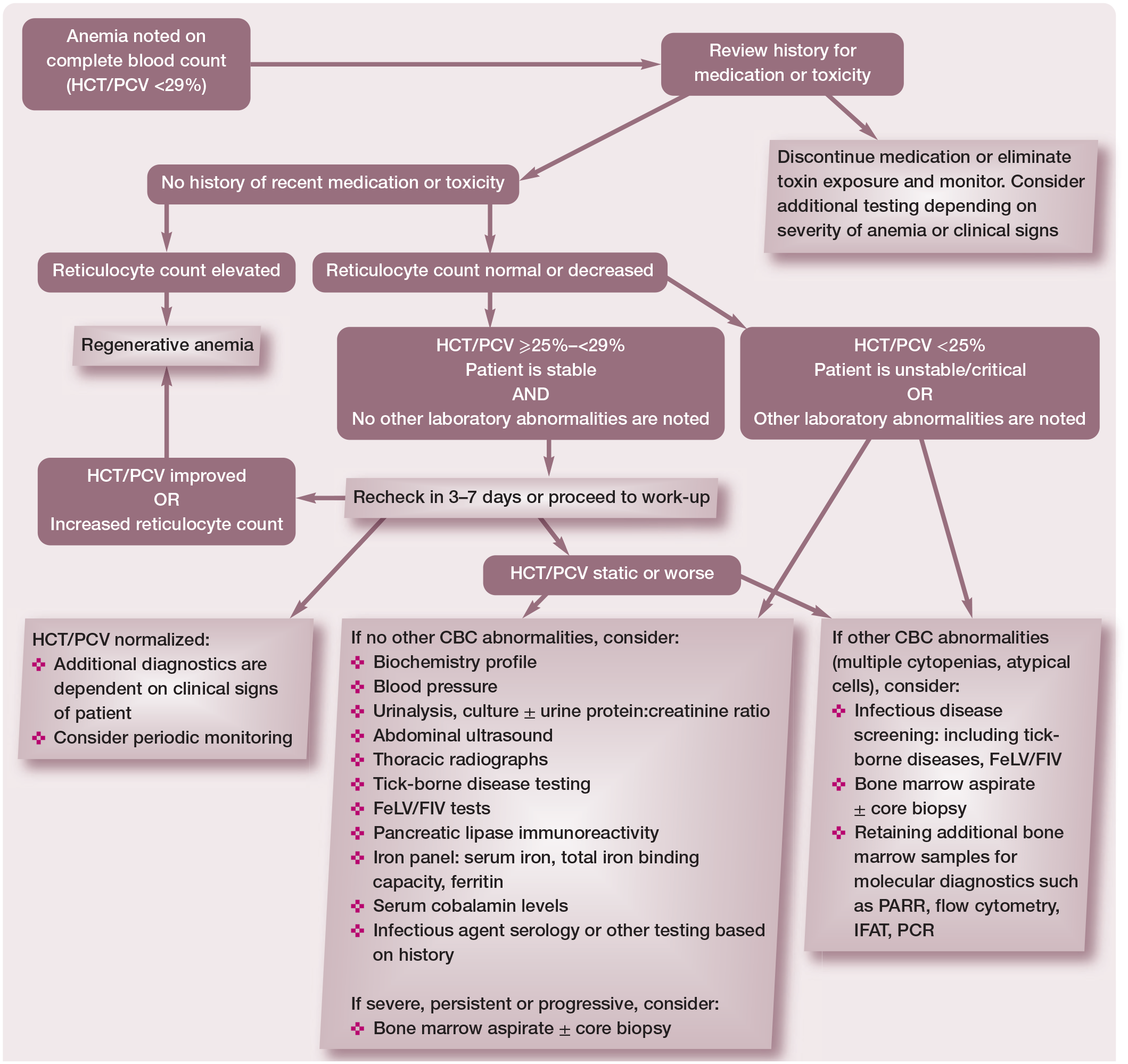
Algorithm for the identification and evaluation of a cat with non-regenerative anemia. HCT = hematocrit; PCV = packed cell volume; CBC = complete blood count; FeLV = feline leukemia virus; FIV = feline immunodeficiency virus; PARR = PCR for antigen receptor rearrangements; IFAT = immunofluorescence antibody test. Adapted from Ettinger SJ, Feldman EC and Cote E. Textbook of Veterinary Internal Medicine, Expert Consult, 8th ed, 2017, with permission of Elsevier
If initial hematology, biochemistry and infectious disease testing and diagnostic imaging do not identify the cause of non-regenerative anemia, the presence of cytopenias, abnormal blood cells identified on a blood smear, organ enlargement or diagnostic imaging abnormalities should prompt additional testing such as blood smear review (Table 3), bone marrow cytology or organ aspiration