Abstract
Objectives
The aim of this study was to compare serum phenobarbital concentrations, adverse events and client satisfaction during 14 weeks of transdermal vs oral phenobarbital administration to epileptic cats.
Methods
This was a prospective, fixed-order, crossover pilot study. Nine client-owned cats with presumptive or diagnosed idiopathic epilepsy were enrolled. Oral phenobarbital (PO-PB) was administered for weeks 1–14 (median starting dosage of 3.8 mg/kg [2.0–5.4 mg/kg/day] q12h); transdermal phenobarbital (TD-PB) was administered for weeks 14–28 (median starting dosage 18.8 mg/kg/day [17.6–24.0 mg/kg/day] q12h). Serum phenobarbital concentrations (S-PB) were measured at weeks 2, 14, 16 and 28. Client satisfaction questionnaires and biochemistry were evaluated at 14 and 28 weeks.
Results
Median S-PB concentrations during oral administration were 21 µg/ml (observed range 11–40 µg/ml) at week 2 and 22 µg/ml (8–35 µg/ml) at week 14, and at the higher TD dosage were 18 µg/ml (0–42 µg/ml) at week 16 and 17 µg/ml (7–50 µg/ml) at week 28. Phenobarbital concentrations were significantly correlated with PO dosage at week 2 (r = 0.75, P = 0.03) but not at weeks 16 and 28. Significantly more dose adjustments were needed during the TD phase (P = 0.03), but 6/9 owners (67%) still preferred TD to PO administration. Adverse effects were mild and comparable in both groups.
Conclusions and relevance
Therapeutic S-PB concentrations were achievable in some cats using TD-PB at 18 mg/kg/day q12h. Poor correlation between TD dosage and S-PB concentrations was observed and more dosage adjustments were required during TD administration. These findings necessitate close therapeutic drug monitoring if TD-PB is prescribed.
Introduction
Phenobarbital (PB) is the most commonly recommended antiepileptic drug for cats with non-metabolic seizures.1–3 When given as prescribed, oral phenobarbital (PO-PB) controls seizures in 93% of these cats if serum phenobarbital (S-PB) concentrations are maintained between 15 and 45 µg/ml. 4 PB is well tolerated in most cats and has a time to steady-state of 10–14 days after PO administration.5,6 However, PO drug administration to cats can be a significant challenge, resulting in stress to the cat, poor compliance and sometimes even injury to the owner. In one study, up to 18% of cat owners elected not to start recommended antiepileptic drug (AED) treatment because of difficulty with PO administration. 7
Transdermal (TD) drug administration has been reported to be better tolerated and easier to administer than PO administration in cats.8,9 In one study comparing PO and TD methimazole administration, 35% of cat owners reported missing at least one PO dose vs 8% during TD administration. 9 During the same study period, all owners reported easier administration by the TD route. 9
The overall purpose of this study was to compare S-PB concentrations, adverse events and client satisfaction during 14 weeks of TD- vs PO-PB administration to epileptic cats. Our primary aim was to determine whether serum concentrations correlated with dosage by both routes of administration. Our secondary aims were to compare the incidence of adverse events and evaluate client preference for both routes of administration.
We hypothesized that S-PB concentrations would correlate with dosage for both routes of administration, adverse events would be minor and comparable, and more clients would prefer TD to PO administration.
Materials and methods
This pilot study was designed as a prospective, fixed-order, crossover clinical trial in cats with idiopathic epilepsy. Cats were considered to have idiopathic epilepsy or presumptive idiopathic epilepsy based on modifications of the recent International Veterinary Epilepsy Task Force recommendations for diagnosing idiopathic epilepsy in dogs. 10
Cats were included if they: (1) met the modified tier 1 level of confidence and had a seizure history for a minimum of 1 year; or (2) met the modified tier II level of confidence. No other AEDs were allowed, but concurrent administration of other drugs such as methimazole, antibiotics, antiparasitic or topical flea/tick products were permitted. Cats receiving glucocorticoids were excluded from the study owing to unknown interactions with PB biotransformation. Cats diagnosed with idiopathic (or presumptive) epilepsy at study inclusion (‘newly diagnosed’) and cats diagnosed and on PB drug therapy for more than 2 weeks prior to inclusion in the study (‘previously diagnosed’) were included.
