Abstract
Objectives
The aim of this study was to evaluate and quantify the changes in neurological status in cats after perineal urethrostomy performed in dorsal and ventral recumbency.
Methods
This was a prospective, randomised study. Twenty male castrated cats with feline lower urinary tract disease presented for perineal urethrostomy were enrolled in this study. Surgery was performed in either dorsal recumbency (group A) or ventral recumbency (group B). Motor response of patellar tendon, gastrocnemius muscle, pelvic limb withdrawal and perineal reflexes, as well as the presence of spinal pain in the lumbosacral region, motor function of the tail and faecal continence, were examined before surgery, and 24 h and 14 days after surgery.
Results
The animals had a mean weight of 5.07 ± 1.08 kg, with a mean age of 6.12 ± 1.85 years. Weight and age were not significantly different between groups A and B (both P = 0.897). All tested parameters of the neurological examination performed prior to surgery were considered normal in both groups (P = 1). The comparison between neurological examinations (perineal reflex and spinal pain) before and 24 h after surgery revealed a significantly decreased briskness of the perineal reflex and an increased occurrence of spinal pain 24 h after surgery (P = 0.043 and P = 0.031, respectively). However, the changes of aforementioned parameters were statistically insignificant (P = 0.249 and P = 0.141) between groups A and B. The other parameters (patellar tendon, pelvic limb withdrawal and gastrocnemius muscle reflexes, motor function of the tail and faecal continence) were statistically insignificant (P = 1) before surgery and 24 h after surgery, as well as between groups A and B 24 h after surgery. Results of all tested parameters were statistically insignificant (P = 1) before surgery and 14 days after surgery, as well as between groups A and B 14 days after surgery.
Conclusions and relevance
The briskness of the perineal reflex was significantly decreased and the occurrence of spinal pain significantly increased 24 h after surgery. A parallel with a low-grade positioning-dependent nerve injury as described in human medicine may be drawn. However, no positioning method was proven to be superior to the other.
Introduction
Urinary tract infection (UTI) is a well-documented complication after perineal urethrostomy (PU) in cats and accounts for 17–57% of all long-term complications associated with this surgical procedure.1–5
The pathophysiology of this condition is still not fully understood and is mostly characterised as multifactorial. 6 Traditionally, suggested possible causes include postoperative use of indwelling catheters, 7 reduction of the urethral length, 6 creation of a stoma with a larger opening and closer proximity to the anus, 6 decrease of the urethral tonus secondary to neuromuscular changes, 8 removal of the part of the urethral mucosal barrier 6 and damage to the neurological structures during dissection. 8
A recently published anatomical cadaveric study by the same authors considered the reduction of the vertebral canal diameter arising from the perioperative positioning of the animal as a possible contributor to neuropathy in in the lumbosacral region. According to the authors, the reduction in the diameter of the vertebral canal between the sixth lumbar and second coccygeal vertebrae may directly or indirectly lead to positioning-dependent iatrogenic nerve injury. 9
The objective of this study was to evaluate the neurological status in cats before and after perineal urethrostomy performed in dorsal and ventral recumbency and to determine the more suitable physiological method of positioning.
Materials and methods
Inclusion criteria
Male, castrated, adult, neurologically normal European Shorthair cats with feline lower urinary tract disease requiring a perineal urethrostomy were enrolled in this study. Indications for surgery were urethral obstructions that could not be conservatively unblocked, recurrence of urethral obstruction despite conservative therapy or urethral trauma secondary to obstruction.
Surgical procedure
The cats were premedicated with midazolam (0.1 mg/kg IV, Midazolam; B Braun Melsungen), ketamine (1 mg/kg IV, Narketan; Vetoquinol) and methadone (0.1 mg/kg IV, Comfortan, Albrecht). General anaesthesia was induced with propofol (2 mg/kg IV, Narcofol; CP-Pharma) and maintained with a mixture of isoflurane (Isofluran; Baxter) and oxygen. Cefazolin (22 mg/kg IV, Cefazolin; Hexal) or amoxicillin clavulanate (12.5 mg/kg IV, AmoxClav; Hexal) was given after induction and lactated Ringer’s solution (Sterofundin; B Braun Melsungen) was administered at a rate of 5–10 ml/kg/h through an IV catheter.
