Abstract
Objectives
The aims of this descriptive study were to identify risk factors for feline Microsporum canis infection at shelter intake, to describe screening test accuracy, and to refine confirmatory testing time frames.
Methods
Database records for the general feline population and intake data, medical records and fungal culture logs for cats diagnosed with M canis at a limited admissions shelter were accessed retrospectively for a period of 2 years.
Results
The feline population at the study shelter had a prevalence of M canis of 1.8% (95% confidence interval [CI] 1.6–2.0%). Kittens were eight (95% CI 4.8–13.5) times more likely to present with dermatophytosis than adults. Although more cats presented with M canis during summer and autumn, season was not significant when the model was controlled for age. Owner-surrendered cats were half as likely (95% CI 0.41–0.77) as transported cats to be diagnosed with M canis. Wood’s lamp examinations had a sensitivity of 66.8% (95% CI 60.2–73.4) and a specificity of 74.8% (95% CI 64.2–85.1) compared with dermatophyte test medium (DTM) culture. In 78.8% (95% CI 61–91) of littermate or household groups with mixed Wood’s lamp results, all cats were DTM culture positive. Under consistent incubation conditions, 202/202 diagnostic DTM plates for M canis-infected cats showed recognizable colony growth before 7 days (median 4 days), and 19/19 fomite carrier cat cultures showed growth before 12 days (median 5 days).
Conclusions and relevance
Applying the results of this study to shelter protocols could optimize diagnostic approaches and shorten the length of stay for shelter cats and kittens, resulting in streamlined shelter operations and improved feline welfare.
Introduction
Feline dermatophytosis, especially infection with Microsporum canis, is a significant concern for animal shelters, veterinary practices and pet owners. The disease, although clinically mild and self-curing, is problematic because it is contagious, zoonotic and environmentally hardy.1–3 To prevent and contain spread, shelters strive to recognize infected cats at intake, 4 first through exposure history and perceived risk factors, by using inexpensive point-of-care screening tools during intake examination, and, secondly, through follow-up diagnostic confirmation. When to isolate, whether to treat while waiting for diagnosis and how to proceed with asymptomatic littermates or housemates can be unclear, as the reported incubation period for M canis is 1–3 weeks. 3 Providing biosecure, humane housing, and socialization and enrichment during long-term isolation 5 is physically and financially challenging, and represents a barrier for many shelters attempting treatment or even pursuing diagnosis.
Previous studies have reported an increased risk of M canis infection in cats <1 year of age,6,7 the Persian breed, 7 and cats with feline immunodeficiency virus infection, 8 and exposure factors such as warm or humid climate,2,3,9 warm season,6,7 living in catteries or shelters2,3 or living stray or outdoors.2,9
Screening at shelter intake is performed using diagnostic procedures such as physical examination and alternative light-source examination of the skin. Under the Wood’s lamp (WL) wavelength of 365 nm, M canis-infected hairs may fluoresce pale green, 4 which can then be plucked for a definitively diagnostic trichogram.2,4 However, not all infected animals fluoresce. Though incorrect, 2 a WL sensitivity of 50% is still commonly repeated.3,10 As other substances can also fluoresce at this wavelength, specificity of diagnosis is dependent on operator experience and cooperation of the cat. 2
Shelters commonly reach for one of two options for confirmation testing.11,12 Real-time PCR available through a commercial laboratory is moderately expensive but offers a diagnostic turnaround of 1–3 days. 13 Recent studies favorably compare PCR with culture for initial diagnosis,11,14,15 but challenges exist in distinguishing mechanical carriers from truly infected cats using PCR. Dermatophyte test medium (DTM) fungal culture with microscopic identification of fungal macroconidia has been considered the gold standard for dermatophytosis detection,3,4 has been used by shelter clinicians to help distinguish infected from fomite carrier cats1,4 and remains the best method for determining mycological cure.11,12,15 However, turnaround time for DTM culture is very slow in a shelter context: the current literature recommends waiting 14–21 days before a plate is considered sufficiently diagnostic.4,16,17
Time spent in the shelter, also known as ‘length of stay’, is a carefully monitored metric because the swift movement of animals from intake to outcome (adoption, transfer, euthanasia, etc) provides better individual welfare and allows the shelter to serve more animals.18–20 Optimizing common M canis screening and diagnostic methods could shorten the length of stay if time to diagnosis for infected cats, mechanical carrier cats, uninfected cats and their housemates can be safely shortened.