The study was approved by the University of Wisconsin-Madison Institutional Animal Care and Use Committee and all owners of enrolled cats provided written informed consent. Owners retained the right to withdraw from study participation for any reason, including refractory seizures.
TD-PB formulation
We previously evaluated S-PB concentrations in a population of healthy cats administered TD-PB using either pluronic lecithin organogel or a proprietary lipophilic liposomal cream vehicle (Lipoderm Activemax; Professional Compounding Centers of America). 8 The lipophilic liposomal cream was considered easier to administer by the owners in this prior study; therefore, it was selected as the vehicle for the current study. The UW-Veterinary Care Pharmacy, which was responsible for compounding the product, adheres to USP <795> compounding standards (www.USP.org). For the current study, we performed strength testing of PB in the lipophilic liposomic cream (250 mg/ml) at 1, 3, 6 and 8 weeks after formulation, using a Professional Compounding Centers of America-certified laboratory with strict adherence to USP compounding standards. (Eagle Analytical, Houston, TX, USA). Strength was maintained between 99% and 101% of the original sample at each time point through 8 weeks. This high uniformity was above the 85% variability considered acceptable for TD medications. 11 Based on these data, we prescribed PB formulated in the lipophilic liposomal cream in 8 week allocations. 8
PB administration
All cats were first treated with PO-PB on weeks 1–14, with a crossover to TD-PB on weeks 14–28 (Figure 1). There was no washout period between the PO and TD portions owing to the risk of breakthrough seizures. PO-PB was administered as generic tablets or liquid suspension from several manufacturers. For the PO phase, newly diagnosed cats were treated with 6 mg/kg/day PO-PB q12h; previously diagnosed cats were permitted to continue at their current dosage if S-PB was within 15–45 µg/ml at enrollment. A therapeutic reference interval for S-PB has not been well established in cats; therefore, the therapeutic reference interval of dogs (15–45 µg/ml) was used.4,12,13

Study flowchart for cats during the 28 week clinical trial. Cats entered the clinical trial at week 0 or week 2 (if already receiving oral phenobarbital). S-PB = serum phenobarbital; TD = transdermal; PO = oral
For the second phase of the study, PB was given TD at a total daily dosage of 18 mg/kg/day q12h. The dosage was selected based on our prior study of TD-PB in healthy cats, 8 in which 4/6 cats (67%) achieved serum concentrations within the therapeutic reference interval (15–45 µg/ml) when dosed at 18 mg/kg/day TD q12h. TD-PB was applied to the hairless portion of the inner pinnae surface by owners wearing disposable exam gloves. Owners were instructed to massage the ear until all observable PB gel was gone from the gloved finger.
Owners maintained a daily log of any observed adverse effects, difficulties with administration or missed doses, and recorded all observed seizures during both the PO and TD phases of the study. However, seizure control was not a specific outcome of the study owing to the relatively short duration of observation. Cats were re-evaluated at weeks 2 and 14 (during PO-PB administration), and weeks 16 and 28 (ie, 2 and 14 weeks after the start of TD-PB administration). At each visit, physical and neurologic examinations were performed, blood was drawn for S-PB concentration measurement and owners reported observational data from daily logs. Dosages were adjusted if S-PB concentrations were ⩽15 µg/ml, if adverse effects were noted, or if one or more seizures every 6 weeks or status epilepticus was noted. S-PB concentrations were evaluated 14 days after any dosage adjustments. On weeks 14 and 28, additional blood was drawn for repeat serum biochemistry and owners completed an online satisfaction questionnaire (Table 1).