cytology (Table 4). The degree of anemia can also help to prioritize differentials. A severe anemia (HCT <14%), especially in a hemodynamically stable patient, suggests a slow onset of anemia and is more commonly associated with primary bone marrow disease, neoplasia and infectious diseases, including FIP, FeLV, FIV and coinfection with FIV and Haemoplasma species. 48
Blood smear evaluation for investigation of underlying causes of non-regenerative anemia in cats

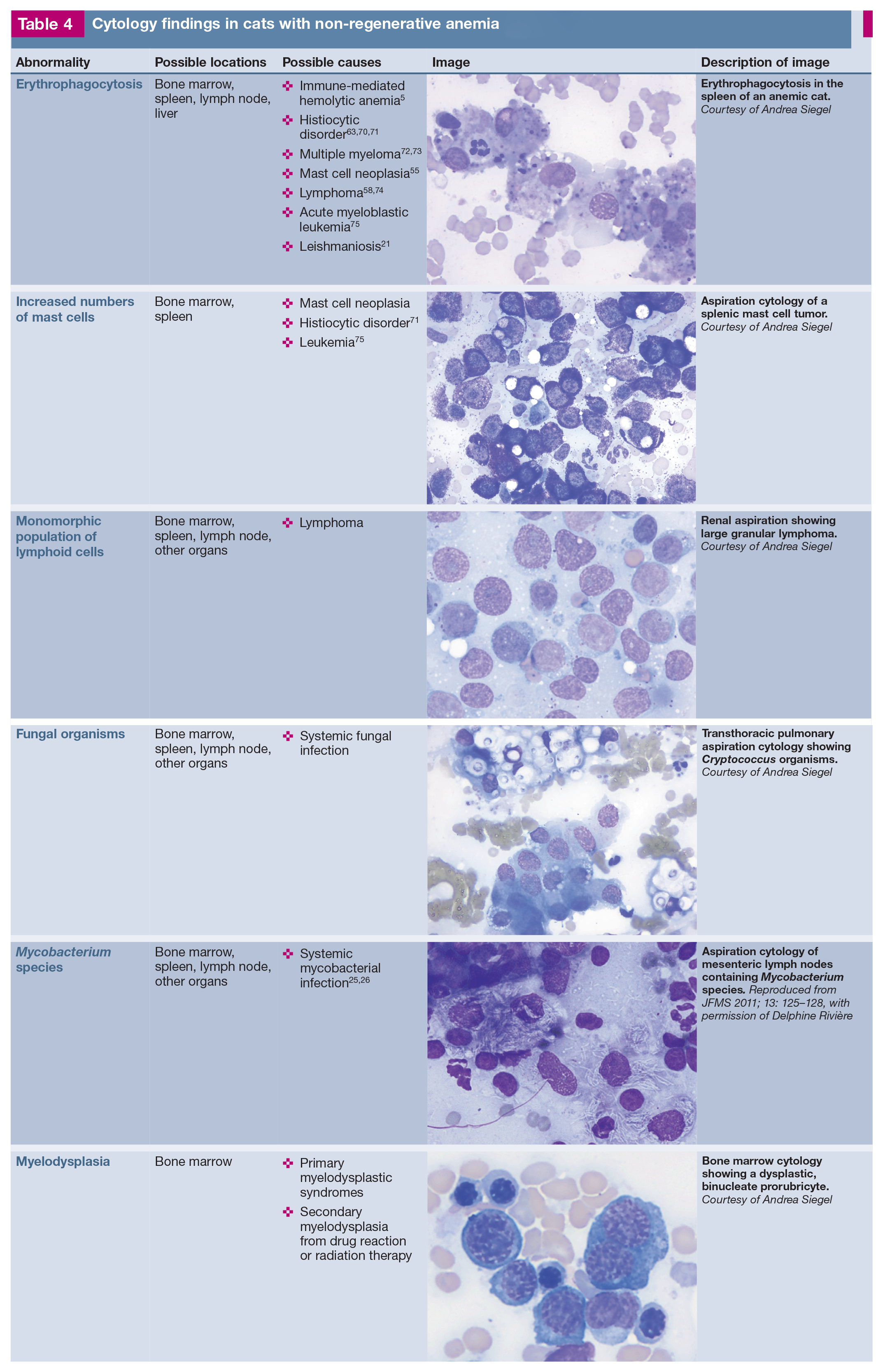
Cytology findings in cats with non-regenerative anemia
Common causes of non-regenerative anemia
Infectious diseases
FeLV and FIV are the most common infectious causes of non-regenerative anemia in the cat (see box on page 615). Some infectious diseases are geographically specific and so it is important to be familiar with the local diseases and to obtain a travel history.