The patient was positioned either in dorsal or ventral recumbency for surgery. The skin was clipped, surgically prepared with povidone-iodine solution (Braunoderm; B Braun Melsungen) and covered with sterile field drapes. Perineal urethrostomy was performed as described by Wilson and Harrison. 10 The procedure was performed either by a board-certified surgeon, senior surgeon or surgical resident. All surgeons had comparable experience with perineal urethrostomy in dorsal, as well as ventral, recumbency. The surgical approach was the same in both positions. Postoperative treatment consisted of fluid therapy with IV crystalloid fluids and pain management with buprenorphine (0.01–0.03 mg/kg q8h, Buprenovet; Bayer) and metamizole (20–40 mg/kg q8h, Vetalgin; MSD). Antibiotic therapy was continued for 5 days after surgery and an Elizabethan collar was worn by all cats.
Perioperative positioning
Perioperative positioning of cats in dorsal or ventral recumbency was performed as described by Slunsky et al. 9 For dorsal recumbency the thoracic limbs were tied in a caudal direction and the pelvic limbs in a cranial direction using gauze. The tail hung free over the surgical table (Figure 1). Ventral recumbency was accomplished by tying the thoracic limbs in a cranial direction and the pelvic limbs over the edge of the table in a ventrolateral position. The tail was hyperextended and secured with gauze to the surgical table, so access to the perineal region was ensured (Figure 2). All operating tables were soft-covered with foam material. The authors considered the surface padding adequate for safe positioning. Additional padding (ie, towels) was not deemed necessary and would have increased the variables of the study by possibly affecting the mechanical forces on the fixated body.

Dorsal recumbency (group A). (a) The thoracic limbs are pulled in a caudal direction and the pelvic limbs in a cranial direction with gauze. (b) The tail hangs free over the surgical table

Ventral recumbency (group B). (a) The thoracic limbs are pulled in a cranial direction and the pelvic limbs are tied over the edge of the table in a ventrolateral position. (b) The tail is hyperextended and secured with gauze to the surgical table so access to the perineal region is ensured
Neurological assessment
Cats were assessed preoperatively, and 24 h and 14 days postoperatively. All neurological examinations were performed by one examiner (PS) under the supervision of an ECVN diplomate (SL), and included: patellar tendon reflex; gastrocnemius muscle reflex; pelvic limb withdrawal reflex; perineal reflex; the presence of spinal pain by palpation of the paraspinal muscles in the lumbosacral region and motor function of the tail; and faecal continence. The patellar tendon reflex and gastrocnemius reflex were tested using a reflex hammer. Pelvic limb withdrawal and perineal reflexes were tested by applying an adequate painful stimuli using arterial forceps as described by de Lahunta and Glass. 11 The examiner evaluated if the reflex was present or absent (0 = absent, 1 = present) and scored the level of reflex briskness using a scoring scale (0 = absent, 1 = reduced, 2 = normal, 3 = exaggerated, 4 = clonus). Spinal pain was assessed by palpating the paravertebral muscles (0 = absent, 1 = present). Motor function of the tail and faecal continence were assessed by visual observation of the animal (0 = absent, 1 = present).
Randomisation and statistical analysis
The method of positioning was drawn at random (drawing black and white balls from a box with replacement) immediately before surgery and the animals were assigned either to group A (surgery in dorsal recumbency) or group B (surgery in ventral recumbency). The Mann–Whitney U-test was used to test for differences in the group parameters (patellar tendon reflex, gastrocnemius muscle reflex, pelvic limb withdrawal reflex, perineal reflex, age, weight). The Wilcoxon test was used to investigate the changes in the parameters in pairs (patellar tendon reflex, gastrocnemius muscle reflex, pelvic limb withdrawal reflex, perineal reflex), between the examination times (pre-surgery, and 24 h and 2 weeks postoperatively). Fisher’s exact test was used to calculate the group differences in the criteria ‘spinal pain’, ‘motor function of the tail’, ‘faecal continence’ at the different examination times. McNemar’s test was utilised to investigate if the criteria ‘spinal pain’, ‘motor function of the tail’, ‘faecal continence’, changed between the examination times (pre-surgery, and 24 h and 2 weeks postoperatively).
