Abstract
Objectives
The aim of this study was to describe the results of two-dimensional (2D) and contrast-enhancement ultrasound (CEUS) in four cats with intestinal ischaemia.
Methods
Data were collected from hospital records of all cats that had intestinal ischaemia between January 2012 and August 2018. The inclusion criteria were complete abdominal ultrasound examination, colour flow Doppler and CEUS of lesions, confirmation of intestinal ischaemia detected by visual assessment of avascular intestinal segment at surgery, and/or necropsy and histopathology. All images and video clips were reviewed by the same experienced operator.
Results
Four cats with different intestinal ischaemic lesions were included in the study: duodenal perforating ulcer, jejunal necrotising enteritis, necrosis secondary to jejunojejunal intussusception and iatrogenic damage of jejunal arteries. On the 2D ultrasound, all intestinal lesions were characterised by non-specific findings: focal hypoechoic wall thickening with loss of normal layering associated with hyperechoic mesentery surrounding the intestinal tract. CEUS showed a reduced or absent enhancement of the intestinal lesions in comparison to the surrounding perfused wall.
Conclusions and relevance
Intestinal ischaemia is a potentially fatal disorder. Grey-scale, colour and power Doppler ultrasonography are not sensitive for evaluating this condition. Our preliminary findings illustrate the usefulness of CEUS for the detection of intestinal wall impaired perfusion in cats.
Introduction
Intestinal ischaemia (IIS) is a potentially fatal event caused by various diseases and characterised by a rapidly progressive course leading to intestinal necrosis, both in humans and animals.1–4Acute arterial mesenteric thromboembolism secondary to hypertrophic cardiomyopathy,3,5 trauma-associated mesenteric ischaemia, 6 bowel strangulation and ischaemia secondary to foreign body obstruction 7 have been reported in the veterinary literature. Clinical presentation is non-specific and includes vomiting and abdominal pain.3,4 A prompt diagnosis of this condition is essential for a favourable outcome. 3 In humans, the gold standard for diagnosis of IIS is abdominal CT angiography (sensitivity: 82–96%; specificity: 94%) or magnetic resonance angiography.1,2,8–11
Abdominal ultrasonography (US) is commonly used in human and small animal patients with acute abdomen; however, this technique is not accurate when evaluating IIS.1,2,5,8,12 Conventional Doppler US is not suitable for the evaluation of intestinal micro-perfusion because flow velocity and amplitude are too low to be detected, even in a normal intestine.1,8,13 Furthermore, severely increased peristalsis associated with abundant luminal gas may also complicate the feasibility of Doppler US. On the contrary, several studies have demonstrated the accuracy of contrast-enhanced US (CEUS) for the detection of human IIS (sensitivity: 85–94.1%; specificity: 100%), characterised by reduced or absent colour signal on the affected intestinal wall after contrast medium (CM) injection.8,14
In the veterinary literature, CEUS of feline normal intestine is described as a rapid intense enhancement of the serosal and submucosal layers, followed by gradual enhancement of the entire wall section during the early phase. At peak enhancement, a loss of demarcation between intestinal wall layers with a gradual washout of the signal from the intestinal wall in the late phase are reported.15–18 To our knowledge, the use of CEUS in the diagnosis of IIS has been described only in two abstracts.7,19 The first assessed the usefulness of CEUS in IIS secondary to foreign-body obstruction in dogs using a subjective scoring system without describing CEUS patterns. 7 The second reported CEUS features in a case series of small animal gastrointestinal wall impaired perfusion as reduced or absent CM enhancement. 19
The purpose of this study is to describe the CEUS patterns in cats with confirmed intestinal impaired perfusion or ischaemia.
Materials and methods
The medical records of cats with IIS admitted to the Veterinary Teaching Hospital at the University of Bologna from January 2012 to August 2018 were reviewed. Inclusion criteria were a complete abdominal US with colour flow Doppler (CFD) and CEUS of intestinal lesions. Confirmation of IIS was obtained by visual assessment of avascular intestinal segment at surgery/necropsy and/or by histological examinations of biopsy. For each case, the following information was recorded: breed, sex, age, history, physical examination and outcome.
