Abstract
Objectives
The aim of this retrospective descriptive study was to determine the effectiveness of using iridium implants in addition to surgery in cats with feline injection-site sarcomas (FISSs) in terms of time to progression and disease-specific survival and to identify prognostic factors for patient outcome.
Methods
Medical records of cats presented at our institution with FISS were reviewed. Inclusion criteria included histologic diagnosis of a tumor type associated with post-injection neoplastic development, tumor located at a site associated with vaccination, no other therapies prior to the administration of brachytherapy with the exception of surgery and adequate follow-up data.
Results
Twenty-two cats with FISS were treated with surgery and brachytherapy delivered by postoperative iridium-192 interstitial implants. Radiation doses ranged from 4000 to 6000 cGy (median dose 5079.55 cGy), with most doses delivered over 7 days. The median number of surgeries prior to brachytherapy was one (range 1–4). The complications associated with postoperative brachytherapy were typically mild, although four cats developed more severe complications. The median time to progression for all cats was 619 days and disease-specific survival time for all cats was 1242 days. The 1 and 2 year tumor-free rates in these cats were 63.6% and 40.9%, respectively. The local failure rate was 54.5% and the distant failure rate was 13.6% due to lung metastasis. There was a significant difference in time to progression of cats that had a single surgery performed prior to brachytherapy and those that had multiple surgeries (undefined vs 310 days; P = 0.01). There were no other statistically significant identified prognostic factors.
Conclusions and relevance
These data suggest that the addition of brachytherapy postoperatively in cats with FISS was well tolerated and is comparable to other forms of adjuvant therapy.
Introduction
Feline injection-site sarcomas (FISSs) are a significant health problem in domestic cats. They are highly invasive neoplasms that require aggressive treatment, which may include surgery, radiation therapy and chemotherapy. In many anatomic locations, complete surgical excision is difficult, with the current recommendations being 5 cm lateral margins and two fascial planes for deep margins based on the positive outcomes in the publication of Phelps et al. 1 Major surgical complications were reported in 11% of cats, with wound dehiscence being the most common complication; however, the regrowth rate in these cats was 14%. 1 The overall survival time (OST) was 901 days, which is much longer than previously reported. 1 Cats that have a fewer number of surgeries and complete excisions will often benefit from improved survival.1–3 Even when located on a distal limb, amputation is often required for complete surgical excision, but local control is improved for this location; for example, in the paper by Hershey et al, if the tumor was on the limb, the time to recurrence (TTR) was 325 days vs the median at 94 days. 3 Despite wide-margin surgery, margins are often incomplete and recurrence rates range from 30% to 70%. 4
Because of this, the best treatment outcomes are associated with combination therapy of aggressive surgery and radiation therapy.5–9 It is unclear if, when radiation therapy is used, there is a benefit to performing preoperative radiation therapy vs postoperative therapy. In the study by Cronin et al, preoperative radiation therapy followed by surgery was performed for feline fibrosarcoma and the disease-free interval (DFI) and OST were 398 days and 600 days, respectively. 5 Cats that had incomplete margins after surgery had a decreased DFI at 112 days. 5 When postoperative electron beam radiation was used to treat cats after surgery, the 1 and 2 year survival rates were 86% and 44%, respectively, with a median survival time (MST) of 730 days. 6 There was a decrease in DFI and MST if there was a larger delay between the start of radiation therapy and surgery. 6 In another study, by Bregazzi et al, TTR was not reached for the cats treated with surgery and radiation therapy and MST was 842 days. 7 Unfortunately, local tumor control is still disappointing, with 28–45% of tumors recurring after multimodal therapy with surgery and radiation therapy, although better outcomes are generally achieved when cats with microscopic disease are treated, complete surgical margins are obtained and a decreased number of surgeries are performed.5–9
It is unclear how the role of chemotherapy benefits cats with FISS. In the study by Eckstein et al a better outcome was achieved if the cats had chemotherapy as part of their treatment protocol. 9 When combinations of doxorubicin and cyclophosphamide were used in the face of gross disease, 50% of the cats had a >50% reduction in total tumor volume vs cats treated with doxorubicin or Doxil alone that had a 39% response rate.10,11 When lomustine was used for gross disease the response rates were 25%. 12 If responses were observed in these studies, in most cases they were short lived. When doxorubicin was used in cats that had surgery and radiation therapy, the TTR was actually shorter than surgery and radiation without chemotherapy (661 days vs unreached), although it was not statistically significant. 7
There is, however, evidence for surgery followed by adjunctive therapy such as brachytherapy using iririum-192 and potentially immunotherapy with recombinant canarypox virus expressing feline interleukin 2, as the cats in the study by Jas et al showed significantly greater time to relapse (>730 days) than cats in their reference control group (286 days). 13 This descriptive retrospective study evaluated the use of iridium-192 interstitial implants alone as adjunctive therapy following surgery in 22 cats with presumed FISS.
