Abstract
Objectives
The aim of this study was to compare the effects of butorphanol and buprenorphine, as part of a multimodal analgesic protocol, on recovery and analgesia in cats undergoing orchiectomy.
Methods
In a prospective, randomized, blind clinical trial, 47 adult male cats were randomly assigned to receive either butorphanol (0.3 mg/kg, n = 24) or buprenorphine (0.02 mg/kg, n = 23) in combination with dexmedetomidine (25 μg/kg) and alfaxalone (2 mg/kg) as a single intramuscular injection for the induction of general anesthesia. Isoflurane carried in oxygen was supplemented as needed during orchiectomy. All cats received lidocaine (2 mg/kg intratesticular), meloxicam (0.3 mg/kg SC) and atipamezole (125 μg/kg IM) postoperatively. Pain and sedation scales were applied at baseline, and 2, 4 and 6 h postoperatively. Time to achieve sternal recumbency and to begin eating were also recorded.
Results
Pain scale scores were low and showed no difference between the treatment groups at all time points (P ⩾0.99, all time points). Four cats exceeded the analgesia intervention threshold for rescue analgesia (butorphanol, n = 3; buprenorphine, n = 1). There was no difference in sedation scores between groups at any time point (P >0.99, all time points). Significantly more cats in the buprenorphine group (n = 12) required isoflurane than in the butorphanol group (n = 2) (P = 0.0013; relative risk 6.3, 95% confidence interval [CI] 1.8–23.5). There was no significant difference in time to achieve sternal recumbency (P = 0.96, 95% CI –20 to 20) between groups or in return to eating (P = 0.48, 95% CI –92.0 to 113.5), with most cats eating within 1 h of surgery (butorphanol, 79%; buprenorphine, 83%).
Conclusions and relevance
There were no significant differences in analgesia or recovery between butorphanol and buprenorphine treatment groups as part of a multimodal injectable anesthetic protocol. Butorphanol is associated with superior depth of anesthesia, facilitating injectable anesthesia.
Introduction
Surveys in the UK, South Africa and Canada have shown improvements in the use of perioperative analgesic techniques in both dogs and cats.1–6 Despite this improvement, however, cats continue to be more frequently undertreated with analgesics than dogs for surgical procedures. 6
Butorphanol, a kappa (κ)-opioid receptor agonist and mu (μ)-opioid receptor antagonist, is an attractive choice for use in short procedures of mild to moderate pain intensity owing to its rapid onset of action, analgesic and sedative properties and ready availability.7–12 However, its use as a sole analgesic has been questioned owing to its limited efficacy in the presence of higher pain levels and short duration of analgesia.7–8,13 Buprenorphine is a partial µ-opioid receptor agonist and a κ-opioid receptor antagonist. It is most frequently used in the perioperative period for mild to moderate pain intensity, such as ovariohysterectomy and orchiectomy.7,14 A previous study reported that butorphanol, in combination with medetomidine, given to cats undergoing ovariohysterectomy did not provide sufficient postoperative analgesia when compared with buprenorphine and medetomidine. 7 All cats in the butorphanol group required rescue analgesia in contrast to the buprenorphine group, in which none of the cats required rescue. 7 General anesthesia was induced with intravenous (IV) propofol and maintained with isoflurane in oxygen.