Two-dimensional US and CFD examinations were performed using an ultrasound unit (iU22 and Epiq5G Ultrasound System; Philips Healthcare) equipped with micro-convex and linear-array probes of different frequencies (C5-8, L3-9 and L5-12 MHz). For CEUS examination, linear-array probes of high frequency (L3-9 and L5-12 MHz) were used. CEUS was performed according to a previously described technique.16,20 Briefly, a second-generation CM (Sonovue; Bracco Diagnostic) was manually administered in the cephalic vein as a single rapid bolus dose of 0.5 ml per cat immediately followed by a bolus of 4 ml normal saline. The CM in the target organ was visualised using dedicated imaging software (Pulse Inversion Harmonic and Power Modulation combined). Still images and video clips were reviewed using a DICOM viewer (Show Case; Trillium Technology) by the same experienced operator and the following parameters were recorded for each intestinal lesion: number, location, wall thickness, layering appearance, echogenicity, CFD and CM distribution within the lesion and in the surrounding intestinal loops. In particular, the distribution of the CM within the intestinal wall was evaluated subjectively as present, reduced or absent based on the degree of intestinal mural enhancement and homogeneity at peak intensity (PI). A commercial software program was used for quantitative computerised analysis of the CM blood pool phase (QLAB Quantification Software; Philips Healthcare).
Regions of interest (ROIs) were manually drawn on the above-described intestinal lesions.
A second ROI was placed on an adjacent normal loop (when available in the same image) or segment of intestinal wall adjacent to the lesion. Raw data for each ROI were plotted in time-intensity diagrams and the following perfusion variables were recorded: time to peak from injection (expressed in seconds) and PI (expressed in arbitrary units [au]).
Any additional US findings were also recorded (eg, pneumoperitoneum, peritoneal effusion, regional lymphadenopathy).
Results
Four cats met the inclusion criteria. Age ranged from 2 to 13 years (median 8.5 years). Two cats were neutered females, one was an intact female, while the other one was a neutered male. Three cats were domestic shorthairs and one was a Maine Coon.
One cat (case 1) had no history of gastrointestinal disease before being referred to the hospital. Two cats (cases 2 and 3) and one cat (case 4) had been previously diagnosed with diffuse inflammatory enteropathy and focal jejunal lymphoma by other veterinarians, respectively. At admission, clinical signs included lethargy (n = 4), anorexia (n = 4), abdominal pain (n = 1), vomiting (n = 2), diarrhoea (n = 1) and hypothermia (n = 1).
The ischaemic lesions were associated with: non-neoplastic duodenal perforating ulcer (case 1), focal jejunal transmural necrotising enteritis (case 2), obstruction secondary to double jejunojejunal intussusception (case 3) and iatrogenic damage from accidental resection of jejunal arteries during lymphadenectomy (case 4).
In three cases the final diagnosis of IIS was histological. Particularly, cases 1 and 2 had a focal colliquative necrosis and a focal necrotising transmural enteritis, respectively. In case 3, two different histological findings were found: a severe and diffuse enteritis at the level of external loop (intussusceptions), a diffuse haemorrhagic, and necrotising enteritis with submucosal thrombosis in the internal portion of jejunal loop (intussusceptum).
In the remaining case (case 4), IIS was visualised during surgical procedure. In particular, the latest cat was scheduled for lymphadenectomy of neoplastic jejunal lymph node, but several jejunal arteries were damaged. At the end of surgery, the intestinal wall of a jejunal loop showed a slightly darker colour.
On two-dimensional US (2D-US), three cats showed an asymmetrical or symmetrical focal hypoechoic wall thickening with loss of normal layering at the level of duodenum (case 1; Figure 1a) and jejunum (cases 2 and 4; Figure 1b,c). Case 3 had a contemporary intussusception of two jejunal loops at the same site, characterised by a typical multi-layered appearance of the wall, representing the superimposed wall layers of the two intussuscepta and intussusceptions (Figure 2). Particularly, both invaginated loops were characterised by mild wall thickening with reduced layering and hypoechoic wall. The mesentery appeared thickened and hyperechoic, focally at the level of the intestinal lesion in two cats (cases 1 and 3) and diffusely in the others two (cases 2 and 4). Mild hypoechoic peritoneal effusion was evident in all cats. At CFD examination, none of the focal lesions showed vascular signals within the intestinal wall.
