Abstract
Papillary endothelial hyperplasia (PEH) is a rare soft tissue lesion arising from excessive reactive endothelial cell proliferation described in humans, dogs, and horses. PEH is considered a diagnostic challenge in humans, in which it is frequently misdiagnosed as angiosarcoma. We describe here PEH that developed at injection sites in 2 cats that were initially misdiagnosed as feline injection-site sarcoma by cytology and as subcutaneous angiosarcoma by histopathology. Morphologic features included sharp demarcation from surrounding tissues, and a layered microscopic architecture with an outer fibrous capsule from which emerged fibrovascular stalks covered by a monolayer of factor VIII–related antigen and CD31-positive flat-to-plump endothelial cells. Both lesions had a cystic core containing abundant erythrocytes and fibrin. PEH lesions did not recur in either case. Immunohistochemistry for α–smooth muscle actin and desmin demonstrated that the capsule was devoid of smooth muscle cells, excluding an intravascular origin. PEH in these cats was hypothesized to have developed extravascularly following trauma related to injection. We wish to provide awareness of PEH in domestic cats and of the risk of misdiagnoses leading to overtreatment.
Keywords
Papillary endothelial hyperplasia (PEH) is a rare disease derived from excessive intravascular endothelial cell proliferation; PEH has been described in humans,1,2,7,12 dogs, 8 and horses. 13 The diagnosis of PEH relies on the microscopic finding of a well-demarcated lesion composed of papillary projections of reactive endothelium supported by fibrovascular stalks and intimately associated with organized fibrin.1,2,7,8,12,13 PEH represents a diagnostic challenge for pathologists because it is often misdiagnosed as malignancy and especially as angiosarcoma (AS) on both cytology5,9,11,14,17,22,24 and histopathology.8,12 Formerly known as “Masson tumor” from the name of the pathologist who first identified this entity, 20 PEH was originally considered a neoplastic lesion. Currently, it is interpreted as an excessive or abnormal response to thrombus organization.1,12,18 PEH development seems to be associated with generic vascular trauma with subsequent inflammation and an excessive tissue repair reaction; the organizing thrombus functions as a matrix for the development of papillary structures.1,7,12,18
Here we describe cases of PEH in 2 cats. An 8-y-old, castrated male domestic shorthaired cat (case 1) was presented with a left thigh intramuscular mass with inguinal lymphadenopathy. May-Grünwald/Giemsa–stained fine-needle aspirate preparations of the mass were moderately cellular, with a bloody background, and a prevalent population of plump, spindle-to-round cells occasionally arranged in cohesive groups (Fig. 1). Cells had an intermediate nuclear:cytoplasmic ratio and light-blue cytoplasm with occasional small clear vacuoles. Nuclei were oval, with coarsely clumped, irregularly distributed chromatin, and occasionally contained multiple, small, prominent nucleoli. Occasional multinucleate giant spindle cells were present (Fig. 1, inset). Mitotic figures were rare and morphologically normal. Anisocytosis and anisokaryosis were moderate. In addition, some macrophages with erythrophagocytosis, and mild lymphocytic inflammation, were observed. A generic diagnosis of soft tissue sarcoma was made.