Intramuscular (IM) administration of drugs to provide both anesthesia and analgesia is a common route of delivery in cats undergoing castration or ovariohysterectomy in clinical practice or in large volume spay–neuter settings.15–17 Ideally, these drug combinations will provide an appropriate depth of general anesthesia, sufficient perioperative analgesia and a rapid recovery. An increasingly popular means of assessing perioperative care in human medicine, one that has been applied in cats, is enhanced recovery after surgery (ERAS).18,19 ERAS is based on optimizing perioperative care and monitoring the impact of different strategies on multiple outcomes, such as pain relief and return to mobility and feeding.20–23 Importantly, the desire for a rapid recovery and return to normal function should not be at the expense of analgesia. Applying the principles of ERAS in cats has shown how anesthetic and analgesic protocol choice can shorten recovery to sternal recumbency and return to eating, while maintaining appropriate postoperative analgesia.18,19
The purpose of this study was to evaluate the effects of two IM anesthesia protocols employing multimodal analgesia on recovery and analgesia following orchiectomy in cats. We hypothesized that the combination of buprenorphine–dexmedetomidine–alfaxalone with an intratesticular lidocaine block and postoperative meloxicam would provide better postoperative analgesia than butorphanol (in place of buprenorphine). A secondary hypothesis was that cats receiving butorphanol would have a shorter anesthetic recovery than cats receiving buprenorphine.
Materials and methods
Cats scheduled for orchiectomy at the City of Calgary Animal Services Centre Clinic (May–August 2016) and enrolled through the on-site animal shelter and the clinic’s no-cost spay and neuter program were eligible for study participation. Exclusion criteria were aggressive behavior, body mass <1.5 kg, cryptorchidism, American Society of Anesthesiologists (ASA) physical status classification >2, incomplete administration of IM premedication, requirement for procedures other than orchiectomy to be performed during the same general anesthetic and postoperative hypothermia (rectal temperature <36°C). The study was approved by the University of Calgary Veterinary Sciences Animal Care Committee, operating under the auspices of the Canadian Council on Animal Care (protocol AC13-0146). Written informed consent was provided for all animals.
All cats underwent a pre-anesthetic physical examination. Age or approximate age, breed, weight and ASA status were recorded. Food and water were withheld for 11 and 2 h, respectively, before anesthesia. Animals were housed individually throughout their stay in the clinic. Housing included a litter box, igloo shelter and bedding.
Cats were randomly assigned to one of two groups (block randomization, sequence stored in folder not accessible to investigators; sealedenvelope.com): one group was scheduled to receive buprenorphine (0.02 mg/kg) in combination with dexmedetomidine (25 μg/kg) and alfaxalone (2 mg/kg) IM and the other was to receive butorphanol (0.3 mg/kg) in place of buprenorphine.
Investigators (raters for pain and sedation) were blinded to the treatment group assignment of cats. Following training with an investigator familiar with the sedation and pain scales used (DSJP), all sedation and pain assessments were performed by one of two trained veterinary students (TA or KLMM).
The sedation scale was a simple descriptive scale ranging from 0 to 4, used previously in a similar setting (0 = no signs of sedation; 1 = can stand but wobbly; 2 = sternal; 3 = head lift; 4 = fast asleep, no response to clicker). 18 Sedation was assessed at baseline, 5 mins after treatment injection and 2, 4 and 6 h postoperatively. Cats that were sourced from the shelter population were also scored at 24 h postoperatively.
The pain scales used were the UNESP-Botucatu multidimensional composite pain scale (U-B MCPS) 24 and the revised composite measures pain scale – feline (RCMPS-F). 25 The U-B MCPS scale consists of three subscales: pain expression (scale range 0–12), comprising assessment of overall behavior, reaction to palpation of the surgical site, reaction to palpation of the abdomen or flank and vocalization; psychomotor (scale range 0–12) comprising assessment of posture, comfort, activity and attitude; and physiologic (scale range 0–6), comprising assessment of systemic arterial blood pressure and appetite. The physiologic subscale was not included as baseline data (preoperative appetite and systemic arterial blood pressure) were not available. The RCMPS-F includes assessment of undisturbed behavior, response to interaction with an observer and palpation around a surgical/potentially painful site (scale range 0–16). For both scales, a higher score reflects increasing pain. Pain was assessed at baseline, 2, 4, 6 and 24 h postoperatively. Cats were also scored for demeanor at each time point. 17 The demeanor scale was applied as in a previous study, omitting questions 3 and 4 as baseline data (preoperative litter box use and appetite) were not available (scale range 0–21). 26 Inter-rater reliability between TA and KLMM for the demeanor and pain scales using an intra-class correlation coefficient (ICC) during training was found to be ‘very good’ for all scales (demeanor ICC = 0.98 [95% confidence interval {CI} 0.97–0.99]; U-B MCPS pain expression and psychomotor ICC = 0.94 [95% CI 0.94–0.95]; RCMPS-F ICC = 0.91 [95% CI 0.89–93]).