Iron deficiency
Iron deficiency is typically related to chronic blood loss in the cat (gastrointestinal, parasitic from severe flea infestation, or anecdotally reported in cases of chronic hematuria). Iron stores are depleted over time as the bone marrow compensates for ongoing losses, resulting in a progressive non-regenerative anemia. In many species, anemia secondary to iron deficiency results in microcytosis and hypochromasia.
Differentiating between iron deficiency anemia and anemia of inflammation can be difficult and, in some cases, both conditions may coexist. In cats, because conventional evaluation of mean cell volume and mean cell hemoglobin concentration for microcytosis and hypochromasia are insensitive markers of iron deficiency, reticulocyte indices are better indicators of iron status. 76 Decreased reticulocyte hemoglobin content (CHR <0.88 fmol) has a 93.8% sensitivity and 76.9% specificity for identifying iron deficiency in cats; 77 however, reticulocyte hemoglobin is not uniformly available.
Measurement of iron parameters may further differentiate iron deficiency anemia from anemia of inflammation, although the distinction can still be difficult. Serum iron levels are low in both conditions. Ferritin, the soluble storage form of iron in tissues, is typically decreased in iron deficiency anemia and increased in anemia of inflammation. Transferrin, the main protein that binds and transports iron in blood, is measured indirectly as total iron binding capacity (TIBC), and TIBC levels are typically normal to increased in cases of iron deficiency, although concurrent inflammation can decrease TIBC and hinder interpretation.78 Iron saturation percentages (serum iron/TIBC) <20% can be suggestive of iron deficiency.78 While iron assays may help to provide insight, in some cases these results may not change therapeutic recommendations.
Bone marrow disorders
Multiple primary (medullary) and secondary (extramedullary) bone marrow disorders cause ineffective erythropoiesis, producing a non-regenerative anemia. These include: PRCA, NRIMHA, AA/pancytopenia, primary and secondary MDSs, myelofibrosis and primary and secondary myelophthisis. While bone marrow disorders as a general category are common causes of non-regenerative anemia in the cat, individual specific diagnoses are uncommon in cats. Bone marrow aspirate and cytology and/or bone marrow biopsy may be required to distinguish between these bone marrow disorders.
PRCA, NRIMHA and AA present to the feline practitioner with a similar clinical picture: a profoundly anemic cat without obvious blood loss and often in need of an urgent RBC transfusion. Each may be idiopathic/immune-mediated or triggered by an underlying cause, such as FeLV infection. These three forms of non-regenerative anemia are differentiated by complete blood count (CBC), and bone marrow cytology and histopathology findings. Both PRCA and NRIMHA, but not AA, may produce increased numbers of lymphocytes in the peripheral blood, lymphocyte aggregates in bone marrow samples and appear to occur in younger cats.44,79–81 Recently an association has been made between IMHA and pancreatitis. 82

Therapeutic options
Identifying the underlying cause of non-regenerative anemia is important for tailoring the patient’s treatment plan. Treatment of non-regenerative anemia includes supportive therapy such as increasing the oxygen-carrying capacity of the blood via RBC transfusion or with ESAs. Specific therapies should target the underlying cause and are not all covered within the scope of this article.
Supportive therapy
Transfusion
RBC transfusion can be used to rapidly increase oxygen-carrying capacity in non-regenerative anemia of any etiology. Approximately one-third of RBC transfusions in cats are for non-regenerative anemia;90,91 this may reflect the high prevalence of chronic kidney disease in cats.