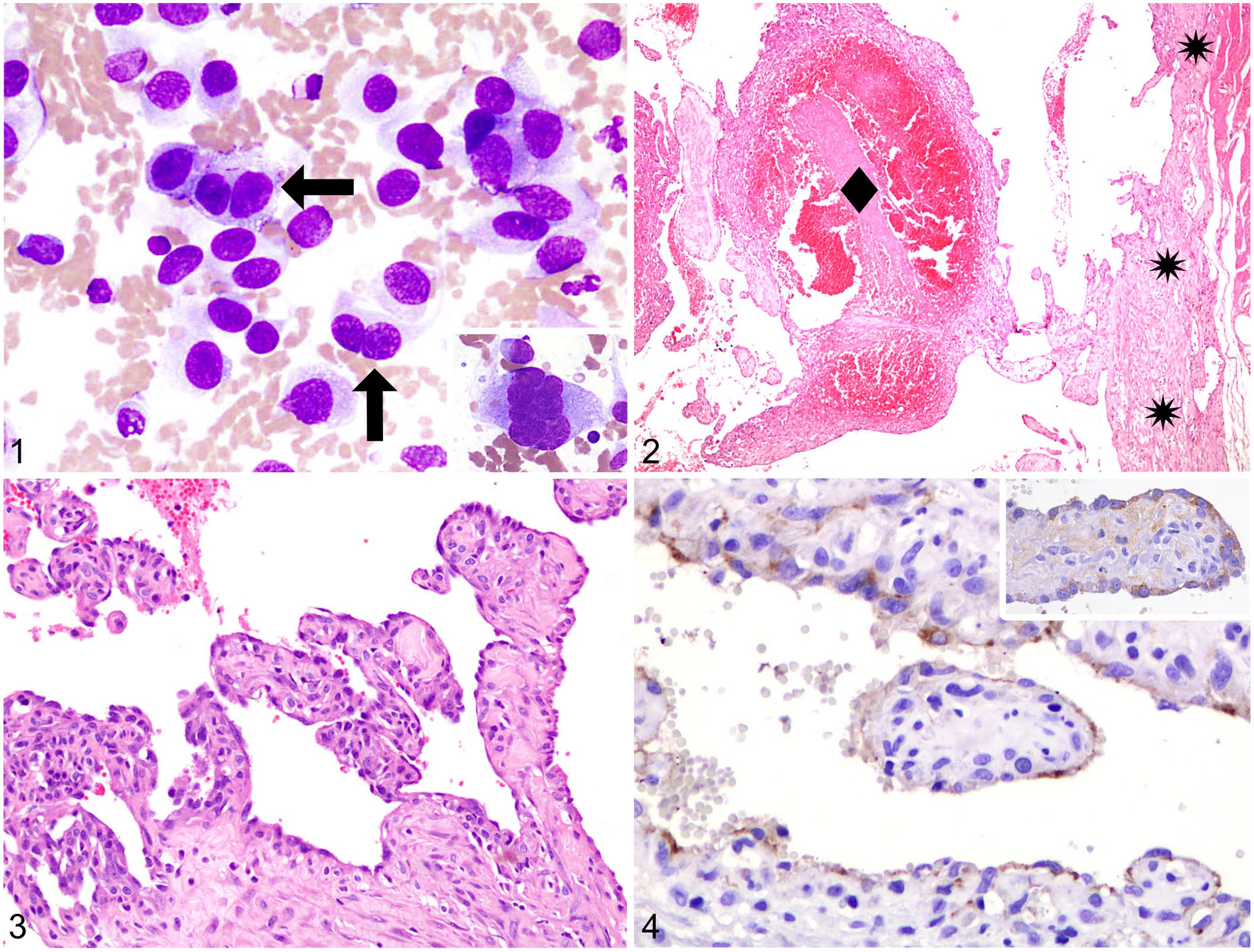
Cytologic, histologic, and immunohistochemical features of feline papillary endothelial hyperplasia (PEH) in 2 cats.