Anesthesia was induced by administration of the predetermined drug combination as a single IM injection into the lumbar epaxial muscles. Time of administration was recorded and considered to be the start of the anesthesia time. Following injection, cats were left undisturbed but visually monitored in a kennel. Five minutes after injection, sedation was assessed using a standardized noise stimulus (i-Click Clicker; i-Click) approximately 30 cm from each cat’s head. Cats were left undisturbed for an additional 3 mins before removal from the kennel for surgical preparation. Oxygen was administered via facemask at 1 L/min. Cats were placed in lateral recumbency and connected to monitoring equipment. Hair at the surgical site was clipped and the skin was aseptically prepared for surgery. A testicular block was performed using lidocaine (2 mg/kg) divided between the two testicles, at least 1 min before surgery. Throughout the anesthetic period, the following variables were monitored and recorded every 5 mins: pulse oximetry (probe placed on digital pad or ear), heart rate (thoracic auscultation), indirect arterial blood pressure (oscillometric technique with a cuff placed on antebrachium) and respiration rate (visual observation).
Surgery was performed by one of three experienced veterinarians via a scrotal approach. Start of surgery was defined as initiation of the first skin incision. Mosquito hemostats were used to ligate the spermatic cord via a figure of eight technique. The end of surgery and the end of anesthesia were defined as return of the second spermatic cord pedicle into the scrotum, at which time meloxicam (0.3 mg/kg SC) and atipamezole (125 μg/kg IM) were administered. Supplemental anesthesia was provided with isoflurane in oxygen delivered through a face mask connected to a Bain non-rebreathing system if purposeful movement was noted in response to surgical stimulation. The end of surgery was considered time 0 for postoperative assessments. Postoperative rectal temperature was measured at this time with a digital thermometer. If a rectal temperature below 36°C was obtained, cats were placed in an incubator and excluded from the study. Cats were monitored on the surgical table until an ear flick was observed and then returned to their kennel, with the igloo and litter box removed, and placed in lateral recumbency.
During recovery, cats were monitored every 5 mins until they achieved sternal recumbency. Sternal recumbency was defined as all four limbs tucked under the body. Once the cat was sternal, the igloo and litter box were replaced in the kennel and food was offered. Time to eating was monitored hourly after food was provided. Rectal temperature was evaluated again 2 h postoperatively. If cats had a rectal temperature of 36–37°C, heated disks were placed under the towels in the kennel. If a rectal temperature <36°C was obtained, cats were placed in an incubator and excluded from the study. Hyperthermia was defined as a temperature >40°C. Cats that reached the predetermined analgesia intervention threshold of ⩾4 (/16, RCMPS-F scale) 25 were assessed by an experienced veterinarian before administration of rescue analgesia (buprenorphine 0.01 mg/kg IM). Cats receiving rescue analgesia were re-assessed approximately 20 mins later and regular intervals during the study period to ensure they were comfortable. Cats that reached the threshold were assigned that score for the remaining time points, and that score was included in the statistical analyses.