All cats should be AB blood typed prior to the first transfusion and undergo crossmatching for subsequent transfusions. 92 Crossmatching in transfusion-naive cats has been recommended, but may not be required for a safe transfusion.93,94
Erythropoiesis-stimulating agents
In cats, ESAs are used commonly to treat non-regenerative anemia secondary to chronic kidney disease with good response.8,95 Use of ESAs for other primary medullary causes of non-regenerative anemia in the cat is mentioned sporadically in the literature without clear consensus regarding their utility.
An increase in HCT is reported with ESA use in cats with non-regenerative anemia due to AA, FIV and FIP.4,96,97 No increase in viral load was reported in cats with FIV. 96 In humans, ESAs are in widespread clinical use and are the most commonly used therapy for anemia secondary to MDSs, despite not being approved by the Food and Drug Administration for this specific use. 98 In one case series there appeared to be no clinical benefit in 2/4 cats with PRCA that received ESAs; however, interpretation of response, or lack thereof, is hindered as the cats were also on immunosuppressive medications, and durations of therapy and response were not reported. 99 The use of ESAs in cats with PRCA secondary to FeLV and in some cases with primary PRCA is not expected to be beneficial as endogenous erythropoietin levels are already elevated. Testing erythropoietin levels in cases of PRCA prior to administration of ESAs may be warranted.99,100
A number of recombinant human ESA products are available on the market including epoetin alfa, epoetin beta and darbepoetin alfa. Significant homology (>80%) between human erythropoietin and feline erythropoietin allows recombinant human products to bind to and interact with erythropoietin receptors in cats.101,102 Darbepoetin is hyperglycosylated compared with epoetin, resulting in a circulating half-life that is three times longer and a reduction in mean clearance rate by over 70% (ml/kg x h). 103 Both products may be associated with secondary PRCA, a refractory anemia after initial response to therapy as a sequela to the formation of anti-erythropoietin antibodies. Literature supports that this may occur in up to 8% of cats receiving darbepoetin or 25–45% cats receiving epoetin.7,8 With cessation of therapy, resolution of the anemia over months, associated with a concurrent reduction in antibody levels, has been reported; however, transfusions may be necessary in the interim. 7 The risk of PRCA, at least with epoetin, seems to increase with duration of therapy. 7 No longer produced, recombinant feline erythropoietin has historically been evaluated and was also shown to induce neutralizing antibodies, causing red cell aplasia in 30% of cats. 30 The choice of which agent to use should be based on numerous factors (see box).

Aside from PRCA, other side effects of these agents include hypertension, seizures and iron deficiency. 9 Concurrent monthly parenteral iron supplementation is recommended (see below). For epoetin specifically, additional adverse events of local reactions at injection sites (which may be predictive of antibody formation), fever, arthralgia and transient cutaneous or mucocutaneous reactions are also possible.7,9
Specific therapies
Targeted therapies to control blood loss
If chronic blood loss is suspected to be the cause of iron deficiency resulting in anemia, treatments to control bleeding and prevent further loss are indicated. Treatment options vary depending on the underlying disease and the source of the bleeding. These may include gastroprotective agents for ulcers, ectoparasite control and environmental decontamination for flea infestation or antiparasitics for chronic intestinal parasitism.