All statistical models were analysed using a software programme for statistical analysis (IBM SPSS Statistics 22). Probability values were reported, with P <0.05 considered statistically significant.
Results
Twenty cats fulfilled the inclusion criteria and were enrolled in the study. The mean weight was 5.07 ± 1.08 kg and the mean age was 6.12 ± 1.85 years. Weight and age were not significantly different between groups A and B (both P = 0.897).
Neurological status of the cats prior to surgery
All tested reflexes (patellar tendon reflex, pelvic limb withdrawal reflex, gastrocnemius muscle reflex and perineal reflex) were considered normal in both groups. None of the cats had spinal pain in the lumbosacral region. None of the cats had loss of motor function of their tail or faecal continence. The comparison between the neurological examinations in groups A and B yielded insignificant results (P = 1).
Neurological status of the cats 24 h after surgery
In group A, the perineal reflex was reduced in one cat (1/10; 10%). The same patient (1/10; 10%) suffered from spinal pain in the lumbosacral region. The perineal reflex in group B was reduced in three cats (3/10; 30%) and completely absent in one cat (1/10; 10%) (Figure 3). Spinal pain was present in five cats (5/10; 50%). Only one cat with postoperative spinal pain had a normal perineal reflex (1/10; 10%) (Figure 4). The comparison of the neurological examinations (perineal reflex and spinal pain) before surgery and 24 h after surgery yielded significant results (P = 0.043 and P = 0.031, respectively), but the changes of aforementioned parameters were statistically insignificant (P = 0.249 and P = 0.141, respectively) between groups A and B at this time point. Results of the other tested parameters of the neurological examinations (patellar tendon reflex, pelvic limb withdrawal reflex, gastrocnemius muscle reflex, motor function of the tail, faecal continence) were statistically insignificant (P = 1) before surgery and 24 h after surgery, as well as between groups A and B 24 h after surgery.
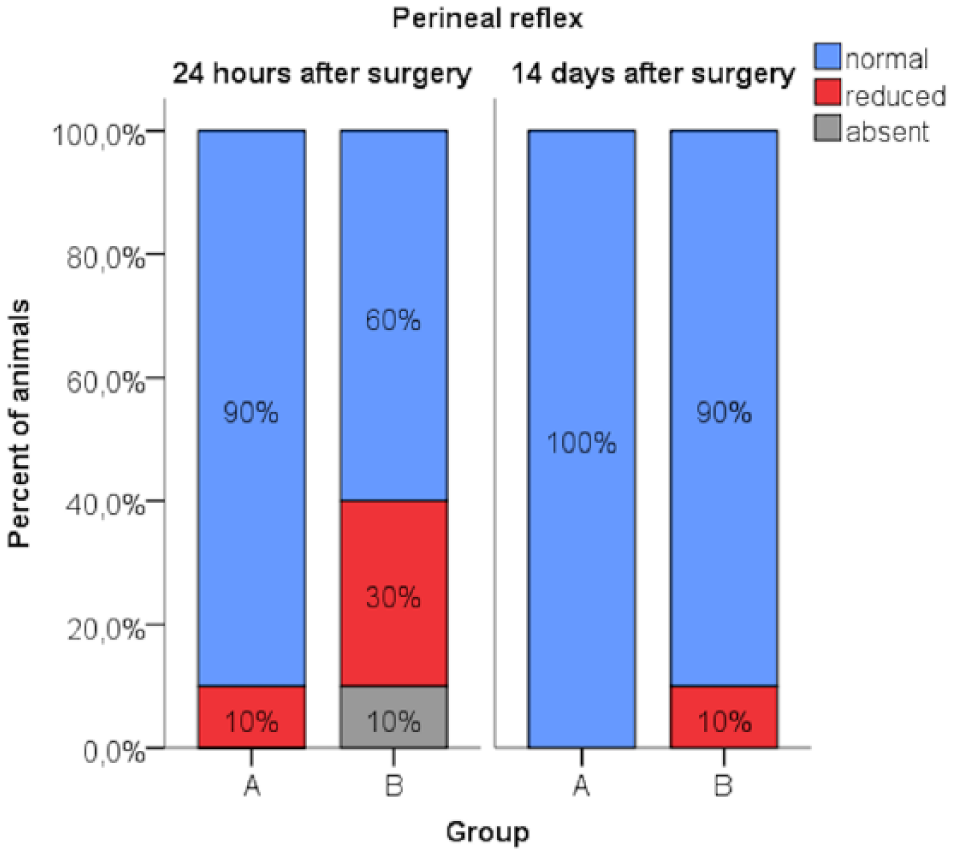
Briskness of the perineal reflex in groups A and B, 24 h and 14 days after surgery

Presence of spinal pain in groups A and B, 24 h and 14 days after surgery
Neurological status of the cats 14 days after surgery
All tested reflexes in the cats in group A were considered normal 14 days after surgery. One cat (1/10; 10%) from group B had a reduced perineal reflex (Figure 3). In this cat the perineal reflex was absent during the examination 24 h after surgery; therefore, the neurological status of this cat had improved slightly but was not normal. None of the cats in either group had spinal pain in the lumbosacral region (Figure 4). Results of all tested parameters of the neurological examinations (patellar tendon reflex, pelvic limb withdrawal reflex, gastrocnemius muscle reflex, perineal reflex, spinal pain, motor function of the tail, faecal continence) were statistically insignificant (P = 1) before surgery and 14 days after surgery, as well as between groups A and B, 14 days after surgery.