An estimated sample size of 20 cats per treatment group was calculated to identify a difference of two units between treatments for each of the three pain scales applied (RCMPS-F, U-B MCPS pain expression and psychomotor subscales), with a SD of 0.6–1.0 (depending on scale), an alpha (α) of 0.05 and a power of 0.9. Data were tested for normality (D’Agostino-Pearson omnibus normality test) and presented as mean ± SD or median (range). The 95% CI for comparisons between groups is presented where available. Differences between treatment groups were compared using the unpaired Student’s t-test for time between treatment injection and start of surgery, duration of anesthesia, postoperative rectal temperatures, mean systemic arterial blood pressure, heart rate and respiratory rate. Surgical duration, time to sternal recumbency and time to eating were compared using a Mann–Whitney U-test. Isoflurane requirement was compared between groups with a Fisher’s exact test and relative risk (RR) calculated (Koopman asymptotic score). Sedation and demeanor scale scores were compared between groups at each time point (Kruskall–Wallis test and Dunn’s multiple comparison test) and sedation scores were also compared to baseline within each treatment group (Friedman test and Dunn’s multiple comparison test). Pain scores from the RCMPS-F and U-B MCPS subscales were compared between groups using the Kruskal–Wallis test and Dunn’s multiple comparison test. Data supporting the results are available in an electronic repository: https://doi.org/10.7910/DVN/6F4X1P.
Results
Fifty-two cats were recruited. Five cats were excluded for the following reasons: postoperative hypothermia (n = 2); mis-injection (n = 1); inadequate sedation for surgery (n = 1); drug protocol deviation (n = 1). No cats were hyperthermic (>40°C). Data from 47 were included in the study: 24 were in the butorphanol group and 23 in the buprenorphine group. Breeds included were domestic shorthair (n = 31), and domestic longhair (n = 16). There were no significant differences between groups for time from drug injection to start of surgery, surgery and anesthesia duration and postoperative temperature (Table 1).
Demographic and surgical variables for 47 cats anesthetized with intramuscular dexmedetomidine (25 μg/kg) and alfaxalone (2 mg/kg) and either butorphanol (0.3 mg/kg) or buprenorphine (0.02 mg/kg) for orchiectomy surgery

Data are expressed as mean ± SD or median (range). 95% CI is the 95% confidence interval for the mean/median difference
In all cats, sedation scores were 0 at baseline. There were no differences in sedation scores between groups at any time point (P >0.99, all time points). Compared with baseline, there was a significant increase in sedation score 5 mins after drug administration in both groups (P <0.0001). At 2 h postoperatively, one cat in the butorphanol group had a sedation score of 1. All other cats had sedation scores of 0 at this time and all subsequent sedation scores in both groups were 0, with no significant difference to baseline (P >0.99, all comparisons).
No cats were hypotensive and there was no significant difference in mean systemic arterial blood pressure between groups (butorphanol; 126.1 ± 24.7 mmHg, buprenorphine; 128.7 ± 22.2 mmHg, P = 0.71, 95% CI –16.7 to 11.4). Heart rates did not differ significantly (butorphanol; 123.0 ± 17.8 beats per minute, buprenorphine; 119.7 ± 20.4 beats per minute [P = 0.55, 95% CI –7.8 to 14.6). Respiratory rates did not differ significantly (butorphanol; 41.0 ± 6.7 breaths per minute, buprenorphine; 43.0 ± 9.6 breaths per minute [P = 0.42, 95% CI –6.9 to 2.9]). Eighteen cats had low values (<90%) for arterial oxygen saturation (butorphanol, n = 10; buprenorphine, n = 8).
Isoflurane requirement for surgery was significantly different between groups, with two cats in the butorphanol group requiring isoflurane supplementation, whereas 12 cats in the buprenorphine required isoflurane (P = 0.0013, RR 6.3, 95% CI 1.8–23.5; Figure 1).

Supplementary isoflurane requirements in cats receiving intramuscular dexmedetomidine–alfaxalone with either butorphanol (n = 24) or buprenorphine (n = 23) for orchiectomy.
There was no significant difference in time taken to return to sternal recumbency between groups (P = 0.96, 95% CI ™20 to 20), though data variability was greater (wider interquartile range) in the butorphanol group (Figure 2).