The first objective of this retrospective descriptive study was to determine the prevalence of M canis infection and investigate risk factors for incoming cats at a large, limited admission animal shelter in Northwestern USA. The second objective was to describe M canis test characteristics in a shelter context in more detail, including sensitivity and specificity of WL testing compared with initial DTM culture at this shelter, and evaluating the prevalence of M canis in affected litters or household groups. Finally, the study investigated initial daily DTM culture growth profiles with the goal of helping clinicians more precisely understand the time frame for and benefits/limitations of fungal culture confirmation.
Materials and methods
Data collection
Data were gathered retrospectively from a single large, limited admissions, non-profit humane society operating in the Pacific Northwestern USA between 15 May 2014 and 15 May 2016. Permission to use this data was given by the shelter’s medical director and vice president of shelter operations. Incoming and outgoing animal reports from the shelter’s population management software (Shelter Buddy; The Animal Shelter Database System, RSPCA Queensland, Australia) were accessed in March of 2017. Data used in this study included animal identification number, date of admittance, age category and intake source, including origin shelter if transferred. Except when re-admitted for dermatophytosis treatment, only the first instance of each cat in the database during the study period was used. Each of the 11,214 adult cats and kittens admitted to the study shelter during the study period had both an incoming and outgoing status and date.
Cats were aged by shelter staff at intake, and placed in the ‘kitten’ category if 6 months old or younger; the term ‘cat’ is used here to indicate all age groups. Intake categories included shelter-to-shelter transport, owner relinquishment, cruelty investigation seizures (including hoarding cases) by humane officers, kittens born to queens in foster care, animals not reclaimed after public assistance holds, cats returned after adoption and strays. Except in select circumstances, per shelter protocol, stray cats were immediately transferred to a neighboring municipal organization holding the jurisdiction’s animal control contract. Animals brought to the study shelter specifically for euthanasia services or dead on arrival were excluded from all aspects of this study.
Handwritten DTM culture monitoring forms were reviewed to identify all feline patients that shelter clinicians had suspected of dermatophytosis during the study period, and to determine daily and final results of all fungal cultures performed during this time. Medical records of all cats for which diagnostic cultures were performed were accessed to determine the results of examination with a standard plug-in WL, when recorded. Equivocal WL results (when cases were ‘borderline’ or the veterinarian was unsure) were coded as positive. Suspect cats were sorted into ‘household groups’ based on pre-intake contact; this included related or unrelated adult cats previously living together in one residence, litters of kittens, or family units made up of a queen and kittens. Data retrieval and curation were performed by the first author.
Diagnostic techniques
Shelter clinicians were alerted to the possibility of feline dermatophyte infection by available history, the presence of gross lesions on intake examination, or fluorescence on routine WL screening by staff. On rare occasions, cats were admitted to the shelter with previous diagnosis or suspicion of M canis infection. Once flagged, a second WL examination was performed by the shelter clinician and an initial DTM fungal culture was performed if dermatophytosis was suspected. Generic commercial DTM culture plates were inoculated with individual animal haircoat samples using the sterile toothbrush technique. Sample plates were kept in a darkened incubator at 25–27°C and 30–35% humidity.