Discussion
Patient positioning as a cause of postoperative neuropathy was first recognised by Halsted in 1908. 12 Current publications from human medicine report incidences of postoperative peripheral neuropathy ranging from 0.03–25%, depending on the criteria used to define neuropathy. 13 In large animals, iatrogenic nerve injuries caused by inappropriate perioperative positioning are well described and usually relate to cases of limb paresis or paralysis.14–17 Information about iatrogenic nerve injuries in small animals caused by inappropriate perioperative positioning have been described experimentally and include studies in rodents.18,19 High-grade neurological deterioration is represented only in a small group of human patients. 20 Low-grade, transitional motor and sensory deficits are much more common in human beings, but may still lead to significant health problems. 21 The diagnosis for this mild neurological deterioration requires a well-aimed neurological examination and often the use of specific diagnostic methods. 22 Consequently, the question to be asked is: do positioning-dependent low-grade nerve injuries occur in cats and is it simply an under-diagnosed condition?
A previously published study by the authors hypothesised that PU in cats performed in dorsal recumbency is superior to PU in ventral recumbency with regard to the risk of positioning-dependent iatrogenic nerve injury. 9 This hypothesis could not be confirmed in this clinical trial. The neurological status was not significantly different between the two groups of animals on which PU was performed (dorsal recumbency, group A; ventral recumbency, group B). The result may reflect the clinical insignificance between dorsal and ventral positioning during PU surgery. However, the lack of statistical significance in this study could also be due to the small animal population or the relatively low sensitivity of the performed neurological examination.23–25
Neurological examination is a semi-quantitative diagnostic method, which is considerably subjective and strongly investigator-dependent.26–28 Other, more sensitive, methods such as electrodiagnostic or urethral pressure profilometry could be more suitable.8,29 Unfortunately, these methods are relatively invasive, requiring general anaesthesia, and are time-consuming. In clinical patients without a medical indication warranting such diagnostic methods, an official application for animal experiments given by state government is necessary, as well as the consent of the owner. In the country of this study’s origin it was not likely to receive such permission and ethical consent; therefore, from a legal and ethical point of view, it was not justifiable to employ such methods on otherwise healthy, privately owned animals. Thus, the authors were constrained to restrict the diagnostics to a neurological examination.