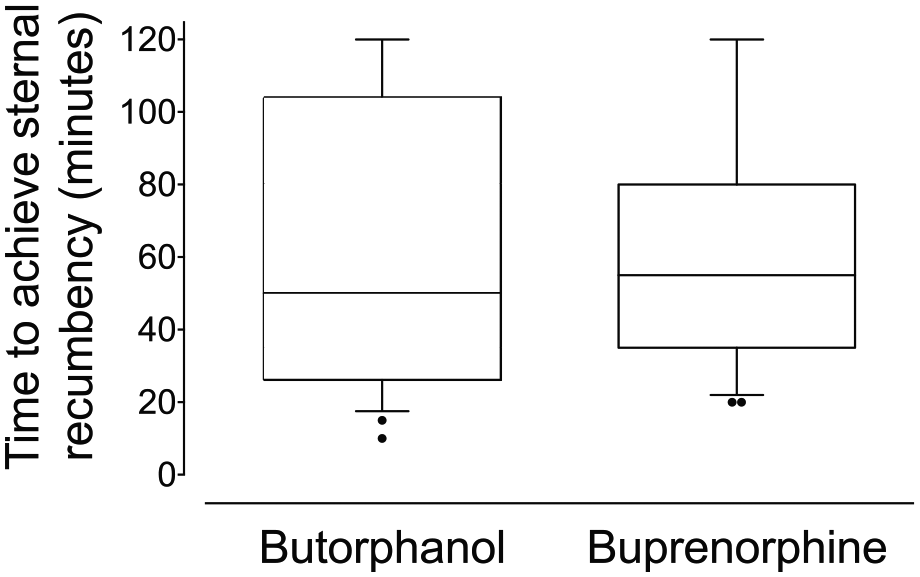
Time to achieving sternal recumbency following orchiectomy surgery in cats receiving intramuscular dexmedetomidine–alfaxalone with either butorphanol (n = 24) or buprenorphine (n = 23). Box and whisker plots show median, interquartile range (box limits) and 10–90 percentiles (whiskers)
There were no significant differences in pain scores between treatment groups, as measured by the RCMPS-F pain scale (P >0.99, all comparisons; Figure 3), U-B MCPS psychomotor (P >0.95, all comparisons) and U-B MCPS pain expression (P > 0.99, all comparisons; see supplementary material) subscales between groups at any time points. Four cats exceeded the analgesia intervention threshold of ⩾4 on the RCMPS-F scale and received rescue analgesia (butorphanol, n = 3; buprenorphine, n = 1). There were no significant differences in demeanor scale scores between groups at any time (P >0.99, all comparisons; Figure 4).
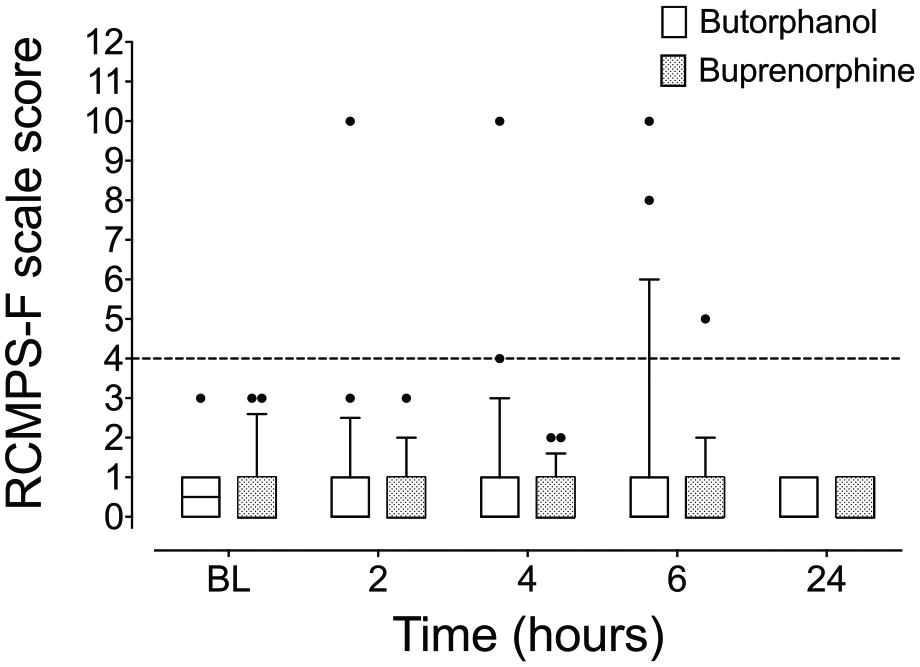
Pain scale scores assessed with the revised composite measure pain scale – feline (RCMPS-F) at baseline (BL) and post-orchiectomy surgery in cats receiving intramuscular dexmedetomidine–alfaxalone with either butorphanol (n = 24) or buprenorphine (n = 23). The analgesia intervention threshold (⩾4/16) is indicated by the broken horizontal line. Four cats exceeded the analgesia intervention threshold. Additional data points reflect that these scores were assigned for remaining time points. Box and whisker plots show median, interquartile range (box limits) and 10–90 percentiles (whiskers)

The behavior (demeanor) of cats at baseline and following orchiectomy surgery. Box and whisker plots show median, interquartile range (box limits) and 10–90 percentiles (whiskers)
There was no significant difference in time to eating between groups 1 h after food was offered (P = 0.48, 95% CI 0–0), with the majority of cats eating by 1 h after surgery (butorphanol, 79%; buprenorphine, 83%).
A limited number of cats were available for the 24 h postoperative pain assessment (butorphanol, n = 6; buprenorphine, n = 5), so statistical analysis was not performed. Pain scores were low for all cats at the 24 h assessment (RCMPS-F score range 0–1, U-B MCPS pain expression score range 0–0, U-B MCPS psychomotor score range 0–6).
Discussion
The main finding of this study was that a single IM injection of butorphanol–dexmedetomidine–alfaxalone in combination with multimodal analgesia provided a more reliable surgical anesthetic plane for orchiectomy in cats compared with buprenorphine–dexmedetomidine–alfaxalone, without significantly compromising postoperative recovery or analgesia.