For each of the 294 initial DTM cultures performed during the study period, daily monitoring logs were filled out by technical staff every weekday. The day of first recognizable red color change or white colony growth (‘recognition of growth’) was recorded for each plate. Microscopic confirmation of dermatophyte species macroconidia or contaminant structures was performed for each plate by clinicians using standard techniques. One cat was diagnosed with Trichophyton mentagrophytes and two with Microsporum gypseum during the study period; owing to small numbers and applicability these cats were excluded from this study. For the remaining 291 culture plates, M canis dermatophytosis was diagnosed if the pretreatment culture plate grew more than four characteristic fungal colonies, a mechanical carrier state was diagnosed if pretreatment culture grew 1–4 colonies,4,21 and a cat was considered negative if the culture plate continued to show no growth or only contaminants grew, during a minimum of 14 days’ incubation. 16
Population management
At the clinician’s discretion, dermatophyte suspect cats were either isolated in place via signage while waiting for diagnosis or moved immediately to a designated ‘ringworm’ treatment ward. All cats in this ward were treated with daily oral and bi-weekly topical antifungal compounds until two consecutive 14 day cultures with no dermatophyte growth were observed.4,16,21 If initial cultures indicated carrier state, showed no growth after 14 days or when infected animals achieved mycological cure, then cats were bathed and moved through the normal sheltering processes into the general population.
Statistical methods
Prevalence is used to describe the frequency of dermatophytosis based on the assumption that most cats were infected or incubating the infection upon intake. As the proposed risk factors (eg, age, source, season) were categorical, association with each factor and diagnosis of dermatophytosis was assessed using the χ2 test of independence. Factors significant at P ⩽0.20 were further evaluated using unconditional logistic regression 22 to assess joint effects and control confounding. Regression coefficients were exponentiated to obtain odds ratios, and those significant at P <0.05 were retained in the final model. Sensitivity, specificity and prevalence-based predictive values of WL examination were evaluated in comparison to DTM fungal culture as the gold standard, using standard formulas. A Kaplan–Meier survival analysis was used to compare time-to-diagnosis for fungal growth between infected and non-infected fomite carrier cats. As no data were censored and positives occurred quickly, significance was tested using the Breslow (generalized Wilcoxon) statistic. χ 2 tests, logistic regression and time-to-event analyses were performed in SPSS (IBM SPSS version 24). Proportions and test characteristics were calculated using Microsoft Excel (version 16) and an online proportion confidence interval (CI) calculator. 23 Significance was set at P <0.05.
Results
Epidemiology of cats with dermatophytosis
Of 11,214 cats admitted to the study shelter during the study period, 202 were diagnosed with M canis dermatophytosis infection, and 19 were diagnosed as M canis carriers. Overall prevalence of M canis infection in the study population was 1.8% (n = 202/11,214; 95% CI 1.6–2.0), which represented 69.4% (n = 202/291; 95% CI 63–74) of the cats suspected by clinicians of having dermatophytosis infection.
Time to recognition
Time until the first culture was taken had a range of 10 days before to 66 days after shelter intake. Median time from intake until recognition was 3 days; 69.8% (n = 141/202; 95% CI 63–76) were cultured within 2 weeks of intake and 90.1% (n = 182/202; 95% CI 85–94%) within 1 month of intake. Five cats were diagnosed after adoption and returned to the shelter for treatment.
Age
Kittens aged 6 months and younger had a significantly higher prevalence (2.9%; 95% CI 2.5–3.3) of dermatophytosis compared with adult cats (0.2%; 95% CI 0.1–0.4% [P <0.001]), as seen in Table 1. In this cohort, kittens made up 92.1% of all feline M canis diagnoses while representing only 56.7% of feline intakes. Kittens were 9.1 (95% CI 5.4–15.2; P <0.001) times more likely to be infected with M canis than adult cats.