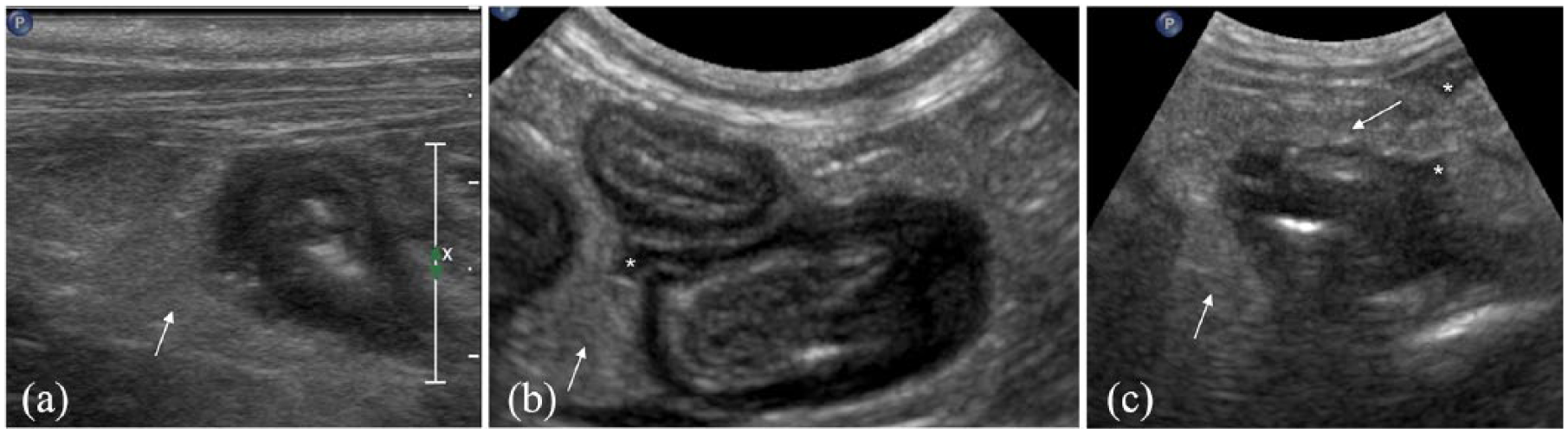
Transverse sonograms of thickened intestinal loops in three cats with intestinal ischaemia due to (a; case 1) duodenal perforating ulcer, (b; case 2) focal jejunal necrotising enteritis and (c; case 4) iatrogenic damage from accidental resection of jejunal arteries. Note the hypoechoic (a,b) asymmetrical and (c) symmetrical intestinal thickening with complete loss of wall layering. The mesentery surrounding the loops appears thickened and hyperechoic (white arrows) and mild effusion is evident (white asterisks)

Transverse sonograms of a double jejunojejunal intussusception in a cat (case 3) obtained with (a) micro-convex and (b) linear array probes, respectively. Note the two inner invaginated loops (white single asterisk) and one outer loop (white double asterisks). Mild wall thickening with reduced layering is evident in one invaginated loop (white arrow) on both images. Lu = lumen; FF = free fluid
CEUS was characterised as severely reduced (case 1) or absent CM enhancement (cases 2–4) of the above-described intestinal lesions compared with the perfused surrounding loops or intestinal wall adjacent to the lesion (Figure 3a–d). Table 1 summarises all 2D-US, CFD and CEUS findings of the intestinal lesion and adjacent loops.