The significantly reduced briskness of the perineal reflex found 24 h after surgery returned to normal 14 days after surgery. The difference between group A and B was statistically insignificant. The perineal reflex evaluates the spinal cord segments S1 and S3. 30 The motor neurons originate in the spinal cord segments between (L7) S1–S2 (S3) for the pudendal nerve 31 and (S1) S2–S3 for the pelvic nerve. 32 The pudendal nerve provides somatic innervation to the periurethral striated muscle at the bladder neck (external urethral sphincter). The external urethral striated muscle remains in a state of steady contraction, contributing to active urethral resistance during urine storage and is inhibited during reflex urination. The parasympathetic pelvic nerve initiates reflex detrusor muscle contraction and subsequent urination. Injury of these nerves lead to a decrease of active urethral resistance in the case of pudendal nerve injury and to urine retention and subsequent urine overflow in the case of pelvic nerve injury. 33
The pathophysiology leading to a reduced briskness of the perineal reflex remains unclear. A reduction of the vertebral canal diameter between L7–S1, 9 stretching of the peripheral nerves, 34 direct pressure on the peripheral nerves, 35 ischaemic myopathy and/or ischaemic neuropathy 36 and dorsal hyperflexion of the tail in cats positioned in ventral recumbency 9 may all be possible ethological factors leading to injury of the aforementioned nerves. The injury of these nerves and the resulting functional deficits may potentially be one of the factors contributing to the development of UTI in cats after perineal urethrostomy, but further clinical evaluation in a larger, long-term study, as well as objective diagnostic methods (electrodiagnostic, urethral pressure profilometry), are needed to confirm this hypothesis.
Spinal pain was present with statistical significance 24 h after surgery in both groups but was not significantly different between the animals in which PU was performed in either dorsal recumbency (group A) or ventral recumbency (group B). Hyperalgesia of the paralumbar musculature in the lumbosacral region may reflect a non-specific lesion of the spinal cord in this area 37 or ischaemic myopathy caused by pressure on the paralumbar muscles. 38 Interestingly – although not statistically significant – more animals were affected in group B (5/10). These were fixated in ventral recumbency with no contact of the lumbar musculature to the surgical table. Only 1/10 animals was affected in group A. This might be indicative of a neurological rather than a muscular problem; however, a larger study utilising electromyography would be necessary to verify this hypothesis. Until then, the aetiology of the hyperalgesia remains unclear.
This study has some potential limitations. This study’s cat population was relatively small. A power analysis had not been performed prior to the study. Statistically, the chance of achieving a significant result increases with the same effect/difference as the sample size increases. Therefore, a non-significant result may not mean that there is no correlation or difference.39,40 Extension forces on the extremities and tail were not quantified and unified, but only forces required for stable positioning and adequate surgical view were applied. Furthermore, a complete neurological examination was not performed on the animals. The reason for this was a rapidly developed antipathy of the cats towards this procedure and a consequent aggressive behaviour during follow-up examinations. Based on this finding, the neurological examination was limited to the hindlimb only. Any postoperative abnormality in urination was evaluated in this study. Based on our experiences, many variables (ie, postoperative pain, mucosal swelling, hematoma formation, underlying cystitis, foreign environment, etc) play a role in the unwillingness of the cats to urinate after perianal urethrostomy. These conditions were not subject of this short-term study.
Conclusions
The briskness of the perineal reflex was significantly decreased and the occurrence of spinal pain significantly increased 24 h after surgery; therefore, a parallel with a low-grade positioning-dependent nerve injury as described in human medicine may be drawn, but neither positioning method was proven to be superior to the other. The presence of aforementioned neurological abnormalities (reduction/absence of perineal reflex, spinal pain) as a cause for postoperative UTI remains unclear and should be assessed in a long-term study.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