The substantial reduction in isoflurane requirement observed in the butorphanol treatment group makes this an attractive anesthetic protocol for this procedure when an injectable anesthetic protocol is desired. These findings are consistent with the results obtained by Ko et al, 15 who reported that butorphanol administered at a dose of 0.2 mg/kg combined with ketamine (3 mg/kg) and dexmedetomidine (25 μg/kg) provided a superior depth of anesthesia than buprenorphine (0.03 mg/kg). 15 In their study, 10% (n = 1/10) of cats in the butorphanol group required isoflurane supplementation, whereas 70% (7/10) of cats in the buprenorphine group required isoflurane supplementation. Similarly, Harrison et al 27 reported that cats receiving buprenorphine–ketamine–medetomidine required isoflurane supplementation for orchiectomy but at a lower incidence than in the present study. 27 Two males (3.7%) in their study population required supplemental isoflurane in contrast with 52.2% in the present study. The time between injection of the drug combination and the start of surgery was comparable between studies. A likely explanation for the difference in isoflurane requirement between studies are the differences in drug dosing: Harrison et al 27 used 100 μg/kg of medetomidine (equivalent to 50 μg/kg dexmedetomidine), which is twice the dose used in our study. 27 Harrison et al 27 also used a higher dose of ketamine (10 mg/kg) than the 3 mg/kg used by Ko et al. 15 The use of ketamine is advantageous in reducing the volume for injection compared to alfaxalone, but has several potential disadvantages. These include interference with the pain assessment scales used here, a longer and lower quality recovery, and a delayed return to eating.18,19,26,28
The onset of action of buprenorphine is approximately 35 mins when administered IM based on results of thermal nociceptive threshold testing. 29 As this is longer than our time from injection to the start of surgery, buprenorphine may not have reached its peak effect by the start of surgery. Ko et al 15 suggested that their isoflurane requirement may have been reduced if they had allowed for more time to elapse between injection and the start of surgery. 15 It is unclear if this approach would have been successful, as although this could have allowed for increasing analgesic effects, buprenorphine is typically associated with a low level of sedation.29,30 A further disadvantage of allowing more time for buprenorphine to take effect could be the waning effects of the other drugs given, particularly alfaxalone. Tamura et al 31 reported that alfaxalone at 2.5 mg/kg IM induced lateral recumbency approximately 3 mins after injection, with a peak effect observed at approximately 15 mins. 31 Using a two-injection dosing strategy (buprenorphine followed by alfaxalone-dexmedetomidine) could circumvent this problem but with the disadvantages of increasing personnel workload, handling and a potentially aversive experience for the cats.