Materials and methods
The case records of 22 cats that received surgery and radiation therapy with iridium-192 implants for presumed FISS were selected for study. These patients were treated during the period of 1995–2008. A board-certified medical oncologist and a consulting board-certified radiation oncologist determined whether or not cats were good candidates for this therapy. These treatments, as well as other options, were offered to owners as part of standard of care. Inclusion criteria included histologic diagnosis of a tumor type associated with post-injection neoplastic development, tumor located in a site associated with vaccination, no other therapies prior to the administration of brachytherapy with the exception of surgery and adequate follow-up data. Data recording was performed by the board-certified radiation oncologist, as well as the certified radiation technician and the board-certified medical oncologist for each case within the medical record for each patient. Vaccination type was not known in all cases but cats where the type of vaccine used was known included feline leukemia vaccine, vaccine for feline rhinotracheitis/calicivirus/panleukopenia combination and rabies vaccine.
Each cat had a cytoreductive surgical procedure with wide margins taken. Surgery was followed with brachytherapy in each case. Adverse events associated with the brachytherapy were recorded and graded based on toxicity criteria of the veterinary radiation therapy oncology group. 14
The recommendation for use of chemotherapy was based on the judgment of the clinicians managing the cases and owner preference. Chemotherapy consisted of doxorubicin administration in all the cases that received medical therapy.
Follow-up for each cat was performed by examination at Texas A&M or the referring veterinarian’s hospital, and correspondence with the owners at 3 month intervals. Data were collected until either death of the cat or loss of the patient to follow-up. The time to progression (TTP) was calculated from the time of implantation of iridium-192 to disease progression (local progression or metastasis). Patients were censored if they were alive at the end of the study period, lost to follow-up or dead without evidence of disease progression. The disease-specific survival (DSS) was calculated from the date of diagnosis to the day of death as a result of tumor progression or treatment-related complications. Patients were censored if they were alive at the end of the study period, lost to follow-up, or if they died of non-tumor/non-treatment-related causes. Risk factors that were accessed included whether or not the patients had recurrence, whether or not the patient had one or multiple surgeries prior to radiation therapy, whether or not the surgery was performed by a board-certified surgeon or general practitioner, clean vs incomplete margins on histopathology, whether or not recurrence, if it occurred, happened at the margin or within the radiation treated field or if the patient received chemotherapy vs no chemotherapy. Recurrence rates and metastatic rates were also determined.
Descriptive statistics were performed using Microsoft Excel for Mac 2011 version 14.7.7. Kaplan–Meier survival proportions were performed using GraphPad Prism 7 for Mac OS X version 7.0c. Significance for survival comparisons was set at a P value of 0.05. Statistical analysis was performed by a board-certified medical oncologist with 13 years’ experience in running and evaluating clinical trial data.
Results
A medical record search for feline patients with a histologic diagnosis of sarcoma between 1 January 1995 and 1 January 2014 in the Texas A&M Veterinary Teaching Hospital VMIS medical records system revealed 154 cases. Of these, 22 cats with a total of 24 distinctly eligible tumors (one cat was included that had three tumors at three distinctly different sites diagnosed at the same time and treated individually) were diagnosed with histologically confirmed soft tissue sarcomas, presumably injection-site associated, and received both surgery and postoperative iridium-192 interstitial implants with adequate follow-up information for inclusion in the study. The population consisted of 19 domestic shorthair cats (86.4%) and three domestic longhair cats (13.6%). There were 11 castrated males and 11 spayed females (1:1) with a mean and median age of 9.89 and 10 years, respectively (range 5–15 years). The mean and median weight of this population was 5.18 kg (range 2.27–8.82 kg).