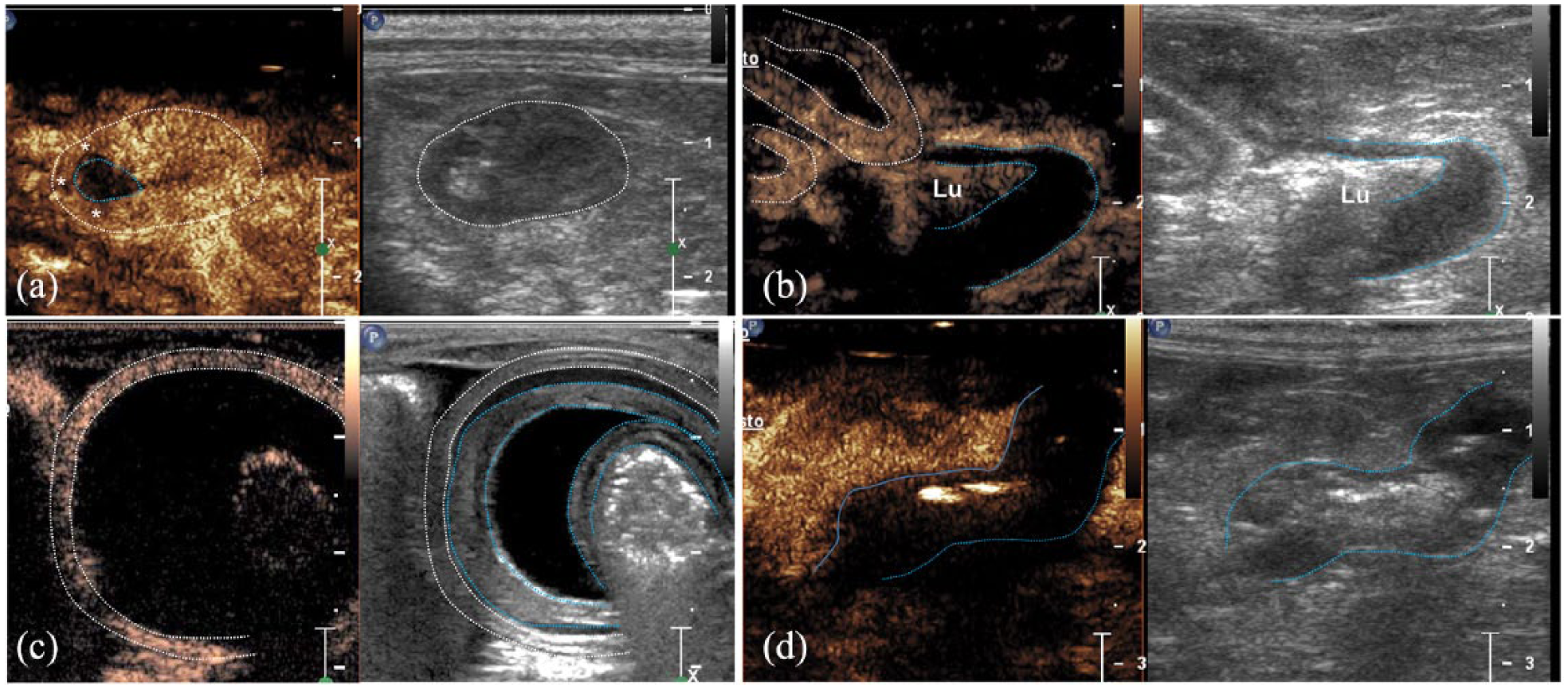
Contrast-enhanced ultrasound images of different intestinal ischaemic lesions in four cats after intravenous injection of contrast medium (CM). Each image illustrates contrast enhancement at peak intensity on the left and grey-scale image on the right. (a) Case 1. Duodenal perforating ulcer: transverse sonogram of duodenum (dotted white ring) showing a reduced CM enhancement of the asymmetric thickening (white asterisks) with a central rounded avascular area (light blue dotted ring) in comparison to the other portion of the wall. (b) Case 2. Focal jejunal necrotising enteritis: oblique sonogram of the same jejunal loop shown in Figure 1b. The asymmetric intestinal wall thickening shows an absence of contrast enhancement (between the light blue dotted lines) vs the normally perfused adjacent loop (between white dotted lines). Lu = lumen. (c) Case 3. Double jejunojejunal intussusception: transverse sonogram of two inner invaginated