Prevalence of infection and odds ratios (ORs) associated with potential risk factors for Microsporum canis in study cats
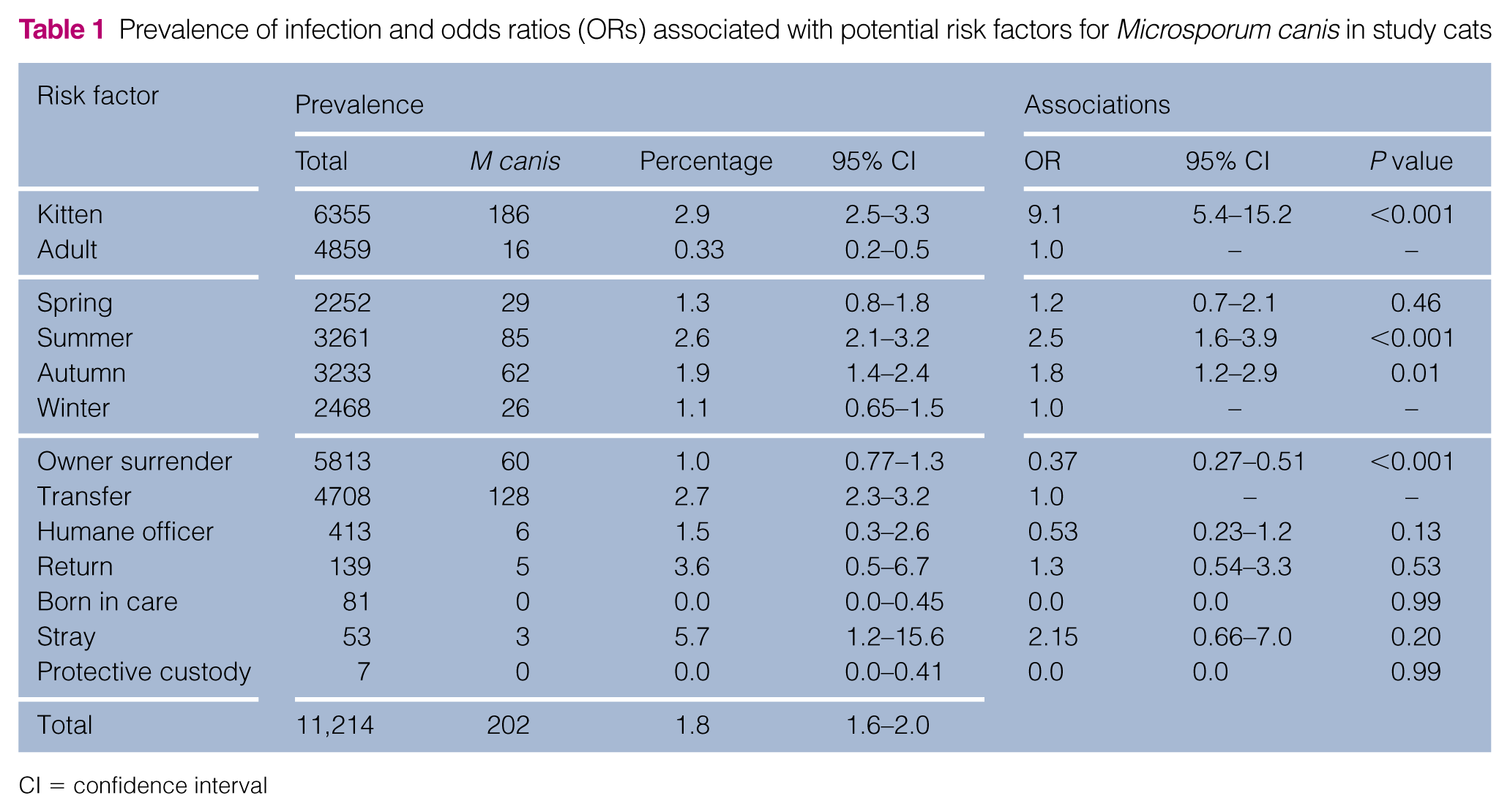
CI = confidence interval
Seasonality
Diagnosis of M canis in cats at the study shelter showed a statistically significant variation by season: those entering the shelter in the summer were 2.5 times more likely (95% CI 1.6–3.9; P <0.001) and those entering in the autumn were 1.8 times more likely (95% CI 1.2–2.9; P = 0.01) to be infected compared with those entering in the winter (Table 1).
Cat source
Cats arrived at the study shelter from a variety of sources (Table 1). The prevalence of dermatophytosis in cats transferred from other sheltering organizations was 2.7% (95% CI 2.25–3.18) vs 1.0 % (95% CI 0.77–1.29) among cats surrendered by their owners. Owner-surrendered cats were less than half as likely (OR 0.37; 95% CI 0.27–0.51 [P <0.001]) to be infected than transported cats. No other sources of intake were found to have a significantly different risk compared with transferred cats.