The tumors were located in the subcutaneous soft tissues in the dorsal cervical and interscapular areas (n = 12/22 [54.5%]), dorsally along the thoracolumbar spine (n = 3/22 [13.6%]), over the hip or caudal flank (n = 4/22 [18.2%]) and laterally along the thoracic and abdominal wall (n = 3/22 [13.6%]). Histologically, the tumors were confirmed as fibrosarcoma (n = 17/22 [77.2%]), undifferentiated sarcoma (n = 1/22 [4.5%]), giant-cell tumor (n = 2/22 [9%]), myxosarcoma (n = 1/22 [4.5%]) and rhabdomyosarcoma (n = 1/22 [4.5%]). The median time from last vaccination to diagnosis was 285 days (range 7–1440 days). The median and mean duration of lesion presence prior to presentation to a veterinarian was 56 and 182 days, respectively (range 14–336 days). Preoperative blood work evaluation included a complete blood count, biochemistry profile and urinalysis in most cases (n = 19/22 [86.4%]). All cases had thoracic radiographs prior to surgery (n = 22/22 [100%]). No evidence of pulmonary metastasis was noted at initial evaluation in any case. In the six cases (27.2%) tested for feline leukemia/feline immunodeficiency virus, each test was negative. Con-trast-enhanced CT was performed in 19 cases (n = 19/22 [86.4%]) to assess the size of the field to be implanted.
Cats had surgery with wide margins taken, performed either at their referring veterinarian or at Texas A&M by a board-certified surgeon. Eighteen (81.8%) cats had surgery at Texas A&M and four (18.2%) had surgery at their local veterinarian’s office. The total number of surgeries prior to idirum-192 implantation ranged from one to four, with a median of one surgery. All cats were implanted with iridium-192 a median of 4.5 days after cytoreductive surgery (mean 15.4 days, range 1–94 days). Radiation doses were computer calculated, using a two-dimensional program (Prowess 3000; Prowess), with the reference dose being the isodose contour located 0.5 cm from and completely encircling the implant plane.
A manual afterloading technique was used to place the iridium-192 seed ribbons in a single plane approximately 0.5 cm below the surface of the skin with the iridium seeds spaced 1 cm apart, center to center of each seed, with the seed ribbons spaced in rows 1 cm apart. Briefly, the fur over the implant region was shaved and sterile prepped. A sterile ruler and surgical pen were used to mark the ribbon entry and exit locations based on CT reconstruction of the previous surgery site (Figure 1a). For cats that did not have CT performed, the proposed implant was graphed and then the coordinates of the implant seeds were entered into the dosimetry computer.15,16 The iridium seed ribbons were placed by inserting stainless steel 16 G implant needles, through which nylon afterloading tubes were placed until all were satisfactorily in position (Figure 1b). The needles were then withdrawn, leaving the tubes in place, after which the iridium ribbons were afterloaded into the nylon tubes (Figure 1c). Lastly, the iridium-containing afterloading tubes were secured in place by stainless steel buttons placed on each end of each tube (Figure 1a).