The limb was amputated and submitted with the inguinal lymph node for histopathology. Grossly, a 5 × 4.8 × 4.3-cm sharply demarcated soft mass was observed in the femoral biceps. On cut surface, the lesion was multiloculated and dark-red, contained blood and fibrin clots, and was bordered by a fibrous capsule. Representative samples were fixed in 10% neutral-buffered formalin and processed routinely for histopathology. H&E-stained tissue sections showed an expansile, encapsulated mass with a highly cellular periphery. The lesion had a specific layered architecture with an external capsule composed of reactive fibroblasts and collagen from which emerged fibrovascular stalks covered by reactive endothelium and delimiting irregular vascular channels (Fig. 2). The central core was composed of blood and fibrin clots representing 80% of the mass (Fig. 2). Vascular papillae were intimately associated with large numbers of fibrin thrombi at various stages of organization (Fig. 2). Vascular channels were lined by, and papillary structures were covered by, a single layer of polygonal-to-spindle–shaped, plump endothelial cells with intermediate-to-high nuclear:cytoplasmic ratio and a variable amount of faintly eosinophilic cytoplasm (Fig. 3). Nuclei were oval, with finely stippled to vesicular chromatin, and 1 or 2 basophilic, round, prominent nucleoli. Less than 5% of cells displayed moderate anisocytosis, anisokaryosis, and multinucleation. Mitotic figures were 2 in ten 400× magnification fields (2.37 mm2) and were morphologically normal. The fibrovascular stalks and capsule were multifocally infiltrated by small mature lymphocytes, plasma cells, and macrophages that occasionally demonstrated erythrophagocytosis. Tissues adjacent to the lesion were diffusely edematous but lesion free. The lymph node was reactive with blood reabsorption. Gross and microscopic findings were initially interpreted as primary intramuscular AS. However, a lack of tissue infiltration with sharp demarcation, a specific layered architecture, vascular structures lined by a single layer of cells, minimal cellular atypia, lack of necrosis, and the abundant fibrin thrombi were incompatible with the initial diagnosis. The case was reviewed thoroughly, and the features of the lesion were consistent with a diagnosis of PEH.1,2,7,8,12,13
The cat was euthanized 9 mo post-surgery, following the detection of pulmonary nodules on thoracic radiographs, consistent with bronchogenic carcinoma in imaging, not confirmed on cytology nor histopathology. An autopsy was not permitted.
Following the diagnosis of one case of PEH in a cat, we searched the electronic archives of the biopsy service of the Department of Veterinary Medicine of Milan (DIMEVET; 1998–2021) for feline cutaneous or subcutaneous AS. We retrieved and reassessed 29 cases. In 1 of 29 cases (designated case 2), microscopic features paralleled the lesions of case 1 and were consistent with PEH. The lesion was a 6 × 7 × 5-cm subcutaneous mass on the left thorax from a 16-y-old, castrated male domestic shorthaired cat. The cat had a history of injection at the site of development. Prior to surgery and histopathology, a fine-needle aspirate was obtained by the referring veterinarian and was diagnosed as feline injection-site sarcoma (FISS). The lesion was removed surgically and submitted for routine histopathology and diagnosed as AS. Three months after surgery, the cat developed a cytologically confirmed cutaneous large-cell lymphoma at the site of the surgical scar and was successively lost to follow-up. Microscopic reassessment of the other cases confirmed AS in 28 of 29 cats. Of these, 12 were cutaneous, 11 were subcutaneous, and 5 were cutaneous-to-subcutaneous. No primary intramuscular AS was recorded.
Sections from representative areas of the lesions in both cases were immunolabeled with anti-factor VIII–related antigen and anti-CD31 antibodies, as described previously. 3 Endothelial cells were diffusely factor VIII–related antigen- and CD31-positive with moderate-to-low intensity (Fig. 4), confirming a vascular origin of lining cells and paralleling PEH staining patterns that have been described previously. 1 Sections were also immunolabeled with anti–α-smooth muscle actin (α-SMA; M0851-1A4, monoclonal mouse, 1:2,000; Dako) and anti-desmin (ab32362, monoclonal rabbit, 1:5,000; Abcam) antibodies to assess their expression and the origin of the spindle cells in the capsule and in fibrovascular stalks. Scattered spindle cells in the capsule and numerous spindle cells in fibrovascular stalks were diffusely and intensely α-SMA–positive and desmin-negative (Figs. 5, 6). Accordingly, the presence of smooth muscle was ruled out, confirming the extravascular origin of the lesions. α-SMA–positive spindle cells were interpreted as myofibroblasts or pericytes (for those in the subendothelial lining).