Owner questionnaire administered after 14 weeks of oral (study week 14) or transdermal (study week 28) phenobarbital in epileptic cats

Serum samples were analyzed for PB concentrations through the University of Wisconsin-Madison, School of Veterinary Medicine, Clinical Pathology Laboratory, using a standard commercial immunoassay (Immulite 2000; Siemens Healthcare), validated for accuracy and precision, with a limit of quantitation of 5 µg/ml; for the purposes of this study, samples measuring <5 µg/ml were reported as undetectable (0 µg/ml).
Statistical analyses
Data are reported as median (with observed range) for demographic characteristics, seizure duration prior to enrollment and S-PB concentrations. Median S-PB concentrations at 2 and 14 weeks after starting and adjusting each dosing regimen were compared between the two routes of administration using a Mann–Whitney U-test. Serum concentrations were also compared after normalizing the given dosage to a standardized total daily dosage (6 mg/kg/day for PO and 18 mg/kg/day for TD). Actual total daily dosages and S-PB concentrations were correlated for each dosing regimen and at each time point using a Spearman correlation coefficient. Statistical analyses were performed using commercial software programs (Prism 7.0; GraphPad Software) with P <0.05 considered significant. Adverse events and client satisfaction are provided as descriptive data.
Results
Nine domestic shorthair, client-owned cats were included in the pilot study. Median body weight was 4.5 kg (range 3.5–6.5 kg) at enrollment, with a median age of 11.1 years (range 1.0–15.8 years). Seven of nine cats had a history of generalized seizures and two had complex partial seizures. At initial enrollment, all cats had normal interictal neurologic examinations.
PO phase