All patients were anesthetized for the duration of the procedure. The radiation treatment area ranged from 30 to 100 mm2, with a median of 72 mm2 and mean of 67.4 mm2. Duration of implantation was either 6 or 7 days (median 7, mean 6.95 days). The number of ribbons implanted ranged from three to 10 (median six, mean 6.57), while the range of number of seeds per ribbon ranged from three to 20 (median 10, mean 10.45). The dosage administered ranged from 4000 to 6000 cGy, with a median of dose of 5000 cGy and mean dose of 5079.55 cGy.
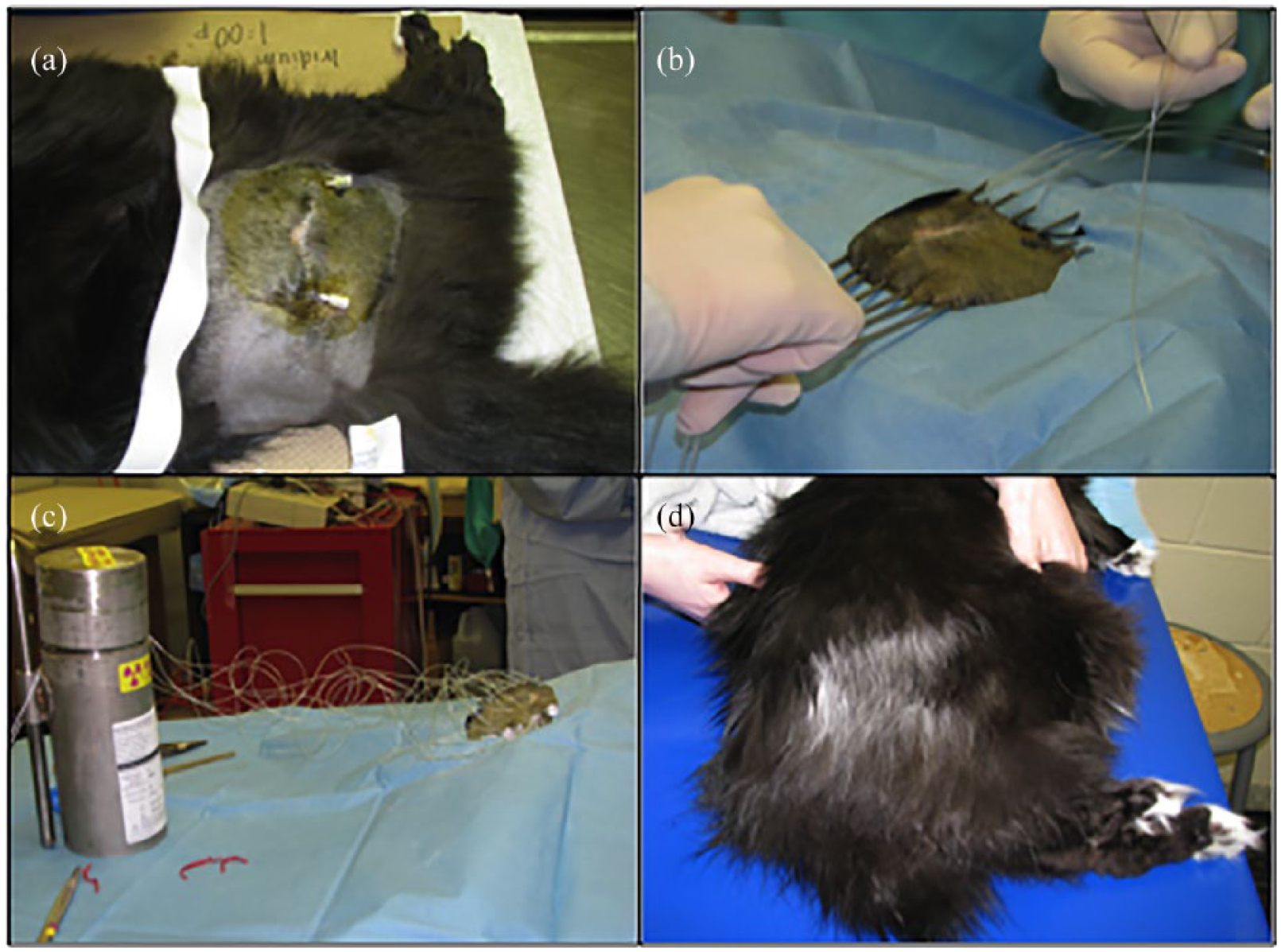
(a) The fur over the implant region was shaved and sterile prepped, and this image shows the implants secured with metallic buttons on either end of the incision. (b) Needle placement for iridium implantation. (c) Iridium-192 implantation. (d) Post-radiation adverse effects (thin haircoat and leukotricia)
Complications associated with postoperative brachytherapy were noted in 20 cats (90.9%). Although they were typically mild, a few developed grade 3 adverse events (Table 1). Acute side effects occurred during the treatment period or shortly after. Five cats (22.7%) developed mild serous drainage from one or more of the cutaneous exits of the afterloading tubes, and four cats (18.2%) developed postoperative seromas. These changes resolved within 2 weeks of explantation in all cases. Two cats (9%) developed infections associated with their implantation sites that resolved after antibiotic treatment. Four cats (18.2%) had tubes that moved from their site of placement or were self-removed by the patient. Most of these tubes were replaced and all were located within the isolation cage. One cat had severe swelling where the needles were placed for implantation of iridium and was likely associated with the disinfectant used; this resolved with supportive care. The most severe complication was postoperative wound dehiscence in three cats (13.6%). One cat exhibited profound anorexia during hospitalization and required percutaneous endoscopic gastrostomy tube placement for feedings. Delayed side effects mostly consisted of chronic skin or haircoat changes. Seventeen of the 22 cats (77.3%) had long-term changes in their haircoat within the treatment field, including altered haircoat color, alopecia, slow regrowth of hair or cutaneous hyperpigmentation (Figure 1d).
Graded adverse events in the 22 cats treated with postoperative iridium-192

Four cats (18.2%) received doxorubicin chemotherapy. Three of these cats had doxorubicin after radiation therapy, but prior to local recurrence. These cases received three, four or five doses. The fourth cat received two doses of doxorubicin after local recurrence occurred and had no response. Doxorubicin therapy was stopped because the cat developed significant gastrointestinal adverse effects related to the chemotherapy itself and not the radiation.