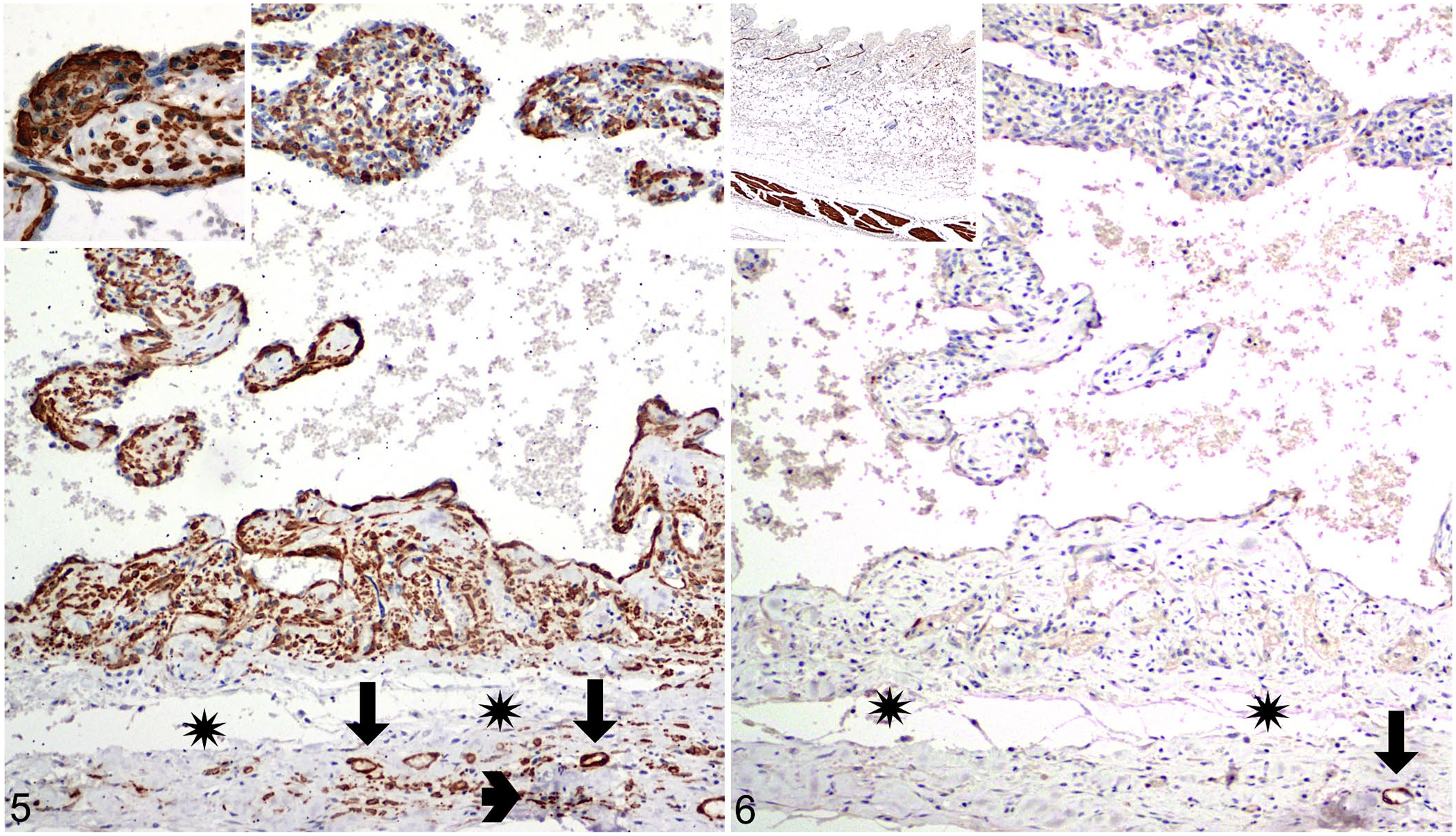
Immunohistochemical expression of alpha–smooth muscle actin (α-SMA) and desmin in a left subcutaneous thoracic mass in a case of feline papillary endothelial hyperplasia (PEH) in case 2.