Iron supplementation
Cats being treated with ESAs or for iron deficiency anemia require iron supplementation. Parenteral iron supplementation is preferable given its more reliable absorption, especially if iron deficiency is secondary to malabsorption. Parenteral supplementation includes use of iron dextran, iron gluconate and iron sucrose. Iron dextran is the most common choice in veterinary medicine. The recommended dosage in cats is 10 mg/kg administered every 3–4 weeks.9,104 To decrease the risk of anaphylactic reaction, which occurs with intravenous iron dextran, intramuscular administration is recommended. Iron is absorbed slowly via the lymphatic system after injection, with approximately 60% absorption within 3 days and up to 90% after 1–3 weeks. 9
Oral iron supplementation, however, is the least expensive form of supplementation and is also considered the safest. Ferrous sulfate is the most common of the oral supplements, with varied dosing reported. The recommended dosage is 50–100 mg/cat/day PO for a total of 8.8–25 mg of elemental iron.9,104 Side effects are typically mild but include gastrointestinal upset. Concurrent administration with food or with calcium may reduce absorption; so will medications that increase gastric pH. 9 Administration of iron orally reduces absorption of antibiotics such as fluoroquinolones and tetracyclines, and dose spacing is recommended. 104 Other oral iron supplements are available including ferrous gluconate dosed at 16.25 mg/kg/day and ferrous fumarate dosed at 2–4 mg/kg/day in the cat. 104
Vitamin B12 supplementation
Anemia as a consequence of hypocobalaminemia requires vitamin B12 supplementation. As with iron, parenteral administration is preferred if the hypocobalaminemia is secondary to malabsorption. In cats with inflammatory bowel disease, the half-life of serum cobalamin is shortened from 12.75 days to 5 days, prompting the recommendation for weekly administration at least initially. 105
For parenteral supplementation vitamin B12 is administered as cyanocobalamin. Dosing protocols vary, with the most recent recommendation being to administer 250 μg SC once weekly for 6 weeks, followed by an additional dose 30 days later; serum cobalamin is reassessed 30 days after the final injection.9,106 Data supports that weekly injections increase serum cobalamin levels to supranormal levels. However, a recent publication demonstrated that following completion of a 6 week course of 250 μg SC once weekly, 95% of cats with hypocobalaminemia and enteropathy experienced significant decreases in serum cobalamin levels at 4 weeks after completion of the protocol, and by 10 weeks 55% of the cats again had levels below the reference interval. 107 Given the recent data, chronic intermittent parenteral supplementation may be required to maintain serum cobalamin levels within the reference interval after the initial weekly regimen. Additional studies are required to determine an appropriate dosing protocol. Anecdotally, for cats where cyanocobalamin does not succeed in increasing serum cobalamin levels, hydroxocobalamin can be used at the same dose and frequency.106,108
Data supports that oral supplementation with cyanocobalamin can also achieve supranormal serum cobalamin levels in cats that are hypocobalaminemic. The dosing recommendation is 250 μg PO q24h with continuous supplementation. 109 Tapering protocols for oral cyanocobalamin administration have not been evaluated in the cat. The efficacy of oral supplementation in comparison to parenteral administration has not yet been established.
Immunosuppressive treatment of primary medullary causes of non-regenerative anemia
Because single case reports and case series predominate in the literature, evidence-based treatments for primary medullary disease are lacking in feline medicine. Multiple classification schemes exist for primary bone marrow diseases, making comparison of treatment outcomes difficult.
Dosages of immunosuppressive agents used in non-regenerative anemia are provided in Table 5.
Dosages for immunosuppressive drugs used to treat non-regenerative anemia

Ciclosporin is available for intravenous and oral administration. Reports of ciclosporin use in cats include administration of the feline oral microemulsion solution as well as human oral solution, ciclosporin in oil and generic products. 80 Compounded transdermal ciclosporin in pluronic lecithin organogel is inconsistently absorbed and is not recommended for use in cats. 113 The feline oral microemulsion solution of ciclosporin administered at the label dosage reaches steady state in 7 days; however, doses needed to achieve clinical remission for PRCA cases have tended to be higher than the labeled dose, with 2–7 weeks required to achieve clinical remission.80,114 The optimal blood level of ciclosporin is unknown, but in a group of cats with PRCA, whole blood trough levels of 96–368 ng/ml were associated with clinical remission. 80
Gastrointestinal signs are among the most common adverse effects associated with ciclosporin administration. These include vomiting, hypersalivation, diarrhea, weight loss, regurgitation and lethargy.115,116 Effects on appetite are variable, with both increases and decreases reported, and anorexia only occurring rarely.114,117 Secondary infections, including toxoplasmosis and salmonellosis, have been reported.80,118–120 Less common adverse reactions include anaphylaxis during intravenous infusion of ciclosporin and hepatotoxicity. 80 Malignancy, most commonly lymphoma, has been reported in cats receiving long-term ciclosporin.114,121,122 Cats receiving chronic ciclosporin therapy following renal transplantation are at six times higher risk of both developing a malignant tumor and of lymphoma. 122
New directions in therapy
Management of non-regenerative anemia of inflammation using iron and ESAs is ineffective for some patients and also there are risks, driving the search for novel therapies to stimulate erythropoiesis. Many of these therapies target the hepcidin–ferroportin axis. Direct antagonists inhibit hepcidin function.131–133 Hepcidin production inhibitors (including IL-6 pathway inhibitors and vitamin D) prevent hepcidin transcription.131–133

Ferroportin agonists and stabilizers promote ferroportin resistance to hepcidin action.131–133 Prolyl hydroxylase inhibitors reduce hepcidin levels and also stabilize hypoxia-inducible factors to promote endogenous erythropoietin production.131,133,134 Other targets include erythropoietin gene therapy.131,134
Many different drugs in these categories are under commercial development and in clinical trials for use in humans. 133 As their efficacy and safety profiles are established, these novel agents will likely also be considered in feline patients to expand our options for long-term management of these challenging cases.