After local recurrence, five cats received additional surgery(s) at the location of tumor recurrence.
Of the 22 cats, eight (36.4%) were lost to follow-up, four (18.2%) died or were euthanized for causes other than their FISS, and 10 (45.4%) died or were euthanized as result of their FISS. Of the cats lost to follow-up, two (9.1%) had recurrence noted at 173 and 191 days following implantation and then were lost to follow-up at 376 and 347 days post-implantation, respectively. The remaining six cats were disease free when they were lost to follow-up.
The local failure rate for these cats was 54.5%. Of the 12 cats that developed recurrence, eight (66.7%) developed recurrence within 1 year of radiation therapy. The remaining four cats developed recurrence at 447, 518, 619 and 833 days post-radiation therapy. In the 12 cats in which local tumor recurrence was noted, six were believed to have clean surgical margins at the time of resection, while the surgical margins were determined to be incomplete in six cases. Of the 10 cases (45.5%) with no evidence of local tumor recurrence, the surgical margins were determined to be complete in six cases (60%) and incomplete in four (40%). Location of tumor recurrence was assessed in each of the 12 cats. Recurrence was within the radiation treatment field in six cats (50%) and at a margin of the treatment fields in five cats (41.7%). The precise location of the recurrence was not evaluable from the medical record in one case.
For these 22 cats the distant failure rate was 13.6%. Of the three cats that developed metastatic lung disease, two were euthanized at 155 and 327 days, respectively, and one was lost to follow-up at 410 days. Two of these cats did not have evidence of local recurrence at the time of euthanasia or loss to follow-up.
The median TTP for all cats was 619 days (range 72–2968) (Figure 2). The number of previous surgeries was the only factor that influenced the median TTP. Cats receiving only one surgery prior to brachytherapy had a median TTP that was undefined (range 92–2968 days, n = 16) vs those that received multiple surgeries with a TTP of 310 days (range 72–518 days, n = 6; P = 0.01) (Figure 3). Whether or not the first surgery was performed by a board-certified surgeon (median 619 days, range 84–2968 days, n = 18) vs a general practitioner (median 259.5 days, range 72–1700 days, n = 6) (P = 0.42), clean (median 553 days, range 84–2792, n = 10) vs incomplete margins (median 518 days, range 72–2968, n = 15) (P = 0.92) and location of recurrence at the margin (median 447 days, range 72–2374, n = 5) or in the field (median 205.5 days, range 84–1867, n = 6) (P = 0.99) did not affect TTP. There was also no difference in TTP for cats that received chemotherapy (median 355.5 days, range 72–756 days, n = 4) vs those that did not (median 518 days, range 84–2968 days, n = 18) (P = 0.4).
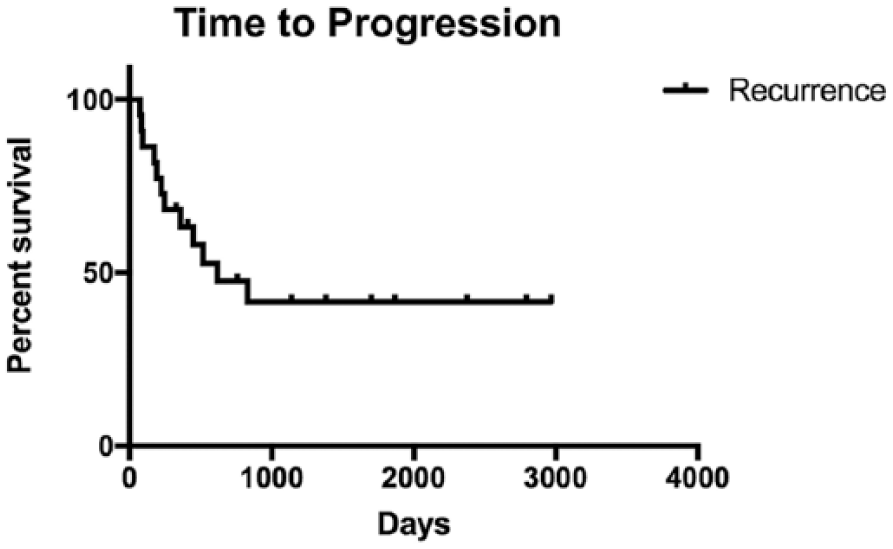
Kaplan–Meier curve of time to progression for all cats. The median time to progression was 619 days

Kaplan–Meier curve of time to progression for cats treated with one surgery prior to iridium-192 and cats that received multiple surgeries. The time to progression for cats that had one surgery prior to iridium-192 treatment is significantly better than cats that had multiple surgeries
The median DSS for all cats was 1242 days (range 152–2968 days) (Figure 4). There were no significant differences in DSS in cats that had no recurrence (undefined, range 250–2968 days, n = 10) vs recurrence (median 1230 days, range 152–1876 days, n = 12) (P = 0.17), cats that had clean margins (median 1242 days, range 152–2968 days, n = 10) vs incomplete margins (median 1242 days range 276–2792 days, n = 15) (P = 0.77), recurrence at the margin (median 1091 days, range 660–1290 days, n = 5) vs in the field (median 361.5 days, range 152–1230 days, n = 6) (P = 0.06), if the cat had a single surgery (undefined, range 152–2968 days, n = 16) vs multiple surgeries prior to radiation (1091 days, range 376–1290 days, n = 6) (P = 0.22), or whether the first surgery was performed by a board-certified surgeon (median 1230 days, range 152–2968 days, n = 18) vs a general practitioner (median 1242 days, range 660–1242 days, n = 6) (P = 0.76). It also made no difference in DSS if the cats received chemotherapy (median 961 days, range 152–1242 days, n = 4) or did not (median 1242 days, range 276–2968 days, n = 18; P = 0.24). At 1 year, 63.6% of the cats were tumor-free; this was reduced to 40.9% at 2 years.