The median total daily PO-PB dosages at weeks 2 and 14 were 3.8 mg/kg (2.0–5.4 mg/kg/day) and 4.6 mg/kg (2.0–7.0 mg/kg/day), respectively, q12h. Median S-PB concentrations at 2 and 14 weeks were 21 µg/ml (observed range 11–40 µg/ml) and 22 µg/ml (observed range 8–35 µg/ml), respectively. Seven of nine cats had S-PB concentrations between 15 and 45 µg/ml at 2 and 14 weeks during PO treatment (Figure 2). S-PB concentrations were significantly correlated with PO dosage at 2 weeks (r = 0.75, P = 0.03), with a weaker non-significant correlation at 14 weeks (r = 0.53, P = 0.15). Two cats required one dose adjustment each during the PO phase because of low S-PB concentrations at week 2.
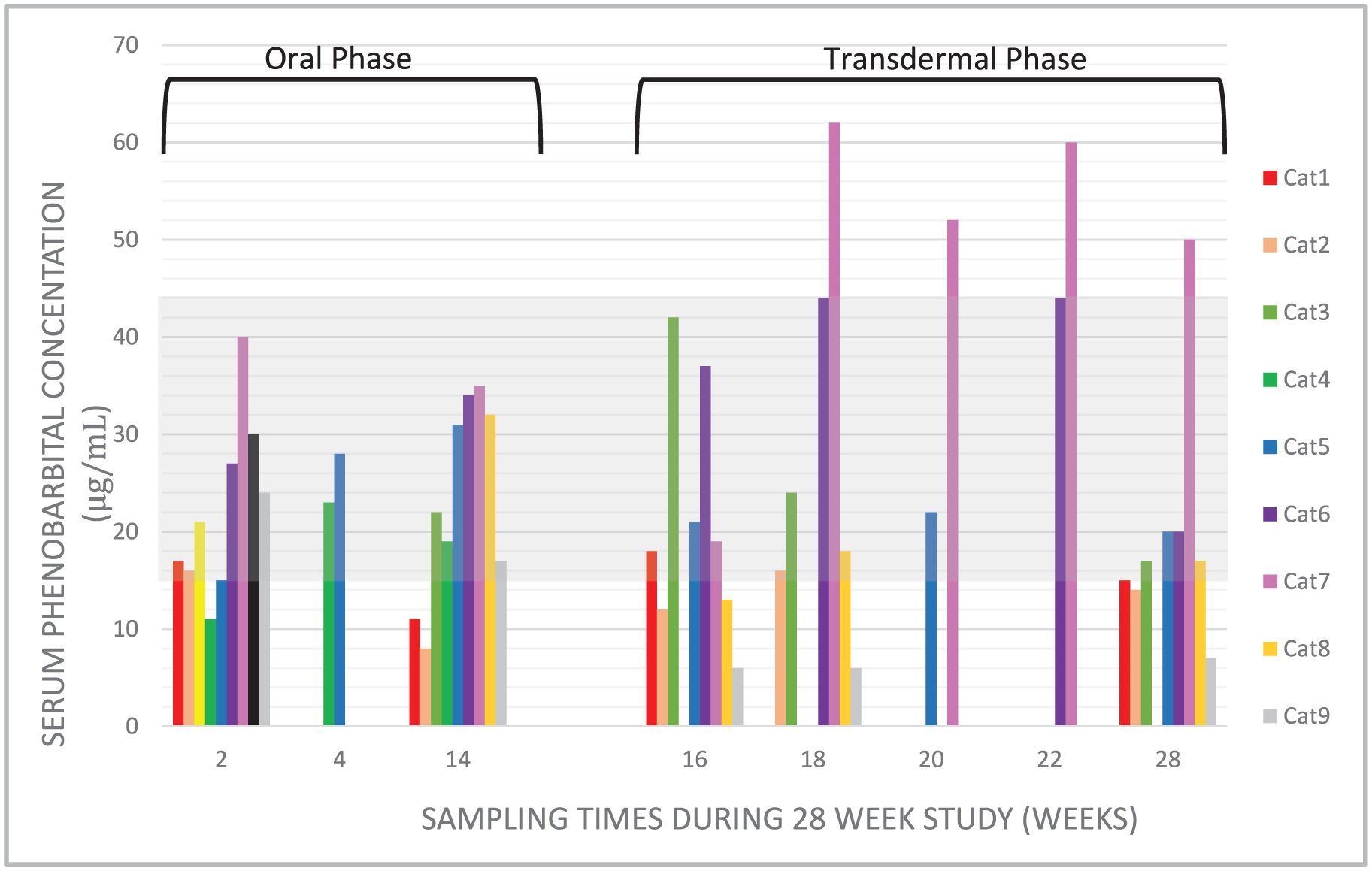
Serum phenobarbital concentrations for nine epileptic cats during oral or transdermal phenobarbital treatment. Cats received oral phenobarbital for weeks 1–14 and transdermal phenobarbital for weeks 15–28. The shaded area represents the canine therapeutic reference interval (15–45 µg/ml). Standard monitoring was at weeks 2, 14, 16 and 28; however, some cats had additional monitoring outside of these times
PO administration was reported as ‘easy’ by 4/9 owners (44%), ‘usually easy’ by 4/9 owners (44%) and ‘usually difficult’ by one owner. PO-PB was administered directly in six cats and in the food for three cats. Missed PO doses were reported ‘never’ by 5/9 owners (56%) over 14 weeks, ‘monthly’ by 2/9 of owners and ‘weekly’ by one owner, with one non-respondent.
Adverse effects were reported in 1/9 cats during PO administration and consisted of increased lethargy, ataxia and weakness during the first 14 days. A 50% dosage reduction led to resolution of clinical signs, with S-PB concentration still within the therapeutic reference interval. Two of nine cats had seizures reported (reported range 4–10 seizures) in the 14 week PO phase. No clinically significant change in serum biochemistry values, including liver enzyme activities, were noted between enrollment and week 14 (data not shown).