For the 4613 cats with recorded information regarding transport, 79 transfer partners were identified. Of those 79 organizations, 28 shelters in five states (CA, OR, WA, UT, KS) transferred cats with M canis to the study shelter. The prevalence of M canis did not differ by state (P = 0.85). Among transport partners who transferred >50 cats to the study shelter (n = 24), average M canis prevalence in the transported population was 3.3%, and ranged from 0% (n = 0/302) to 15.1% (n = 8/53; two litters of four kittens transported 1 year apart.)
Multivariable model
Age, season and source were further evaluated in a multivariable logistic regression model. Age distribution of cats varied significantly by season, and these factors showed co-linearity in the regression model. As age was a stronger predictor, season was removed from the model. Intake categories with no M canis cats were also removed (born in care, protective custody). Controlling for source, kittens were 8.0 times more likely to be diagnosed with M canis than adults (95% CI 4.8–13.4, P <0.001), and owner-surrendered cats were about half as likely to be diagnosed with M canis as transport cats (95% CI 0.41–0.77; P <0.001), as shown in Table 2. Transport kittens represented 30.1% (n = 3455/11,214) of the shelter cat population during the study period.
Final multivariable model for Microsporum canis infection in study cats

OR = odds ratio; CI = confidence interval
Test characteristics
Sensitivity and specificity
Of 291 cats with diagnostic DTM cultures, 266 had recorded WL results (Table 3). For this analysis, carrier cats and those with no fungal growth were considered uninfected.
Wood’s lamp (WL) and dermatophyte test medium (DTM) culture test results for 266 cats

In this population of dermatophytosis suspects, the sensitivity of WL examination was 66.8% (95% CI 60.3–73.4%), and the specificity was 74.8% (95% CI 64.2–85.1%). The prevalence of positive DTM culture results in the suspect population was 74.8% (n = 199/266), so the predictive value of a positive WL result was 88.7% (95% CI 82.5–93.3) and the predictive value of a negative result was 43.1% (95% CI 33.9–52.6). At the sensitivity and specificity calculated above, Figure 1 shows the predictive values of negative and positive WL results at varying M canis prevalence levels, including the 2.9% (prevalence within kittens) and 1.8% (total population prevalence).

Predictive values of Wood’s lamp examination at different prevalence levels of Microsporum canis
Household penetration
Shelters frequently admit litters of kittens or cats from multi-cat households, and the history, physical examination or WL results from one cat cast clinical suspicion on the others. In total, 226 cats in the dermatophyte suspect population were taken into the shelter with littermates or co-housed adults, representing 65 household groups. The average group size was 3.4 cats. Within 33 household groups, member cats showed a mixture of positive and negative WL results. In 26/33 (78.8%, 95% CI 61–91%) of these mixed WL groups, all cats tested positive for M canis on fungal culture; in 4/33 groups culture results were also mixed, and in 3/33 groups all members were culture negative (Table 4).
Dermatophyte test medium (DTM) culture results in household groups with mixed Wood’s lamp results

CI = confidence interval
Of the WL-negative individual cats that were members of mixed WL result groups (n = 66), 69.2% (95% CI 55.6–80.1) were infected with M canis.
DTM culture growth parameters
Pretreatment culture plates that grew >four colonies (cats considered infected with M canis) had colony growth recognized at a median of 4 days (95% CI 3.7–4.3). All 202 plates in this group were positive by 7 days (Table 5). Plates that grew 1–4 colonies during the 14 day plate incubation and monitoring period (cats considered to be M canis fomite carriers) had colony growth recognized at a median of 5 days (95% CI 3.9–6.1). Only 4/221 plates (1.8%; 95% CI 0.5–4.6) that grew any colonies took longer than 7 days to grow (range 8–12 days). Figure 2 shows a Kaplan–Meier curve of time to diagnosis for plates in the infected and fomite carrier categories.