Kaplan–Meier survival curve of disease specific survival for all cats. The median disease specific survival for all cats was 1242 days
Discussion
This study aimed to describe clinical outcomes of cats treated for FISS with wide-margin surgery followed by iridium-192 brachytherapy. To our knowledge, there has been limited use of iridium-192 in domestic animals such as cats for postoperative brachytherapy, although it has been used in large animals for the treatment of both equine and bovine tumors. Interstitial brachytherapy for treatment of fibrosarcoma in cats has been reported.15–17 The information in the article by Turrel and Koblik mainly involved the description of the technique for treating with iridium-192 and types of tumors to treat with iridium brachytherapy; their results showed a 1 year tumor-free rate of 50% in cats with fibrosarcoma treated with iridium-192, indicating a valid potential adjuvant therapy role for this disease. 17 In France, a group of researchers have been treating cats that have FISS with brachytherapy and when they combined surgery, brachytherapy and immunotherapy into a treatment protocol, the cats had significantly longer times to relapse, again solidifying a place for brachytherapy to be used in the treatment of patients with FISS. 13
Aggressive surgical excision has been considered the most effective treatment for soft tissue sarcomas and the current recommendations, especially for infiltrative FISS, are based on a study in which 91 cats with tumors in various locations were treated with surgery alone using 5 cm lateral margins and two fascial planes for the deep margins. 1 Complete histologic resection was achieved in 97% of cases and local recurrence occurred in 14% of these cats. 1 In a study by Romanelli et al, cats received wide-margin surgical excision where possible with 4–5 cm lateral margins and one fascial plane deep to the tumor. 18 Complete histological resection was achieved in 95% of cases; however, 19/54 (35%) cats with complete resection had local recurrence. 18 This is a higher rate of regrowth than that published by Phelps et al, 1 suggesting that even with complete excision some cats may need adjunctive therapy and it is difficult to know which cats these will be. 18 They also suggested this may reflect the difficulty in histologically assessing all of the surgical margins from a surgical section. 18 Indeed, long-term remissions and cures are possible when the tumors are very small or on limbs where amputation may obtain wide clean surgical margins. Wide-margin first excision results in a longer DFI than marginal first excision, and cats with sarcomas located in the limbs had a median DFI of 325 days vs 66 days for cats with sarcomas in other sites.2,3 There was a longer time to first recurrence in cats that had wide excision than in those with marginal excision, as well as cats that had radical excision (amputation) having a longer time to recurrence than marginal and wide excisions combined. 3
FISSs infiltrate along fascial planes and often have inapparent extensions of neoplastic tissue; therefore, surgery alone is often inadequate to control this disease. In a previous study of 61 cats receiving surgery alone for presumed FISS, the median postoperative DFI was 94 days, with the interval being 66 days for cases coming from a referring veterinarian and 274 days for cases operated at a referral institution, indicating a significant difference if the first surgery is performed by a board-certified surgeon. 3 In the current study, there was no difference in TTP or DSS in these cats whether a general practitioner or board-certified surgeon performed the surgery. However, all cats received adjuvant therapy, some received multiple surgeries and there are too few cats in the general practitioner surgery group to make any conclusions. Despite the lack of significance in this study, it would still be recommended to consult a board-certified surgeon regarding the surgical management of this disease.