TD phase
The median total daily TD-PB dosage at study week 16 (2 weeks after switching to TD-PB) was 18.8 mg/kg/day (17.6–24.0 mg/kg/day), and at week 28 (14 weeks into TD-PB) was 18.6 mg/kg/day (13.2–32.4 mg/kg/day). The median volume of TD-PB per dose was 0.18 ml (0.11–0.26 ml) q12h. Median S-PB concentrations at 16 and 28 weeks were 18 µg/ml (observed range 0–42 µg/ml) and 17 µg/ml (observed range 7–50 µg/ml), respectively (Figure 2). Removal of cat 4 from the analysis (serum concentration 0) resulted in an adjusted median S-PB of 18.5 µg/ml (observed range 6–42 µg/ml) at week 16. Five of nine (56%) and 4/9 (44%) cats at 16 and 28 weeks, respectively, had S-PB concentrations within the range of 15–45 µg/ml (Figure 2).
During the TD phase, S-PB concentrations were not positively correlated with dosage at study week 16 (r = −0.15, P = 0.71) or 28 (r = −0.70, P = 0.06). Owing to a dose calculation error, one cat was started on TD-PB at 24 mg/kg/day; however, this cat still had undetectable S-PB concentrations at week 16. This cat was removed from the study owing to a marked increase in seizure frequency on TD-PB, and was returned to PO-PB administration. The dispensed syringes of TD-PB gel from this cat were sent for analysis and were found to have strength of 106% of the original dispensed 250 mg/ml concentration. This cat was included in outcome analyses but not long-term analyses of adverse effects.
All owners reported TD administration as ‘easy’ or ‘usually easy’. Missed TD doses were reported ‘never’ by 6/9 owners (67%) or only ‘monthly’ by 2/9 of owners. However, multiple owners reported difficulty dispensing the TD gel from the syringe owing to high viscosity.
Adverse effects were reported in 2/9 cats (22%) during TD-PB administration, and consisted of increased ataxia, weakness and lethargy. Serum PB concentrations measured for these cats were 35 µg/ml and 62 µg/ml, respectively. Dose adjustments for TD-PB were made once over 14 weeks in 6/9 cats, and two or more times in 2/9 cats (median one adjustment per cat, range 1–4). Dose adjustments were indicated owing to S-PB concentrations outside of the therapeutic reference interval (5/9 cats), adverse effects (2/9 cats) and poor seizure control (one cat). No irritation to the pinnae or excessive ear grooming was noted during TD dosing. Four of nine (44%) cats had observed seizures (range 1–4 seizures) during the TD phase. The cat with undetectable S-PB at week 16 experienced cluster seizures and status epilepticus and was removed from the study as reported above. No clinically significant change in serum biochemistry values was noted between weeks 14 and 28 (ie, during TD-PB administration).
Serum PB concentrations did not differ significantly between TD and PO administration when normalized for individual differences in dosage within routes, both at first steady-state sample (study week 2 for PO and study week 16 for TD; P = 1.0), and after chronic administration (study week 14 for PO and study week 28 for TD; P = 0.74). There were significantly more dosage adjustments during the TD phase compared with the PO phase (P = 0.03).
Overall, 6/9 owners (67%) preferred TD-PB administration to PO-PB administration. The remaining three owners preferred PO-PB administration owing to fewer requirements for dose adjustments (n = 2) and ease of administration for their cat (n = 1).
Discussion
The overall goal of this pilot study was to evaluate TD-PB administration vs PO-PB administration in epileptic cats. While most cats attained therapeutic S-PB concentrations during TD dosing, S-PB concentrations did not correlate with TD dosage. This may have been secondary to inconsistent owner application (eg, difficulty expelling the lipophilic liposomal gel from the syringe), drug diversion by other cats grooming the treated cat, or variable TD bioavailability. PB has several properties that make it a good candidate for TD administration:14,15 it is a small, lipophilic molecule (232 g/mole; log P <2.6), allowing absorption across the lipophilic stratum corneum, and it can be given at relatively small doses (<50 mg per dose). However, TD absorption is dependent on emulsification of the stratum corneum, and possibly formation of a drug depot in the subcutis, 16 and this may have been inconsistent among cats.