Day of recognition of first growth on dermatophyte test medium culture by diagnostic category

NG = no growth
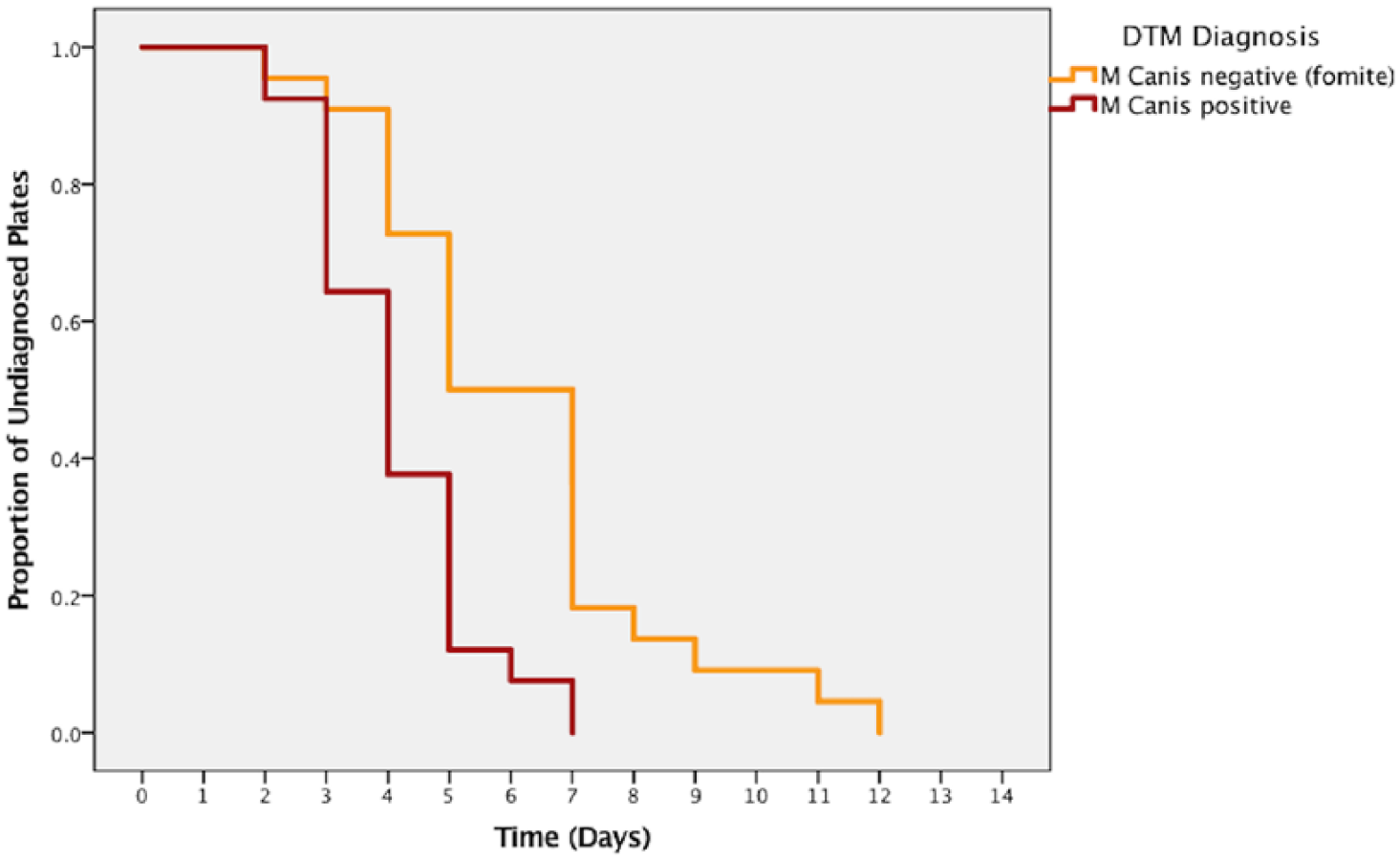
Time to first day of recognized Microsporum canis colony growth by diagnostic category. DTM = dermatophyte test medium
Breslow (generalized Wilcoxon) analysis showed a significantly shorter (P <0.001) time until the appearance of culture growth for plates that indicated infection with M canis and those that indicated fomite carriage.