To our knowledge, PEH has not been reported previously in domestic cats. PEH develops frequently in soft tissues of the extremities and represents 2–4% of soft tissue vascular lesions of the skin in humans and dogs.2,8,12 Despite its putative rarity, the true frequency of PEH in cats is unknown because of a lack of awareness of its existence. In our 2 cases, the histopathologic findings prompted the misdiagnosis of primary AS, paralleling what is frequently reported for PEH in humans. 12 However, the microscopic aspects of AS, including lack of demarcation, infiltrative growth, endothelial multi-layering, possible solid tumor growth, necrosis, and marked cellular atypia with high mitotic activity and atypical mitoses,8,15 were missing in both of our cases of feline PEH. The main features that prompted the suspicion of a benign lesion were the sharp demarcation from normal tissue associated with a fibrous capsule and the lack of infiltrative growth. Diagnostic microscopic findings of PEH in both cats included a papillary proliferative pattern and the association of endothelial cells with the thrombotic material, all features that closely resembled those reported in human cases.1,2,7,8,12,13 Notably, the lesions had a specific layered architecture with an external capsule from which emerged fibrovascular stalks covered by reactive endothelium, and a central core composed of blood and fibrin clots. Moreover, the lesions had a characteristically low cellularity, given that the hemorrhagic and thrombotic core comprised >80% of the lesion.
Three types of PEH have been described in humans: a primary or “pure” form arising in a normal-to-dilated blood vessel, most often a vein; a secondary or “mixed” form occurring as a focal change within a preexisting vascular lesion; and a rare “extravascular” form arising within a hematoma. 12 In primary human PEH, the capsule has been reported to contain smooth muscle cells or elastic fibers as residual components of a preexisting vessel wall. 7 Immunohistochemistry demonstrated the absence of smooth muscles in the capsule in both of our cases, excluding an intravascular origin of the PEH lesions.
In humans, hemangioma is the most common preexisting vascular lesion from which PEH can develop. 12 Additionally, PEH can morphologically resemble sinusoidal hemangioma, a type of hemangioma composed of thin-walled cavernous vessels arranged in a sinusoidal pattern in which papillae covered by endothelial cells are frequently present. 4 Interestingly, descriptions of the coexistence of sinusoidal hemangioma and PEH have been published recently, showing that these lesions share microscopic features and pathogenesis. 6 Cutaneous hemangioma has been reported rarely in cats 21 ; intramuscular hemangioma, sinusoidal hemangioma, or hemangiomas with papillary formation appear not to have been described in cats. Histologically, well-formed, orderly, blood-filled vascular structures closely resembling normal blood vessels were not identified in either of our cases.4,8,21 Additionally, the history in both cats was that of a lesion developing following injection, and no preexisting lesions were reported by owners or referring practitioners. Thus, hemangioma was reasonably excluded as a possible preexisting lesion or differential diagnosis for PEH in both cases. PEH arising from vascular malformations has been reported in humans. 7 No microscopic findings consistent with a preexisting vascular malformation were found in either of our cases. Ultimately, both lesions were interpreted as extravascular and most likely derived from injection-related trauma with hematoma development. 12 The shared history of an injection at the site of development is consistent with this hypothesis, and the injections may have induced the emergence of factors facilitating PEH development. Based on the reactive nature of these lesions, we hypothesized that α-SMA–positive cells could be either myofibroblast (most of the cells forming the stalks) or pericytes (cells in the subendothelial lining of the lesion).