loops (between the light blue dotted lines) and one outer loop (between the white dotted lines) with absent and normal CM uptake, respectively. (d) Case 4. Iatrogenic damage from accidental resection of jejunal arteries: longitudinal sonogram of a thickened jejunal loop (between the light blue dotted lines) showing an absence of CM enhancement
Two-dimensional ultrasound (US), colour flow Doppler (CFD) and contrast enhanced ultrasonographic (CEUS) findings in a group of cats with gastrointestinal ischaemia
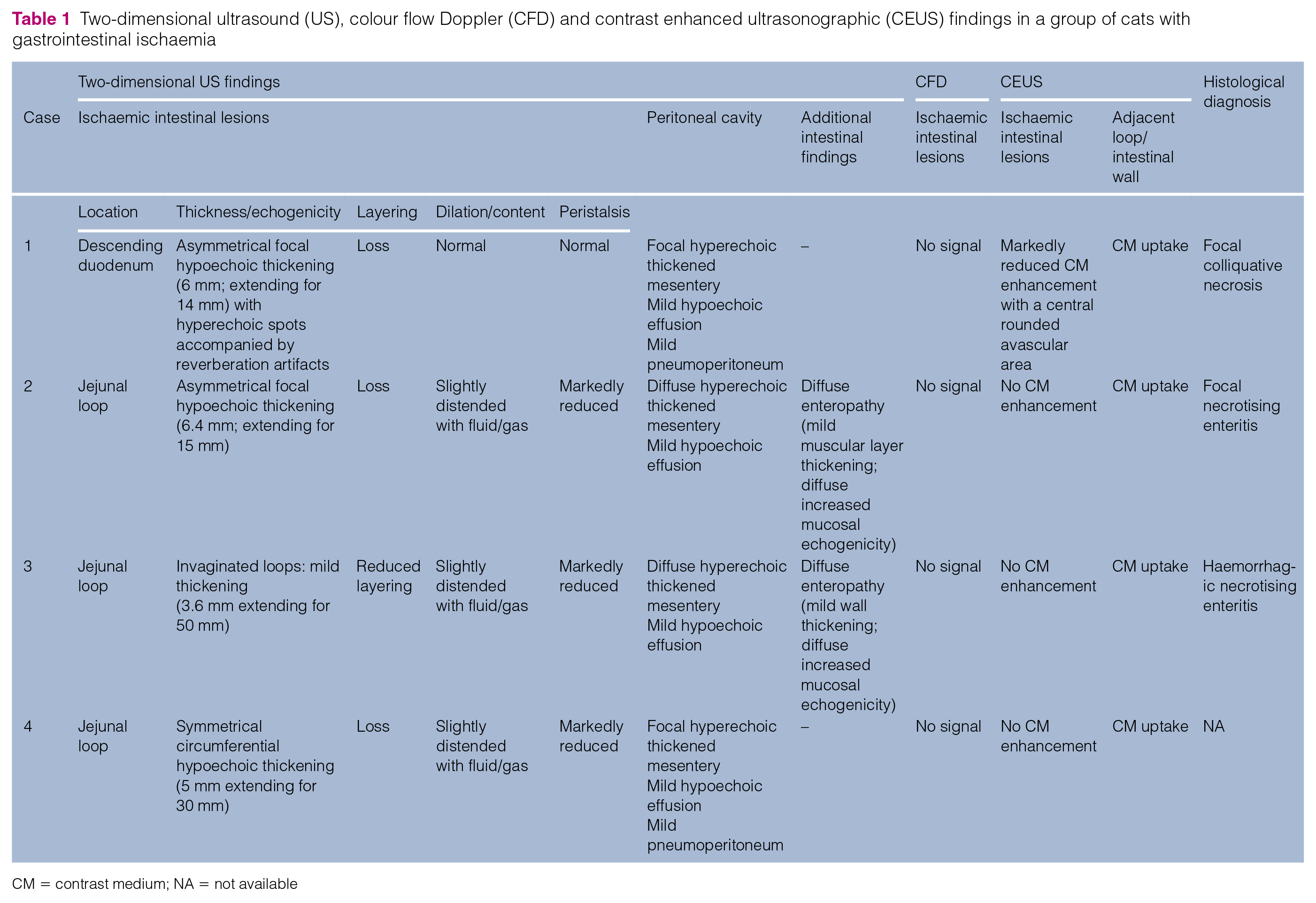
CM = contrast medium; NA = not available
Quantitative computerised analysis of the intestinal wall enhancement was performed in three cats (cases 1, 2 and 4; Table 2). Briefly, duodenal lesion showed reduced enhancement at the PI (3 au) associated with a rounded central avascular area vs the adjacent intestinal wall (18 au) (Figure 4). As they were completely avascular, in the other two intestinal lesions PI was undetectable.