It is recognized that the best opportunity for surgical success is with an aggressive first surgery in combination with neoadjuvant or adjuvant radiation therapy.3–9,19,20 The cats in this study with the longest TTP had only one surgery prior to adjuvant radiation therapy, although there was no difference in DSS in these cats and those undergoing multiple surgeries. However, these results are biased as only those cats with recurrence received additional surgery. Many cats with recurrence did not have a second surgery, but their survival numbers are bolstered by those that did have additional surgeries and therefore lived longer. It did not appear that there was any difference in TTP or DSS after the first surgery whether the margins were clean or incomplete. This is not surprising as all cats, regardless of margins, received postoperative brachytherapy.
Owing to the difficulty of complete surgical excision in locations commonly associated with the development of FISS, adjunctive therapy with radiation, chemotherapy or a combination has been recommended.4–9,13,17,19–21 Even with histologically confirmed complete surgical margins, the local recurrence rate may be as high as 50%. 4 In general, the combination of surgery and radiation is appropriate for tumors that have a low cure rate with either modality alone, when there is substantial risk for local recurrence, or when the combination may better preserve normal tissue functionality or appearance. 22 However, it remains unclear if the most beneficial radiation delivery method is pre- or postoperative teletherapy, high-energy electron therapy or postoperative brachytherapy implants.
Preoperative irradiation of soft tissue sarcomas has been utilized.5,8,23 The advantages of preoperative radiation therapy include: a greater anti-tumor effect due to a smaller population of resistant hypoxic cells as local blood supply is not surgically disturbed; a smaller radiation field size decreasing the complication probability; a decreased possibility for autotransplantation of tumor cells that occurs during surgical manipulation; making a surgical excision more feasible by decreasing the field, and, potentially, tumor size; and a decreased number of potentially clonogenic cells that might metastasize during surgical manipulation.22–24 The primary disadvantage of preoperative irradiation is that it may impede postoperative healing of irradiated tissues after surgery and may delay surgery.22–24 Thirty-three cats with fibrosarcoma treated preoperatively with cobalt-60 showed a median DFI of 398 days. 5 These cats were treated daily for 16 fractions, receiving 3 Gy per fraction for a total dose of 48 Gy, and they tolerated the therapy with minimal toxicity. 5
Postoperative external beam irradiation may treat residual disease in the operative field if resection was limited and may make it feasible to deliver higher doses of radiation than is possible preoperatively.22,24 The disadvantages of postoperative external beam irradiation include a delay in radiotherapy until wound healing has occurred, a larger area of treatment field required compared with a preoperative protocol and the potential for a decreased radiation effect due to a subsequent surgically disrupted vascular supply to the healed operative site. 22
One preliminary report of postoperative cobalt radiotherapy for fibrosarcoma in nine cats receiving 200 cGy fractions three times weekly to a total dose of 63 Gy, resulted in a median DFI of 256 days with a mean of 266 days. 25 A study of postoperative electron beam therapy delivering 400 cGy three times weekly for a total dose of 52 Gy reported a mean DFI of 405 days. 6 The recurrence rate in these cats was 41%. 6 The DFI was found to decrease as time between surgery and the start of radiotherapy increased, and if metastasis occurred. 6 An additional study reported a comparison of cats treated with FISS by combining surgery, external beam radiation therapy and chemotherapy vs surgery and external beam radiation therapy alone. 7 In seven cats treated with surgery and radiation administered 5 days weekly for 19 fractions to a total dose of approximately 57 Gy, the DFI ranged from 83 to >1119 days. 7 In 18 cats that received surgery, radiation and doxorubicin chemotherapy, the median DFI was 661 days, which was not statistically different from the group that did not receive chemotherapy. 7
Postoperative interstitial implant brachytherapy has potential advantages over teletherapy radiation. With interstitial implants, the patient needs to be anesthetized no more than twice, once for the implant procedure and potentially again for the explant, although heavy sedation may be adequate for this. In some cases, a higher total dose of radiation may be delivered over a shorter time interval, and perhaps, with the exception of electron therapy, lower radiation doses may be delivered to the underlying normal tissues. 16 The biological effect of brachytherapy with such treatments as iridium-192 varies rapidly with dose rate. 26 A maximum dose achieved that avoids damage to the normal tissues depends on the dose rate. 26 This dose rate depends on the source that is used, as well as how the implants are distributed. 