Notably, the lesions were associated with lymphoplasmacytic infiltrates in the capsule and the fibrovascular stalks. This finding is unusual in human PEH, 12 but inflammation is commonly reported in injection-site lesions in cats. 16 Additionally, infiltration of macrophages was observed in our 2 cases, and this is a finding specifically implicated in human PEH development. 18 Following generic vascular bed injuries, migrating macrophages might release fibroblast growth factor (FGF), thus stimulating endothelial cell proliferation. 18 Endothelial cells, in turn, secrete more FGF, exacerbating the effect through an autocrine loop. 18
Noteworthy, the second cat developed lymphoma at the surgical scar, paralleling descriptions of primary cutaneous lymphoma developing after surgery 19 or at injection sites in cats. 23
Fine-needle aspiration cytology is commonly used to investigate superficial and deep masses in domestic animals 10 ; however, cytologic descriptions of PEH are lacking in veterinary medicine. Cytologic features of our case paralleled those reported in human PEH and included moderate cellularity, hemorrhagic background, pleomorphic spindle-to-round cells, hyperchromatic nuclei, and prominent nucleoli.5,9,11,14,17,22,24 In human medicine, these findings are nonspecific and have driven various interpretations including reactive-to-malignant vascular lesions or various types of carcinoma.5,9,11,14,17,22,24 In our 2 cases, initial cytologic findings led to the erroneous but understandable diagnostic hypothesis of FISS because cytologic findings were consistent and because FISS represents a frequent sequela of injection in cats.16,25 The finding of multinucleate giant cells, also observed on cytology from a human PEH case, 11 and lymphocytic inflammation, were observed in both cats and are common cytologic features of FISS. 16 Moreover, the cytologic observation of erythrophagocytosis, and neoplastic aggregates of cohesive cells, are cytologic criteria described in epithelioid AS in dogs, thus explaining the inclusion of AS among the list of differentials in these cats. 3
According to the human literature, there are some cytologic characteristics that, taken together, albeit nonspecific, are considered diagnostic clues of PEH. These include the hemorrhagic background,5,9,11,14,17,22,24 variable presence of fibrin,5,24 acinar-to-papillary architecture with cells surrounding hyaline stalks,9,11,14,17 and the coexistence of spindle and round-to-polygonal cells.9,11,17,22,24 The spindle cells line the capillary networks or are scattered in the background; round cells often surround hyaline cores. 11 The cytology of PEH in the 2 cats had a hemorrhagic background and a population of plump spindle-to-round cells occasionally arranged in loosely cohesive groups, not arranged in an acinar or papillary architecture surrounding fibrin cores. In human reports, the cytologic finding of spindle mono- and multinucleate cells is described but not interpreted.5,9,11,14,17,22,24 Based on immunohistochemical results (α-SMA–positive, desmin-negative cells), the origin of spindle cells described in our cases is consistent with myofibroblasts. Our report confirms that, as in human medicine, cytology alone is nonspecific, does not warrant a definitive diagnosis, and might lead to false-positive diagnoses.
In our 2 cases, cytology and histology led to misdiagnoses of FISS and AS, respectively. FISS is a well-known, malignant, and locally aggressive tumor that has been associated most commonly with injections in cats (it develops in 1–10 of every 10,000 vaccinated cats),16,25 thus a misdiagnosis is possible. On the other hand, primary feline cutaneous and subcutaneous ASs are rare, and to our knowledge, primary intramuscular AS has not been reported in domestic cats. 15 These observations were confirmed by review of our caseload, in which no intramuscular AS were found, and only 29 cases of cutaneous and subcutaneous ASs were found over a 24-y period (in a total of 6,333 feline biopsies). In domestic cats, the systematic data regarding the frequency of local recurrence and metastasis, treatment, and prognosis of ASs are fragmentary. 15 Despite this, researchers have suggested that AS could warrant multimodal therapy, including aggressive surgical resection, radiotherapy, and chemotherapy. 15 On the contrary, PEH has an excellent prognosis after simple surgical excision 7 ; thus, the identification of PEH, and more specifically its differentiation from FISS and AS on cytology and from AS on histopathology, is critical to avoid overtreatment in domestic cats.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