Results of quantitative contrast-enhanced ultrasonography (CEUS) of three cats with gastrointestinal ischaemia

TTPinj = time to peak from injection; PI = peak intensity; au = arbitrary units; ND = not detectable

Signal intensity in arbitrary units (au) as a function of time (s) for an ischaemic intestinal lesion and surrounding jejunal loop from case 2. Two regions of interest have been drawn in order to analyse the enhancement pattern, both from intestinal ischaemic lesion (red line) and surrounding jejunal loop (yellow line). Contrast enhancement from the wall of jejunal loop increases until it reaches a peak intensity around 9 s after bolus injection, while signal intensity from the ischaemic lesion remains at the baseline intensity as a consequence of absent contrast medium uptake. BI = baseline intensity; PI = peak intensity
Two cats were euthanased immediately after US examination owing to poor prognosis (cases 2 and 4). The other two cats (cases 1 and 3) survived surgical treatment and were eventually discharged.
Discussion
Our study illustrates the 2D and CEUS findings in bowel ischaemia in four cats. A diagnosis of IIS might be suspected ultrasonographically, but conventional US features of ischaemia are non-specific and similar to those of traumatic, inflammatory or neoplastic process.21,22 The 2D-US findings of our cases were similar to those previously reported.3,21 A focal hypoechoic intestinal wall thickening associated with reduced/loss of layering, focal or diffuse steatitis and peritoneal effusion were present in almost all cats in this study. In one cat, we also found several hyperechoic speckles associated with reverberation artefacts in the intestinal wall, suggesting the presence of ulceration.23,24 On CFD examination there were no signals within the affected intestinal wall in any cat, because, most likely, diameter and slow blood flow are limiting factors for the detection of blood vessels, even in a normal feline bowel wall. 16 In our cases, CEUS provided more information than 2D-US and CFD. A reduced or absent enhancement after contrast injection in the intestinal wall is consistent with bowel ischaemia similar to that previously reported in humans.8,11,14 Bowel strangulation and superior mesenteric artery embolism are the most important causes of IIS in human medicine.8,14 The focal absence of flow within the intestinal wall was related to necrotic changes secondary to inflammatory conditions (ie, duodenal ulcer and jejunal enteritis) in two cats, while in one case an ischaemic lesion was associated with bowel obstruction secondary to intussusception. PI values of all our intestinal lesions confirm the severe reduction or absence of CM uptake in comparison with adjacent perfused intestinal walls.
There were some limitations in this study. Histopathological diagnosis was available in 3/4 cases, although IIS was strongly suspected because of severe iatrogenic damage to several jejunal arteries in one cat. The heterogeneous causes of IIS, including iatrogenic damage rather than spontaneous disease, associated with a low number of our cases imply that our findings could not be generalised to animals with intestinal wall impaired perfusion. Further studies with a greater number of animals are needed to definitively determine the role of CEUS for the diagnoses of IIS.
Conclusions
Our experience suggests that CEUS can be a useful imaging modality for the assessment of IIS in cats with US evidence of lost or altered intestinal layering. By taking the non-invasive nature of this technique into account, CEUS has potential applications in daily practice as a diagnostic method for IIS.
Footnotes
Author note
Preliminary results were presented as an abstract at the 20th EVDI Annual Conference, Wroclaw, Poland, 31 August to 3 September 2016.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