26 In order to achieve a consistent response, the total dose should be varied according to the dose rate used. 26 Total radiation doses of 50–70 Gy in 5–9 days, at dose rates of approximately 30 cGy/h, are typically delivered when an implant is the sole treatment.26,27 The low-dose-rate implants may help spare some of the late responding normal tissues from the potential effects of the irradiation.26,27 Although low-dose-rate irradiation tends to allow for cells to repair radiation damage (sublethal radiation repair), some tissues have been shown to demonstrate an inverse dose-rate effect, resulting in increased cell kill due to the cells accumulating in the relatively radiosensitive late G2 and G1-S boundary phases of the cell cycle.26,27 The sparing effect seen in late-responding tissues may result from the low alpha/beta ratios of slowly responding tissues, implying greater capacity to repair, and with slower rates of repair than acutely responding tissues.26,27 Thus, this differential between late-responding normal tissues and acutely responding tumor is increased if the radiation dose-rate is low enough to allow for the normal tissue to repair.26,27
Disadvantages of interstitial brachytherapy in comparison to teletherapy include the need for an operative procedure, exposure of the operator to radiation during the implant and explant, and the need to isolate the patient while implanted with radioactive material. 16 However, cats may be more tolerant of this than other domestic species. Adverse effects associated with the interstitial brachytherapy seemed tolerable overall, with most of the acute effects occurring possibly as a result of the insertion of the tubes and maybe not truly due to radiation toxicity. Timing of the implants may be important, as each of the three cats that had wound dehiscence were implanted prior to postoperative wound healing. These cats were implanted 1, 4 and 5 days after surgery. Interestingly, these three cats had a long TTP (619, 1381 and 2968 days, respectively) and DSS (680, 1381 and 2968 days, respectively). This is reminiscent of dogs that develop postoperative infections following their limb-sparing surgery for osteosarcoma as they appear to have extended TTP and DSS. 28 This may warrant further investigation. It is also possible that the improved TTP and DSS in these cats could be due to the short time between surgery and brachytherapy decreasing the amount of tumor cell repopulation to occur. Our recommended protocol is to delay brachytherapy until there is complete healing of the surgical incision, typically 14–21 days postoperatively. This may be a potential disadvantage as with postoperative external beam radiation therapy, allowing the tumor time to recover and increasing potential risk for recurrence.
In this study, the TTP and DSS for these cats receiving postoperative brachytherapy appears comparable to surgical excision followed by postoperative external beam radiation therapy. Unfortunately, the rate of recurrence was still high, though comparable at 54.5%. There was no difference in DSS if cats had recurrence or did not have recurrence (undefined vs 1230 days P = 0.17). The lack of statistical difference could be because cats with recurrence received additional therapies such as additional surgeries and/or chemotherapy. Even if a difference exists, there may not have been the power to detect it. Although there was no difference in TTP whether it was in the radiation field or at the margin, the P value for this comparison was 0.06. It is possible that recurrence within the radiation therapy field might represent a more aggressive tumor and be a negative prognostic factor. Additional studies with more animals would be needed to validate this finding. At least five of the tumors in this study recurred at the margins of the radiation field. It is possible that the incidence of local recurrence would have been lower if larger fields had been implanted. However, this approach would be associated with increased expense and potential toxicity. Increasing the average dose to the higher end of the range (6000 cGy) may result in decreased rates of recurrence, taking into consideration size of field and potential risk of adverse effects. Another approach to local recurrence after brachytherapy would be additional surgical resection. Four cats had additional surgery(s) after recurrence that resulted in DSSs of 576, 660, 1091 and 1290 days, respectively.
In the present study, chemotherapy did not appear to make a difference in TTP or DSS in these cats. The number of cats receiving adjuvant doxorubicin chemotherapy was small, and there is some incorporated bias as 75% of cats that received doxorubicin were cats that had recurrence. In other studies, chemotherapy has not been shown to improved survival.6,7
Conclusions
Iridium-192 interstitial implants appear to be an acceptable form of adjunctive radiation therapy for FISS. The therapy was well tolerated and most complications were cosmetic. Three cats developed wound dehiscence; this complication may be prevented by delaying implantation to 14–21 days postoperatively. Brachytherapy with iridium-192 implants resulted in TTP and DSS that are comparable to other reports of adjunctive radiation therapy following aggressive surgery for cats with FISS.
Footnotes
Acknowledgements
The authors would like to thank Dr Claudia Barton for her advice in regards to conception and designing of the manuscript. The authors would also like to thank Ms Vicki Weir for her help with data acquisition and caring for the patients.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