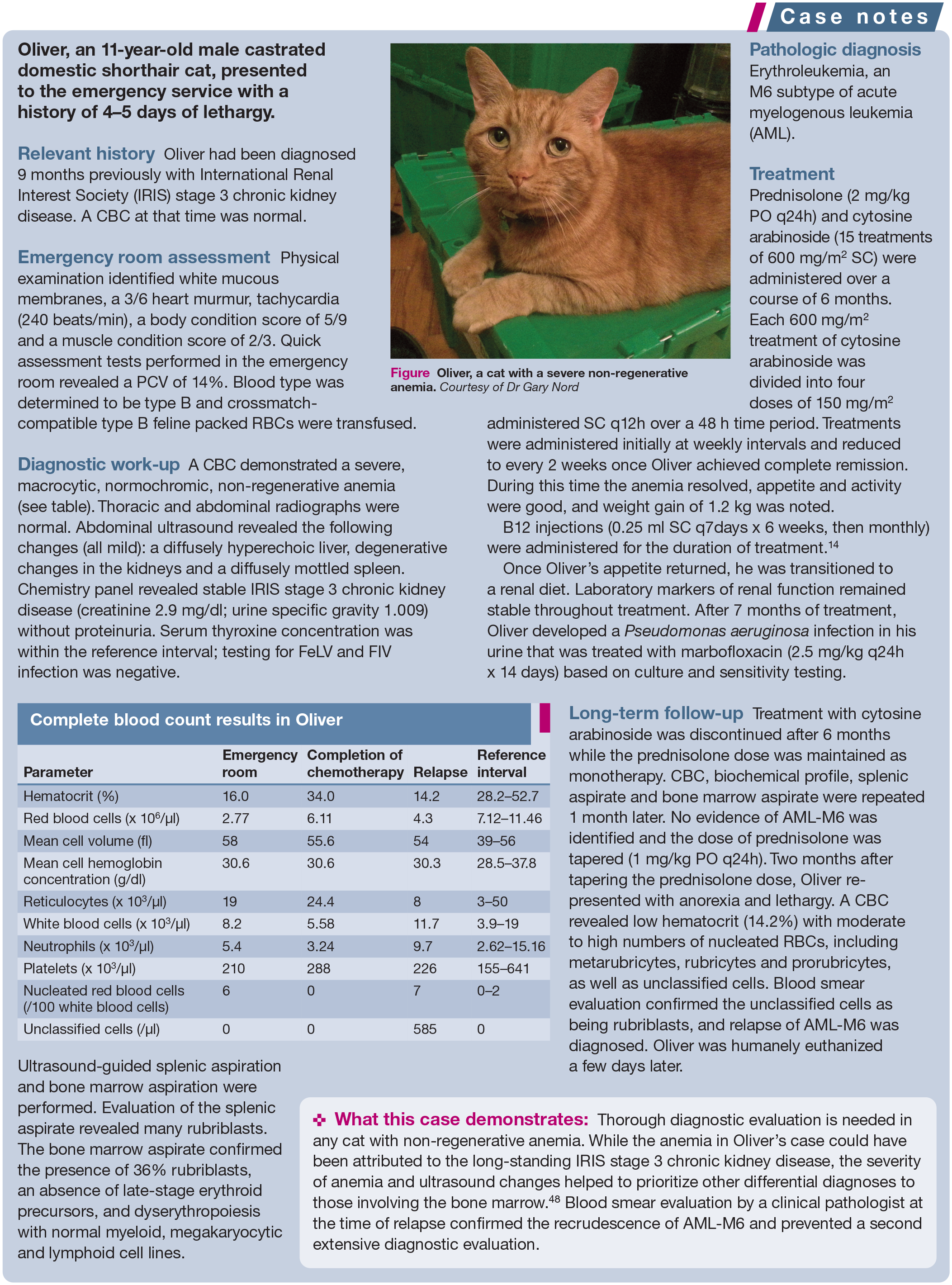

Oliver, a cat with a severe non-regenerative anemia. Courtesy of Dr Gary Nord
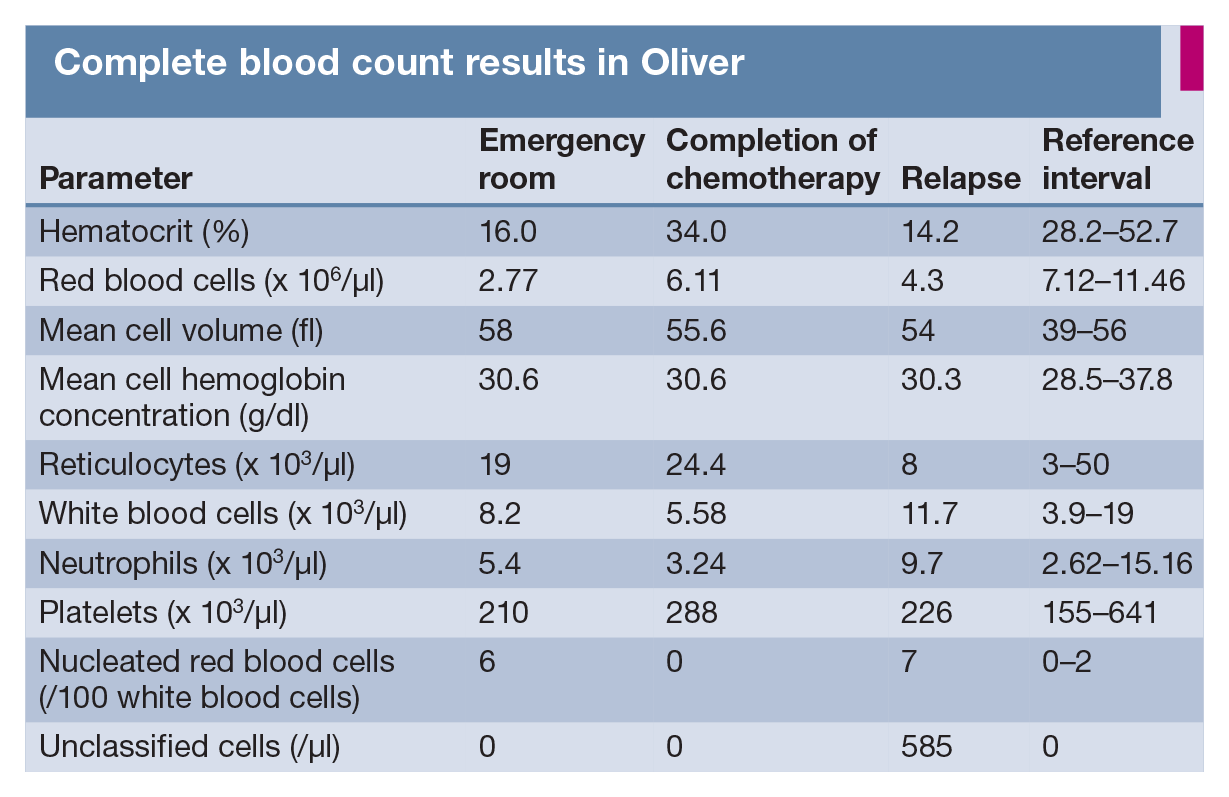
Complete blood count results in Oliver
Key Points
Obtain an aggregate reticulocyte count for every cat diagnosed with anemia.
Use all components of the CBC to direct the diagnostic plan.
Review the results of a minimum database, CBC, biochemical profile and urinalysis to direct further diagnostic testing.
A blood film should be reviewed by a clinical pathologist or trained technician for clues to the underlying cause of anemia.
Anemia of inflammation is a diagnosis of exclusion.
Once extramedullary causes of anemia are ruled out, a bone marrow aspirate/biopsy is appropriate.
Some diseases are geographically specific – know your local diseases and do not forget to obtain a travel history.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