Discussion
This study included a large number of individual cats but only captures the population characteristics and practices of one sheltering organization in one region of the USA over a short time span. This shelter differs from many others in that it: (1) has a robust feline transport program; (2) does not routinely admit stray or feral cats; and (3) occasionally admits cats known or suspected of having M canis infection. Therefore, results presented here, while instructive, may not be generalizable to all shelters.
Overall prevalence of M canis in cats admitted to this Pacific Northwest shelter during the 2 year study period (n = 202/11,214 [1.8%]; 95% CI 1.6–2.0) was higher than the prevalence described in several other studies, including cats presenting to a teaching hospital in upstate New York (n = 45/22,135 [0.2%]), 24 those admitted over 2 years at a Dutch shelter (n = 8/1335, n = 5/1413 and n = 5/1549; 95% CI 0.32–0.6) 25 and those seen by general practitioners in the UK (n = 2/154 [1.3%]). 26 This prevalence was very similar to that reported in cats admitted to a Midwestern US shelter over 2 years (n = 94/5644 [1.7%]), 1 but less than half of that reported in a study of cats in four shelters in California and Washington (n = 11/200 [5.5%]) 27 Differences in prevalence may relate to different methods of classification or diagnosis, length of study period, type of organization, regional climate, proportion and source of kittens, and other factors.
In the final model, kittens were 8.0 (95% CI 4.8–13.5) times more likely to be diagnosed with dermatophytosis than cats over 6 months of age, consistent with previous studies that found that cats under 1 year of age had an increased risk.6,28 Increased susceptibility to dermatophytosis in young animals can be explained by a number of factors, including immune system immaturity, lack of previous immunity, skin microtrauma from siblings or ectoparasites, 2 and frequent close contact with other cats during socialization periods. Increased stress, from living in the shelter and/or the strain of transport, may also contribute to increased susceptibility in this population. 29 Although an increase in the prevalence of dermatophytosis during summer and autumn months has been previously described 28 and was also observed in this study, in this shelter seasonal variation was a cofactor of the seasonal distribution of kitten intake.
The higher prevalence among transported adults and kittens in the final model (odds ratio 1.78, 95% CI 1.30–2.44) could be related to warmer or more humid source climates, endemic/enzootic dermatophytosis in source shelter populations,1,30 or the original intake category of the cat (free-roaming vs owned) 2 at the source shelter. Further analysis found that dermatophytosis prevalence did not differ by state of intake, and no transfer partner transported a particularly high proportion of infected cats.
At least a dozen cats admitted to the study shelter were transferred by partner shelters specifically for the treatment of suspected M canis, although many turned out to be DTM culture negative or fomite carriers. Excluding these known cases failed to meaningfully alter the results presented above, and as this was routine practice for the study shelter and suspicion history was not available for all incoming cats, these cases were retained in the study population.
A number of studies have found that free-roaming or stray cats are more likely to present with dermatophytosis to animal shelters than cats surrendered by owners. For example, a Portuguese study found that 17/136 (12.5%) hair samples taken from stray cats in a trap–neuter–return program were positive for M canis. 28 Moriello et al isolated M canis from 4.0% (n = 8/200) of healthy-appearing stray cats from a southern US shelter, but none in a northern US shelter. 9 Information about original intake source from partner shelters was not available in this study. Furthermore, intake source information from different shelters should be compared cautiously, as caretakers, municipalities or even different admissions staff may classify loosely owned cats differently. 31 As the practice of shelter-to-shelter feline transport is increasing nationally, 32 we believe these findings are still instructive.
In the population of dermatophyte suspect cats tested by clinicians at this shelter, the sensitivity of WL examination was 66.8% (95% CI 60.3–73.4%), similar to the figure of 72% suggested by analysis of pooled data from several studies reviewed by Moriello et al in 2017. 2 Specificity of WL examination was 74.8% (95% CI 64.2–85.1%) in this study. This low figure may be related to the level of experience and training of new clinicians at the study shelter: education levels ranged from supervised veterinary medical students and shelter medicine interns to boarded diplomates. The 17 cats declared falsely positive on WL exam included those with medical records in which the clinician documented being unsure about the WL result.