There was no significant difference in pain scores between butorphanol or buprenorphine administration at any time point and no significant difference in rescue analgesia requirements. This finding, using feline-specific pain scales developed according to psychometric principles for validation, confirms that of Slingsby et al, 32 who reported low pain scores using an interactive visual analog score and mechanical nociceptive testing for cats following orchiectomy. 32 Their protocol consisted of medetomidine (20 μg/kg, IM) combined with either methadone (0.5 mg/kg), butorphanol (0.4 mg/kg) or buprenorphine (0.02 mg/kg) followed by IV induction with propofol and maintenance with inhaled isoflurane.
It is important to note that all cats in the study reported here received multimodal analgesia with the inclusion of dexmedetomidine, meloxicam and intratesticular lidocaine. Without this approach, it is likely that the number of cats requiring rescue analgesia would have been different. The study was not powered for rescue analgesia as an outcome measure. The choice of IM buprenorphine as a rescue analgesic was based on experience with these multimodal analgesic protocols and predicted potential levels of pain with this procedure. It would not be a suitable rescue analgesic for more invasive procedures, when higher levels of pain could be encountered.
The benefit of multimodal analgesia is supported by the findings of Warne et al, 7 in which pre- and postoperative administration of butorphanol (0.4 mg/kg) in combination with medetomidine (15 μg/kg) did not provide sufficient analgesia for cats undergoing ovariohysterectomy as measured using the U-B MCPS. In contrast, buprenorphine (0.02 mg/kg) administered pre- and postoperatively provided sufficient analgesia. 7 Compared with the current study, these differences likely reflect the nature of the procedures and variations in protocols. Orchiectomy is less invasive, has a shorter operative time and is associated with less tissue handling and trauma when compared with ovariohysterectomy. The present study included a higher dose of dexmedetomidine (25 μg/kg, equivalent to 50 μg/kg medetomidine) which may have further improved intraoperative antinociception. Additionally, Warne et al 7 did not administer locoregional anesthesia and although meloxicam (0.2 mg/kg) was provided, it was not given until 6 h after surgery. 7 Pain assessment was initiated 20 mins after extubation by Warne et al, 7 and sedation was not scored. Pain and sedation were both assessed in the current study but not until 2 h after the end of surgery. Sedation has been identified as a confounding factor that inflates pain scores collected using the U-B MCPS, 33 and it is likely that cats in the study by Warne et al 7 were still sedated this soon after the end of surgery and atipamezole administration. 34
To our knowledge, there are no other studies that combined the triple drug combination used here with an intratesticular lidocaine block and a non-steroidal anti-inflammatory drug perioperatively. This multimodal approach limits direct comparisons between studies but resulted in the low levels of pain observed.
A further obstacle to comparisons with older studies is the relatively recent availability of validated composite measure pain scales developed specifically for acute pain in cats.24,25 In principle, these scales represent a valid and reliable reflection of the pain experience, though it is important not to assume generalizability. 33 Inter-rater reliability was confirmed before starting this study and the concurrent use of demeanor and sedation scales allowed identification of factors known to confound these scales.26,33,34
The effect of postoperative hypothermia on pain scores in cats has not been studied. Klune et al 35 established that postoperative hypothermia interferes with pain scoring in rats using the Rat Grimace Scale. 35 To control for this potential confound, temperature was recorded immediately and 2 h postoperatively in the present study.