In contrast to TD-PB, during PO dosing, S-PB concentrations correlated well with PO dosage at week 2, suggesting consistent administration and bioavailability. This correlation was weaker at week 14 of PO administration, which may have been due to missed medication, as this was reportedly higher during PO vs TD dosing. Vomiting was reported in one cat during PO-PB, and it was within 24 h of blood sampling at week 14, which may have decreased PB absorption. Enhanced biotransformation of PB secondary to hepatic enzyme induction has not been found with chronic PO administration in cats, 5 so this mechanism is considered unlikely.
When normalized for individual differences in dosage, S-PB concentrations were similar for the two routes of administration after 14 weeks of each treatment. However, this was a pilot study with a small sample size, and was ultimately underpowered to detect the observed serum concentrations at week 14 (22 vs 17 µg/ml, a 22.7% difference) as significantly different. A post-hoc power calculation determined that 54 cats would have been necessary to detect this difference as significant, with 80% power. Therefore, these data should be considered preliminary and primarily descriptive. Our findings do suggest that an approximately three-fold higher TD-PB dosage yields S-PB concentrations that are clinically comparable to those reached with a standard PO dose; therefore, this fold-increase may be a good starting point when initiating TD-PB treatment or transitioning from PO-PB to TD-PB treatment.
The requirement for a higher TD dosage suggests lower bioavailability for TD-PB than PO-PB; however, bioavailability was not directly determined in this study. Importantly, dosage adjustments were indicated more often during TD administration vs PO administration. This finding may have been biased because some cats were already receiving chronic PO-PB before starting this study and, therefore, dose adjustments may have occurred in the initial adjustment period prior to study inclusion. Even so, veterinarians and owners should be prepared for careful and continued monitoring when initiating TD-PB in epileptic cats and for the possibility of frequent dosage adjustments to maintain therapeutic S-PB concentrations and adequate seizure control.
TD-PB was well accepted by owners in this study, even with more frequent visits for dosage adjustments. Administration was reported to be ‘easy’ or ‘usually easy’ by all owners, which is similar to previous studies of TD methimazole.9,17,18 Minor adverse effects reported by either administration route were consistent with previous reports of increased S-PB concentrations in cats and dogs during PO administration.2,19 The cats in our study with sedation and ataxia had S-PB concentrations ⩾35 µg/ml and showed improved clinical signs after dose reduction. No pinna irritation was noted with TD-PB in the lipophilic liposomic cream.
There were several limitations to this pilot study, notably the small number of enrolled cats and the non-randomized crossover design. We allowed prior treatment with PO-PB in order to increase the eligible pool of cats, and designed the study to examine the effects of transitioning from PO- to TD-PB; however, a randomized crossover design would have been preferred
Conclusions
The results from this pilot study demonstrate that therapeutic S-PB concentrations are achievable with TD-PB in some cats; however, S-PB concentrations did not reliably correlate with TD dose. Therefore, consistent monitoring of seizure control and S-PB concentrations, along with appropriate client counseling, is recommended when prescribing TD-PB. Client satisfaction was greater and fewer doses were missed with TD-PB vs PO-PB. Prescribing veterinarians should be aware that individual variation in TD absorption or administration might affect the utility of TD-PB. Additionally, variable compounding standards of individual pharmacies may affect the absorption and therefore serum concentrations achieved in individual cats.
Monitoring of S-PB concentrations 14 days after initiation of therapy, and after every dose adjustment, is strongly recommended when managing an epileptic cat with TD-PB. PB is considered a Drug Enforcement Administration class IV controlled substance; therefore, prescribers should select patients carefully and educate clients fully on the risks of repeated human or non-epileptic animal contact during the course of treatment for the epileptic cat and the risk of casual contact with PB during TD application.
Footnotes
Acknowledgements
The authors thank Drs Mackenzie Jeffers, Sandra Sawchuck, Roberta Duresa, Calico Schmidt, Muna Qahwash, Helena Rylander, Jodi Bohl and Ms Laurie Martin for referral of the cats included in this study.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding for this study was provided by the University of Wisconsin-Madison School of Veterinary Medicine Companion Animal Grant Fund.