At a low total population prevalence of 1.8%, the predictive value of a negative WL examination is extremely high (99.2%), making this a valuable population screening tool. For populations of cats with a higher prevalence (eg, those with risk factors, history of exposure, lesions or other reasons to suspect dermatophytosis) WL negativity becomes a less useful predictor. Additionally, in this shelter population, close to three-quarters of the population of cats suspected of having M canis presented to the shelter as a family group or with a housemate. Although it was common for household groups to have mixed WL examination results, it was uncommon for groups to have mixed results on DTM culture: 78.8% (95% CI 61–91%) of the mixed WL groups had complete penetration of dermatophytosis.
The most significant result of this study is that through careful attention to daily examination of pending fungal culture plates, we found that 202/202 (100%; 95% CI 98.2–100) infected cat cultures showed colony growth consistent with M canis by day 7 of inoculation, with a median of 4 days, and 15/19 (78.9%; 95% CI 54.4–93.9%) of fomite carrier cat cultures showed growth by day 7, with a median wait time of 5 days. The daily growth results presented here could include up to 2 days’ lag period until culture recognition if first growth occurred on a weekend, so true median time until diagnosis may have been even shorter than reported.
A recent short report by Stuntebeck et al found that 674/1076 (62.6%) M canis-positive screening cultures from seven animal shelters showed results by day 7, and 98.2% were diagnostic by day 14. 16 In that report, toothbrush fungal culture was used as a screening test rather than a confirmation test, which could have picked up higher proportions of environmental M canis on incoming feline haircoats; fomite carriers and infected cats were not distinguished. Though daily examination of cultures was similarly performed, results were only reported on days 7, 14 and 21, making detailed comparison difficult. Indeed, both the present study and Stuntebeck et al support the finding that waiting 21 days for DTM culture finalization in shelters is excessive. 16 Recent studies in the human medical literature have also shortened recommended incubation times for human dermatophytosis diagnosis by about half. 33 Figure 1 in the supplementary material gives an example of how the results from this study could be used to inform a shelter’s dermatophytosis diagnostic protocol.
Conclusions
Transport kittens emerged as the population at greatest risk for M canis infection in this animal shelter. Results presented here suggest that shelter resources would be well spent targeting litters of kittens and transport cats for careful WL screening by trained staff and clinicians, as this inexpensive, efficient test was found to be reasonably reliable.
The results of this study support isolating WL-positive cats and starting treatment during the initial diagnostic culture incubation period. These findings also support presumptive isolation and initiation of treatment of all members of a litter or household group when some individuals are WL positive. Although this practice will result in holding a few M canis-negative cats for a little longer, in the majority of cases presumptive isolation and treatment will lead to earlier treatment completion for positive cats and prevent potential in-shelter spread of dermatophytosis. Results presented here also suggest that, given a similar population, testing and incubation conditions, shelters could consider releasing cats from treatment and isolation if their initial culture plate shows no sign of growth after 7–10 days.
The length of time an animal stays in the shelter is a significant animal welfare concern. The results of this report should be used by shelters to decrease feline length of stay by helping predict which cats are most likely to present with dermatophytosis, understanding the reliability of commonly used screening tests in shelter populations, and optimizing waiting times associated with fungal culture diagnosis.
Supplemental Material
Supplementary Figure 1
Example shelter dermatophytosis diagnostic rubric
Footnotes
Acknowledgements
The authors would like to thank Kris Otteman DVM, CAWA, DABVP-SMP for allowing the use of Oregon Humane Society (OHS) data for this paper; Erika Simms and Ed Pashkin for helping retrieve data; and the OHS cattery staff for their hard work caring for infected cats. The authors would also like to acknowledge the family of Vladimir (Vova) Dubrovsky, and honor the contributions of their late son, husband and brother.
Supplementary material
The following file is available online: Figure 1: Example shelter dermatophytosis diagnostic rubric.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