ERAS is an important consideration for anesthetic protocols in high-volume clinics to optimize recovery without compromising pain management. It was hypothesized that use of butorphanol would result in improved recovery after surgery in comparison with buprenorphine owing to its shorter duration of action. However, no difference in recovery variables, such as time to sternal recumbency and time to eating, could be identified between groups. The concept of ERAS is to achieve a rapid and smooth return to normal function. This is done by applying a multidisciplinary approach to optimizing numerous aspects of perioperative care, such as analgesia, nutritional state, fluid therapy, physiotherapy and minimally invasive surgical techniques. Applying these principles has been highly effective for multiple procedures and populations in human medicine, resulting in shorter hospital stays, reduced surgical site infections, improved comfort and reduced blood loss.20–23 The application of ERAS in veterinary medicine is clearly in its infancy;18,19 the successes achieved in human medicine warrant further investigation of the benefits for veterinary species.
Limitations of our study include the use of only two of the three subscales of the U-B MCPS. The physiological subscale was omitted. Brondani et al 24 acknowledged that the physiological subscale had limited practicality in a clinical setting and suggested that it could be excluded without compromising the pain assessment. 24 As is common in clinical pain research, aggressive cats were excluded. This, combined with the recently identified interference of aggression with the U-B MCPS and RCMPS-F scales, highlights a limitation in our current ability to assess pain in this population. 33 A further limitation is that because pain scores were generally low and both treatment groups received multimodal analgesia, the relative contributions of individual analgesic agents is unknown. While it is possible that analgesia would have been acceptable with the omission of the opioids, the goal of the study was not to investigate opioid-free analgesia. Buprenorphine enhances the antinociceptive effects of medetomidine 30 and butorphanol is a useful sedative adjunct. 36
Conclusions
The combination of butorphanol–dexmedetomidine–alfaxalone with an intratesticular lidocaine block and postoperative meloxicam provides acceptable postoperative analgesia for orchiectomy surgery and is associated with a superior depth of anesthesia than buprenorphine (in place of butorphanol). Both protocols offered comparable recoveries, though the anesthetic-sparing effects of butorphanol, characterized by lower inhalant anesthetic requirement, make it a more attractive choice.
Supplemental Material
Figure S1
Pain scale scores from the pain expression and psychomotor subscales of the UNESP-Botucatu multidimensional composite pain scale (U-B MCPS)
Footnotes
Acknowledgements
The authors thank Arlene Johnson, Doris Porter, Karen Brick, Tiffany Ramsden, Sonja Gavora and Wendy Weed of the City of Calgary Animal Services Centre Clinic (Calgary, AB, Canada) for technical assistance.
Supplementary material
The following file is available online:
Figure S1: Pain scale scores from the pain expression and psychomotor subscales of the UNESP-Botucatu multidimensional composite pain scale (U-B MCPS).
Author note
This paper was presented in part at the University of Calgary Faculty of Veterinary Medicine Summer Undergraduate Research Experience Research Day, Calgary, Canada, August 26, 2016.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Financial support for this study was provided by the Office of Community Partnerships of the University of Calgary, Faculty of Veterinary Medicine and a Zoetis Investment in Innovation Fund (DP). TA and KLMM received summer studentships from the Veterinary Clinical and Diagnostic Sciences Department (University of Calgary, Faculty of Veterinary Medicine) and Summer Undergraduate Research Experiences Program (University of Calgary).
Ethical approval
This work involved the use of nonexperimental animals only (owned or unowned), and followed established internationally recognized high standards (‘best practice’) of individual veterinary clinical patient care. Ethical approval from a committee was not necessarily required. The study was approved by the University of Calgary Veterinary Sciences Animal Care Committee, operating under the auspices of the Canadian Council on Animal Care (protocol AC13-0146).
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work for the procedure(s) undertaken. No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
